Abstract
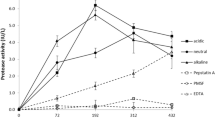
An extracellular laccase capable of oxidizing ABTS (the diammonium salt of 2,2′-azinobis-3-ethylbenzothiazoline-6-sulfonic acid) was detected in ligninolytic cultures of Penicillium chrysogenum. By contrast, no lignin peroxidase, manganese-dependent peroxidase or aryl-alcohol oxidase was detected at any time during culturing. Both ABTS laccase activity and mineralization of dehydrogenative polymerizate of coniferyl alcohol were regulated by the C/N ratio in the medium and partially inhibited in the presence of thioglycolic acid, suggesting that both events are associated. In the presence of several known laccase inducers neither ABTS laccase activity nor mineralization rates were enhanced. However, a new laccase was detected in P. chrysogenum, able to oxidize 2,6-dimethoxyphenol but not involved in lignin mineralization. Studies with the known ligninolytic basidiomycete Trametes villosa suggest that lignin degradation by this fungus also involves the action of laccase.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 6 July 1995/Received revision: 28 October 1995/Accepted: 6 November 1995
Rights and permissions
About this article
Cite this article
Rodríguez, A., Falcón, M., Carnicero, A. et al. Laccase activities of Penicillium chrysogenum in relation to lignin degradation. Appl Microbiol Biotechnol 45, 399–403 (1996). https://doi.org/10.1007/s002530050702
Issue Date:
DOI: https://doi.org/10.1007/s002530050702