Abstract
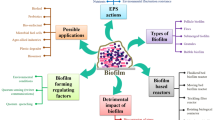
There is an increasing need for application of biofilm process in the upcycling of wastewater treatment plants all around the world in recent years, yet there are few literatures on summarizing wastewater biofilm during the life cycle. In particular, there is a vacancy on characterization at various stages of biofilm and its regulation. This review provided a whole look at biofilm formation and its development, accompanied by microbial physiology, ecology, and activity, where the initialization of biofilm formation and its characterization were stressed. The new progresses on biofilm physio-ecology analysis and methods on evaluating microbial activity were summarized, while it is worth mentioning that the concept of aging biofilm was also presented. Furthermore, regulations methods of biofilm were reviewed and future research trends on biofilm control were prospected, aiming at guiding biofilm control in biofilm-based wastewater treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biofilms are complex biostructures which adhere to surfaces of carriers (George et al. 2000). A biofilm consists of mixed microbes such as yeasts, fungi, and protozoa, and associated deposits enclosed in a self-produced extracellular polymeric substances (EPS). The presence of biofilms may have a harmful impact on a broad range of areas, specifically in the food, environmental, and biomedical fields (Flint et al. 1997; Maukonen et al. 2003; Sihorkar and Vyas 2001); however, it can be used beneficially in biological wastewater treatment. The biofilm provides structural integrity, bacterial protection of critical and sensitive microorganisms, intercellular communication, formation and maintenance of the microcolony, and capturing and consumption of nutrients, and is of vital importance for the performance of biofilm processing system (Boltz et al. 2017).
Biofilm process (also known as attached-growth process) has been widely used in biological wastewater treatment in the past few decades. In the biofilm wastewater treatment process, biofilms are attached to biocarriers and substrates such as biochemical oxygen demand (BOD), ammonia nitrogen, nitrate, dissolved O2, and so on, and are delivered from bulk liquid to the interface. And then, the nutrients supplied are exploited to synthesize new generations of microbes and for metabolic consumption; thus, contaminants in the wastewater are removed. A wide variety of biofilm reactors have been developed and applied to deal with domestic sewage and a variety of industrial wastewater (Andreottola et al. 2002; Odegaard et al. 1993), such as biological contact oxidation tank (Zhang et al. 2015), biological rotating disc (Visscher et al. 2013), biological aerated filter (BAF), biological fluidized bed, moving bed biofilm reactor (MBBR), and integrated fixed-film activated sludge reactor (IFAS) (Boltz et al. 2017). In general, biofilm reactors have several advantages such as strong adaptability, high removal rate of organics and nitrogen, low excess sludge production, and convenient operation management, resulting in an increasing need for application in the upcycling of wastewater treatment plants around the world (Escudie et al. 2011). In recent years, with the development of anaerobic ammonium oxidation (ANNAMMOX), autotrophic denitrification and other biofilm-based technologies (Augusto et al. 2018; Jiang et al. 2018; Li et al. 2016), and gradual improvement of wastewater discharge standards in many countries and regions, the biofilm process has become as a hot spot in the aspects of advanced wastewater treatment, water reuse, and the upgrading and reconstruction of wastewater treatment plants. However, there are few literatures on summarizing wastewater biofilm during the life cycle. In particular, there is a vacancy on characterization at various stages of biofilm and its regulation.
In order to fill knowledge gaps in the above fields and offer effective guidance for the innovative application of biofilm process, this review provided a whole look at biofilm formation and its affecting factors, development of biofilm accompanied by microbial physiology, ecology and activity especially emphasized on the initialization formation, and characterization of biofilm. Recent studies about biofilm physio-ecology analysis and methods on evaluating microbial activity were summarized, and the concept of aging biofilm was also presented to outline the entire life cycle of biofilm. Furthermore, regulation methods of biofilm were reviewed, and future research trends on biofilm control were prospected, aiming at offering guidelines on biofilm control in biofilm-based wastewater treatment.
Biofilm formation and its affecting factors
Biofilm formation process
Formation and development of biofilm usually can be summarized into the following four stages based on the previous studies (Fig. 1): initial adsorption of macromolecules (e.g., protein, polysaccharide) to surfaces of carriers, microbes adhesion, biofilm development and maturity, and biofilm aging (Derlon et al. 2013a; Huang et al. 2014; Simões et al. 2010). Initial adsorption of macromolecules to surfaces triggers the whole process of biofilm formation and creates conditions for bacterial cells colonization on biocarrier surfaces. On awareness of this, we systematically investigated deposition behaviors of soluble pollutants (prepared by real and synthetic wastewaters with different configurations of model macromolecules) on model carriers by using a quartz crystal microbalance with dissipation monitoring (QCM-D). Moderate concentrations of calcium ion and rhamnolipid were proved to have a promoting effect on macromolecular deposition which has important implications for regulating biofilm formation (Huang et al. 2015, 2018a). Microbe adhesion stage includes two circumstances: One is nonselective adhesion includes adhesion and aggregation of bacteria to carriers and other bacteria mediated by high-affinity adhesion factors (membrane transport protein, viscous polysaccharide, extracellular DNA) or accessory structure (i.e., flagellum, pili) of bacteria surface; the other is specific adhesion that is adhesion triggered by the recognition of specific adhesion protein on the surface of bacteria to surface receptors (i.e., glycoprotein and glycolipid) (Jefferson 2004; Verstraeten et al. 2008). Development and maturity of biofilm mainly include the growth and accumulation of microbes, with the operation of biofilm system, the biofilm gets thicker and thicker and then the nutrient transfer is hindered, leading to decrease of biofilm activity and treatment efficiency, which is called aging biofilm state. The detachment of aging biofilm caused by the erosion and sloughing is vital to the recovery of biofilm activity.
Affecting factors
Biofilm formation is a very complex process, and a variety of factors contribute an impact to this process which can be concluded as three types: biocarrier surface properties, interface fluid characteristics, and cell properties (Jefferson 2004; Shen et al. 2015; Simões et al. 2010) as illustrated in Table 1.
In general, attachment of microorganisms occurs more commonly on surfaces that are rougher, more hydrophobic, and coated by conditioning films (Shen et al. 2015). There is an electrostatic repulsion between negative organic molecules of carriers and the bacteria which makes it difficult for bacteria to attach to the surfaces. Increasing the interface fluid velocity which is below the critical velocity or the nutrient concentrations properly can also promote bacteria adhesion (Paul et al. 2012; Simões et al. 2010).
As studies have shown, pH value can influence bacterial surface charge characteristics. When pH value in liquid phase is higher than isoelectric point of bacteria, bacterial surfaces show electronegativity due to amino acids’ ionization. Otherwise, bacterial surfaces show electropositivity; pH-induced changes of bacterial surface electrical behavior influence the dynamics of bacterial adhesion directly. Liu (Liu 1995) applied the colloidal stability theory to explain influences of liquid pH to nitrobacteria fixed rate. Stable electric double layer or solvation structures are formed around the electriferous bacteria in the presence of Zeta potential and hinders the effective contact between bacteria and surfaces. Besides, the solvation structures can lead to steric hindrance among bacteria which will do harm to bacterial adherence to carriers. Zeta potential of bacterial surfaces tends to be zero, and surface solvation structures almost disappear when bacteria are in the isoelectric-point environment. Microorganisms in liquid phase are in an extreme stable state under this circumstance and would adhere to the carriers or gather together to decrease surface free energy and reach a new stable state.
Bacterial extracellular appendants (i.e., flagella and pilus) are also necessary for their adhesion and aggregation (Jefferson 2004; Sauer and Camper 2001; Simões et al. 2010). In general, there will be a repulsion between bacteria and surfaces when the contact distance is 10–20 nm. If bacteria can use the mentioned appendants to overcome this repulsion and make the distance lower than 1 nm, a strong attraction will exist between the surfaces of carriers and bacteria which could promote the adhesion (Sauer and Camper 2001). Extracellular polymeric substances (EPS) secreted by microbials can aid in the adhesion of microorganisms to carrier surface. EPS can bind the free cells and ions in the wastewater together and protect microbes from toxic heavy mental and organic pollutants (Cao et al. 2011; Lai et al. 2018). Thus, the production of EPS has a significant impact on the formation of biofilm.
In recent years, bacterial quorum sensing (QS) gradually attracts much attention in wastewater treatment field. For example, Shrout and Nerenberg (2012) summarized bacterial quorum sensing theory and its regulation in wastewater treatment biofilm. Ren et al. (2013) verified that N-acy-l-homoserine lactones (AHLs) produced by aerobic granular sludge have effects in the formation of Escherichia coli K12 biofilm. They found AHL degrading enzyme activity in the activated sludge and concluded that quorum quenching and quorum sensing exist at the same time. In a recent study, we also found that distribution of QS signaling molecules displayed significant positive relationship with the concentration of EPS, providing a potential method for improving biofilm formation (Wang et al. 2018a).
As a kind of second messenger, intracellular c-di-GMP also plays a critical role in biofilm formation and its shedding, which was first identified to be an important factor that participates in the biosynthesis of cellulose (Ross et al. 1987). Then, it was discovered that c-di-GMP was associated with the phenotypes regulation in some bacteria (Tischler and Camilli 2004). It was also reported that extracellular matrix components such as polysaccharides, pili, adhesins (e.g., LapA is a kind of large adhesive protein that promotes microorganisms attachment and biofilm formation in Pseudomonas putida), and extracellular DNA can be regulated by c-di-GMP (Hinsa et al. 2003; Irie et al. 2012; Jain et al. 2012; Ueda and Wood 2010) through specific targets, while all these substances contribute to biofilm formation and its three-dimensional structure. In the meantime, c-di-GMP also take part in the dispersal of biofilm, for example, increased c-di-GMP levels enable BalA protein activation which was identified to be necessary for the biofilm dispersion of Pseudomonas aeruginosa (Petrova and Sauer 2012). Generally, increased concentration of intracellular c-di-GMP promotes surface attachment and the formation of biofilm, while the decreasing concentration of intracellular c-di-GMP can cause biofilm dispersal. However, research also showed that high level of c-di-GMP can promote the production of polysaccharide, while it may suppress the quorum sensing-dependent biofilm formation (Schmid et al. 2017). And, a relatively high concentration (200 μm) of extracellular c-di-GMP inhabits intercellular interactions and reduces biofilm formation of Staphylococcus aureus (Karaolis et al. 2005). The regulation of biofilm by regulating the content of c-di-GMP may be a promising research direction in the field of wastewater biofilm treatment, while a more precise relationship between biofilm formation and c-di-GMP remains to be ascertained.
Based on the knowledge of quorum sensing, the effect of exogenous AHLs on microbial adhesion of high ammonia nitrogen wastewater was investigated in our recent study with the help of QCM-D. Interesting results were obtained, which indicated that the addition of exogenous AHLs, especially N-octanoyl-l-homoserine lactone, improved microbial adhesion to surfaces of carriers, deposition amount, and thus the formation of biofilm, suggesting that exogenous AHLs might be potential in accelerating the startup process of biofilm formation in high ammonia nitrogen wastewater treatment systems (Peng et al. 2018b).
Biofilm physic-ecology and its characterization
In the field of microbial physiological and ecological research, with the help of microelectrodes (He et al. 2017; Zhou et al. 2011), electron microscopy (Fu et al. 2011; Zhou et al. 2011), atomic force microscope (AFM) (Zhang et al. 2011; Zhu et al. 2015), and modern molecular biology techniques, such as PCR-DGGE (He et al. 2017; Zhang et al. 2011), FISH (Persson et al. 2014) and high-throughput sequencing (Lu et al. 2014; Peng et al. 2014), biofilm morphology, internal mass transfer, and microbial community, etc., have been effectively characterized.
Biofilm structure
The macrostructure of biofilm is the common result of biofilm growth and hydraulic shear. Two typical biofilm structure models are “heterogeneous Mosaic structure” model and “mushrooms or tulip” model (Wimpenny et al. 2000; Zhou et al. 2011), both formed by the random combination and attachment of independent accumulations or communities. The “mushroom or tulip model” is a structure resembling a mushroom or tulip shaped by a micro colony and the bottom of which is narrower than the top. Furthermore, there are water channels around these colonies to transport nutrients, enzymes, metabolic products, and discharged wastes since the aqueous solution can continuously flow and circulate in the channels.
During the process of substrates utilization by microbes, the thickness of biofilm increases. Since dissolved oxygen can only spread to a certain area of biofilm, the anaerobic zone is formed in the inner biofilm close to the carriers. According to the diffusion of substrates, biofilm can be divided into two parts in functional structure: One is the substrates utilization area that directly exposed to wastewater, and the other is microbial hunger area that close to the surface of carriers since the substrates are almost utilized by the microbes in the outer layer. Microorganisms in the hunger area have to take advantages of energy from metabolism of their own cells to maintain their biological activities and usually lose the ability of adhering to carriers which lead to the shedding from the surfaces (Jefferson 2004; Paul et al. 2012; Verstraeten et al. 2008).
The team led by Paul Etienne is one of the most representative group focusing on the structure of biofilms (Coufort et al. 2007; Derlon et al. 2008; Derlon et al. 2013a; Marcato-Romain et al. 2012; Ochoa et al. 2007; Paul et al. 2012; Ras et al. 2011). They studied the effects of hydrodynamic and growth conditions (electron donor and receptor, C/N ratio, etc.) on the physical and chemical properties of biofilms by using Couette-Taylor reactors, and put forward the hierarchical (stratification) model of biofilm. It is believable that biofilm is composed of the basal layer and the outer layer under the action of the shear stress, where the base is compacted and the bonding layer is easy to fall off. Furthermore, the effect of specific centrifugal forces on biofilm structures was also reported in our group that three different fractions of biofilms could be divided under different centrifugal forces (Wang et al. 2018a).
Biofilm biophase
Biofilm is mainly composed of biophase and surrounding EPS. Biophase in biofilm is very abundant, and it forms a complex ecosystem composed of bacteria, fungi, algae, protozoa, and metazoan (Sudo 1988). As a functional organism, the distribution of biophase is not a simple combination among microbes, but an organic configuration based on the optimization principle of the whole metabolism function of organism, and can serve as biological indicators to inspect and judge operation conditions and wastewater treatment effects of the biofilm reactor (Derlon et al. 2013a).
Bacteria are the principal part of the biophase in biofilm and EPS produced by them establishes the foundation for biofilm structure. The presence and dominance of bacteria are usually related to their growth rates, wastewater qualities, and environmental conditions, such as nutrition, attachment growth conditions, dissolved oxygen supply, and temperature. Heterotrophic bacteria are the main type of bacteria in biofilm, who can gain sufficient energy substrates from water flowing through the biofilm surface. According to the demand of oxygen, heterotrophic bacteria can be divided into aerobic heterotrophic bacteria, anaerobic respiration heterotrophic bacteria, anaerobic heterotrophic bacteria, and facultative anaerobes. The common species of heterotrophic bacteria in biofilm include Sphaerotilus, Zoogloeas, Thiobacillus, Alcaligenes, Pseudomonas, Nocardias, Sarcinas, Streptococcus faecalis, Escherichia coli, Nitrosations, and Bacillus (Kim et al. 2015b; Tang et al. 2018; Wang et al. 2018b). In addition, fungi such as filamentous bacteria will appear in biofilm under specific circumstances (i.e., composition change of organic matter in sewage, increase of load, decrease of temperature, etc.) and common species of which include Subbaromyces splendens and Trichosporon cutaneum. Algae are not the main microorganism population in biofilm; thus, their function of purifying wastewater is little. The common species of algae in biofilm include Chlorella, Chlorococcum, Oscillatoria, Stigeoclonium, and Circumfili. Protozoa are the lowest unicellular animals in the Animalia. In the mature biofilm, protozoa feed on bacteria, playing a positive role on the physical activity state of biofilm. The common species of protozoa include Flagellates (i.e., Oikomonas termo), Sarcodina (i.e., Amoeba, Vahlkampfia, and Arcella) and Ciliates (i.e., Opercularia microdiscum, Vorticella convallaria, and Opercularia coarctata) (Dopheide et al. 2011). Metazoan are multicellular animals, which belong to the invertebrate. The common species of metazoan in biofilm include Rotifera, Nemata, Oligochaeta, and insects and their larva (Derlon et al. 2013b).
Generally, the morphology and chemical constituents of biofilm can be characterized using microscopy, spectroscopy, and microelectrode technology (details are shown in Table 1). Furthermore, emerging in situ monitoring techniques such as ultrasonic time-domain reflectometry (UTDR) have been developed for biofilm monitoring so that they can provide more information about the actual and dynamic process about the absorption and accumulation of biofilm. Initial adherence, reversible adhesion, and irreversible adhesion during the initial biofilm formation process could be successfully distinguished by the UTDR measurement in our recent study (Wang et al. 2018a). Biotechnologies have been also widely used in this research area. The currently and commonly used combined applications of quantitative polymerase chain reaction (qPCR), fluorescent in situ hybridization (FISH), advanced 2-D microscopy, and micro-scale chemical sensors have facilitated researchers to obtain a better vision of biofilm composition including both the cellular matter and their excretions than ever before (Boltz et al. 2017). To explore the microbial composition of biofilms, phospholipid fatty acid (PLFA) analysis has been employed in different environmental samples such as soil and wastewater treatment, for PLFAs can be biomarkers to characterize microorganisms (Amir et al. 2008), for instance, fungus, protozoa, Gram-negative, and Gram-positive bacteria can be identified through this method. With the development of biological analysis technology, 16s rRNA sequence analysis gradually becomes the main analytical tool for applications since it can make systematic classification of bacteria in biofilm (Zhu et al. 2015). Meanwhile, it is foreseeable that the proteomics and functional genomics technologies, describing structures, functions, and interactions of specific genes and proteins, will play an important role in biofilm research in the near future (Herschend et al. 2017; Hu et al. 2016; Tang et al. 2016).
In the future, research on biofilm formation and its structure or biophase will remain the focus in biofilm studies. In situ and real-time monitoring methods, such as QCM-D and OCT, will attract more attention. Especially for OCT, it is easy to obtain physical characteristics on macro- and mesoscales in a visual way (Wagner et al. 2010). Furthermore, multiple parameter monitoring such as a combination of optical, electrochemical, acoustic, and microbial community information will be a trend in biofilm analysis.
Biofilm activity
Biofilm activity is the objective basis of wastewater purification and the basic guarantee for the normal operation of biofilm process, leading to the degradation of pollutants through physical absorption, biochemical actions, and classified predations of biophase in biofilm (de Assis et al. 2017; Laureni et al. 2015). Currently, biofilm activity indicators are mainly composed of total solid and volatile solid content, oxygen uptake rate, adenosine triphosphate content, dehydrogenase activity, deoxyribonucleic acid content, etc. Table 2 compares various methods for estimation of biofilm activity.
Constrained by testing conditions in the previous studies, biofilm activity evaluation methods generally lack biological response. Along with the advance of modern biotechnologies, bio-driven biofilm activity from different scales (i.e., molecular level, bacterial populations, and biological communities) can be well characterized. Additionally, the study of aging biofilm evaluation is inadequate. According to the literatures, generation and consumption of ATP correspond to the internal energy charge state of the cells (Blagodatskaya and Kuzyakov 2013; Huenken et al. 2005; Xiao et al. 2015), i.e., the ATP generation process is suppressed and the utilization of ATP is stimulated under a high energy charge state, while the effect is the opposite when the energy charge state is relatively low. The content of ATP directly reflects the activity of biofilm communities. In actual wastewater treatment, aging biofilm may decrease the efficiency of biofilm treatment and collapse the whole system. Therefore, the effective regulation of aging biofilm on carriers is an important and urgent issue in the field of biofilm-based wastewater treatment (Yu et al. 2016). Some preliminary results on basic characteristics, chemical and enzymatic treatments, and regenerations towards aging biofilm have also been conducted in our previous studies (Hu et al. 2013a, b; Huang et al. 2014, 2018b). Even so, biofilm activity evaluation based on biological response and the activation of aging biofilm still need to be further explored in the context of biological wastewater treatment.
Regulation methods of biofilm
The formation and aging of biofilm in wastewater treatment is a series of complex processes, and effective regulation towards the processes according to the actual situation is the only way to achieve the best performance of biofilm reactors.
In the early stages of biofilm formation, it is needed to create a good adhesion condition which can promote the formation of biofilm and subsequent activity. Except for the conventional affecting factors list in Table 3, quorum sensing caused by AHLs also can be employed to regulation the biofilm formation. Once the biofilm is developed and mature, the biophase is synergistic to achieve the metabolic transformation of the pollutants. For the biofilm that reaches the aging state, it is necessary to be removed and activated. There are many approaches reported which are mainly composed of physical, chemical, and biological methods. The conditions, objectives, and effects of different approaches are shown in Table 4. It was worth noting that rhamnolipids can not only be used for activity recovery of aging biofilm but can also be used to promote biofilm formation and improve the treatment efficiency of biofilm process under low concentration (i.e., 20 and 50 mg/L), as reported in our recent study (Peng et al. 2018a). Generally speaking, related researches on aging biofilm control were mainly focused on traditional hydraulic shear method and chemical sterilization method. There are few studies on the application of economically efficient and environmentally friendly methods for activity recovery of aging biofilm in wastewater treatment systems, which should be paid more attention to in the future research.
Conclusions and prospects
This article reviewed wastewater biofilm from four main aspects: formation and its affecting factors, characterization, activity, and regulation. Further investigations are still needed: (1) specific and feasible methods for shortening biofilm formation in refractory wastewater treatment; (2) revealing the characteristics of biofilm from a more microscopic point of view through molecular biology technologies (i.e., macroproteomics and metagenomic approaches) and in situ monitoring techniques; (3) recovery of aging biofilm by cost-effective and environmentally friendly regulation methods. Moreover, novel biofilm reaction principles and technologies are continuing to inspire people’s interest to meet the increasing requirements of pollutants removal and lower energy consumption.
References
Amir S, Merlina G, Pinelli E, Winterton P, Revel JC, Hafidi M (2008) Microbial community dynamics during composting of sewage sludge and straw studied through phospholipid and neutral lipid analysis. J Hazard Mater 159(2–3):593–601. https://doi.org/10.1016/j.jhazmat.2008.02.062
Andreottola G, Foladori P, Ragazzi M, Villa R (2002) Dairy wastewater treatment in a moving bed biofilm reactor. Water Sci Technol 45(12):321–328. https://doi.org/10.2166/wst.2002.0441
de Assis LR, Calijuri ML, do Couto EA, Assemany PP (2017) Microalgal biomass production and nutrients removal from domestic sewage in a hybrid high-rate pond with biofilm reactor. Ecol Eng 106:191–199. https://doi.org/10.1016/j.ecoleng.2017.05.040
Augusto MR, Camiloti PR, Souza TSO (2018) Fast start-up of the single-stage nitrogen removal using anammox and partial nitritation (SNAP) from conventional activated sludge in a membrane-aerated biofilm reactor. Bioresour Technol 266:151–157. https://doi.org/10.1016/j.biortech.2018.06.068
Awong J, Bitton G, Koopman B (1985) ATP, oxygen- uptake rate and INT-dehydrogenase activity of Actinomycete foams. Water Res 19(7):917–921. https://doi.org/10.1016/0043-1354(85)90151-4
Bai J, Kim YT, Ryu S, Lee JH (2016) Biocontrol and rapid detection of food-borne pathogens using bacteriophages and endolysins. Front Microbiol 7:474. https://doi.org/10.3389/fmicb.2016.00474
Banat IM, De Rienzo MAD, Quinn GA (2014) Microbial biofilms: biosurfactants as antibiofilm agents. Appl Microbiol Biotechnol 98(24):9915–9929. https://doi.org/10.1007/s00253-014-6169-6
Bellucci M, Bernet N, Harmand J, Godon J-J, Milferstedt K (2015) Invasibility of resident biofilms by allochthonous communities in bioreactors. Water Res 81:232–239. https://doi.org/10.1016/j.watres.2015.05.051
BinAhmed S, Hasane A, Wang Z, Mansurov A, Castrillon SR-V (2018) Bacterial adhesion to ultrafiltration membranes: role of hydrophilicity, natural organic matter, and cell-surface macromolecules. Environ Sci Technol 52(1):162–172. https://doi.org/10.1021/acs.est.7b03682
Blagodatskaya E, Kuzyakov Y (2013) Active microorganisms in soil: critical review of estimation criteria and approaches. Soil Biol Biochem 67:192–211. https://doi.org/10.1016/j.soilbio.2013.08.024
Blenkinsopp SA, Lock MA (1990) The measurement of electron transport system activity in river biofilms. Water Res 24(4):441–445. https://doi.org/10.1016/0043-1354(90)90226-v
Boltz JP, Smets BF, Rittmann BE, van Loosdrecht MCM, Morgenroth E, Daigger GT (2017) From biofilm ecology to reactors: a focused review. Water Sci Technol 75(8):1753–1760. https://doi.org/10.2166/wst.2017.061
Cao B, Ahmed B, Kennedy DW, Wang Z, Shi L, Marshall MJ, Fredrickson JK, Isern NG, Majors PD, Beyenal H (2011) Contribution of extracellular polymeric substances from Shewanella sp HRCR-1 biofilms to U(VI) immobilization. Environ Sci Technol 45(13):5483–5490. https://doi.org/10.1021/es200095j
Cao B, Majors PD, Ahmed B, Renslow RS, Silvia CP, Shi L, Kjelleberg S, Fredrickson JK, Beyenal H (2012) Biofilm shows spatially stratified metabolic responses to contaminant exposure. Environ Microbiol 14(11):2901–2910. https://doi.org/10.1111/j.1462-2920.2012.02850.x
Chau NP, Chung NH, Jeon JG (2015) Relationships between the antibacterial activity of sodium hypochlorite and treatment time and biofilm age in early Enterococcus faecalis biofilms. Int Endod J 48(8):782–789. https://doi.org/10.1111/iej.12376
Chen X, Stewart PS (2000) Biofilm removal caused by chemical treatments. Water Res 34(17):4229–4233. https://doi.org/10.1016/s0043-1354(00)00187-1
Chen P, Zhong Z, Liu F, Xing W (2015) Cleaning ceramic membranes used in treating desizing wastewater with a complex-surfactant SDBS-assisted method. Desalination 365:25–35. https://doi.org/10.1016/j.desal.2015.01.037
Coufort C, Derlon N, Ochoa-Chaves J, Line A, Paul E (2007) Cohesion and detachment in biofilm systems for different electron acceptor and donors. Water Sci Technol 55(8–9):421–428. https://doi.org/10.2166/wst.2007.286
Cresson R, Carrère H, Delgenès JP, Bernet N (2006) Biofilm formation during the start-up period of an anaerobic biofilm reactor—impact of nutrient complementation. Biochem Eng J 30(1):55–62. https://doi.org/10.1016/j.bej.2006.02.002
Derlon N, Masse A, Escudie R, Bernet N, Paul E (2008) Stratification in the cohesion of biofilms grown under various environmental conditions. Water Res 42(8–9):2102–2110. https://doi.org/10.1016/j.watres.2007.11.016
Derlon N, Coufort-Saudejaud C, Queinnec I, Paul E (2013a) Growth limiting conditions and denitrification govern extent and frequency of volume detachment of biofilms. Chem Eng J 218:368–375. https://doi.org/10.1016/j.cej.2012.11.061
Derlon N, Koch N, Eugster B, Posch T, Pernthaler J, Pronk W, Morgenroth E (2013b) Activity of metazoa governs biofilm structure formation and enhances permeate flux during gravity-driven membrane (GDM) filtration. Water Res 47(6):2085–2095. https://doi.org/10.1016/j.watres.2013.01.033
Desmond P, Best JP, Morgenroth E, Derlon N (2017) Linking composition of extracellular polymeric substances (EPS) to the physical structure and hydraulic resistance of membrane biofilms. Water Res 132:211–221. https://doi.org/10.1016/j.watres.2017.12.058
Dheilly A, Linossier I, Darchen A, Hadjiev D, Corbel C, Alonso V (2008) Monitoring of microbial adhesion and biofilm growth using electrochemical impedancemetry. Appl Microbiol Biotechnol 79(1):157–164. https://doi.org/10.1007/s00253-008-1404-7
Dong H, Zhang K, Han X, Du B, Wei Q, Wei D (2017) Achievement, performance and characteristics of microbial products in a partial nitrification sequencing batch reactor as a pretreatment for anaerobic ammonium oxidation. Chemosphere 183:212–218. https://doi.org/10.1016/j.chemosphere.2017.05.119
Dopheide A, Lear G, Stott R, Lewis G (2011) Preferential feeding by the Ciliates chilodonella and Tetrahymena spp. and effects of these protozoa on bacterial biofilm structure and composition. Appl Environ Microbiol 77(13):4564–4572. https://doi.org/10.1128/aem.02421-10
Dusane DH, Nancharaiah YV, Zinjarde SS, Venugopalan VP (2010) Rhamnolipid mediated disruption of marine Bacillus pumilus biofilms. Colloids Surf B 81(1):242–248. https://doi.org/10.1016/j.colsurfb.2010.07.013
Ellenbroek FM, Cappenberg TE (1991) DNA synthesis and tritiated thymidine incorporation by heterotrophic freshwater bacteria in continuous culture. Appl Environ Microbiol 57(6):1675–1682
Escudie R, Cresson R, Delgenes J-P, Bernet N (2011) Control of start-up and operation of anaerobic biofilm reactors: an overview of 15 years of research. Water Res 45(1):1–10. https://doi.org/10.1016/j.watres.2010.07.081
Fang Y, Deng C, Chen J, Lu J, Chen S, Zhou S (2018) Accelerating the start-up of the cathodic biofilm by adding acyl-homoserine lactone signaling molecules. Bioresour Technol 266:548–554. https://doi.org/10.1016/j.biortech.2018.07.095
Feres M, Louzoun Y, Haber S, Faveri M, Figueiredo LC, Levin L (2018) Support vector machine-based differentiation between aggressive and chronic periodontitis using microbial profiles. Int Endod J 68(1):39–46. https://doi.org/10.1111/idj.12326
Filion-Cote S, Melaine F, Kirk AG, Tabrizian M (2017) Monitoring of bacterial film formation and its breakdown with an angular-based surface plasmon resonance biosensor. Analyst 142(13):2386–2394. https://doi.org/10.1039/c7an00068e10.1039/
Fish K, Osborn AM, Boxall JB (2017) Biofilm structures (EPS and bacterial communities) in drinking water distribution systems are conditioned by hydraulics and influence discolouration. Sci Total Environ 593-594:571–580. https://doi.org/10.1016/j.scitotenv.2017.03.176
Flint SH, Bremer PJ, Brooks JD (1997) Biofilms in dairy manufacturing plant - description, current concerns and methods of control. Biofouling 11(1):81–97. https://doi.org/10.1080/08927019709378321
Fu B, Liao XY, Liang R, Ding LL, Xu K, Ren HQ (2011) COD removal from expanded granular sludge bed effluent using a moving bed biofilm reactor and their microbial community analysis. World J Microbiol Biotechnol 27(4):915–923. https://doi.org/10.1007/s11274-010-0534-0
George RP, Muraleedharan P, Parvathavarthini N, Khatak HS, Rao TS (2000) Microbiologically influenced corrosion of AISI type 304 stainless steels under fresh water biofilms. Mater Corros 51(4):213–218. https://doi.org/10.1002/(sici)1521-4176(200004)51:4<213:aid-maco213>3.0.co;2-j
Gikas P, Livingston AG (1993) Use of ATP to characterize biomass viability in freely suspended and immobilized cell bioreactors. Biotechnol Bioeng 42(11):1337–1351. https://doi.org/10.1002/bit.260421111
Gilbert EM, Agrawal S, Schwartz T, Horn H, Lackner S (2015) Comparing different reactor configurations for partial Nitritation/Anammox at low temperatures. Water Res 81:92–100. https://doi.org/10.1016/j.watres.2015.05.022
Guo K, Freguia S, Dennis PG, Chen X, Donose BC, Keller J, Gooding JJ, Rabaey K (2013) Effects of surface charge and hydrophobicity on anodic biofilm formation, community composition, and current generation in bioelectrochemical systems. Environ Sci Technol 47(13):7563–7570. https://doi.org/10.1021/es400901u
Guo JS, Zhang P, Chen YP, Shen Y, Hu X, Yan P, Yang JX, Fang F, Li C, Gao X, Wang GX (2015) Microbial attachment and adsorption-desorption kinetic of tightly bound extracellular polymeric substances on model organic surfaces. Chem Eng J 279:516–521. https://doi.org/10.1016/j.cej.2015.05.016
Ham SY, Kim HS, Cha E, Park JH, Park HD (2018) Mitigation of membrane biofouling by a quorum quenching bacterium for membrane bioreactors. Bioresour Technol 258:220–226. https://doi.org/10.1016/j.biortech.2018.03.007
Han A, Tsoi J, Matinlinna J, Chen Z (2017) Influence of grit-blasting and hydrofluoric acid etching treatment on surface characteristics and biofilm formation on zirconia. Coatings 7(8):130. https://doi.org/10.3390/coatings7080130
He Q, Zhu Y, Li G, Fan L, Ai H, Huangfu X, Li H (2017) Impact of dissolved oxygen on the production of nitrous oxide in biological aerated filters. Front Environ Sci Eng 11(6). https://doi.org/10.1007/s11783-017-0964-0
Herrling MP, Weisbrodt J, Kirkland CM, Williamson NH, Lackner S, Codd SL, Seymour JD, Guthausen G, Horn H (2017) NMR investigation of water diffusion in different biofilm structures. Biotechnol Bioeng 114(12):2857–2867. https://doi.org/10.1002/bit.26392
Herschend J, Damholt ZBV, Marquard AM, Svensson B, Sorensen SJ, Hagglund P, Burmolle M (2017) A meta-proteomics approach to study the interspecies interactions affecting microbial biofilm development in a model community. Sci Rep 7(1):16483. https://doi.org/10.1038/s41598-017-16633-6
Hinsa SM, Espinosa-Urgel M, Ramos JL, O'Toole GA (2003) Transition from reversible to irreversible attachment during biofilm formation by Pseudomonas fluorescens WCS365 requires an ABC transporter and a large secreted protein. Mol Microbiol 49(4):905–918. https://doi.org/10.1046/j.1365-2958.2003.03615.x
Hoang V, Delatolla R, Abujamel T, Mottawea W, Gadbois A, Laflamme E, Stintzi A (2014) Nitrifying moving bed biofilm reactor (MBBR) biofilm and biomass response to long term exposure to 1 C. Water Res 49:215–224. https://doi.org/10.1016/j.watres.2013.11.018
Hu XB, Xu K, Wang Z, Ding LL, Ren HQ (2013a) Characteristics of biofilm attaching to carriers in moving bed biofilm reactor used to treat vitamin C wastewater. Scanning 35(5):283–291. https://doi.org/10.1002/sca.21064
Hu XB, Wang Z, Xu K, Ren HQ (2013b) Biofilm regeneration on carriers in MBBR used for vitamin C wastewater treatment. Water Sci Technol 67(6):1310–1316. https://doi.org/10.2166/wst.2013.695
Hu Q, Zhang XX, Jia SY, Huang KL, Tang JY, Shi P, Ye L, Ren HQ (2016) Metagenomic insights into ultraviolet disinfection effects on antibiotic resistome in biologically treated wastewater. Water Res 101:309–317. https://doi.org/10.1016/j.watres.2016.05.092
Huang H, Ren HQ, Ding LL, Geng JJ, Xu K, Zhang Y (2014) Aging biofilm from a full-scale moving bed biofilm reactor: characterization and enzymatic treatment study. Bioresour Technol 154:122–130. https://doi.org/10.1016/j.biortech.2013.12.031
Huang H, Ding LL, Ren HQ, Geng JJ, Xu K, Zhang Y (2015) Preconditioning of model biocarriers by soluble pollutants: a QCM-D study. ACS Appl Mater Interfaces 7(13):7222–7230. https://doi.org/10.1021/acsami.5b00324
Huang H, Lin Y, Peng PC, Geng JJ, Xu K, Zhang Y, Ding LL, Ren HQ (2018a) Calcium ion- and rhamnolipid-mediated deposition of soluble matters on biocarriers. Water Res 133:37–46. https://doi.org/10.1016/j.watres.2018.01.010
Huang H, Yu QS, Ren HQ, Geng JJ, Xu K, Zhang Y, Ding LL (2018b) Towards physicochemical and biological effects on detachment and activity recovery of aging biofilm by enzyme and surfactant treatments. Bioresour Technol 247:319–326. https://doi.org/10.1016/j.biortech.2017.09.082
Huenken M, Karsten U, Wiencke C (2005) Determination of the adenylate energy charge (AEC) as a tool to determine the physiological status of macroalgal tissues after UV exposure. Phycologia 44(3):249–253. https://doi.org/10.2216/0031-8884(2005)44[249:DOTAEC]2.0.CO;2
Irie Y, Borlee BR, O'Connor JR, Hill PJ, Harwood CS, Wozniak DJ, Parsek MR (2012) Self-produced exopolysaccharide is a signal that stimulates biofilm formation in Pseudomonas aeruginosa. Proc Natl Acad Sci U S A 109(50):20632–20636. https://doi.org/10.1073/pnas.1217993109
Jahid IK, Mizan MF, Ha AJ, Ha SD (2015) Effect of salinity and incubation time of planktonic cells on biofilm formation, motility, exoprotease production, and quorum sensing of Aeromonas hydrophila. Food Microbiol 49:142–151. https://doi.org/10.1016/j.fm.2015.01.016
Jain R, Behrens AJ, Kaever V, Kazmierczak BI (2012) Type IV pilus assembly in Pseudomonas aeruginosa over a broad range of cyclic di-GMP concentrations. J Bacteriol 194(16):4285–4294. https://doi.org/10.1128/jb.00803-12
Jefferson KK (2004) What drives bacteria to produce a biofilm? FEMS Microbiol Lett 236(2):163–173. https://doi.org/10.1016/j.femsle.2004.06.005
Jeffrey WH, Paul JH (1988) Underestimation of DNA synthesis by thymidine incorporation in marine bacteria. Appl Environ Microbiol 54(12):3165–3168
Jiang Y, Zhang B, He C, Shi J, Borthwick AGL, Huang X (2018) Synchronous microbial vanadium (V) reduction and denitrification in groundwater using hydrogen as the sole electron donor. Water Res 141:289–296. https://doi.org/10.1016/j.watres.2018.05.033
Juhlin A, Svensson S, Thomsen P, Trobos M (2017) Staphylococcal biofilm gene expression on biomaterials—a methodological study. J Biomed Mater Res Part A 105(12):3400–3412. https://doi.org/10.1002/jbm.a.36171
Kang SJ, Pugh LB, Borchardt JA (1983) ATP as a measure of active biomass concentration and inhibition in biological wastewater treatment processes. Proc. 38th Ind. Waste Conf. Purdue University, West Lafayette, pp 751–759
Kang ST, Subramani A, Hoek EMV, Deshusses MA, Matsumoto MR (2004) Direct observation of biofouling in cross-flow microfiltration: mechanisms of deposition and release. J Membr Sci 244(1–2):151–165. https://doi.org/10.1016/j.memsci.2004.07.011
Kappachery S, Paul D, Kweon JH (2012) Effect of N-acetylcysteine against biofouling of reverse osmosis membrane. Desalination 285:184–187. https://doi.org/10.1016/j.desal.2011.09.053
Karaolis DKR, Rashid MH, Chythanya R, Luo WS, Hyodo M, Hayakawa Y (2005) c-di-GMP (3 '-5 '-cyclic diguanylic acid) inhibits Staphylococcus aureus cell-cell interactions and biofilm formation. Antimicrob Agents Chemother 49(3):1029–1038. https://doi.org/10.1128/aac.49.3.1029-1038.2005
Kim LH, Jung Y, Kim SJ, Kim CM, Yu HW, Park HD, Kim IS (2015a) Use of rhamnolipid biosurfactant for membrane biofouling prevention and cleaning. Biofouling 31(2):211–220. https://doi.org/10.1080/08927014.2015.1022724
Kim LH, Jung Y, Yu HW, Chae KJ, Kim IS (2015b) Physicochemical interactions between rhamnolipids and Pseudomonas aeruginosa biofilm layers. Environ Sci Technol 49(6):3718–3726. https://doi.org/10.1021/es505803c
Kumar A, Ting YP (2016) Streptomycin favors biofilm formation by altering cell surface properties. Appl Microbiol Biotechnol 100(20):8843–8853. https://doi.org/10.1007/s00253-016-7793-0
Lai CY, Dong QY, Chen JX, Zhu QS, Yang X, Chen WD, Zhao HP, Zhu L (2018) Role of extracellular polymeric substances in a methane based membrane biofilm reactor reducing vanadate. Environ Sci Technol 52:10680–10688. https://doi.org/10.1021/acs.est.8b02374
Laureni M, Weissbrodt DG, Szivak I, Robin O, Nielsen JL, Morgenroth E, Joss A (2015) Activity and growth of anammox biomass on aerobically pre-treated municipal wastewater. Water Res 80:325–336. https://doi.org/10.1016/j.watres.2015.04.026
Li X, Li J, Wang J, Wang H, Cui C, He B, Zhang H (2014) Direct monitoring of sub-critical flux fouling in a horizontal double-end submerged hollow fiber membrane module using ultrasonic time domain reflectometry. J Membr Sci 451:226–233. https://doi.org/10.1016/j.memsci.2013.09.060
Li R, Morrison L, Collins G, Li A, Zhan X (2016) Simultaneous nitrate and phosphate removal from wastewater lacking organic matter through microbial oxidation of pyrrhotite coupled to nitrate reduction. Water Res 96:32–41. https://doi.org/10.1016/j.watres.2016.03.034
Liang ZX (2015) The expanding roles of c-di-GMP in the biosynthesis of exopolysaccharides and secondary metabolites. Nat Prod Rep 32(5):663–683. https://doi.org/10.1039/C4NP00086B
Liu Y (1995) Adhesion kinetics of nitrifying bacteria on various thermoplastic supports. Colloids Surf B 5(5):213–219. https://doi.org/10.1016/0927-7765(95)01215-x
Liu S, Gunawan C, Barraud N, Rice SA, Harry EJ, Amal R (2016) Understanding, monitoring, and controlling biofilm growth in drinking water distribution systems. Environ Sci Technol 50(17):8954–8976. https://doi.org/10.1021/acs.est.6b00835
Lu H, Chandran K, Stensel D (2014) Microbial ecology of denitrification in biological wastewater treatment. Water Res 64:237–254. https://doi.org/10.1016/j.watres.2014.06.042
Ma R, Qiu S, Jiang Q, Sun H, Xue T, Cai G, Sun B (2017a) AI-2 quorum sensing negatively regulates rbf expression and biofilm formation in Staphylococcus aureus. Int J Med Microbiol 307(4–5):257–267. https://doi.org/10.1016/j.ijmm.2017.03.003
Ma W, Han Y, Ma W, Han H, Zhu H, Xu C, Li K, Wang D (2017b) Enhanced nitrogen removal from coal gasification wastewater by simultaneous nitrification and denitrification (SND) in an oxygen-limited aeration sequencing batch biofilm reactor. Bioresour Technol 244:84–91. https://doi.org/10.1016/j.biortech.2017.07.083
Marcato-Romain CE, Pechaud Y, Paul E, Girbal-Neuhauser E, Dossat-Letisse V (2012) Removal of microbial multi-species biofilms from the paper industry by enzymatic treatments. Biofouling 28(3):305–314. https://doi.org/10.1080/08927014.2012.673122
Maukonen J, Matto J, Wirtanen G, Raaska L, Mattila-Sandholm T, Saarela M (2003) Methodologies for the characterization of microbes in industrial environments: a review. J Ind Microbiol Biotechnol 30(6):327–356. https://doi.org/10.1007/s10295-003-0056-y
Nouvion N, Block JC, Faup GM (1987) Effect of biomass quantity and activity on TOC removal in a fixed-bed reactor. Water Res 21(1):35–40. https://doi.org/10.1016/0043-1354(87)90096-0
Ochoa JC, Coufort C, Escudie R, Line A, Paul E (2007) Influence of non-uniform distribution of shear stress on aerobic biofilms. Chem Eng Sci 62(14):3672–3684. https://doi.org/10.1016/j.ces.2007.03.023
Odegaard H, Rusten B, Badin H (1993) Small wastewater treatment plants based on moving bed biofilm reactors. Water Sci Technol 28(10):351–359
Oh YJ, Lee NR, Jo W, Jung WK, and Lim JS (2009) Effects of substrates on biofilm formation observed by atomic force microscopy. Ultramicroscopy 109(8):874–80. https://doi.org/10.1016/j.ultramic.2009.03.042
Oh HS, Tan CH, Low JH, Rzechowicz M, Siddiqui MF, Winters H, Kjelleberg S, Fane AG, Rice SA (2017) Quorum quenching bacteria can be used to inhibit the biofouling of reverse osmosis membranes. Water Res 112:29–37. https://doi.org/10.1016/j.watres.2017.01.028
Ono K, Oka R, Toyofuku M, Sakaguchi A, Hamada M, Yoshida S, Nomura N (2014) cAMP signaling affects irreversible attachment during biofilm formation by Pseudomonas aeruginosa PAO1. Microbes Environ 29(1):104–106. https://doi.org/10.1264/jsme2.ME13151
Paul JH, Jeffrey WH, DeFlaun MF (1987) Dynamics of extracellular DNA in the marine environment. Appl Environ Microbiol 53:170–179
Paul E, Ochoa JC, Pechaud Y, Liu Y, Line A (2012) Effect of shear stress and growth conditions on detachment and physical properties of biofilms. Water Res 46(17):5499–5508. https://doi.org/10.1016/j.watres.2012.07.029
Peng X, Guo F, Ju F, Zhang T (2014) Shifts in the microbial community, nitrifiers and denitrifiers in the biofilm in a full-scale rotating biological contactor. Environ Sci Technol 48(14):8044–8052. https://doi.org/10.1021/es5017087
Peng PC, Huang H, Ren HQ (2018a) Effect of adding low-concentration of rhamnolipid on reactor performances and microbial community evolution in MBBRs for low C/N ratio and antibiotic wastewater treatment. Bioresour Technol 256:557–561. https://doi.org/10.1016/j.biortech.2018.02.035
Peng PC, Huang H, Ren HQ, Ma HJ, Lin Y, Geng JJ, Xu K, Zhang Y, Ding LL (2018b) Exogenous N-acyl homoserine lactones facilitate microbial adhesion of high ammonia nitrogen wastewater on biocarrier surfaces. Sci Total Environ 624:1013–1022. https://doi.org/10.1016/j.scitotenv.2017.12.248
Persson F, Sultana R, Suarez M, Hermansson M, Plaza E, Wilen BM (2014) Structure and composition of biofilm communities in a moving bed biofilm reactor for nitritation-anammox at low temperatures. Bioresour Technol 154:267–273. https://doi.org/10.1016/j.biortech.2013.12.062
Petrova OE, Sauer K (2012) Dispersion by Pseudomonas aeruginosa requires an unusual posttranslational modification of BdlA. Proc Natl Acad Sci U S A 109(41):16690–16695. https://doi.org/10.1073/pnas.1207832109
Piculell M, Suarez C, Li C, Christensson M, Persson F, Wagner M, Hermansson M, Jonsson K, Welander T (2016) The inhibitory effects of reject water on nitrifying populations grown at different biofilm thickness. Water Res 104:292–302. https://doi.org/10.1016/j.watres.2016.08.027
Ras M, Lefebvre D, Derlon N, Paul E, Girbal-Neuhauser E (2011) Extracellular polymeric substances diversity of biofilms grown under contrasted environmental conditions. Water Res 45(4):1529–1538. https://doi.org/10.1016/j.watres.2010.11.021
Ren TT, Li XY, Yu HQ (2013) Effect of N-acy-L-homoserine lactones-like molecules from aerobic granules on biofilm formation by Escherichia coli K12. Bioresour Technol 129:655–658. https://doi.org/10.1016/j.biortech.2012.12.043
Roosjen A, Busscher HJ, Nordel W, van der Mei HC (2006) Bacterial factors influencing adhesion of Pseudomonas aeruginosa strains to a poly(ethylene oxide) brush. Microbiol-Sgm 152:2673–2682. https://doi.org/10.1099/mic.0.29005-0
Ross P, Weinhouse H, Aloni Y, Michaeli D, Weinberger-Ohana P, Mayer R, Braun S, de Vroom E, van der Marel GA, van Boom JH, Beziman M (1987) Regulation of cellulose synthesis in Acetobacter xylinum by cyclic diguanylic acid. Nature 325:279–281. https://doi.org/10.1038/325279a0
Sauer K, Camper AK (2001) Characterization of phenotypic changes in Pseudomonas putida in response to surface-associated growth. J Bacteriol 183(22):6579–6589. https://doi.org/10.1128/jb.183.22.6579-6589.2001
Saur T, Morin E, Habouzit F, Bernet N, Escudie R (2017) Impact of wall shear stress on initial bacterial adhesion in rotating annular reactor. PLoS One 12(2):e0172113. https://doi.org/10.1371/journal.pone.0172113
Schmid N, Suppiger A, Steiner E, Pessi G, Kaever V, Fazli M, Tolker-Nielsen T, Jenal U, Eberl L (2017) High intracellular c-di-GMP levels antagonize quorum sensing and virulence gene expression in Burkholderia cenocepacia H111. Microbiol-Sam 163(5):754–764. https://doi.org/10.1099/mic.0.000452
Shen Y, Monroy GL, Derlon N, Janjaroen D, Huang C, Morgenroth E, Boppart SA, Ashbolt NJ, Liu WT, Nguyen TH (2015) Role of biofilm roughness and hydrodynamic conditions in Legionella pneumophila adhesion to and detachment from simulated drinking water biofilms. Environ Sci Technol 49(7):4274–4282. https://doi.org/10.1021/es505842v
Shrout JD, Nerenberg R (2012) Monitoring bacterial twitter: does quorum sensing determine the behavior of water and wastewater treatment biofilms? Environ Sci Technol 46(4):1995–2005. https://doi.org/10.1021/es203933h
Si X, Quan X (2017) Prevention of multi-species wastewater biofilm formation using vanillin and EPS disruptors through non-microbicidal mechanisms. Int Biodeterior Biodegrad 116:211–218. https://doi.org/10.1016/j.ibiod.2016.11.009
Sihorkar V, Vyas SP (2001) Biofilm consortia on biomedical and biological surfaces: delivery and targeting strategies. Pharm Res 18(9):1247–1254. https://doi.org/10.1023/a:1013073508318
Sillankorva S, Oliveira R, Vieira MJ, Sutherland IW, Azeredo J (2004) Bacteriophage Phi S1 infection of Pseudomonas fluorescens planktonic cells versus biofilms. Biofouling 20(3):133–138. https://doi.org/10.1080/08927010410001723834
Simões M, Pereira MO, Vieira MJ (2005) Effect of mechanical stress on biofilms challenged by different chemicals. Water Res 39(20):5142–5152. https://doi.org/10.1016/j.watres.2005.09.028
Simões M, Simões LC, Vieira MJ (2010) A review of current and emergent biofilm control strategies. LWT-Food Sci Technol 43(4):573–583. https://doi.org/10.1016/j.lwt.2009.12.008
Stewart PS, Costerton JW (2001) Antibiotic resistance of bacteria in biofilms. Lancet 358(9276):135–138. https://doi.org/10.1016/S0140-6736(01)05321-1
Sudo R (1988) Environmental monitoring by using microorganisms. J Environ Conserv Eng 17(11):715–720
Takada K, Hashimoto K, Soda S, Ike M, Makio T, Nakayama Y, Miyamoto H, Yamashita K, Hashimoto T (2018) Microbial communities on the submerged membranes in full-scale membrane bioreactors treating municipal wastewater. J Environ Eng 144(1):04017084. https://doi.org/10.1061/(ASCE)EE.1943-7870.0001294
Tang JY, Bu YQ, Zhang XX, Huang KL, He XW, Ye L, Shan ZJ, Ren HQ (2016) Metagenomic analysis of bacterial community composition and antibiotic resistance genes in a wastewater treatment plant and its receiving surface water. Ecotoxicol Environ Saf 132:260–269. https://doi.org/10.1016/j.ecoenv.2016.06.016
Tang CC, Tian Y, He ZW, Zuo W, Zhang J (2018) Performance and mechanism of a novel algal-bacterial symbiosis system based on sequencing batch suspended biofilm reactor treating domestic wastewater. Bioresour Technol 265:422–431. https://doi.org/10.1016/j.biortech.2018.06.033
Tischler AD, Camilli A (2004) Cyclic diguanylate (c-di-GMP) regulates Vibrio cholerae biofilm formation. Mol Microbiol 53(3):857–869. https://doi.org/10.1111/j.1365-2958.2004.04155.x
Tomczyk-Zak K, Szczesny P, Gromadka R, Zielenkiewicz U (2017) Taxonomic and chemical assessment of exceptionally abundant rock mine biofilm. PeerJ 5:e3635. https://doi.org/10.7717/peerj.3635
Trojanowicz K, Wojcik W, Styka W, Baczynski T (2011) Evaluation of a novel control method on biofilm parameters in an aerated submerged fixed-bed biofilm reactor. Clean-Soil Air Water 39(1):7–12. https://doi.org/10.1002/clen.201000111
Ueda A, Wood TK (2010) Tyrosine phosphatase TpbA of Pseudomonas aeruginosa controls extracellular DNA via cyclic diguanylic acid concentrations. Environ Microbiol Rep 2(3):449–455. https://doi.org/10.1111/j.1758-2229.2010.00171.x
Verstraeten N, Braeken K, Debkumari B, Fauvart M, Fransaer J, Vermant J, Michiels J (2008) Living on a surface: swarming and biofilm formation. Trends Microbiol 16(10):496–506. https://doi.org/10.1016/j.tim.2008.07.004
Villaverde S, GarciaEncina PA, FdzPolanco F (1997) Influence of pH over nitrifying biofilm activity in submerged biofilters. Water Res 31(5):1180–1186. https://doi.org/10.1016/s0043-1354(96)00376-4
Violant D, Galofre M, Nart J, Teles RP (2014) In vitro evaluation of a multispecies oral biofilm on different implant surfaces. Biomed Mater 9(3):035007. https://doi.org/10.1088/1748-6041/9/3/035007
Visscher F, van der Schaaf J, Nijhuis TA, Schouten JC (2013) Rotating reactors—a review. Chem Eng Res Des 91(10):1923–1940. https://doi.org/10.1016/j.cherd.2013.07.021
Wagner M, Taherzadeh D, Haisch C, Horn H (2010) Investigation of the mesoscale structure and volumetric features of biofilms using optical coherence tomography. Biotechnol Bioeng 107(5):844–853. https://doi.org/10.1002/bit.22864
Wang H, Wang H, Xing T, Wu N, Xu X, Zhou G (2016) Removal of Salmonella biofilm formed under meat processing environment by surfactant in combination with bio-enzyme. LWT-Food Sci Technol 66:298–304. https://doi.org/10.1016/j.lwt.2015.10.049
Wang C, Liu S, Xu X, Zhao C, Yang F, Wang D (2017) Potential coupling effects of ammonia-oxidizing and anaerobic ammonium-oxidizing bacteria on completely autotrophic nitrogen removal over nitrite biofilm formation induced by the second messenger cyclic diguanylate. Appl Microbiol Biotechnol 101(9):3821–3828. https://doi.org/10.1007/s00253-016-7981-y
Wang JF, Ren HQ, Li XH, Li JX, Ding LL, Geng JJ, Xu K, Huang H, Hu HD (2018a) In situ monitoring of wastewater biofilm formation process via ultrasonic time domain reflectometry (UTDR). Chem Eng J 334:2134–2141. https://doi.org/10.1016/j.cej.2017.11.043
Wang X, Bi X, Hem LJ, Ratnaweera H (2018b) Microbial community composition of a multi-stage moving bed biofilm reactor and its interaction with kinetic model parameters estimation. J Environ Manag 218:340–347. https://doi.org/10.1016/j.jenvman.2018.04.015
Wei Q, Ma LZ (2013) Biofilm matrix and its regulation in Pseudomonas aeruginosa. Int J Mol Sci 14(10):20983–21005. https://doi.org/10.3390/ijms141020983
Wen ZZT, Burne RA (2003) Functional genomics approach to identifying genes required for biofilm development by Streptococcus mutans (vol 68, pg 1196, 2002). Appl Environ Microbiol 69(1):722–722. https://doi.org/10.1128/aem.69.1.722.2003
White DC, Bobbie RJ, Herron JS, King JD, Morrison SJ (1979) Biochemical measurements of microbial mass and activity from environmental samples. ASTM Spec Tech Publ (695):13
Wimpenny J, Manz W, Szewzyk U (2000) Heterogeneity in biofilms. FEMS Microbiol Rev 24(5):661–671. https://doi.org/10.1111/j.1574-6976.2000.tb00565.x
Xiao Y, De Araujo C, Sze CC, Stuckey DC (2015) Toxicity measurement in biological wastewater treatment processes: a review. J Hazard Mater 286:15–29. https://doi.org/10.1016/j.jhazmat.2014.12.033
Yang Y, Hoe YW, Zheng Q, Chung HJ, Yuk HG (2017) Biofilm formation by Salmonella Enteritidis in a simulated liquid egg processing environment and its sensitivity to chlorine and hot water treatment. Food Control 73:595–600. https://doi.org/10.1016/j.foodcont.2016.09.008
Young B, Delatolla R, Kennedy K, Laflamme E, Stintzi A (2017) Low temperature MBBR nitrification: microbiome analysis. Water Res 111:224–233. https://doi.org/10.1016/j.watres.2016.12.050
Yu QS, Huang H, Ren HQ, Ding LL, Geng JJ (2016) In situ activity recovery of aging biofilm in biological aerated filter: surfactants treatment and mechanisms study. Bioresour Technol 219:403–410. https://doi.org/10.1016/j.biortech.2016.07.122
Zhang R, Han ZY, Chen CJ, Shi DZ, Huang XX, Wu WX (2011) Microstructure and microbial ecology of biofilm in the bioreactor for nitrogen removing from wastewater: a review. Shengtaixue Zazhi 30(11):2628–2636
Zhang P, Guo JS, Shen Y, Yan P, Chen YP, Wang H, Yang JX, Fang F, Li C (2015) Microbial communities, extracellular proteomics and polysaccharides: a comparative investigation on biofilm and suspended sludge. Bioresour Technol 190:21–28. https://doi.org/10.1016/j.biortech.2015.04.058
Zhang P, Chen YP, Wang W, Shen Y, Guo JS (2016) Surface plasmon resonance for water pollutant detection and water process analysis. TrAC Trends Anal Chem 85:153–165. https://doi.org/10.1016/j.trac.2016.09.003
Zhou XH, Yu T, Shi HC, Shi HM (2011) Temporal and spatial inhibitory effects of zinc and copper on wastewater biofilms from oxygen concentration profiles determined by microelectrodes. Water Res 45(2):953–959. https://doi.org/10.1016/j.watres.2010.09.035
Zhu Y, Zhang Y, Ren HQ, Geng JJ, Xu K, Huang H, Ding LL (2015) Physicochemical characteristics and microbial community evolution of biofilms during the start-up period in a moving bed biofilm reactor. Bioresour Technol 180:345–351. https://doi.org/10.1016/j.biortech.2015.01.006
Funding
This work was supported by the National Natural Science Foundation of China (51878336, 51608254), Jiangsu Natural Science Foundation (BK20160655), National Science and Technology Major Project (2017ZX07204001), and the Fundamental Research Funds for the Central Universities (021114380046).
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
This article contains no studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Huang, H., Peng, C., Peng, P. et al. Towards the biofilm characterization and regulation in biological wastewater treatment. Appl Microbiol Biotechnol 103, 1115–1129 (2019). https://doi.org/10.1007/s00253-018-9511-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9511-6