Abstract
Objective
The aim of this study was to test reproducibility of the CT Pico method in a cadaveric model and to compare CT Pico measurements with a high-precision laser probe for optical scanning.
Materials and methods
The glenoid surface of ten dried cadaveric scapulae (with intact surface) was measured by and high-precision laser probe for optical scanning, the latter being assumed as a reference standard. Measurements were done according to the Pico technique, using a circle-shaped region of interest (ROI) that was placed on the inferior glenoid rim. Measurements obtained using the CT Pico method (three readers) and with laser were compared to assess differences between radiological assessment and the reference standard. Each observer performed two repeated measurements from each scapulae (20 for each observer).
Results
Mean differences between laser measurements and each CT reader were 18.4 % (range, −4 to 61 %) for reader 1, 12.4 % (range, −15 to 64 %) for reader 2, and 11 % (range, −14 to 58 %) for reader 3. Considering all the 60 measurements made by the three readers, 39 measurements out of 60 (65 %) were outside the range [−5 %; +5 %] while 26 measurements (43 %) are outside the range [−10 %; +10 %]. The largest differences (positive and negative) were +64 and −14 %, respectively. Intra-operator reproducibility was high in most cases (intraclass correlation coefficient (ICC) =0.93, ICC = 0.91, ICC = 0.93 and Lin’s Concordance correlation coefficient (CCC) = 0.92, CCC = 0.90, CCC = 0.92 for reader 1 to 3, respectively. However, in five cases the CT Pico measurements showed absolute differences between the first and second measurements that exceeded 10 %.
Conclusions
The inter-observer variability for CT measurement of the glenoid surface using the CT Pico method was high when compared with laser, in the assessment of glenoid surface in cadaveric specimens, thus the CT Pico method is not reliable and could cause errors in the clinical management of the patient.
Level of evidence
Level II, Development of diagnostic criteria on consecutive patients (with universally applied reference “gold” standard)
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The quantification of bone loss is a crucial factor for correct preoperative planning and for explaining failed soft tissue reconstructive operations for shoulder instability in order to reduce the risk of recurrent dislocation of the shoulder after surgery [1].
The prevalence of glenoid bone loss in traumatic unidirectional shoulder instability, also termed TUBS (traumatic, unidirectional, Bankart lesion, surgical treatment) ranges from 8 to 90 %, depending on the series [2, 3]. Because TUBS involves antero-inferior humeral dislocation, the anterior aspect of glenoid surface is usually damaged (bony Bankart lesion) [4].
Surgery is the elective treatment for TUBS, and various surgical techniques are used according to the amount of bone loss. If there is significant glenoid bone loss, a coracoid transfer (Latarjet procedure) is performed, rather than arthroscopic treatment [5–7].
According to Burkhart et al., glenoid bone loss greater than 25 % when, calculated with the laser arthroscopic method, correlates with a recurrence of dislocation after arthroscopic surgery in approximately 67 % of patients, whereas in the presence of minor defects the recurrence rate drops to 4 %, especially in the athletic population [7].
These data led the same authors to state that with a 25 % or more glenoid bone deficit, arthroscopic repair by itself cannot restore the glenohumeral joint to stability [7].
Arthroscopic assessment of glenoid bone loss is considered the gold standard by orthopedic surgeons for quantification of glenoid defects [8]. This invasive method is based on the measurement of antero-posterior diameter of the inferior glenoid surface. Less invasive techniques for the assessment of the antero-posterior diameter by radiological measurement have been shown to produce similar results to arthroscopy [9]. Among others, the CT Pico method is currently used for quantification of glenoid bone loss because of its simplicity and ease of comparison with arthroscopic measurements. This method uses CT with a slice thickness (0.6–1.0 mm) in the sagittal plane followed by 2-D multiplanar reconstructions. The glenoid surface is calculated by means of a circle-shaped region of interest (ROI) placed on the inferior glenoid rim, first in the unaffected glenoid, after transfer the same circle to a reconstructed sagittal image of the affected side; finally the difference of the diameter of both circles is evaluated [10]. Some authors suggest using the CT Pico method by magnetic resonance imaging (MRI) [11]. However, little is known about the reproducibility of the CT Pico method and no comparison between Pico and reference standard quantitative measurement has been reported in the literature [6]. Notably, the main weakness of the CT Pico method can be found in its geometrical two-dimensional approach that could be a limitation in the assessment of surfaces with intrinsic anatomical and post-traumatic variability; in fact, the inscription of the inferior glenoid in a circle using 2-D CT images, could not correspond to the real anatomy of the glenoid surface, which, besides being curve, morphologically is difficult to compare to a circle. The aim of this study was to test the reproducibility of the CT Pico method in a cadaveric model and to compare CT Pico measurements with a high-precision laser probe for optical scanning.
Materials and methods
The hospital ethics board did not require family approval or informed consent for radiologic examination of specimens from the collection of our Institute of Anatomy.
The glenoid surface of ten dried cadaveric scapulae (with intact surface) was measured by CT and laser methods (Fig. 1). Inclusion criteria for anatomical specimens were the presence of a whole glenoid bone surface and integrity of the glenoid bone edge. Scapulae with an impaired glenoid surface or damaged edge were excluded from this study.
Laser measurement
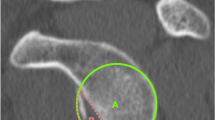
A laser probe for optical scanning system (CAM2, Faro Technologies, Lake Mary, www.faro.com) was used to measure the glenoid area. This certified high-precision device provides measurement of surfaces and volumes with a precision of 10−6 meter. A laser probe on the top of a seven-axis robotic arm allows detection of surface features of the scanned objects via coupled registration of laser feedback and robotic arm. A three-dimensional cloud of points is created and converted into the physical reconstruction by dedicated software (Geomagic Studio, version 10; Geomagic, Morrisville, NC; hyperMILL, Open Mind Technologies, Needham, MA; and Autodesk 3ds Max, Autodesk, San Rafael, CA). The volumetric model is stored as Initial Graphics Exchange Specification (IGES file). Geometric features of this volumetric model can be calculated. Laser calibration was provided by the manufacturer by means of metal cylinder with a nominal diameter of 25.420 mm. The technical data supplied with the unit: accuracy of the measurement (diameter) (0.01 mm) and repeatability (0.022 mm). The glenoid surface of each specimen was laser-scanned by a mechanical engineer with a 4-year experience in CAM2 practice in the setting of mechanical parts 3D scanning and reproduction. The same reader measured glenoid surfaces on an IGES file using CAD software (AutoCAD® 2013), according to the Pico technique, and then tracing a circumference of the circle in the region of interest (ROI) in a sagittal plane. This employs a circumferential region of interest (ROI) that is placed on the inferior glenoid rim using a 3-D model (Fig. 3b, c) [7, 12]. Measurements were performed twice, 2 weeks apart.
CT measurements
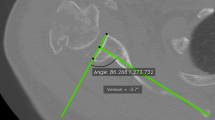
Volumetric CT scan was done with a 64-detector row CT scanner (SOMATOM Sensation 64; Siemens, Erlangen, Germany). Acquisition parameters were as follows: collimation 64 × 0.6 mm, tube current 120 mA, tube voltage 120 kV, rotation time 500 ms, and pitch 1. Reconstruction parameters were as follows: slice thickness 0.6 mm, slice increment 0.3 mm, high spatial frequency kernel (B90 for bone reconstruction), and bone window WL200 WW2000. The adopted acquisition and reconstruction protocol allowed for 0.3-mm spatial resolution of CT images. CT images were reviewed on a Volume Zoom Wizard workstation (Siemens, Erlangen, Germany) by three radiologists (>10-years of experience in musculoskeletal radiology) with the aim of measuring the glenoid surface (Fig. 4). First, the CT Pico method was used to quantify the inferior glenoid surface on volumetric CT images (slice thickening 0.6 mm) [10]. Because the CT Pico method relies on the anatomical assumption that the “bare spot” of articular glenoid surface is in the center of a circumference overlaying the inferior glenoid, oblique sagittal flat multiplanar reformatting (fMPR) was obtained to allow an “en face” view of the glenoid surface. Each observer created his or her own MPR planes for each measurement.
The landmarks of the observers, to position the circle, were the anterior and posterior margins of the equatorial region of the glenoid. The equator was angled so that it would be perpendicular to the true north–south axis of the glenoid. An electronic circumference of the circle in the region of interest (ROI) was placed on the sagittal CT glenoid surface, with particular attention to overlaying this geometric circumference on the inferior glenoid bone edge. The circle did not extend beyond the cortical bone of the inferior glenoid surface (Fig. 4). To assess intra and inter-observer agreement, the three radiologists repeated the measurements 1 month apart on each glenoid specimen [7, 13]. Overall, the three readers carried out 60 measurements using the CT Pico Method (20 for each reader, two for each scapular glenoid). Pico and laser measurements were compared to assess differences between radiological assessment and the reference standard.
Statistical analysis
Glenoid surface measurements were reported as absolute values (cm2), whereas the difference between CT and laser measurements was reported as percentage of the laser value.
Mean, standard deviation, standard error, minimum, maximum, and range, were calculated for each set of CT measurements performed by the three readers. Difference between the repeated CT and laser measurements was assessed by factorial ANOVA and non-parametric tests. The significance level α = 0.05 was used throughout. Intra-operator and inter-operator agreement were assessed by Lin’s concordance correlation coefficient (CCC) [14] and intra-class correlation coefficient (ICC) [15].
Statistical analyses were performed by IBM-SPSS v.20 and by the widely known open source statistical software R, version 3.1.3. Preliminary Power analysis has been performed by the statistical software GPower 3.0.10 (http://www.psycho.uni-duesseldorf.de/abteilungen/aap/gpower3/) and by R v. 3.1.3. with the additional packages “pwr” v.1.1–2 and “Hmisc” v.3.16–0 (http://biostat.mc.vanderbilt.edu/Hmisc) [16]. Using previous information on these measurements from which we could assume an effect size (Cohen’s d [17]) around 1, or more. Power analysis: with an effect size d = 1, α = 0.05, power (1-β) = 0.80 (80 %), 95 % confidence interval (CI), we obtained a sample size equal to 10. The range [−5 %; +5 %] of the reader’s difference values respect to the reference standard was considered excellent, while the range [−10 %; +10 %] was considered moderate.
Results
The summary of the first and second measurements of the glenoid surface are shown in Table 1.
Factorial ANOVA (repeated measures) (SPSS, General Linear Model (GLM), repeated measure) found a statistically significant difference between the results from the laser and the readers measurements (p = 0.010), the largest difference coming from reader 1 (Fig. 1). There is a generalized overestimation of glenoid area for all three CT readers with respect to laser measurement.
Mean differences between laser measurements and each CT reader were 18.4 % (range, −4 to 61 %) for reader 1, 12.4 % (range, −15 to 64 %) for reader 2, and 11 % (range, −14 to 58 %) for reader 3 (Table 2).
Consistency among the readers was considered high, while the absolute agreement measured by the intraclass correlation coefficient was ICC = 0.94 (CI 95 %: 0.83; 0.99) for average measurements and ICC = 0.81 (CI 95 %: 0.54; 0.94) for single measurements.
When evaluating the most discrepant measurement among the two repetitions from each reader, the agreement fell to ICC = −0.01, showing no agreement with the reference. Moreover, considering all 60 measurements made by the three readers, 39 measurements out of 60 (65 %) were outside the range [−5 %; +5 %] while 26 measurements (43 %) were outside the range [−10 %; +10 %] (Fig. 2).
The largest differences (positive and negative) were +64 and −14 %, respectively (Table 2).
Intra-operator reproducibility (same operator in two occasions) of the CT Pico method was moderate-high in most cases (ICC = 0.93, ICC = 0.91, ICC = 0.93 and CCC = 0.92, CCC = 0.90, CCC = 0.92 for reader 1 to 3, respectively) (Table 3).
However, in five cases, the CT Pico measurements found absolute differences between the first and second measurements that exceeded 10 %).
Discussion
The most important findings of this study were the presence of a significant difference between the measurements by the CT Pico method and laser measurements (p = 0.010) and a large inter-observer variability for assessment of glenoid bone surface in the cadaveric model. The inter-observer variability for CT measurement of the glenoid surface using the CT Pico method was unacceptably high when compared with laser, up to 64 % greater than laser, thus the CT Pico method could cause errors in the clinical management of the patient.
The inscription of the inferior glenoid in a circle, which occurs on flat MPR images, did not correspond to the real anatomy of the glenoid surface, which, besides being curved, is difficult to compare to a circle, thus reducing the accuracy of the CT Pico method (Fig. 3c–d).
One normal glenoid specimen (a) and the model derived from laser scanning (b) are illustrated. The measurement of the glenoid surface by the CT Pico method is reported on laser (c) and CT data (d). Note as a part of the anterior glenoid is not included in the circle using the CT Pico method (arrows in c and d). The inscription of the inferior glenoid in the circle, which occurs on flat MPR images (d), could not correspond to the real anatomy of the glenoid surface, which, besides being curved, is never comparable to a circle; this obviously reduces the accuracy of the CT Pico method
Since reliability of such a method appears to be weak when considering the glenoid surface of cadaveric specimens, it is probably not accurate for measuring the amount of bone loss in cases where the glenoid is deficient (Figs. 3 and 4)
However, further studies “in vivo” are needed to confirm this theory. Baudi et al. originally proposed the CT Pico method for the assessment of the glenoid bone surface in TUBS.
The CT Pico method is remarkably fast and simple because of the relatively intuitive multiplanar reformatting planes (MPR) in spite of complex volume rendering and image subtraction [10]. However, the reliability of the CT Pico method is still to be tested, as suggested by several authors [7, 10]. Several other methods for glenoid bone surface quantitation have been proposed, based on morphological evaluation of the inferior bone surface of the glenoid in the sagittal plane [13, 18]. Abnormalities of the inferior aspect of the glenoid were detected in 90 % of TUBS shoulders [9]. For this method, discrete inter-observer agreement was shown by Huijsmans et al. on CT and MRI images [19–21]. Barchilon et al. proposed an alternative method for assessment of glenoid bone loss based on trigonometric calculation of depth of bone loss and glenoid radius [22].
We believe that the CT Pico method has been more successful than the other methods because it is simpler to apply. The aforementioned methods, including Pico, however, were not tested with a reference standard. In our study, a high-precision laser was used as a reference standard for the assessment of the glenoid surface. This technique allowed fine quantitation of the bone surface accounting for geometrical complexity of the anatomical surface. Our study shows a statistically significant difference between the results from the laser and the CT readers measurements (p = 0.010).
As mentioned above, the degree of glenoid bone loss has a significant impact on the outcome of arthroscopic Bankart repair [5]. Several cadaveric studies have shown that an osseous defect with a width approximately 20 % of the glenoid length or 25 % of the glenoid width remains unstable even after Bankart repair, and bone grafting is recommended in such cases [5].
In our study, a large number of CT readers’ measurements (43 %) were outside the range [−10 %; +10 %] of laser measurements, and it is possible that this interobserver variability could also be present in the evaluation of the glena with deficiency and may cause errors in the surgical management of the patient.
Mean differences between laser and CT measurements were shown by our results with generalized overestimation of glenoid surface of the radiological method over the reference standard.
These data suggest low reliability for the CT Pico method, seemingly accountable to oversimplification of the anatomical geometry. Indeed, the anatomical references were not reproducible between readers for the assessment of the inferior glenoid surface. The CT Pico method presents a variability that is too large to be acceptable without concern. Furthermore, glenoid concavity is not accounted for by the CT Pico method, which is based on an en face view.
A reliable method for anatomical surface quantitation should take into account irregularities detected by different planes. In a recent study, some authors suggested curved MPR for the measurement of the glenoid surface by CT [22]. Measurements obtained with curved MPR were more accurate than normal MPR in comparison with the reference standard (laser) and also showed a better interobserver reproducibility; it is necessary to use thicker reconstructions with curved MPR because of the concave morphology of the glenoid that results in partial volume effects at the edges, a source of error with the standard MPR [22]. However, in a clinical setting, it is possible that the differences are not so significant.
Our study has limitations related to the fact that we limited it to the assessment of ten cadaveric specimens. This study does not include clinical cases of TUBS, which should be an area for further investigation. Moreover, it could be possible that by using the CT Pico method, the accuracy of the circle obtained measuring the normal glenoid surface in the “flat” sagittal section may not necessarily influence the accuracy of the ratio between the two diameters, considering the glenoid surface with bone loss as the second one.
However, as already alluded to, the inscription of the inferior glenoid in a circle using the CT Pico method could not correspond to the real anatomy of the glenoid surface, which, besides being a curve, is morphologically difficult to compare to a circle.
Conclusions
The present study shows a significant difference between the measurements by the CT Pico method and laser measurements in the assessment of glenoid surface in cadaveric specimens (p = 0.010). The inter-observer variability for CT measurement of the glenoid surface using the CT Pico method was high when compared with laser measurements, with a positive bias up to +64 %; the CT Pico method seems not very reliable and could cause errors in the clinical management of the patient.
References
Chuang TY, Adams CR, Burkhart SS. Use of preoperative three-dimensional computed tomography to quantify glenoid bone loss in shoulder instability. Arthroscopy. 2008;24(4):376–82.
Boileau P, Villalba M, Henry JY, Balg F, Ahrens P, Neyton L. Risk factors for recurrence of shoulder instability after arthroscopic Bankart repair. J Bone Joint Surg Am. 2006;88:1755–63.
Rowe CR, Patel D, Southmayd WW. The Bankart procedure: a long-term end result study. J Bone Joint Surg Am. 1978;60:1–16.
Griffith J, Antonio G, Yung P, et al. Prevalence, pattern, and spectrum of glenoid bone loss in anterior shoulder dislocation: CT analysis of 218 patients. AJR Am J Roentgenol. 2008;190:1247–54.
Itoi E, Yamamoto N, Kurokawa D, Sano H. Bone loss in anterior instability. Musculoskelet Med. 2013;6:88–94.
Jean K. Classifications of glenoid dysplasia, glenoid bone loss and glenoid loosening: a review of the literature. Eur J Orthop Surg Traumatol. 2013;23:301–10.
Burkhart SS, De Beer JF. Traumatic glenohumeral bone defects and their relationship to failure of arthroscopic Bankart repairs: significance of the inverted-pear glenoid and the humeral engaging Hill-Sachs lesion. Arthroscopy. 2000;16:677–94.
Burkhart SS, Debeer JF, Tehrany AM, Parten PM. Quantifying glenoid bone loss arthroscopically in shoulder instability. Arthroscopy. 2002;18(5):488–91.
Magarelli N, Milano G, Sergio P, Santagada DA, Fabbriciani C, Bonomo L. Intra-observer and interobserver reliability of the ‘Pico’ computed tomography method for quantification of glenoid bone defect in anterior shoulder instability. Skeletal Radiol. 2009;38:1071–5.
Baudi P, Righi P, Bolognesi D, et al. How to identify and calculate glenoid bone deficit. Chir Organi Mov. 2005;90:145–52.
Souza PM, Brandão BL, Brown E, Motta G, Monteiro M, Marchiori E. Recurrent anterior glenohumeral instability: the quantification of glenoid bone loss using magnetic resonance imaging. Skeletal Radiol. 2014;43(8):1085–92.
Bois AJ, Fening SD, Polster J, Jones MH, Miniaci A. Quantifying glenoid bone loss in anterior shoulder instability: reliability and accuracy of 2-dimensional and 3-dimensional computed tomography measurement techniques. Am J Sports Med. 2012;40:2569–77.
Sugaya H, Moriishi J, Dohi M, Kon Y, Tsuchiya A. Glenoid rim morphology in recurrent anterior glenohumeral instability. J Bone Joint Surg Am. 2003;85:878–84.
Lin LI-K. A concordance correlation coefficient to evaluate reproducibility. Biom (Int Biom Soc). 1989;45:255–68.
McGraw KO, Wong SP. Forming inferences about some intraclass correlation coefficients. Psychol Methods. 1986;1:30-46.
Thibaut J. adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics. 2008;24:1403–5.
Jacob C. Statistical power analysis for the behavioral sciences. 2nd ed. Hardcover: Lawrence Erlbaum Associates; 1988.
De Wilde LF, Berghs BM, Audenaert E, Sys G, Van Maele GO, Barbaix E. About the variability of the shape of the glenoid cavity. Surg Radiol Anat. 2004;26:54–9.
Gyftopoulos S, Hasan S, Bencardino J, et al. Diagnostic accuracy of MRI in the measurement of glenoid bone loss. AJR. 2012;199:873–8.
Lee R, Griffith J, Tong M, Sharma N, Yung P. Glenoid bone loss: assessment with MR imaging. Radiology. 2013;267:496–502.
Huijsmans PE, Haen PS, Kidd M, Dhert WJ, van der Hulst VP, Willems WJ. Quantification of a glenoid defect with three-dimensional computed tomography and magnetic resonance imaging: a cadaveric study. J Shoulder Elbow Surg. 2007;16:803–9.
De Filippo M, Castagna A, Steinbach LS, et al. Reproducible noninvasive method for evaluation of glenoid bone loss by multiplanar reconstruction curved computed tomographic imaging using a cadaveric model. Arthroscopy. 2013;29:471–7.
Acknowledgments
Prof. Lynne S. Steinbach, MD, University of California, San Francisco, Department of Radiology and Biomedical Imaging, United States.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
De Filippo, M., Saba, L., Negrini, G. et al. Inter-observer agreement of CT measurement of the glenoid bone surface by the CT Pico method: Comparison with laser in a cadaveric model. Skeletal Radiol 44, 1491–1497 (2015). https://doi.org/10.1007/s00256-015-2199-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-015-2199-0