Abstract
Cyanobacteria, which perform oxygenic photosynthesis, have drawn attention as hosts for the direct production of biofuels and commodity chemicals from CO2 and H2O using light energy. Although cyanobacteria capable of producing diverse chemicals have been generated by metabolic engineering, anaerobic non-photosynthetic culture conditions are often necessary for their production. In this study, we conducted cell type-specific metabolic engineering of the filamentous cyanobacterium Anabaena sp. PCC 7120, which forms a terminally differentiated cell called a heterocyst with a semi-regular spacing of 10–15 cells. Because heterocysts are specialized cells for nitrogen fixation, the intracellular oxygen level of heterocysts is maintained very low even when adjacent cells perform oxygenic photosynthesis. Pyruvate decarboxylase of Zymomonas mobilis and alcohol dehydrogenase of Synechocystis sp. PCC 6803 were exclusively expressed in heterocysts. Ethanol production was concomitant with nitrogen fixation in genetically engineered Anabaena sp. PCC 7120. Engineering of carbon metabolism in heterocysts improved ethanol production, and strain ET14, with an extra copy of the invB gene expressed from a heterocyst-specific promoter, produced 130.9 mg L−1 of ethanol after 9 days. ET14 produced 1681.9 mg L−1 of ethanol by increasing the CO2 supply. Ethanol production per heterocyst cell was approximately threefold higher than that per cell of unicellular cyanobacterium. This study demonstrates the potential of heterocysts for anaerobic production of biofuels and commodity chemicals under oxygenic photosynthetic conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cyanobacteria can produce all carbon compounds that are essential for their growth from CO2 using light energy. In the last decade, cyanobacteria capable of producing useful materials such as biofuels and commodity chemicals from CO2 have been developed by introducing engineered metabolic pathways (Angermayr et al. 2015). Metabolic engineering of cyanobacteria has succeeded in generating strains that produce diverse alcohols such as ethanol (Dexter and Fu 2009), isopropanol (Kusakabe et al. 2013), 1-butanol (Lan and Liao 2011), isobutanol (Atsumi et al. 2009), 2-methyl-1-butanol (Shen and Liao 2012), 1,2-propanediol (Li and Liao 2013), and 2,3-butanediol (Oliver et al. 2013; Savakis et al. 2013). Biofuel production from CO2 using genetically engineered cyanobacteria is a promising process to reduce CO2 emissions and dependency on petroleum.
Fermentation generally occurs under conditions of oxygen deprivation and includes oxygen-sensitive enzymes that are incompatible with oxygenic photosynthesis. Replacing an oxygen-sensitive enzyme involved in the 1-butanol synthesis pathway with an oxygen-tolerant enzyme enhanced photosynthetic 1-butanol production (Lan et al. 2013). Conflict between oxygen-sensitive enzymes and photosynthesis can also be overcome by spatial or temporal separation. For temporal separation, cyanobacteria grown under photosynthetic conditions are transferred to dark and anaerobic conditions, and then fermentation with oxygen-sensitive enzymes is conducted. Meanwhile, spatial separation by dedicating a part of cells of filamentous cyanobacteria to fermentation enables chemical production using oxygen-sensitive enzymes under oxygenic photosynthetic conditions. Anabaena sp. strain PCC 7120 (hereafter Anabaena) is a filamentous cyanobacterium that forms chains of a few hundred cells (Fig. 1a). In the presence of combined nitrogen in the medium, Anabaena filaments comprise only vegetative cells, whereas when combined nitrogen is depleted, heterocysts differentiate from vegetative cells with a semi-regular spacing of 10–15 cells (Flores and Herrero 2010). Heterocysts function in the fixation of dinitrogen, which is catalyzed by the oxygen-sensitive enzyme nitrogenase. To protect nitrogenase from oxygen, heterocysts provide micro-oxic intracellular environments by inactivating the oxygen-evolving photosystem II, forming a thick envelope outside the cell wall, and enhancing respiration, which enable Anabaena to simultaneously perform oxygenic photosynthesis and nitrogen fixation. Because heterocysts lose their photosynthetic ability, vegetative cells supply heterocysts with photosynthetically produced carbohydrate, probably in the form of sucrose (Cumino et al. 2007; Nurnberg et al. 2015). In heterocysts, sucrose is catabolized to generate ATP and NADPH, which are required for nitrogen fixation (Fig. 1b). In addition to sucrose, alanine and glutamate can be transferred from vegetative cells to heterocysts (Jüttner 1983). Cell growth of heterocysts ceases and they are unable to return to a vegetative cell state. Thus, heterocysts are specialized for nitrogen fixation as if they are cell factories for the production of combined nitrogen compounds.
The metabolic pathway for ethanol production in heterocysts of engineered Anabaena PCC 7120. a The filamentous cyanobacterium Anabaena PCC 7120 grown without combined nitrogen. Heterocysts are indicated by arrowheads. b Vegetative cells synthesize sucrose and Ala from CO2 by photosynthesis and provide them for heterocysts. In the heterocysts that express the exogenous ethanol synthesis pathway, ethanol is produced from sucrose and Ala. GAPDH, glyceraldehyde-3-phpsphate (G3P) dehyderogenase (encoded by gap1); PYK, pyruvate kinase (encoded by pyk1); ALDH, alanine dehydrogenase (encoded by ald); PDC, pyruvate decarboxylase (encoded by pdc); ADH, alcohol dehydrogenase (encoded by adhA)
In this study, we conducted heterocyst-specific metabolic engineering of Anabaena. An Anabaena strain that expresses the ethanol synthesis pathway only in heterocysts was developed, so that the ethanol production process was separated from photosynthesis. The Anabaena strain produced ethanol concurrently with nitrogen fixation, and ethanol production was improved by enhancing the pyruvate supply to heterocysts and optimizing the culture conditions. This study shows the potential of heterocysts as a cell factory for chemical production.
Materials and methods
Culture and production conditions
Anabaena sp. strain PCC 7120 and its derivatives were grown in BG-11 medium (containing NaNO3 as a nitrogen source) as previously described (Ehira and Ohmori 2006). Spectinomycin and neomycin were added to the medium at a final concentration of 10 and 30 μg mL−1, respectively, when required. For ethanol production, the cells that were grown in BG-11 medium until they reached an OD750 of 1.0–1.5 were washed three times with sterile water and then resuspended to OD750 of 0.5 in nitrogen-free BG-11 medium that contained 1 mM NaHCO3. In a 100-mL Erlenmeyer flask with a vent cap, a 30-mL aliquot of nitrogen-free culture was incubated at 30 °C under continuous illumination that was provided using a fluorescent lamp at 30 μmol photons m−2 s−1. The cultures were shaken at 100 rpm using a rotary shaker NR-3 (TAITEC, Saitama, Japan).
Expression of the ethanol synthetic pathway in heterocysts using the pET1 plasmid
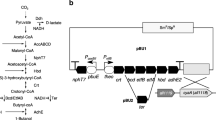
The plasmids constructed and used in this study are listed in Table 1. Primers used in this study were designed based on the genome data obtained from CyanoBase (Fujisawa et al. 2017) and are listed in Table 2. The plasmid pET1 that was used for expressing the ethanol synthetic pathway in heterocysts was constructed as follows. The pdc gene of Zymomonas mobilis ATCC 29191 was amplified by PCR using the primer pair InFZmpdc-F and InFZmpdc-R. The adhA gene of Synechocystis sp. PCC 6803 was amplified by PCR using the primer pair InFSyadhA-F and InFSyadhA-R. The pdc and adhA genes were tandemly cloned into SpeI- and SmaI-digested pBluescript II KS+ (Agilent Technologies, Santa Clara, CA) using In-Fusion HD Cloning Kit (Takara Bio, Shiga, Japan). The promoter region of the hupS gene of Anabaena PCC 7120 was amplified by PCR using the primer pair PhupS-F and PhupS-R and cloned between the SacI and SpeI sites of the pdc and adhA-containing pBluescript II KS+. A SacI-SalI fragment that contained the hupS promoter, pdc and adhA, was cloned between the SacI and SalI sites of the shuttle vector pAM505 (Yoon and Golden 1998) to construct the pET1 plasmid.
Plasmids for metabolic engineering of heterocysts
To integrate synthetic genes into chromosomes, an integration vector, pSU102, was constructed as follows. The nptII gene of pSU101 (Ehira et al. 2017) was eliminated by PCR using the primer pair pSU101-F and pSU101-R, and the PCR fragment was ligated with a spectinomycin and streptomycin resistance cassette, which was excised from pDW9 (Golden and Wiest 1988) with HincII to construct pSU102.
Transcriptional fusion genes of the nifB promoter and the invA, invB, or sigE genes were constructed as follows. The genomic region between alr2882 and all2883 and the nifB promoter region were amplified by PCR using the primer pair NSI-F2 and NSI-R and the primer pair PnifB-F and PnifB-R, respectively, and sequentially cloned between the SalI and EcoRI sites of pBluescript II KS+ and between the EcoRI and SacI sites. The cloned fragment was excised with SalI and SacI and cloned between the SalI and SacI sites of pSU102. The invA gene was amplified by PCR using the primer pair invA-F and invA-R and inserted downstream of the nifB promoter using the SacI and KpnI sites to construct pET13. Similarly, the invB and sigE genes were amplified by PCR using the primer pair invB-F and invB-R and the primer pair sigE-F and sigE-R, respectively, and inserted downstream of the nifB promoter to construct pET14 and pET11, respectively. A DNA fragment that contained the ald gene and its promoter region was amplified using the primer pair ald-F and ald-R and cloned into pSU102 with the DNA fragment of the intergenic region between alr2882 and all2883 to construct pET12.
Strain construction
The strains constructed in this study are listed in Table 1. Transformation of Anabaena PCC 7120 was performed according to the method of Elhai et al. (1997). In brief, strain ET1 was constructed by introducing plasmid pAHpa. Strains ET11, ET12, ET13, and ET14 were constructed by recombination of plasmids pET11, pET12, pET13, and pET14, respectively, into the chromosome of strain ET1.
Ethanol quantification
Culture samples (600 μL) of ethanol-producing strains were centrifuged at 21,500×g for 5 min, and the supernatants (500 μL) were mixed with 1 mg mL−1 of 2-propanol (50 μL) as the internal standard. The mixtures were transferred to 1.5-mL vials and incubated at 60 °C for 20 min. The ethanol concentration in the headspace was measured using gas chromatography (GC-2014; Shimadzu, Kyoto, Japan) with a flame ion detector and a 2-m stainless steel column (internal diameter, 3 mm) filled with Gaskuropack 54 60/80 (GL Sciences, Tokyo, Japan). The column temperature was maintained at 140 °C and the injector and detector were maintained at 180 °C.
Nitrogen fixation activity
The nitrogenase activity was determined by the acetylene reduction assays as previously described (Ehira and Ohmori 2011).
Results
Ethanol tolerance of Anabaena sp. PCC 7120
Nitrogen fixation requires 16 ATP molecules and eight electrons to convert one N2 molecule into two NH3 molecules. Growth under nitrogen-fixing conditions is considered to be susceptible to adverse environmental conditions such as growth in the presence of ethanol. Figure 2 shows the effect of ethanol on the growth of Anabaena with or without combined nitrogen. Addition of 0.75% (v/v) ethanol to the culture medium containing nitrate as the nitrogen source slightly retarded the growth of Anabaena (Fig. 2a). Growth was also affected by 0.75% ethanol under nitrogen-fixing conditions, but cellular proliferation was not suppressed (Fig. 2b). A higher ethanol concentration impaired the growth of Anabaena; the growth rates were obviously decreased in the presence of 1.5% ethanol, and cellular proliferation was completely inhibited by 3.0% ethanol under both growth conditions. These results indicate that the growth of Anabaena in the presence of ethanol under nitrogen-fixing conditions is comparable with that with combined nitrogen, although ethanol at concentrations of > 0.75% showed negative effects on Anabaena growth. In the unicellular non-nitrogen-fixing cyanobacterium Synechocystis sp. PCC 6803, the addition of 2.0% ethanol reduced the growth rate to approximately one fourth (Zhang et al. 2015), whereas Synechococcus elongates PCC 7942, also a unicellular non-nitrogen-fixing cyanobacterium, ceased growth in the presence of approximately 0.4% ethanol (Arai et al. 2017). Therefore, Anabaena has a relatively high tolerance to ethanol.
The effect of ethanol on the growth of Anabaena PCC 7120. Anabaena PCC 7120 was grown with nitrate as the nitrogen source (a) or without combined nitrogen (b) in the absence of ethanol (circles), in the presence of 0.75% (triangles), 1.5% (squares), and 3% (crosses) ethanol. The culture was aerated with air that contained 1.0% CO2 at a rate of about 15 mL min−1
Ethanol production in heterocysts
To generate the ethanol-producing strain ET1, plasmid pET1 was introduced to Anabaena (Fig. 3a). In ET1, the pdc gene that encodes pyruvate decarboxylase and the adhA gene that encodes alcohol dehydrogenase are expressed from the promoter of hupS, which is exclusively activated within heterocysts (Happe et al. 2000; Holmqvist et al. 2009). In heterocysts, NADPH is actively generated to supply a reducing power to nitrogenase through the oxidative pentose phosphate pathway (Summers et al. 1995). Therefore, we used the adhA gene of Synechocystis PCC 6803 that encodes NADP-dependent alcohol dehydrogenase (Vidal et al. 2009). Filaments of the strain ET1 grown with nitrate were transferred to the nitrogen-free medium supplemented with 1 mM NaHCO3 to induce heterocyst differentiation and ethanol production. ET1 produced 59.5 mg L−1 ethanol after 10 days, showing that heterocysts that were genetically engineered to express the ethanol synthetic pathway were capable of ethanol production (Fig. 3b).
Ethanol production by genetically modified Anabaena PCC 7120. a Plasmid map of pET1 used to express the ethanol synthesis pathway in heterocysts. b Ethanol production by strains ET1 (closed circles), ET11 (ET1/PnifB::sigE, open triangles), and ET12 (ET1/Pald::ald, open squares) under nitrogen-fixing conditions with 1 mM NaHCO3. c Nitrogenase activities of the control strain harboring pAM505 (white bars) and ET1 (black bars). The Student’s t test showed a significant difference between the control strain and ET1 levels at day 3 (p < 0.001)
Enhancement of ethanol production by increasing pyruvate supply in heterocysts
Pyruvate is the starting material for the ethanol synthetic pathway introduced in ET1, which is supplied by the intrinsic metabolism of heterocysts (Fig. 1b). To enhance ethanol production, pyruvate supply was increased by activating glycolysis or directly supplying pyruvate from alanine. A group 2 sigma factor, SigE, upregulates the expression of gap1 and pyk1 that encode glyceraldehyde-3-phosphate dehydrogenase and pyruvate kinase, respectively (Ehira and Ohmori 2011). Hence, SigE overexpression can activate glycolysis to increase pyruvate generation. The sigE gene, which was cloned downstream of the heterocyst-specific nifB promoter (Elhai and Wolk 1990; Thiel and Pratte 2014), was integrated into the ET1 chromosome, resulting in ET11. ET11 produced 74.9 mg L−1 ethanol after 10 days, which was 25% higher than that produced by ET1 (Fig. 3b). Duplication of the ald gene that encodes alanine dehydrogenase also improved ethanol production. Alanine dehydrogenase mainly catalyzes the catabolic reaction, namely the conversion of alanine into pyruvate, in heterocysts, and the expression of the ald gene is highly induced in heterocysts (Pernil et al. 2010). In strain ET12, an extra copy of the ald gene with its original promoter was introduced into the ET1 chromosome to increase its expression level in heterocysts. Introducing the extra copy of the ald gene increased the ethanol concentration to > 180% after 10 days (109.2 mg L−1) compared with ET1 (Fig. 3b). Thus, increasing the pyruvate supply in heterocysts promotes ethanol production.
Enhancement of ethanol production by activating sucrose catabolism in heterocysts
Because both ethanol synthesis and nitrogen fixation use NADPH as a reductant, introducing the ethanol synthetic pathway into heterocysts would cause competition between these two reactions for NADPH. To assess the correlation between ethanol production and nitrogen fixation, nitrogenase activities during ethanol production were determined (Fig. 3c). The highest nitrogenase activity was detected on the second day after induction of heterocyst differentiation, both in the control strain and ET1. On the third day, the nitrogenase activity of the control strain was remained high but decreased by half in ET1. Because the ethanol production rate between the second and third day (20.6 mg day−1) was increased more than fourfold compared with that between the first and second day (4.8 mg day−1) (Fig. 3b), competition between nitrogen fixation and ethanol production for NADPH is likely to have occurred. Heterocysts are provided with sucrose from adjacent vegetative cells and use it for generation of NADPH (Fig. 1b). To increase the supply of NADPH, sucrose degradation in heterocysts was activated with an invertase. The invA or invB genes that encode invertases were cloned downstream of the nifB promoter and integrated into the chromosome of ET1, resulting in ET13 or ET14, respectively. Ethanol accumulation in the ET13 culture reached 75.4 mg L−1 in 10 days, but the increase in ethanol concentration ceased on the sixth day (Fig. 4). The maximum ethanol concentration produced by ET14 was 130.9 mg L−1, which was 120% higher than that produced by ET1 (Fig. 4). These results indicate that the activation of sucrose catabolism in heterocysts enhances ethanol production.
Ethanol production with a gas stripping-based recovery system
Ethanol productivity of ET14 was increased more than twofold compared with the original ET1 strain (Fig. 4). However, the ethanol production rate was stagnated after the sixth day. There were two possible causes for the decrease in production rate. The first possibility was inhibitory effects of accumulated ethanol and the second was reduction of photosynthetic activity caused by CO2 limitation. To evaporate ethanol from the culture media and supply CO2, cultures were aerated with air that contained 1% CO2. Evaporated ethanol was recovered in collection tubes that contained cold water. ET14 cultivated with a gas stripping-based recovery system produced 1681.9 mg L−1 of ethanol in 23 days (Fig. 5). Ethanol concentration on the 10th day was 990.1 mg L−1, which was more than seven times the ethanol concentration produced without gas stripping. Thus, the gas stripping-based recovery system substantially improved the ethanol production by ET14.
Discussion
In this study, pyruvate decarboxylase of Z. mobilis and alcohol dehydrogenase of Synechocystis PCC 6803 were expressed from the heterocyst-specific promoter, PhupS, in the filamentous cyanobacterium Anabaena (Fig. 3a). The strain ET14, which contains the ethanol synthesis plasmid pET1 and expresses the invB gene from the nifB promoter in heterocysts, produced 1.7 g L−1 ethanol over 23 days (73 mg L−1 day−1) (Fig. 5). Ethanol-producing strains of the unicellular cyanobacterium Synechocystis PCC 6803 were constructed by introducing the same genes used in this study (Gao et al. 2012). One of the strains, Syn-HZ24, produced 5.5 g L−1 ethanol over 26 days (212 mg L−1 day−1). The ethanol yield of Syn-HZ24 was 2.9-fold higher than that of ET14. However, ET14 produces ethanol only in heterocysts, which account for approximately 10% of the total cells, indicating that ethanol production per heterocyst cell is approximately threefold higher than that of Syn-HZ24. The heterocyst-to-vegetative cell ratio increases with the inactivation of the patN gene (Risser et al. 2012), and an increased heterocyst frequency results in an increase in the total activity of heterocyst metabolism (Masukawa et al. 2017). Thus, ethanol productivity of ET14 could be improved by genetic modification that leads to an increased heterocyst frequency.
Metabolic engineering of heterocysts that resulted in an increased supply of pyruvate enhanced ethanol production (Figs. 3 and 4). Optimization of heterocyst metabolism to ethanol production would be effective for increasing the ethanol yield. Because heterocyst function is specialized for nitrogen fixation and their growth is ceased, heterocysts are amenable to metabolic engineering that cannot be applied to vegetative cells. Phosphoenolpyruvate carboxylase (PEPC) is essential for the vegetative growth of cyanobacteria (Luinenburg and Coleman 1990), but its elimination can redirect carbon metabolism toward pyruvate synthesis. Hence, exclusive PEPC downregulation in heterocysts is a future target of metabolic engineering for increasing ethanol production. The ethanol yield of ET14 drastically increased by aeration with air that contained 1% CO2 (Fig. 5). The maximum ethanol concentration produced by ET14 was < 0.3%, which is lower than the concentration inhibitory to the growth of Anabaena (Fig. 2). Thus, an increased supply of CO2 by aeration is likely to enhance ethanol production by activating carbon assimilation in vegetative cells. The overexpression of key enzymes of the Calvin-Benson cycle activates photosynthesis and biomass production in cyanobacteria (Liang and Lindblad 2016). Genetic manipulation that activates carbon assimilation in vegetative cells could further increase ethanol production. Genetic tools for gene regulation in Anabaena have been developed in recent years. The tight regulation of gene expression was accomplished by applying an inducible promoter with an antisense RNA, a riboswitch, and clustered regularly interspaced short palindromic repeats interference (CRISPRi) (Higo et al. 2016, 2017). These regulatory systems in combination with heterocyst or vegetative cell-specific promoters enable cell type-specific genetic manipulation.
Heterocysts provide the micro-oxic environment required for nitrogen fixation, which is also suitable for fermentation processes that are conducted under oxygen deprivation conditions. Ethanol production was accompanied with nitrogen fixation (Fig. 3), indicating that the micro-oxic environment within heterocysts was maintained during ethanol production. The micro-oxic intracellular environment and the active catabolism of sucrose may contribute to the higher ethanol productivity per heterocyst cell. In addition, heterocysts enable the use of oxygen-sensitive enzymes that are usually inactivated within cyanobacterial cells. For example, the oxygen-sensitive formate dehydrogenase was expressed in heterocysts, resulting in direct formate production from CO2 (Ihara et al. 2013). Thus, using heterocysts as a biocatalyst provides a new possibility for biofuel and chemical production with cyanobacteria.
References
Angermayr SA, Gorchs Rovira A, Hellingwerf KJ (2015) Metabolic engineering of cyanobacteria for the synthesis of commodity products. Trends Biotechnol 33(6):352–361. https://doi.org/10.1016/j.tibtech.2015.03.009
Arai S, Hayashihara K, Kanamoto Y, Shimizu K, Hirokawa Y, Hanai T, Murakami A, Honda H (2017) Alcohol-tolerant mutants of cyanobacterium Synechococcus elongatus PCC 7942 obtained by single-cell mutant screening system. Biotechnol Bioeng 114(8):1771–1778. https://doi.org/10.1002/bit.26307
Atsumi S, Higashide W, Liao JC (2009) Direct photosynthetic recycling of carbon dioxide to isobutyraldehyde. Nat Biotechnol 27(12):1177–1180. https://doi.org/10.1038/nbt.1586
Cumino AC, Marcozzi C, Barreiro R, Salerno GL (2007) Carbon cycling in Anabaena sp. PCC 7120. Sucrose synthesis in the heterocysts and possible role in nitrogen fixation. Plant Physiol 143(3):1385–1397. https://doi.org/10.1104/pp.106.091736
Dexter J, Fu P (2009) Metabolic engineering of cyanobacteria for ethanol production. Energy Environ Sci 2(8):857. https://doi.org/10.1039/b811937f
Ehira S, Ohmori M (2006) NrrA, a nitrogen-responsive response regulator facilitates heterocyst development in the cyanobacterium Anabaena sp. strain PCC 7120. Mol Microbiol 59(6):1692–1703. https://doi.org/10.1111/j.1365-2958.2006.05049.x
Ehira S, Ohmori M (2011) NrrA, a nitrogen-regulated response regulator protein, controls glycogen catabolism in the nitrogen-fixing cyanobacterium Anabaena sp. strain PCC 7120. J Biol Chem 286(44):38109–38114. https://doi.org/10.1074/jbc.M111.289124
Ehira S, Shimmori Y, Watanabe S, Kato H, Yoshikawa H, Ohmori M (2017) The nitrogen-regulated response regulator NrrA is a conserved regulator of glycogen catabolism in β-cyanobacteria. Microbiology. https://doi.org/10.1099/mic.0.000549
Elhai J, Wolk CP (1990) Developmental regulation and spatial pattern of expression of the structural genes for nitrogenase in the cyanobacterium Anabaena. EMBO J 9(10):3379–3388
Elhai J, Vepritskiy A, Muro-Pastor AM, Flores E, Wolk CP (1997) Reduction of conjugal transfer efficiency by three restriction activities of Anabaena sp. strain PCC 7120. J Bacteriol 179(6):1998–2005. https://doi.org/10.1128/jb.179.6.1998-2005.1997
Flores E, Herrero A (2010) Compartmentalized function through cell differentiation in filamentous cyanobacteria. Nat Rev Microbiol 8(1):39–50. https://doi.org/10.1038/nrmicro2242
Fujisawa T, Narikawa R, Maeda S, Watanabe S, Kanesaki Y, Kobayashi K, Nomata J, Hanaoka M, Watanabe M, Ehira S, Suzuki E, Awai K, Nakamura Y (2017) CyanoBase: a large-scale update on its 20th anniversary. Nucleic Acids Res 45(D1):D551–D554. https://doi.org/10.1093/nar/gkw1131
Gao Z, Zhao H, Li Z, Tan X, Lu X (2012) Photosynthetic production of ethanol from carbon dioxide in genetically engineered cyanobacteria. Energy Environ Sci 5(12):9857–9865. https://doi.org/10.1039/c2ee22675h
Golden J, Wiest D (1988) Genome rearrangement and nitrogen fixation in Anabaena blocked by inactivation of xisA gene. Science 242(4884):1421–1423
Happe T, Schütz K, Böhme H (2000) Transcriptional and mutational analysis of the uptake hydrogenase of the filamentous cyanobacterium Anabaena variabilis ATCC 29413. J Bacteriol 182(6):1624–1631. https://doi.org/10.1128/JB.182.6.1624-1631.2000
Higo A, Isu A, Fukaya Y, Hisabori T (2016) Efficient gene induction and endogenous gene repression systems for the filamentous cyanobacterium Anabaena sp. PCC 7120. Plant Cell Physiol 57(2):387–396. https://doi.org/10.1093/pcp/pcv202
Higo A, Isu A, Fukaya Y, Hisabori T (2017) Designing synthetic flexible gene regulation networks using RNA devices in cyanobacteria. ACS Synth Biol 6(1):55–61. https://doi.org/10.1021/acssynbio.6b00201
Holmqvist M, Stensjö K, Oliveira P, Lindberg P, Lindblad P (2009) Characterization of the hupSL promoter activity in Nostoc punctiforme ATCC 29133. BMC Microbiol 9(1):54. https://doi.org/10.1186/1471-2180-9-54
Ihara M, Kawano Y, Urano M, Okabe A (2013) Light driven CO2 fixation by using cyanobacterial photosystem I and NADPH-dependent formate dehydrogenase. PLoS One 8(8):e71581. https://doi.org/10.1371/journal.pone.0071581
Jüttner F (1983) 14C-labeled metabolites in heterocysts and vegetative cells of Anabaena cylindrica filaments and their presumptive function as transport vehicles of organic carbon and nitrogen. J Bacteriol 155(2):628–633
Kaneko T (2001) Complete genomic sequence of the filamentous nitrogen-fixing cyanobacterium Anabaena sp. strain PCC 7120. DNA Research 8(5):205–213
Kusakabe T, Tatsuke T, Tsuruno K, Hirokawa Y, Atsumi S, Liao JC, Hanai T (2013) Engineering a synthetic pathway in cyanobacteria for isopropanol production directly from carbon dioxide and light. Metab Eng 20:101–108. https://doi.org/10.1016/j.ymben.2013.09.007
Lan EI, Liao JC (2011) Metabolic engineering of cyanobacteria for 1-butanol production from carbon dioxide. Metab Eng 13(4):353–363. https://doi.org/10.1016/j.ymben.2011.04.004
Lan EI, Ro SY, Liao JC (2013) Oxygen-tolerant coenzyme A-acylating aldehyde dehydrogenase facilitates efficient photosynthetic n-butanol biosynthesis in cyanobacteria. Energy Environ Sci 6(9):2672–2681. https://doi.org/10.1039/C3EE41405A
Li H, Liao JC (2013) Engineering a cyanobacterium as the catalyst for the photosynthetic conversion of CO2 to 1,2-propanediol. Microb Cell Factories 12(1):4. https://doi.org/10.1186/1475-2859-12-4
Liang F, Lindblad P (2016) Effects of overexpressing photosynthetic carbon flux control enzymes in the cyanobacterium Synechocystis PCC 6803. Metab Eng 38:56–64. https://doi.org/10.1016/j.ymben.2016.06.005
Luinenburg I, Coleman JR (1990) A requirement for phosphoenolpyruvate carboxylase in the cyanobacterium Synechococcus PCC 7942. Arch Microbiol 154(5):471–474. https://doi.org/10.1007/BF00245230
Masukawa H, Sakurai H, Hausinger RP, Inoue K (2017) Increased heterocyst frequency by patN disruption in Anabaena leads to enhanced photobiological hydrogen production at high light intensity and high cell density. Appl Microbiol Biotechnol 101(5):2177–2188. https://doi.org/10.1007/s00253-016-8078-3
Nurnberg DJ, Mariscal V, Bornikoel J, Nieves-Morison M, Kraus N, Herrero A, Maldener I, Flores E, Mullineaux CW (2015) Intercellular diffusion of a fluorescent sucrose analog via the septal junctions in a filamentous cyanobacterium. MBio 6(2):1–12. https://doi.org/10.1128/mBio.02109-14
Oliver JWK, Machado IMP, Yoneda H, Atsumi S (2013) Cyanobacterial conversion of carbon dioxide to 2,3-butanediol. Proc Natl Acad Sci U S A 110(4):1249–1254. https://doi.org/10.1073/pnas.1213024110
Pernil R, Herrero A, Flores E (2010) Catabolic function of compartmentalized alanine dehydrogenase in the heterocyst-forming cyanobacterium Anabaena sp. strain PCC 7120. J Bacteriol 192(19):5165–5172. https://doi.org/10.1128/JB.00603-10
Risser DD, Wong FCY, Meeks JC (2012) Biased inheritance of the protein PatN frees vegetative cells to initiate patterned heterocyst differentiation. Proc Natl Acad Sci U S A 109(38):15342–15347. https://doi.org/10.1073/pnas.1207530109
Savakis PE, Angermayr SA, Hellingwerf KJ (2013) Synthesis of 2,3-butanediol by Synechocystis sp. PCC6803 via heterologous expression of a catabolic pathway from lactic acid- and enterobacteria. Metab Eng 20:121–130. https://doi.org/10.1016/j.ymben.2013.09.008
Shen CR, Liao JC (2012) Photosynthetic production of 2-methyl-1-butanol from CO2 in cyanobacterium Synechococcus elongatus PCC7942 and characterization of the native acetohydroxyacid synthase. Energy Environ Sci 5(11):9574–9583. https://doi.org/10.1039/c2ee23148d
Summers ML, Wallis JG, Campbell EL, Meeks JC (1995) Genetic evidence of a major role for glucose-6-phosphate dehydrogenase in nitrogen fixation and dark growth of the cyanobacterium Nostoc sp. strain ATCC 29133. J Bacteriol 177(21):6184–6194. https://doi.org/10.1128/jb.177.21.6184-6194.1995
Thiel T, Pratte B (2014) Regulation of three nitrogenase gene clusters in thec yanobacterium Anabaena variabilis ATCC 29413. Life 4(4):944–967. https://doi.org/10.3390/life4040944
Vidal R, López-Maury L, Guerrero MG, Florencio FJ (2009) Characterization of an alcohol dehydrogenase from the cyanobacterium Synechocystis sp. strain PCC 6803 that responds to environmental stress conditions via the Hik34-Rre1 two-component system. J Bacteriol 191(13):4383–4391. https://doi.org/10.1128/JB.00183-09
Yoon H, Golden JW (1998) Heterocyst pattern formation controlled by a diffusible peptide. Science 282(5390):935–938. https://doi.org/10.1126/science.282.5390.935
Zhang Y, Niu X, Shi M, Pei G, Zhang X, Chen L, Zhang W (2015) Identification of a transporter Slr0982 involved in ethanol tolerance in cyanobacterium Synechocystis sp. PCC 6803. Front Microbiol 6:487. https://doi.org/10.3389/fmicb.2015.00487
Acknowledgements
This work was supported in part by a Grant-in-aid for Young Scientists (B) 26870472 from the Japan Society for the Promotion of Science, the Precursory Research for Embryonic Science to Technology program from the Japan Science and Technology Agency, and by the Institute for Fermentation, Osaka.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical statement
This study does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ehira, S., Takeuchi, T. & Higo, A. Spatial separation of photosynthesis and ethanol production by cell type-specific metabolic engineering of filamentous cyanobacteria. Appl Microbiol Biotechnol 102, 1523–1531 (2018). https://doi.org/10.1007/s00253-017-8620-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8620-y