Abstract
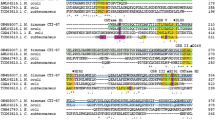
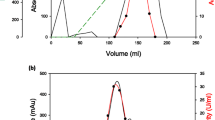
The amylopullulanse produced by Bacillus sp. DSM 405 was purified to homogeneity. It exhibited dual activity, cleaving the α1-4 bonds in starch, releasing a range of malto-oligosaccharides, and also cleaving the α1-6 bonds in pullulan, releasing maltotriose as the sole end-product. The enzyme was a glycoprotein and had a relative molecular mass of 126 000 and an isoelectric point of 4.3. While the enzyme was optimally active on starch at pH 6.5 and at pH 6.0 on pullulan, activity on both substrates was maximal at 70 °C. Kinetic analyses of the enzyme in a system that contained both starch and pullulan as two competing substrates demonstrated the dual specificity of the enzyme. Chemical modification of the carboxyl groups within the active centre of the protein showed that one active site was responsible for hydrolysis of the α1-4 and α1-6 bonds in starch and pullulan respectively. This is the first comprehensive investigation of an amylopullulanse produced by an aerobic bacterium, showing a single active site responsible for both activities.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 3 August 1998 / Received revision: 13 October 1998 / Accepted: 16 October 1998
Rights and permissions
About this article
Cite this article
Brunswick, J., Kelly, C. & Fogarty, W. The amylopullulanase of Bacillus sp. DSM 405. Appl Microbiol Biotechnol 51, 170–175 (1999). https://doi.org/10.1007/s002530051378
Issue Date:
DOI: https://doi.org/10.1007/s002530051378