Abstract
Cellulose was treated with different extracellular microbial enzymes. The treatment of cellulose with the enzymes can improve alkaline solubility. Both endoglucanase and crude cellulase decreased the average degrees of polymerization (\(\overline{{DP}} \)) and improved the alkaline solubility of cellulose most efficiently. The composition of the enzyme, the type of cellulosic materials, pretreatment, and the treatment conditions are the key factors for its effective processing, using the enzymes to improve on alkaline solubility of cellulose. The improvement in the alkaline solubility is caused by the decrease in \(\overline{{DP}} \) and hydrogen bond because of enzymatic hydrolysis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cellulose is the most abundant and renewable biopolymer on Earth (Schurz 1999). It is a demand for the environmentally friendly processes of regenerated cellulose. Acid and high-temperature degradations are unsatisfactory because waste impurities in cellulosic materials generate unwanted byproducts under the harsh conditions. Due to the potential benefits in cellulosic materials, the enzymatic modification of cellulose is a challenge in their applications. (Amano and Kanda 2002; Bhat 2000) Currently, the most widely exploited cellulase applications are in the textile industry, for example, stonewashing of jeans and finishing of cotton fabrics (Cortez et al. 2001; Pere et al. 2001). In the pulp and paper industry, cellulases and hemicellulases are used to improve the running ability of paper machines and enhance the deinking of recycled fibers (van Wyk and Mohulatsi 2003; Bhat 2000; Subramaniyan and Prema 2002).
Extracellular microbial enzymes have the potential to be powerful tools for modifying cellulose (Rabinovich et al. 2002; Schulein 2000; Bhat 2000). Cellulases are the enzymes which hydrolyze the β-1,4 linkages in cellulose. A cellulosic enzyme system consists of three major components: endo-β-glucanase (EC 3.2.1.4), exo-β-glucanase (EC 3.2.1.91), and β-glucosidase (EC 3.2.1.21). The first two enzymes act directly on cellulose, generating mainly cellobiose and glucose as reaction products. The cellobiase hydrolyze cellobiose to glucose. Endoglucanases and cellobiohydrolases degrade soluble cellodextrins and amorphous cellulose. However, it is only the cellobiohydrolases that degrade crystalline cellulose efficiently.
To date, many studies on the action of cellulases or purified cellulases on the cellulose revealed that the enzymes degrade cellulose (Gan et al. 2003; Bhat 2000). Thus, the effect of extracellular microbial enzymes on properties of cellulose need further study. The aim of the present study was to investigate if the treatment of cellulosic materials with different enzymes would improve the alkaline solubility or be highly reactive of them.
Materials and methods
Dissolving pulp
The dissolving pulps were obtained from eucalyptus (hardwood) in Brazil and fir (softwood) in Canada. The linter pulp was obtained from Baoding Swan Co., Ltd. in China. The characteristics of the materials are presented in Table 1. Prior to enzymatic treatments, the materials were disintegrated in an apparatus at respective consistency and agitated with 3,000 rpm for about 1 h.
Enzymes
Crude cellulase was derived from Humicola insolens. The monocomponent endoglucanase was prepared and purified from a genetically modified Aspergillus microorganism. The monocomponent cellobiohydrolase was prepared and purified from H.insolens. Xylanase prepared from a modified Bacillus sp. Mannase was purified from Trichoderma reesei.
The activities of enzymes are shown as international units (IU), in which one unit of activity is defined as the amount of enzyme required to liberate 1 μmol of product per minute.
The endoglucanase (CMCase), xylanase, mannase, and filter paper activities were measured on carboxymethylcellulose (1% CMC, Sigma), xylan (1% birthwood xylan, Sigma), galactomannan (1% local bean, Sigma), and filter paper (No. 1 Whatman), respectively, using methods described previously (Sharrock 1988; Wood and Bhat 1988).
The amount of reducing sugar release was estimated by the dinitrosalicylic acid method, with glucose as a standard. (Wood and Bhat 1988)
Enzymatic treatment of pulp
The enzyme concentration added to the pulp treatments was based on the mass of oven-dry pulp. All enzyme treatments were incubated at 50°C under continuous agitation at 175 rpm and at respective pulp consistency in 50 mM phosphate buffer (pH 7.0), but mannase treatment was in acetic acid and sodium acetate buffer (pH 5.0). Boiling for 15 min to inactivate the enzyme terminated reactions. Control pulp treatments were run in parallel under similar conditions, except that no enzyme was added.
Analyses of pulp
The viscosity and characteristics of the pulp were measured according to the related methods of the International Standards Organization (ISO)/TC6 (5351/1, 1762,535). The carbohydrate composition of the samples was analyzed by high-performance liquid chromatography (HPLC) after acid hydrolysis (Olkkonen et al. 2000).
Determination of the degrees of polymerization (\(\overline{{DP}} \))
The degrees of polymerization were measured viscosimetrically in CuEn (copper ethylene diamine solution), and the obtained intrinsic viscosities were converted into the respective values of \(\overline{{DP}} \) in the following equation (Brown and Wikstrom, 1965):
.
Alkaline solubility of the pulp
The solubility of the reference and enzyme-treated pulps was determined by a modification of the method. The pulps were dissolved under vigorous stirring (1,500 rpm) in 9% (wt.) sodium hydroxide at −5°C at 5% pulp consistency for 3 h. Thereafter, the solutions were centrifuged at 1,000×g for 10 min. The dissolved part was discarded, and the undissolved part was washed with 9% sodium hydroxide and centrifuged at 1,000×g for 10 min. The washed cellulose was treated with 3% hydrochloric acid and then washed with deionized water in ceramic sinter. The solubility (S) of cellulose was calculated from the following equation (Cao and Tan 2002):
-
S=(W−WU)/W×100%, where
-
W is the weight [dry weight (dw)] of enzymatically treated or control cellulose sample, and
-
WU is the weight (dw) of undissolved cellulose.
Determination of the degree of crystalline of cellulose
The crystalline structure of the cellulose component, which was obtained under both control and enzymatic treatments, was measured by X-ray diffraction. After freeze-drying, two slices of pulp were prepared by pressing from every sample and tested for reproducibility. Samples were scanned and recorded using the D/max-2400 X-ray diffractometer (RIGAKU) with a D-5000 rotating anode X-ray generator from10 to 40 of 2θ (scattering angle), using Cu/Kα radiation generated at 30 mA and 40 kV.
The crystalline indices of cellulose samples were calculated from the X-ray diffraction patterns by the following equation (Rogovina et al. 1989):
, where
-
I 002 is the peak intensity from the (002) lattice plane (2θ=22.6°), and
-
I am is the peak intensity of amorphous phases (2θ=19.0°).
Apparent crystallite size (ACS) was estimated through use of the Scherrer equation (Yen and Mo 2001):
, where
-
is the wavelength of the incident X-ray (1.5418Å),
-
is the Bragg angle corresponding to the (002) plane, and
-
is the half-height width of the peak angle of the 002 reflection.
Fourier transform infrared spectra spectroscopy
Fourier transform infrared spectra (FTIR) were obtained with a PerkinElmer 2000 FTIR system spectrometer. The wave number range scanned was 4,000–350 cm−1. After milling, the powder of the samples was compacted into KBr disks and analyzed.
Determination of cellulose accessibility
In the heterogeneous phase, the chemical reactions are influenced by the accessibility of the reagents to cellulose. The IR spectroscopy method was used for the determination of cellulose accessibility (Kamide 2001):
, where
-
I 1,429 is the intensity of the band at 1,429(cm−1 (CH2 scissoring), and
-
I 893 is the intensity of the band at 893(cm−1 (asymmetric out-of-phase ring stretching).
Result and discussion
Enzymes activities
Before evaluating the enzymes for their potential to modify the pulp properties, the enzymes were assessed for their ability to hydrolyze standard substrates (Table 2). Crude cellulase contains high levels of xylanase activity. The endoglucanase also contains low, but significant, levels of xylanase activity. The mannase and xylanase contain very low levels of CMCase activity.
Effects of materials on the \(\overline{{DP}} \) and alkaline solubility of the pulps
The changes of the viscosity average degree of polymerization (\(\overline{{DP}} \)) and alkaline solubility of the samples are shown in Table 3. After treatment with cellulase and endoglucanase, the \(\overline{{DP}} \) of the hardwood pulp and the softwood pulp decrease rapidly, and alkaline solubility increased observably. The \(\overline{{DP}} \) values of linter show a slight decrease, but an insignificant change in alkaline solubility is observed. The treatments with endoglucanase bring out a lower \(\overline{{DP}} \) and a higher alkaline solubility than the treatment with cellulase for all samples. The results indicate that wood pulp is effectively treated by enzymes in the view of the changes in the \(\overline{{DP}} \) and alkaline solubility.
Effects of enzymes on the \(\overline{{DP}} \) and alkaline solubility of the pulp
Both endoglucanase and crude cellulase decrease the \(\overline{{DP}} \) of cellulose and improve the alkaline solubility, even at low enzyme dosages, as seen in Table 4. Cellobiohydrolase has a less pronounced effect on the \(\overline{{DP}} \) and alkaline solubility. The observed changes in the \(\overline{{DP}} \) and alkaline solubility were in keeping with the specific activities of CMCase of endoglucanase and cellobiohydrolase. Table 4 shows the \(\overline{{DP}} \) decrease with the increase in enzyme dosage. The improvement of alkaline solubility accords with the decrease in the \(\overline{{DP}} \) of cellulose.
However, as was expected, the \(\overline{{DP}} \) of the samples are not affected by the xylanase and mannase treatments. The alkaline solubilities of the pulps are also unaffected in the xylanase and mannase treatments from Table 4. This is in accordance with the results of many studies (Rahkamo et al. 1998a,b; Rahkamo et al. 1996).
Effects of enzyme hydrolysis on the \(\overline{{DP}} \) and alkaline solubility of the pulp
Figure 1 shows the decrease in the \(\overline{{DP}} \) of cellulose treated with cellulase, endoglucanase, and cellobiohydrolase. Every curve has a different characteristic. In the experiments with cellobiohydrolase, the decrease in \(\overline{{DP}} \) is limited to 15%. In the case of endoglucanase, cellulose was hydrolyzed strongly at the beginning, with a slow up to some degree in the hydrolysis as time increases. In the treatment with cellulase, the decrease in \(\overline{{DP}} \) is moderate in the beginning of the degradation of cellulose and afterwards become lower.
The changes of cellulose under the enzymatic treatment were caused by the mechanism of its hydrolysis. Cellobiohydrolase does degrade cellulose by an endwise one, therefore causing only a slight diminution of \(\overline{{DP}} \). On the contrary, endoglucanase cleaves the cellulose chains statistically, provoking a relatively strong degradation. The crude cellulase is a multicomponent enzyme and comprises the two enzymes, which act synergistically.
After enzymatic treatment, the alkaline solubility of the samples was investigated. As seen in Fig. 2, endoglucanase is found to be the most efficient in improving the alkaline solubility among the enzymes. Endoglucanase improves strongly the alkaline solubility just in the beginning of the treatment, and the improvement of the alkaline solubility stops in some degree of hydrolysis. However, cellobiohydrolase has a less pronounced effect on the alkaline solubility. It is found that the alkaline solubility of the samples increase rapidly with the addition of the treatment time within 5 h, and the enzymatic treatment time has a little effect on the alkaline solubility over 6 h.
Effect of pretreatment before enzymatic treatment on the \(\overline{{DP}} \) and alkaline solubility of the pulp
Before enzymatic treatment, the pulp was disintegrated under vigorous stirring (3,000 rpm) for 1 h. The necessity of the pretreatment of pulp before enzymatic treatment is shown in the results presented in Table 5. Some additives for improving the mechanical pretreatment of the pulp were also used (Table 5). It was found that alkaline solubility of the dissolving pulp was enhanced, while the viscosity was similar by adding poly(ethoxy alkylamine). As a nonionic surfactant, it contains many polar groups and is water-soluble. The poly(ethoxy alkylamine) can permeate pulp and activate cellulose. Pretreatment of pulp should be one of the key factors for alkaline solubility of the cellulose, and the function of poly(ethoxy alkylamine) is to reduce surface tension, improve cellulose surface spreading and sticking, and enhance alkaline solubility.
It is known that there are three processes before the enzymatic hydrolysis of cellulose: the diffusion of enzyme in the liquid, the transference of enzyme from liquid to the surface of substrate, and the enzymatic absorption of cellulose and the form of the enzyme–cellulose complex. The sample, without disintegration, hinders the transference of enzyme so that the enzyme does not sufficiently touch the substrate. Finally, it affects the hydrolysis of cellulose.
Effect of enzymatic treatment on the crystal structures of cellulose
Figure 3 shows typical X-ray diffraction intensity profiles of the original and the hydrolyzed cellulose samples. For softwood cellulose by enzymatic treatments with cellulase and endoglucanase, the peak for the plane (002) (2θ=22.5°) in the intensity profiles of all the treated cellulose becomes sharper than that of the original samples, and the shoulder of the peak around 2θ=20° has a tendency to increase in intensity. It shows that the enzymes preferentially attack the crystal planes (002) for cellulose I in Fig. 3. These results indicate that the number of small crystallites decreases during enzymatic hydrolysis. The separation of the two peaks observed at 2θ=14.7° and 16.3° [(10Ī) plane] becomes better for softwood cellulose by enzymatic treatments with cellulase and endoglucanase. It is found that the crystallinity of samples was not affected by the xylanase or mannase treatment.
Effects of the different enzymatic treatments on cellulose (softwood) crystallinity [The enzymatic treatments of cellulose were performed at 4% substrate consistency at 50°C for 4 h, with dosage of the enzymes at 2.5 mg/g pulp (dw)]. a indicates xylanase; b, mannase; c, cellulase; d, endoglucanase; and e, control
The crystallinity index and the ACS of cellulose increased during hydrolysis in Table 6. The increase in the crystallinity index and that in ACS of cellulose are good evidence that the amorphous portion of the cellulose was more readily and quickly hydrolyzed than the crystalline portion. The crystallinity of cellulose was not affected by the xylanase or mannase treatments, but cellulase has the most pronounced effect on the crystallinity of cellulose.
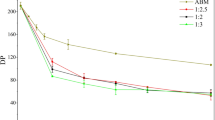
Comparing with the ball-milling method, which can transform the crystalline of cellulose (Ago et al. 2004; Stubicar et al. 1998), both endoglucanase and cellulase decrease the \(\overline{{DP}} \) of cellulose and increase the crystallinity of partially crystalline cellulose.
Effect of enzymatic treatment on the molecular structures of cellulose
All the FTIR spectra in Fig. 4 indicate that enzymatic treatments have some effects on the structure of cellulose. The FTIR spectra show characteristic cellulose peaks around 1,000–1,200 cm−1. The band near 1,160 cm−1 is representative of the antisymmetric bridge stretching of C–O–C groups in cellulose and hemicellulose, and the band near 1,318 cm−1 could be ascribed to CH2-wagging vibrations in cellulose and hemicellulose. The 893-cm−1 band, which is characteristic for anomere C-group deformation and ring valence vibration, especially in hemicelluloses (Baeza and Freer 2001), was reduced after enzyme treatment. There was also a substantial reduction in the band at 1,635–1,640 cm−1, which has been attributed to C=O groups.
The bands near 3,400 cm−1 are representative of OH vibrations. The band of OH vibrations is wide and moves to a lower wavenumber (3,370 cm−1) because of intermolecular and intramolecular hydrogen bonds. After the enzymatic hydrolysis, the bands near 3,400 cm−1 became narrow and moved to a high wavenumber (3,400 cm−1). There is good evidence that a portion of the hydrogen bonds are broken in the enzymatic hydrolysis. Such an effect leads to an improvement on the alkaline solubility of the pulp while the crystalline index increases. However, there are no varieties in the treatment with xylanase and mannase in Fig. 4.
In Table 7, the cellulose accessibility increases in the enzymatic hydrolysis with crude cellulase and endoglucanase. The improvement of alkaline solubility accords with the increase in the accessibility of cellulose. However, the accessibility of the samples is not affected by the xylanase and mannase treatments. It shows that crude cellulase and endoglucanase treatments improve the chemical activity of cellulose because enzymatic hydrolysis decreases the DP of cellulose and breaks a portion of the hydrogen bonds in cellulose.
Effects of structural properties of cellulose on alkaline solubility
It shows that there are no obvious differences in carbohydrate compositions of the original sample and the dissolved part of samples from Table 8. It found the correlation between the decrease in \(\overline{{DP}} \) and the alkaline solubility in Fig. 5. At least, in part corresponding to such a \(\overline{{DP}} \) decrease, alkaline solubility increases from about 20 to over 90%. A portion of the Hydrogen bonds was broken in the enzymatic hydrolysis. It is one of the key factors for improvement on the alkaline solubility of the pulp. In terms of the old and plausible concept (Elias 1984; Yen and Mo 2001), the solubility or swelling ability of polymer is controlled by the crystallinity. But it does not clarify an improvement on the alkaline solubility of the pulp while the crystalline index is increasing (Table 6). Thus, the improvement in the alkaline solubility is caused by the decrease in DP and hydrogen bond because of enzymatic hydrolysis.
Conclusions
The enzymes were assayed and used to modify the cellulose materials. We have examined the effects of enzymatic hydrolysis on the viscosity average degree of polymerization \(\overline{{DP}} \), solubility towards aqueous alkali solution, the crystalline, and the variety of hydrogen bonds analyzed by X-ray and FTIR spectra. The following conclusions can be summarized briefly from this study: (1) The softwood pulp is more effectively treated by enzymes in view of the changes in the \(\overline{{DP}} \) and alkaline solubility. (2) Both endoglucanase and cellulase decrease the \(\overline{{DP}} \) of cellulose and improve the alkaline solubility of cellulose at low-enzyme dosages, but cellobiohydrolase has a less pronounced effect on the \(\overline{{DP}} \) and the alkaline solubility. Xylanase and mannase have no effect on the \(\overline{{DP}} \) and the alkaline solubility. (3) The crystallinity of partially crystalline cellulose increases, and the number of small crystallites decreases during the hydrolysis with endoglucanase and cellulase, using X-ray analysis. (4) Hydrogen bonds of OH groups were partly broken by the treatment with enzyme. (5) The cellulose accessibility increases in the enzymatic hydrolysis with crude cellulase and endoglucanase. (6) The improvement in the alkaline solubility is caused by the decrease in \(\overline{{DP}} \) and hydrogen bond because of enzymatic hydrolysis.
References
Ago M, Endo T, Hirotsu T (2004) Crystalline transformation of native cellulose from cellulose I to cellulose II polymorph by a ball-milling method with a specific amount of water. Cellulose 11:163–167
Amano Y, Kanda T (2002) New insights into cellulose degradation by cellulases and related enzymes. Trends Glycosci Glycotechnol 14:27–34
Baeza J, Freer J (2001) Chemical characterization of wood and its components. In: Hon DNS, Shiraishi N (eds) Wood and cellulosic chemistry. Dekker, New York, pp 275–383
Bhat MK (2000) Cellulases and related enzymes in biotechnology. Biotechnol Adv 18:355–383
Brown W, Wikstrom R (1965) A viscosity–molecular weight relationship for cellulose in cadoxen and a hydrodynamic interpretation. Eur Polym J 1:1–10
Cao Y, Tan H (2002) The properties of enzyme-hydrolyzed cellulose in aqueous sodium hydroxide. Carbohydr Res 337:1453–1457
Cortez JM, Ellis J, Bishop DP (2001) Cellulase finishing of woven, cotton fabrics in jet and winch machines. J Biotechnol 89:239–245
Elias H-G (1984) Part I, structure and properties. In: Macromolecules. Plenum, New York
Gan Q, Allen SJ, Taylor G (2003) Kinetic dynamics in heterogeneous enzymatic hydrolysis of cellulose: an overview, an experimental study and mathematical modelling. Process Biochem 38:1003–1018
Kamide K (2001) Characterization of chemicaly modified cellulose. In: Hon DNS, Shiraishi N (eds) Wood and cellulosic chemistry. Dekker, New York, pp 621–663
Olkkonen C et al (2000) Degradation of model compounds for cellulose and ligno-cellulosic pulp during ozonation in aqueous solution. Holzforschung 54:397–406
Pere J, Puolakka A, Nousiainen P, Buchert J (2001) Action of purified Trichoderma reesei cellulases on cotton fibers and yarn. J Biotechnol 89:247–255
Rabinovich ML, Melnik MS, Boloboba AV (2002) Microbial cellulases (Review). Appl Biochem Microbiol 38:305–321
Rahkamo L et al (1996) Modification of hardwood dissolving pulp with purified Trichoderma reesei cellulases. Cellulose 3:153–163
Rahkamo L, Siika-aho M, Viikari L, Leppanen T, Buchert J (1998a) Effects of cellulases and hemicellulase on the alkaline solubility of dissolving pulps. Holzforschung 52:630–634
Rahkamo L, Viikari L, Buchert J, Paakkari T, Suortti T (1998b) Enzymatic and alkaline treatments of hardwood dissolving pulp. Cellulose 5:79–88
Rogovina SZ, Zhorin VA, Shashkin DP, Yenikolopyan NS (1989) X-ray diffraction study of cellulose after plastic flow under pressure. Polym Sci USSR 31:1376–1380
Schulein M (2000) Protein engineering of cellulases. Biochim Biophys Acta, Prot Struct Mol Enzymol 1543:239–252
Schurz J (1999) A bright future for cellulose. Prog Polym Sci 24:481–483
Sharrock KR (1988) Cellulase assay methods: a review. J Biochem Biophys Methods 17:81–105
Stubicar N et al (1998) An X-ray diffraction study of the crystalline to amorphous phase change in cellulose during high-energy dry ball milling. Holzforschung 52:455–458
Subramaniyan S, Prema P (2002) Biotechnology of microbial xylanases: enzymology, molecular biology, and application. Crit Rev Biotechnol 22:33–64
van Wyk JPH, Mohulatsi M (2003) Biodegradation of wastepaper by cellulase from Trichoderma viride. Bioresour Technol 86:21–23
Wood TM, Bhat GR (1988) Measurement of cellulase activity. Methods Enzymol 160:87–116
Yen J, Mo Z (2001) Modern polymer physics. Science, Beijing
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Cao, Y., Tan, H. Improvement of alkali solubility of cellulose with enzymatic treatment. Appl Microbiol Biotechnol 70, 176–182 (2006). https://doi.org/10.1007/s00253-005-0069-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-005-0069-8