Abstract.
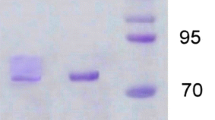
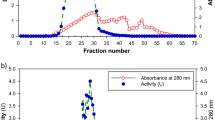
Ganoderma lucidum, a medicinal white-rot basidiomycete, produces many laccase isozymes in liquid culture. Three laccase isozymes (GaLc 1, 2, 3) have been purified 32.4-fold from the crude enzyme protein through anion exchange chromatography, preparative gel electrophoresis, and electroelution. Their estimated molecular weights are 65–68 kDa, and they contain 7–10% N-linked carbohydrates. The three isozymes have identical N-terminal amino acid sequences: G-I-G-P-T. The optimum pH and temperature both for each isozyme singly and the isozyme mixture are pH 3.5 and 20°C, respectively. One isozyme (GaLc 3) is quite stable at pH 4.0–10.0, and shows good stability when incubated at temperatures lower than 40°C. The K m values of GaLc 3 for o-tolidine and 2,2'-azino-bis-(3-ethylthiazoline-6-sulfonate) (ABTS) are 401.6 µM and 3.7 µM respectively, and the V max of GaLc 3 for these substrates is 0.0198 OD min–1unit–1 and 0.0142 OD min–1unit–1, respectively.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received revision: 18 April 2001
Electronic Publication
Rights and permissions
About this article
Cite this article
Ko, .EM., Leem, .YE. & Choi, .H. Purification and characterization of laccase isozymes from the white-rot basidiomycete Ganoderma lucidum . Appl Microbiol Biotechnol 57, 98–102 (2001). https://doi.org/10.1007/s002530100727
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002530100727