Abstract
Primary leiomyosarcoma of bone is a rare malignant tumor of smooth muscle. We report a case of low-grade subperiosteal primary bone leiomyosarcoma in the tibial diaphysis, which radiologically appeared to be osteoid osteoma. A 35-year-old man presented with a several-year history of a palpable hard nodule in the distal left leg, which had enlarged and become painful over the previous 2 years. Radiographs showed solid periosteal reaction with a well-defined lytic lesion in the posteromedial cortical border of the left tibial diaphysis. Computed tomography demonstrated a small, well-defined lytic lesion, not calcified, in a subperiosteal location, surrounded by solid periosteal bone formation. The lesion was excised en bloc and the histological diagnosis of a low-grade leiomyosarcoma was made. To the best of our knowledge, the surface location of primary bone leiomyosarcoma has not been previously described in the literature.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Leiomyosarcoma is a malignant neoplasm of smooth muscle cells that occurs predominantly in the gastrointestinal tract and uterus. Primary bone leiomyosarcoma is a rare tumor, which occurs primarily in the medullary cavity, most commonly in the long bones of the lower extremities [1, 9]. Only a few studies on the radiological features of primary bone leiomyosarcoma have been reported. Primary bone leiomyosarcoma appears radiologically as an osteolytic lesion with aggressive features, arising within the medullary cavity. We report a case of subperiosteal leiomyosarcoma of the tibial diaphysis, which radiologically appeared to be osteoid osteoma. Leiomyosarcoma on the surface of bone has, to the best of our knowledge, not previously been reported in the English literature.
Case report
A 35-year-old man presented with a several-year history of a palpable hard nodule in the distal left leg, which had enlarged and become painful over the previous 2 years. The patient denied either traumatic antecedent or constitutional symptoms.
Physical examination revealed a hard non-movable tender mass located in the medial aspect of the distal left leg. Laboratory data were within normal limits.
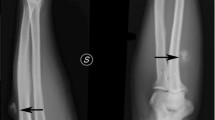
Radiographs showed a well-defined lytic lesion in the posteromedial cortical border of the left distal tibial diaphysis, surrounded by considerable reactive sclerosis as well as an uninterrupted, solid periosteal reaction (Fig. 1A, B). Computed tomography (CT) demonstrated a lytic lesion with sharply defined, lobulated borders, which was located in a subperiosteal location (Fig. 2A, B). The lesion eroded the cortex, and was surrounded posteriorly by a thin periosteal bone layer, whereas the rest of the lesion was surrounded by a thick periosteal solid reaction (Fig. 2B, C). The size of the lesion was 6×12 mm. There was no matrix calcification.
CT scans through lesion show a lytic lesion with lobulated borders, surrounded by reactive sclerosis and mature periosteal new formation (arrowheads) posteromedially, and by a thin periosteal shell posteriorly. Note the lesion’s location at the outer margin of cortical bone, indicating a subperiosteal location. C CT scan through the inferior margin of lesion shows solid periosteal reaction posteromedially in the tibia
The patient was treated in a community hospital with an en bloc resection of the tumor with the presumed diagnosis of osteoid osteoma. Histological examination of the whole specimen revealed a low-grade leiomyosarcoma. The patient was then transferred to our hospital, a teaching tertiary institution, for further management.
Pathology review revealed an unencapsulated lesion, which eroded the adjacent dense cortical bone. It consisted of interlacing bundles of large pleomorphic spindle cells with tapered eosinophilic cytoplasm (Fig. 3A). Nuclei were elongated, cigar-shaped, with blunt ends and prominent nucleoli (Fig. 3B). Only scattered mitoses were seen. In the central portion of the lesion there were areas of hyalinization but necrosis was not seen. The diagnosis of leiomyosarcoma was confirmed by the positive immunostaining of vimentin and smooth-muscle-specific actin (Fig. 4). Desmin reactivity was also present. Tumor cells showed negative immunostaining of S-100 protein, low-molecular-weight keratin (CAM5.2) and epithelial membrane antigen.
Photomicrograph of a histological section shows a tumor composed of large pleomorphic spindle cells arranged in fascicles (hematoxylin and eosin, ×100). B Photomicrograph of a histological section at higher magnification shows spindle cells with tapered eosinophilic cytoplasm. Note the blunt-ended, elongated nuclei. Destruction of bone is evidenced by the scalloping along the edges of the portion of dense, cortical-type bone included (hematoxylin and eosin, ×400)
Postoperative chest and whole abdominal CT scans were performed in order to exclude a primary gastrointestinal leiomyosarcoma or pulmonary metastases, and there was no evidence of other tumoral lesions. 99mTc-MDP scintigraphy did not reveal radiotracer uptake at other skeletal sites.
Although there was no residual tumor identified on postoperative radiographs and MR imaging of the left leg, a second surgical intervention was performed at our hospital; biopsies of the cortex and medullary cavity did not show tumoral infiltration. The patient did not receive further treatment.
Five years after treatment, the patient is well without any evidence of recurrence.
Discussion
Leiomyosarcomas are malignant neoplasms of smooth muscle cells that occur predominantly in the uterus and gastrointestinal tract. The diagnosis of a primary bone leiomyosarcoma is only possible after the exclusion of either a soft tissue mass extending into the bone or the presence of a leiomyosarcoma elsewhere [1, 2]. This is especially important in female patients with uterine lesions that have been previously treated as leiomyomas [1]. However, bone metastases of uterine leiomyosarcoma are rare and occur late in the course of the disease, with an incidence of 1–5% [1, 2]. Bone involvement in soft tissue leiomyosarcoma may also occur by hematogenous spread.
Primary bone leiomyosarcoma is a rare tumor. Since the first description in 1965 by Evans and Sanerkin [3], around 100 cases of extra-facial primary bone leiomyosarcoma have been reported in the literature [4, 5, 6, 7, 8, 9, 10]. Leiomyosarcoma of face bones and skull are excluded in most series because in these locations soft tissue involvement is the rule, it being very difficult to assess the primary origin of the tumor [2].
Primary bone leiomyosarcoma is mainly observed in the fourth to seventh decades of life [2, 4, 7, 10], but all ages may be affected [8]. There is no gender predilection. Pain and palpable mass are the most frequent clinical manifestations. This condition typically involves long bones, most frequently around the knee. Lesions are metaphyseal or metaphyseal-epiphyseal. Pelvic bones and thorax flat bones are less frequently involved. Most lesions are solitary and only a few cases have been described with multiple sites involved [2, 8].
Histologically, leiomyosarcomas are characterized by fusiform cells arranged in bundles and fascicles, less commonly showing a cartwheel pattern or totally disorganized growth [7, 8]. Immunohistochemically, widespread cytoplasmic positivity for smooth muscle actin and vimentin are typically seen [7, 8]. There are two main hypotheses about the cell of origin of primary bone leiomyosarcoma: a possible vascular smooth muscle cell origin [2, 3, 8], or an intermediate cellular form capable of smooth muscle differentiation [2, 8].
The radiological appearance of these lesions has received less attention in the literature. Only three series [4, 10, 11], one of them including also soft tissue leiomyosarcoma [11], and several case reports [1, 12, 13, 14, 15, 16, 17] have been published in the English radiological literature.
In a review of the English literature, most lesions were described as osteolytic lesions with a moth-eaten or permeative pattern of bone destruction. Perilesional sclerosis has been described in low-grade primary leiomyosarcoma of bone [4], as well as in the case presented here.
Cortical destruction and a soft tissue mass are frequent, typically with subtle or non-periosteal reaction [8, 10].
Sundaram et al. reported two characteristics of primary bone leiomyosarcoma that may be helpful in differentiating these from other aggressive osteolytic lesions [10]. First, lesions had a considerable length, extending along the longitudinal axis of the bone, and second, the signal intensity of these lesions on T2-weighted MR images is intermediate to low (decreased signal intensity with respect to normal bone marrow), whereas most osteolytic lesions are hyperintense on T2-weighted images. These two features of primary leiomyosarcoma of bone are common to primary lymphoma of bone [10].
The subperiosteal location of the lesion is the most interesting feature of our case. A cortical origin could be postulated for the lesion in our case, but when the center of the lesion was analyzed on CT images following the method described by Kaiser et al. [18], it was localized in the outer border of the cortex, clearly indicating a subperiosteal origin. All previously reported bone leiomyosarcomas are intramedullary in origin, most often centrally located [4, 8]. To our knowledge, this is the first description of a surface bone leiomyosarcoma.
In our case, the location and small size of the lesion, compact periosteal reaction and long duration of symptoms were thought to be suggestive of osteoid osteoma.
The small size of the lesion in our case allows the exclusion of osteoblastoma, which may have an intracortical location. Intracortical abscess may also be considered in the radiological differential diagnosis.
Other uncommon features of the lesion in our case are the low histological grade and the recurrence-free period. Since surface bone sarcomas generally have a more favorable prognosis than their intramedullary counterparts [19], this more favorable prognosis in our case may be related to the location of the lesion on the bone surface.
Pathological differential diagnosis of high-grade leiomyosarcoma includes fibrosarcoma, malignant fibrous histiocytoma and myofibrosarcoma [8]. Fibrosarcoma and malignant fibrous histiocytoma may be differentiated by immunohistochemistry because both tumors lack the expression of muscle markers. However, the distinction between a poorly differentiated leiomyosarcoma and myofibrosarcoma may be more difficult because both tumors variably express immunoreactivity for smooth muscle actin and, to a lesser extent, desmin [8]. On electron microscopy, the cells composing myofibrosarcoma usually have a well-developed, variably dilated, branching rough endoplasmic reticulum, and the parallel arrays of microfilaments are thin and generally restricted to the ectoplasm. A collagenous stroma also is usually more commonly seen in myofibrosarcomas. In contrast, the rough endoplasmic reticulum is inconspicuous in leiomyosarcoma, whereas microfilaments with interspersed fusiform densities are found in all regions of the cytoplasm [8].
References
Fornasier VL, Paley D. Leiomyosarcoma of bone: primary or secondary? Skeletal Radiol 1983; 10:147―153.
Jeanrot C, Ouaknine M, Anract P, Carlioz A, Forest M, Tomeno B. Leiomyosarcome primitif osseux. Presentation de cinq observations anatomo-cliniques et revue de la litterature. Rev Chir Pathol 2000; 86:63―73.
Evans DMD, Sanerkin NG. Primary leiomyosarcoma of bone. J Pathol Bacteriol 1965; 90:348―350.
Berlin O, Angervall L, Kindblom LG, Berlin IC, Stener B. Primary leiomyosarcoma of bone. A clinical, radiographic, pathologic-anatomic, and prognostic study of 16 cases. Skeletal Radiol 1987; 16:364―376.
Myers JL, Arocho J, Bernreuter W, Dunham W, Mazur MT. Leiomyosarcoma of bone. A clinicopathologic, immunohistochemical, and ultrastructural study of five cases. Cancer 1991; 67:1051―1056.
Jundt G, Moll C, Nidecker A, Schilt R, Remagen W. Primary leiomyosarcoma of bone: report of eight cases. Hum Pathol 1994; 25:1205―1212.
Khoddami M, Bedard YC, Bell RS, Kandel RA. Primary leiomyosarcoma of bone. Report of seven cases and review of the literature. Arch Pathol Lab Med 1996; 120:671―675.
Antonescu CR, Erlandson RA, Huvos AG. Primary leiomyosarcoma of bone: a clinicopathologic, immunohistochemical, and ultrastructural study of 33 patients and a literature review. Am J Surg Pathol 1997; 21:1281―1294.
Wirbel RJ, Verelst S, Hanselmann R, Remberger K, Kubale R, Mutschler WE. Primary leiomyosarcoma of bone: clinicopathologic, immunohistochemical, and molecular biologic aspects. Ann Surg Oncol 1998; 5:635―641.
Sundaram M, Akduman I, White LM, McDonald DJ, Kandel R, Janney C. Primary leiomyosarcoma of bone. AJR Am J Roentgenol 1999; 172:771―776.
Bush CH, Reith JD, Spanier SS. Mineralization in musculoskeletal leiomyosarcoma: radiologic-pathologic correlation. AJR Am J Roentgenol 2003; 180:109―113.
Meister P, Konrad E, Gokel JM, Remberger K. Case report 59. Leiomyosarcoma of the humerus. Skeletal Radiol 1978; 2:265―267.
Abdelwahab IF, Hermann G, Kenan S, Klein MJ, Lewis MM. Case report 794. Primary leiomyosarcoma of the right femur. Skeletal Radiol 1993; 22:379―381.
Akata D, Aralasmak A, Ozmen MN, Akhan O, Altundag K, Gullu I. US and CT findings of multicentric leiomyosarcomatosis. Eur Radiol 1999; 9:711―714.
Inoue S, Tanaka K, Sakamoto A, Matsuda S, Tsuneyoshi M, Iwamoto Y. Primary leiomyosarcoma of the patella. Skeletal Radiol 2001; 30:530―533.
Shen SH, Steinbach LS, Wang SF, Chen WY, Chen WM, Chang CY. Primary leiomyosarcoma of bone. Skeletal Radiol 2001; 30:600―603.
Ganau S, Tomas X, Mallofre C, Macho JM, Pomes J, Combalia A. Leiomyosarcoma of sacrum: imaging and histopathologic findings. Eur Radiol 2002; 12 (Suppl 3):S35―S39.
Kaiser F, Resnick D, Haghighi P. Evidence of the subperiosteal origin of osteoid osteoma in tubular bones: analysis by CT and MR imaging. AJR Am J Roentgenol 1998; 170:609―614.
Seeger LL, Yao L, Eckardt JJ. Surface lesions of bone. Radiology 1998; 206:17―33.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Narváez, J.A., De Lama, E., Portabella, F. et al. Subperiosteal leiomyosarcoma of the tibia. Skeletal Radiol 34, 42–46 (2005). https://doi.org/10.1007/s00256-004-0838-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-004-0838-y