Abstract
Immunoglobulin light (IGL) chain genes encoding σ and λ from channel catfish, Ictalurus punctatus, and λ from Atlantic cod, Gadus morhua, were identified by mining of expressed sequence tag databases, 5′-RACE and RT-PCR protocols. cDNAs for each of these IGL chains encode typical variable (V), joining (J), and constant (C) regions and Southern blot analyses and genomic sequencing show that genes encoding these isotypes, like other teleost IGL genes, are found in a cluster organization of one or two V gene segments, followed by single J and C gene segments, all in the same transcriptional orientation. However, unlike the teleost κ genes, genes encoding catfish σ and λ are few in number and the two isotypes are each encoded by only two clusters. Similarly, Atlantic cod λ genes are predicted to be encoded by two or three clusters. As expected, sequence and phylogenetic analyses comparisons demonstrate that catfish Vσ and Cσ genes are most similar to Vσ and Cσ genes of other ectothermic vertebrates. Although catfish and Atlantic cod Vλ genes cluster with other vertebrate Vλ genes, their Cλ sequences cluster in a distinct group separate from other vertebrate IGL C sequences. However, support for classifying these sequences as λ, is their V and J recombination signal sequence (RSS) organization. The catfish and Atlantic cod genes have typical λ-like RSS with the Vλ RSS consisting of heptamer-23 bp spacer-nonamer and the Jλ RSS consisting of heptamer-12 bp spacer-nonamer. This is the first report demonstrating the presence of Igλ in teleosts.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
All studied jawed vertebrate species, except for birds, as represented by the galliforms (chicken, Gallus gallus) and anseriforms (ducks, Anas platyrhynchos, Cairina moschata), express more than one immunoglobulin light (IGL) chain isotype (Magor et al. 1994; McCormack et al. 1989; Reynaud et al. 1983, 1987; Lundqvist et al. 2006). The two mammalian IgL isotypes are classically referred to as κ and λ and were initially identified by serology approximately 40 years ago (Edelman and Gally 1962; Wu and Kabat 1970). As IGL chain isotypes were initially identified in different vertebrates there were attempts to classify them as κ- or λ-like. For example, based on sequence and recombination signal sequence (RSS) orientation, the IGL of birds was found to be related to mammalian Igλ (Gadol et al. 1976; Kubo et al. 1970) and Igκ isotypes were found in elasmobranchs and bony fish (Greenberg et al. 1993, and reviewed in Bengtén et al. 2000; Criscitiello and Flajnik 2007). However, it is now clear that many ectothermic vertebrates express more than two types of IGL chains and due to difficulties with resolving their phylogenetic relationships several nomenclatures exist in parallel. Three IGL isotypes, κ (originally termed ρ), σ and λ, have been identified in the amphibian Xenopus (Haire et al. 1996; Schwager et al. 1991; Zezza et al. 1992), and in the elasmobranchs, four IGL isotypes have been described. Type I or sigma-cart is unique to cartilaginous fish (Criscitiello and Flajnik 2007; Fleurant et al. 2004). Type II is considered λ-like, and type III, is considered a κ-type (Hohman et al. 1992; Rast et al. 1994). More recently, a fourth IGL isotype was identified in nurse shark, Ginglymostoma cirratum, and found to be orthologous to the Xenopus Igσ isotype (Criscitiello and Flajnik 2007). In several species of teleost, three IGL isotypes have been identified, type L1, type L2, and type L3 (Coscia et al. 2008; Daggfeldt et al. 1993; Haire et al. 2000; Partula et al. 1996; Saha et al. 2004 and reviewed in Solem and Stenvik 2006). While teleost L1 and L3 (including channel catfish, Ictalurus punctatus, isotypes F and G; Ghaffari and Lobb 1993, 1997) clearly represent distinct isotypes with amino acid identities of ~30% to each other, phylogenetic analyses demonstrated them to be orthologs of κ light chains (Criscitiello and Flajnik 2007). In contrast, teleost L2 is orthologous to the σ isotype. Teleost Igκ and Igσ genes are organized in clusters of VL-JL-CL and for Igκ, the Vκ genes are in opposite transcriptional orientation to the Jκ and Cκ genes, while the Vσ, Jσ, and Cσ genes are all in the same transcriptional orientation. Notably, this opposite orientation for Vκ implies rearrangement occurs by inversion rather than deletion. Another difference between teleost Igκ and Igσ isotypes is that Igσ genes tend to be fewer in number than Igκ genes in the different fish examined. For example, in the rainbow trout, Onchorynchus mykiss, the Igκ locus is estimated to contain ~20 gene clusters (Daggfeldt et al. 1993) and the Igσ locus is estimated to consist of at least ten gene clusters (Timmusk et al. 2000). Thus, in teleost, the IGL chain genes of the κ linage have undergone extensive duplications and diversifications (Hsu and Criscitiello 2006) and are also the most abundantly expressed in peripheral blood leukocytes (PBL). It was estimated that in catfish, the F and G isotypes together represents 90% of the IGL (Lobb and Clem 1982; Lobb et al. 1984). Similarly, the ratio of type L1 to type L2 expression in rainbow trout PBL is estimated to be 85:15 (Timmusk et al. 2000). Recently, an Igσ sequence from blue catfish, I. furcatus, was found (Hsu and Criscitiello 2006) and annotated (Criscitiello and Flajnik 2007) and it was suggested that while primordial Igκ, Igλ, and Igσ light chain isotypes are all present in cartilaginous fish, some of these light chain lineages have been lost in different vertebrate groups. For example, Igλ appears to be present in all groups of jawed vertebrates with the exception of teleost fish (Criscitiello and Flajnik 2007), while Igκ appears to be lost among birds and microbats (microchiropterans) (Das et al. 2008) and similarly, Igσ was suggested to be present in all cold-blooded vertebrates but absent among mammals and birds (Criscitiello and Flajnik 2007). However, a caveat is that although Igκ and Igλ have been identified in the green lizard (Anolis carolinensis) thus far, Igσ has not (Das et al. 2008). Here, we describe the identification, genomic organization, expression and phylogenetic relationship of Igσ and Igλ in the channel catfish and an Igλ in the Atlantic cod, Gadus morhua. Genes encoding each of these IGL chains are arranged in clusters consisting of one or two V, a single J and C, all in the same transcriptional orientation. However, unlike the catfish F and G isotypes, catfish Igσ, and Igλ isotypes are encoded by only one or two genomic clusters. Similarly, Atlantic cod Igλ appears to be encoded by one to three genomic clusters.
Materials and methods
Experimental animals
Channel catfish (1–2 kg) were obtained from a commercial source (ConAgra, Isola, MS) and maintained in individual tanks as previously described (van Ginkel et al. 1992). Catfish PBL were isolated from heparinized blood by centrifugation on a cushion of Ficoll-Hypaque (Lymphoprep, Accurate Chemical Corp. Westbury, NY) as described previously (Miller et al. 1994). Atlantic cod were caught by netting in the fjord Gullmaren on the Swedish west coast and spleen, pronephros (head kidney), ovary and testis were immediately snap frozen in liquid nitrogen and stored at −70°C until used.
Identification of catfish Igσ and Igλ and Atlantic cod Igλ cDNAs
A catfish Igσ sequence was obtained by RT-PCR using primers (supplementary Table 1) based on a blue catfish Igσ chain (accession number CK403931; (Hsu and Criscitiello 2006)). Using the IgT/Z C domains of rainbow trout and zebrafish (Danilova et al. 2005; Hansen et al. 2005) as queries to search catfish EST databases, a catfish Igλ-like EST (accession number CBPO4528.b1) consisting of an unrearranged V-intron-J-C was identified from an LPS-stimulated PBL cDNA library. This EST was one of four that encoded an Igλ-like sequence. The cDNA library designated CBPO (JxLPS1) was made as part of the Joint Genome Community Sequencing (JGI) program 2006: I punctatus, I furcatus EST project (http://www.jgi.doe.gov/sequencing/). Full-length transcripts for both catfish isotypes were subsequently obtained using 5′ and 3′-RACE protocols. An Atlantic cod Igλ fragment was obtained by using short degenerate primers corresponding to conserved regions found in vertebrate TRBV and IGL V framework (FR) 2 and FR3 regions. A larger Atlantic cod cDNA fragment was obtained by anchored PCR from an Atlantic cod amplified spleen λgt-10 cDNA library using a gene-specific forward primer (GmVLF) and a reverse λgt-10 phage primer (see supplementary Table 1). This spleen cDNA library was prepared from poly(A)-selected mRNA using a Promega cDNA Synthesis Kit (Promega, Madison, WI) and consisted of ~10.9 × 106 independent recombinants before amplification. Full-length and larger unrearranged Atlantic cod Igλ cDNAs were then obtained by screening the spleen cDNA library using standardized methods. All cDNAs were sequenced on both strands using forward and reverse gene-specific primers (supplementary Table 1). The Atlantic cod Igσ (ES474588, EX732557, EX727050, EX727984, EY975775), rainbow trout Igλ (BX861772, CA257088), blue catfish Igλ (CK403484) and green anole Igλ (FG746006) and green anole Igκ (FG748778) sequences were identified by Basic Local Alignment Search Tool (BLAST; Altschul et al. 1990) against the National Center for Biotechnology Information (NCBI; www.ncbi.nlm.nih.gov) databases.
Sequence analysis
Nucleotide and amino acid sequences were analyzed using DNASTAR software (Madison, WI) and similarity searches were performed using BLAST analysis against the NCBI non-redundant database. Immunoglobulin domains and signal peptides were predicted using SOSUI (signal) Beta Version, SMART (http://smart.embl-heidelberg.de/) and CBS Prediction servers (http://www.cbs.dtu.dk/services). The IMGT/DomainGapAlign TOOL (http://imgt.cines.fr) or when possible IMGT/V-QUEST (Lefranc et al. 2005) were used to determine complementarity determining regions (CDR), framework (FR) and strand delimitations. Pairwise alignments were made using CLUSTALW (Thompson et al. 1997) in the MegAlign module of DNASTAR. Neighbor-joining trees with pairwise gap deletions were constructed based on the CLUSTALW alignments using MEGA 4.0 allowing bootstrap values of 1,000.
Southern blotting and genomic PCR
Genomic DNA was prepared using erythrocytes from outbred and homozygous gynogenetic catfish as described previously (Hogan et al. 1999; Miller et al. 1988). Catfish Southern blots were prepared with 5 μg genomic DNA that was digested to completion with EcoR I, Xba I or Pst I restriction enzymes, separated on 1% agarose gels and transferred by capillary action onto Hybond-N+ membranes (GE Healthcare Bio-Sciences AB, Piscataway, NJ) using standard techniques. Hybridizations were performed in Rapid-hyb buffer (GE Healthcare Bio-Sciences AB, Piscataway, NJ) at 65°C according to manufacturer's instructions and membranes were washed at high stringency (65°C with 0.1× SSC, 0.1% SDS). The catfish V probes encompassed the FR1-FR3 region of either Igσ or Igλ and the C probes consisted of the entire C Ig domain through the stop codon of either Igσ or Igλ (see supplementary Table 1). Probes were amplified by PCR using IDPol DNA polymerase (ID Labs Biotechnology, London, Ontario) according to the manufacturer's protocol. Parameters were: 3 min 94°C, followed by 30 cycles of 94°C 30 s, 60°C 30 s, 72°C 1 min, then extension at 72°C for 10 min. The probes were random primed labeled with [32P] dCTP by Megaprime labeling (GE Healthcare Bio-Sciences AB).
Atlantic cod Southern blots were prepared with 10 μg of sperm genomic DNA digested to completion with BamHI, EcoR I or Hind III. The digested DNA was electrophoresed on 0.8% agarose gels and transferred to Biodyne B nylon filters (Pall Corporation, East Hills, NY) following the manufacturer's instructions. The filters were prehybridized and hybridized in a buffer modified from Church and Gilbert (1984) at 65°C overnight and washed at high stringency conditions. Atlantic cod V and C probes were generated by digesting an Atlantic cod Igλ cDNA (#S1-41) with restriction enzymes to obtain V and C specific fragments. These fragments were then recovered on glass wool (Heery et al. 1990) and labeled with [32P] dCTP using the Megaprime Labeling System (GE Healthcare Bio-Sciences AB).
IGL gene clusters were amplified from genomic DNA from a homozygous gynogenetic catfish (G25) and a total of 66 ng of DNA was used in each reaction. Amplification was performed with HotStart-IT FideliTAq DNA polymerase (USB Corporation, Cleveland OH) and parameters were: 2 min 94°C followed by 30 cycles of 94°C 30 s, 61°C 30 s 72°C 2 min followed by a final extension of 72°C 10 min (see Supplementary Table 1 for primers used). The resulting PCR products ranging from 2–2.4 kb were separated on 0.8% agarose gels, cloned into pCR4-TOPO (Invitrogen Life Technologies, Carlsbad, CA) and sequenced on both strands using vector- and gene-specific primers. Sequence analyses were performed with the DNASTAR software (Madison, WI) as described above.
Monoclonal antibodies, PBL isolation, magnetic activated cell sorting and flow cytometry
Hybridoma culture supernatant was used directly as the monoclonal antibody (mAb) source and all supernatants contained equivalent Ig concentrations as determined by ELISA (Southern Biotechnology, Birmingham, AL) and Western blot: mouse anti-catfish IgM 9E1 (IgG1,κ) reacts with catfish Igμ chain, (Miller et al. 1987); mouse anti-catfish IgL 3F12 (IgG1, κ) reacts with the IGL F isotype (Lobb and Clem 1982; Lobb et al. 1984); mouse anti-catfish IgL 11A2 (IgG2b, κ) reacts with IGL G isotype (Shen et al. 2003); and mouse anti-trout IgM 1.14 (IgG1, κ) reacts with trout Igμ chains (DeLuca et al. 1983), which was used as an isotype control.
Catfish PBL were isolated from heparinized blood by centrifugation as described above and resuspended to 1 × 107 cells/ml in ice cold RPMI-1640 adjusted to catfish tonicity with 10% deonized water (cfRPMI) containing 0.03% sodium azide. To obtain specific cell populations, PBL were sorted into surface IgM enriched (sIgM+) and surface IgM depleted (sIgM−) fractions by magnetic activated cell sorting (MACS) with anti-catfish IgM 9E1 mAb according to the manufacturer's protocol. Briefly, 2 × 108 cells were incubated with 1 ml mAb 9E1 for 30 min on ice. After washing with cfRPMI, cells were then resuspended in 300 μl of degassed cfRPMI containing 0.5% BSA and 2 mM EDTA. Twenty microliters of goat anti-mouse IgG microbeads (Miltenyi Biotec, Gladbach, Germany) were added to 1 × 107 cells and cells were incubated for 25 min at 4°C, washed with cfRPMI, and separated into fractions using MACS LS columns (Miltenyi Biotec) according to the manufacturer’s protocol. The resulting fractions were washed in cfRPMI and analyzed by flow cytometry analysis (FACS). Sub-samples of the sorted cells were also used for RNA, which was prepared as described below.
For FACS, aliquots of 1 × 106 PBL or sorted populations were incubated with 50 μl of mAb supernatant for 30 min at 4°C, washed, and then incubated with 50 μl goat anti-mouse Ig (H + L) secondary antibodies (Southern Biotechnology Associates) conjugated to phycoerythrin (PE) diluted 1:80 (v/v) for 30 min. Finally, the cells were washed, suspended in 0.3 ml of cfRPMI and analyzed on a FACScan flow cytometer (Becton Dickinson, Franklin Lakes, NJ) for single staining.
RNA preparation, RT-PCR, and 5′-RACE
Total RNA from various catfish tissues, PBL and sorted PBL populations was prepared using RNA-Bee (Tel-test Inc, Friendswood, TX) and treated with DNAse I (Invitrogen Life Technologies) according to the manufacturer's recommended protocols. One microgram was subsequently reverse transcribed into cDNA using an oligo-T primer and 200 units of Superscript III reverse transcriptase (RT; Invitrogen Life Technologies). RT-PCR was performed with specific primers for catfish membrane (m) IgM, mIgD, Igσ, Igλ, and the housekeeping gene elongation factor alpha (EF1-α; see supplementary Table 1). Typical parameters were: 3 min 94°C, followed by 30 cycles of 94°C 30 s, 60°C 30 s, 72°C 1 min, and a final extension cycle at 72°C 10 min. Annealing temperatures varied between 58–60°C depending on what primer pair was used. Amplified products were electrophoresed on 1% agarose gels, excised, and cloned and sequenced as above. Sequence analyses were performed with the DNASTAR software (Madison, WI). For 5′-RACE analysis, PBL RNA was prepared as above and 5′-RACE was performed using the GeneRacer Kit (Invitrogen Life technologies) and gene-specific primers according to the manufacturer's recommended protocol.
Results and discussion
Catfish Igσ
Catfish Igσ transcripts were isolated from PBL using 5'-RACE and RT-PCR protocols. Of 36 RACE products, 13 contained only unspliced Cσ genes and 23 contained a Jσ gene spliced to Cσ, preceded by an intron sequence. All of the sterile JCσ transcripts contained an identical J gene consisting of a nonamer, 23-bp spacer, heptamer preceding a J segment encoding 11 amino acids. Thus, to obtain a full-length Igσ cDNA, forward primers based on the 5′-end of Vσ-like sequences identified in the EST searches were designed and used in RT-PCR in combination with reverse Cσ specific primers. The representative full-length rearranged IpIgσ1 transcript consists of 732 nucleotides encoding a 244-amino-acid polypeptide with a predicted molecular weight of 23.7 kDa. The cDNA encodes a typical leader sequence, followed by V, J and C regions (see sFig. 1a). Both the Vσ and Cσ region sequences contain the conserved cysteine residues (CYS 23, CYS 104) that form the intrachain disulfide bonds and the conserved cysteine in the C region (CYS 126) that forms the interchain disulfide bond with the IgH chain (Fig. 1a). Also, as noted for other Vσ genes (Criscitiello and Flajnik 2007), CDR1 was shorter in length than CDR2, i.e., five vs nine amino acids. The Jσ region is 11 amino acids in length and contains a typical J-glycine bulge motif of FGQG. Multiple alignments and similarity searches show that the catfish Igσ has the highest amino acid similarity to teleost L2 isotypes and Xenopus and horn shark Igσ, hence, the σ designation. As expected, the IpIgσ1 V was most similar to other teleost Vσ genes (L2 Vs), with amino acid sequence identities ranging from 58–72% when compared to rainbow trout, carp (Cyprinus carpio) and zebrafish L2 Vs. In comparison, amino acid identities with horn shark and Xenopus Vσ sequences were lower at 46% and 30%, respectively. A similar trend also occurs when comparing the Jσ and Cσ regions of the different species. Jσ amino acid identities range from 55–91%, with the teleost Jσ regions sharing the highest amino acid identities. Similarly, catfish Cσ exhibits 53–58% amino acid identity with the L2 Cσ of the three bony fish, but drops to <36% amino acid identity with the horn shark and Xenopus Cσ regions (Fig. 1b).
Igσ amino acid sequence comparisons. a Amino acid alignments of catfish Vσ, Jσ, and Cσ regions with teleost L2, amphibian and shark Vσ and Cσ regions. The conserved cysteine residues (CYS 23 and CYS 104) that form the intrachain disulfide bonds are labeled (*) and the conserved C region cysteine (CYS 126) that forms the interchain disulfide bond with IgH is marked by (↓). Residues identical to the catfish sequence are shown as (.) and (–) represents gaps in the alignment. The amino acids numbers for the catfish sequence according to the standardized rules of the IMGT unique numbering are given above (Lefranc et al. 2005). b Comparison of Igσ percent amino acid identities from pairwise alignments of J and C regions. Abbreviations and GenBank accession numbers are: catfish (Ipσ EU872023); carp (CcL2 V and J (AB091113) and C (BAC81202)); zebrafish (DrL2, AAG31698); rainbow trout (OmL2 AAB41310); Atlantic cod (Gmσ V (EX732557), J and C (EC907978)); X. laevis (Xlσ AAH82413); and horn shark (Hfσ EF114760)
Southern blot analyses indicate that catfish Igσ genes, like other teleost IGL genes are found in a cluster organization. However, unlike catfish F and G genes, the Igσ genes are few in number. Previously, Southern blot analyses showed there were at least 30 V segments and 15 C segments for the G isotype, and at least 50 different fragments hybridized with V and C probes for the F isotype (Ghaffari and Lobb 1993, 1997). Briefly, in this study, genomic DNA from five individual outbred and one gynogenetic (homozygous) catfish was hybridized with either a Vσ or Cσ probe and depending upon the restriction enzyme used, 2-6 Vσ and 2-4 Cσ hybridizing bands were observed. The representative Southern blot of an Xba I digest indicates a typical Ig gene cluster organization with most of the same restriction fragments hybridizing with both V and C probes (Fig. 2a). Two Igσ clusters were amplified from gynogenetic catfish G25 genomic DNA using a Vσ forward primer and a reverse Cσ primer (Fig. 2b). The IpIgσ 3C gene cluster is 2,057 bp in length and consists of single V, J, and C genes, termed IGSV1-1, IGSJ1, and IGSC1. In contrast, the IpIgσ 5A gene cluster is 2,397 bp in length and contains two V genes, followed by single J and C genes. IGSV1-2 is 85% identical at the nucleotide level to IGSV1-1, and IGSV1-3 is a pseudogene truncated at the 5′ end. All are in the same transcriptional orientation and sequencing shows that the J and C genes in the two clusters are identical. Each of the V genes has a typical RSS of heptamer, 12-bp spacer, nonamer (Fig. 2c). Additionally three other Vσ cDNAs were amplified from outbred fish using RT-PCR and 5′-RACE (sFig. 2). Two (IGSV1-1*02 and IGSV1-1*03) are ~99% identical at the nucleotide level to IGSV1-1 and likely are alleles of this sequence. A third (IGSV1-2*02) represents an allele of IGSV1-2. Together Southern blots performed with homozygous gynogenetic DNA combined with sequencing data indicate that only two Igσ gene clusters are present in the catfish genome. This notion is further supported by sequencing of 23 RACE products from a single outbred fish, where only three different Cσ sequences, presumably alleles, with ~95% amino acid identity were identified (sFig. 3). Overall, the catfish Igσ repertoire appears very limited with two gene clusters each containing a single identical J (as well as an identical C), and either one or two V gene segments, all belonging to the same Vσ family. IGSV1-1 is located in one cluster (IpIgL σ 3C) and IGSV1-2 and IGSV1-3P are located in the second cluster (IpIgL σ 5A). In comparison, two distinct Vσ families have been identified in carp and limited sequencing suggests possibly four Jσ segments and two Cσ cDNAs, which are ~90% identical at the amino acid level (Ishikawa et al. 2004). Also, Southern blots indicate that only two to four carp Igσ clusters are present. In contrast, nine Vσ (L2) genes representing at least three Vσ families were sequenced in the rainbow trout and Southern blotting and genomic sequencing indicated that two to three Vσ genes occur in an Igσ (L2) cluster (Timmusk et al. 2000). Also, as with the catfish Igσ genes, the rainbow trout Igσ genes are in the same transcriptional orientation, and the Jσ and Cσ genes, while not identical, are quite similar with only a few amino acid differences. More recently, two different studies characterizing IGL chain gene organizations in zebrafish have shown that there are two Cσ genes in tandem with each C preceded by four or five functional Vσ genes (Hsu and Criscitiello 2006; Zimmerman et al. 2008). This discrepancy in gene segment number is likely because the annotation was performed on bacterial artificial chromosomes (BACs) from two different zebrafish libraries. However, in both studies, only two Vσ families were identified and genes from both families were associated with each of the two Cσ genes. Again, all the gene segments were in the same transcriptional orientation. Altogether, five teleost Vσ subgroups based on sequences sharing more than 75% nucleotide identity (Giudicelli and Lefranc 1999) could be identified from pairwise alignments of IGSV nucelotide sequences from catfish, carp, Atlantic cod, rainbow trout, and zebrafish (sFig. 4). The largest subgroup is represented in catfish, carp, rainbow trout, and zebrafish, while another subgroup is currently only found in carp and zebrafish and two Vσ subgroups are only present in the rainbow trout. Also, the five Atlantic cod Vσ sequences, which were identified from BLAST searches of ESTs, formed one single subgroup not represented in the other species.
Genomic analysis of IpIgσ. a Representative Southern blot analysis of genomic DNA from one homozygous gynogenetic (G25) and five outbred catfish. DNA was digested with Xba I and hybridized with either Vσ or Cσ specific probe. Size markers in kilobase pairs are on the right. b Representative schematics of two different IpIgσ genomic clusters that were amplified using Vσ and Cσ specific primers on gDNA isolated from gynogenetic catfish G25. Cluster IpIgLσ 3C contains a single V gene followed by a J gene segment and a C gene. Cluster IpIgLσ 5A contains two V genes located upstream of a single J gene segment and C gene. The genes are in the same transcriptional orientation. c Igσ V and J genes utilize typical RSS. The nucleotide sequence from the 3′ end of the V through the J gene segment of genomic cluster IpIgLσ3C is shown. Conserved heptamer and nonamer sequences are underlined and number of nucleotides spacing them are indicated above the sequence. Inferred amino acids of the partial V and J genes are shown below and numbers of nucleotides are on the right. GenBank accession number for IpIgLσ 3C (EU872024) and IpIgLσ 5A (EU872023)
Catfish and Atlantic cod IgL λ chains
RT-PCR was also used to clone and sequence catfish Igλ cDNAs. As above, forward V and reverse C region primers were designed based upon the identified catfish Igλ EST (CBPO4528.b1). Three different-sized Igλ products were amplified by RT-PCR and verified by sequencing. The longest was 916 bp and consisted of a V gene, 240-bp intron, followed by a J segment spliced to a C gene. Similarly, another product of 702 bp, was the result of mispriming by the V primer and consisted of the 3′ end of the V gene, intron, J spliced to C. As with mammalian Vλ and Jλ genes, the catfish Vλ RSS consisted of a heptamer and nonamer separated by a 23-bp intron and the Jλ heptamer and nonamer separated by a 12-bp intron. The smallest PCR product, 646 bp was a rearranged Igλ cDNA and based upon this sequence, additional 5′-RACE primers were made and used to obtain full-length catfish Igλ transcripts. The representative full-length catfish Igλ transcript consists of 696 nucleotides and encodes a 232-amino-acid polypeptide with a predicted molecular weight of 22.5 kDa. The cDNA encodes a typical leader sequence, followed by V, J, and C region. Both the Vλ and Cλ region sequences contain conserved cysteine residues that form the intrachain disulfide bonds and the conserved cysteine (CYS 126) in the constant region that forms the interchain disulfide bond with the IgH chain (Fig. 3a). Similarly, the Atlantic cod Igλ cDNA (originally termed L2) contains all the typical IGL features. It is 847 nucleotides in length and encodes a 240-amino-acid polypeptide with a predicted molecular of 23 kDa. The catfish and Atlantic cod Jλ genes are 13 and 12 amino acids in length, respectively, and like other Ig and TCR chains they encode a J-glycine bulge motif of FGxG (see sFig. 1b and c). As shown in Fig. 3a catfish Vλ also contains 2 additional cysteines at position 106 and 110 that are found conserved in rainbow trout and Atlantic cod Vλ sequences. Notably, database searches using the catfish Vλ predicted amino acid sequence identified the Atlantic cod Vλ sequence as the top related sequence (E-value, 3e−19) followed by various human, chicken, and horse Igλ V regions (E-values 9e−16 to 2e−15). Also, the CDR1 and CDR2 lengths were similar to the Vλ CDR1 and CDR2 lengths of most other species with CDR1 being longer than CDR2 (Criscitiello and Flajnik 2007). For example, the average human Vλ CDR1 length is nine amino acids and CDR2 length is four amino acids. For different teleost Vλ segments, the CDR1 length is 7-9 amino acids and CDR2 length is 2–3. We believe that CDR1 and CDR2 lengths, as suggested by Criscitiello and Flajnik 2007, combined with the RSS spacing supports the classification of this catfish IGL isotype as λ. The RSS spacer criteria has been widely used to categorize IGL isotypes and most recently it was used along with 21 other cladistic molecular markers to classify IGL chains from tetrapod species into three groups corresponding to the κ, λ, and σ isotypes (Das et al. 2008. In addition, it was recently used in combination with synteny to classify the Xenopus tropicalis Igλ locus (Qin et al. 2008).
Igλ amino acid sequence comparisons. a Amino acid alignment of catfish Vλ, Jλ and Cλ regions with teleost Igλ, amphibian type III, chicken, and mammalian Igλ J and C regions. The conserved cysteine residues that form the intrachain disulfide bonds are labeled (*) and the CL region cysteine that forms the interchain disulfide bond with IgH chain is marked by (↓). Residues identical to the catfish sequence are shown as (.) and (–) represents gaps in the alignment The amino acids numbers are given above according to the standardized rules of the IMGT unique numbering (Lefranc et al. 2005). b Comparison of Igλ percent amino acid identities from pairwise alignments of Jλ and Cλ regions from different species. Abbreviations and Genbank accession numbers are: catfish (Ipλ, EU872025); rainbow trout (Omλ, V and J (BX861772) and C (CA257088)); Atlantic cod (Gmλ, AJ293808); sandbar shark (CpII, M81314), X. laevis type III (XlIII, L76575); Green anole (Acλ FG746006); chicken (Ggλ K00678); and human (Hsλ ABU90549)
In order to identify additional teleost Igλ sequences database searches using the Atlantic cod Igλ chain were performed and Igλ-like sequences from several salmonids (Oncorhynchus nerka, Oncorhynchus tshawytscha, Salvelinus fontinalis, Salmo salar), were identified and a rainbow trout Igλ sequence was included in the phylogenetic analyses. As with the catfish Igσ chains, the catfish Igλ sequences have the highest amino acid identities with the rainbow trout and Atlantic cod Vλs, Jλs and Cλs, at 55–64%, 46–67%, and 50–59%, respectively. Also, as expected, when the catfish Vλ, Jλ, and Cλ are compared to a representative V, J and C of sandbar shark Type II IgL, Xenopus Type III, chicken Igλ and human Igλ the amino acid identities are lower (Fig. 3b). Interestingly, Igλ transcripts were not found among the 1,609,630 zebrafish ESTs or the 32,000 carp ESTs.
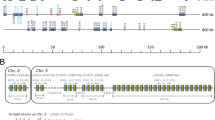
Southern blots of catfish and Atlantic cod genomic DNA with Vλ and Cλ probes indicate that the Igλ locus in these two species is also of the cluster type since some of the restriction fragments hybridized with both V and C probes (Fig. 4a). Similar to the catfish Igσ locus, only two Igλ clusters have been identified in catfish. Using 5'-RACE and RT-PCR protocols three different functional Igλ VL were identified from an individual catfish. Two, IGLV1-1*02 and IGLV1-1*03, are 98% identical to each other at the nucleotide level and appear to be allelic while the third, IGLV1-2*01, with less identity (85%) is from the second Igλ cluster inferred by the Southern blot analyses (sFig. 5). The existence of two λ genomic clusters were confirmed by using two unique Vλ forward primers (O-669 and O-685) in combination with the reverse Cλ primer (O-671; sTable 1) to amplify the λ clusters from gynogenetic catfish G25 genomic DNA (Fig. 4b). One cluster IpIgλ 2A is 1,878 in length and contains single V, J, and C genes all in the same transcriptional orientation. They are designated IGLV1-1, IGLJ1, and IGLC1. The second cluster IpIgλ2E is 98% identical to IpIgλ2A at the nucleotide level and differs mainly in the V region (89% nt identity). There are also two nucleotide differences in the J sequences, resulting in amino acid changes and four nucleotide differences in the C region, one of which results in an amino acid change. Both clusters have the mammalian Igλ-type RSS and the RSS of the Atlantic cod and rainbow trout Vλ and Jλ are also of the Igλ-type. An alignment of the Igλ clusters of catfish, Atlantic cod and rainbow trout show them to be of similar size (Fig. 4c).
Genomic analysis of IpIgλ. a Representative Southern blot analysis of genomic DNA from one homozygous gynogenetic (G25) and five outbred catfish. DNA was digested with Xba I and hybridized with either a catfish Vλ or Cλ specific probe (left panel). In the right panel Atlantic cod genomic DNA was digested with either EcoR I, or Hind III and hybridized with either an Atlantic cod Vλ or Cλ specific probe. Size markers in kilobase pairs are on the right. b Representative schematic of an IpIgL λ genomic cluster that was amplified using Vλ and Cλ specific primers on gDNA isolated from gynogenetic catfish G25. Cluster IpIgλ 2A contained a single VL and JL gene segment followed by a CL gene, all in the same transcriptional orientation. c Teleost IgL Vλ and Jλ genes utilize typical λ RSS. Nucleotide alignment from the 3′ end of a Vλ through the Jλ gene segment of a catfish, Atlantic cod, and rainbow trout Igλ genomic sequences are shown. The conserved heptamer and nonamer sequences are underlined and number of nucleotides spacing them are indicated above the sequence. Inferred amino acids of the partial Vλ and Jλ genes are shown below and numbers of nucleotides are indicated on the right. Accession numbers for IpIgLλ 2A (EU872025), rainbow trout Igλ (BX861772), and Atlantic cod Igλ (AJ293808)
IgL phylogenetic analyses
It was recently proposed that vertebrate IGL chains can be classified into four distinct ancestral clans: one restricted to elasmobranchs (cartilaginous σ/type I), one in all vertebrates except teleost (λ/elasmobranch type II), one found in all ectothermic vertebrates (σ/teleost L2/elasmobranch type IV), and one in all vertebrates except birds (κ/elasmobranch type III/teleost L1 and L3; (Criscitiello and Flajnik 2007). However, as presented in this study, when teleost V regions from the previously isolated L1, L2 and L3 isotypes were compared with the newly identified catfish and Atlantic cod Vσ and Atlantic cod, rainbow trout and catfish Vλ sequences in phylogenetic analyses, three distinct clusters supported by high bootstraps values were formed (Fig. 5a). Importantly, while Vσ and Vλ sequences clustered in individual groups, the V sequences found associated with the L1 and L3 C regions were intermingled on a third branch consistent with the notion that L1/G, L3/F share a common origin and are of the κ class. In contrast, as expected, phylogenetic analyses of the different teleost IGLC sequences resulted in four individual clusters corresponding to the four teleost isotypes L1/G, σ/L2, L3/F, and λ. Moreover, the topology of the V phylogenetic tree did not change significantly when elasmobranch and tetrapod V sequences were included in the analyses; however, the bootstrap values dropped (Fig. 6a). Even so, the fact that teleost Vλs cluster with the shark type II, Xenopus Type III and the traditional Vλ sequences from chicken and mammals support the concept that these new teleost IGL isotypes are Igλ. Similarly, when the different vertebrate IGL C sequences were analyzed, the C of the teleost σ (type L2) chains clustered with shark and Xenopus Cσ sequences (Fig. 6b). Again, while not supported by high bootstrap values, the representative cartilaginous Cκ (type III) and Cλ (type II) sequences clustered with the mammalian Cκ sequences and the mammalian, Xenopus, and bird Cλ sequences, respectively. In contrast, the teleost Cλ as well as the κ linage C sequences (type L1/G and type L3/F), clustered in distinct groups separately from other vertebrate C sequences and appear to have diverged further from their mammalian, bird, Xenopus, and cartilaginous fish counterparts. Since phylogenetic analyses of IGL isotypes result in trees with low bootstrap values, Das et al. 2008 recently redefined the IGL isotypes based on 21 cladistic molecular markers. Briefly, when the molecular markers were applied to a data set of ~1,300 IGLV, 129 IGLJ and 78 IGLC sequences from 12 tetrapod species, three groups corresponding to Igκ, Igλ, and Igσ isotypes were identified (sTable 2). Here, when the same criteria were applied to the teleost IGL sequences four of the five markers identified in the IGLV region were shown to be valid (sFig. 6). For example, Igλ can be distinguished from Igκ and Igσ by the presence of a gap in amino acid position 7, Igκ has PHE at position 87 and Igσ has TYR, while Igλ does not exhibit PHE or TYR at this position. Also, from our analyses, Igσ can be distinguished by the presence of ILE at position 90 (highlighted in green in sFig. 6). However, the defined markers for IGLJ and IGLC were not so clear-cut when the teleost sequences were included in the analysis. The only valid IGLJ marker was that J sequences of the κ linage have a THR at position 117, while Jσ and Jλ sequences do not. As for the IGLC sequences, only two unique Igσ markers are observed: the absence of PHE/TYR/GLY at position 28 and PRO at position 30. Comparatively, Cλ sequences contained ARG /LYS at positions 84a combined with GLN at position 84b. Finally, the C regions of the κ isotypes, are distinguished by the presence of certain amino acid residue combinations at positions 13, 28, 81, 84a, 84g, 108, and 121. Whether these markers will be valid for a larger all-inclusive data set is not known. Nevertheless, together, our sequence and phylogenetic analyses demonstrate that teleosts possess four IgL isotypes. As previously suggested, two of these, type L1 and type L3 (which include catfish F and G) have diverged from the Igκ lineage and teleost type L2 IgL chains are of Igσ origin (Bengtén et al. 2000; Criscitiello and Flajnik 2007; Ota et al. 2000). More importantly, these findings support the prediction by Criscitiello et al. 2007 that λ light chains may be present in all vertebrate groups, but not necessarily in each species of each group.
Phylogenetic analyses of teleost IGL a V sequences and b C sequences. The neighborhood-joining trees with pairwise gap deletions were drawn using MEGA4 with 1,000 bootstrap replications. Channel catfish β2-microglobulin was used as an outgroup and clusters of sequences are marked by blocks. All Genbank accession numbers are listed in the figure, except for catfish B2-microglobulin (AAC67230). Species abbreviations are: Cc carp, Dr zebrafish, Om rainbow trout, Ip channel catfish, If blue catfish, Gm Atlantic cod
Phylogenetic analyses of vertebrate IGL a V sequences and b C sequences. The neighborhood-joining trees with pairwise gap deletions were drawn using MEGA4 with 1,000 bootstrap replications Channel catfish β2-microglobulin was used as an outgroup. Clusters of sequences are marked by blocks and GenBank accession numbers are: human λ (ABU90549); sheep λ (AAB94914); chicken λ (K00678); duck λ (M25726); dog λ (ABY55585); mouse λ (J00587); catfish λ (EU872022); Atlantic cod L2 (λ; CAC03754); rainbow trout λ (BX861350); X. laevis III (L76579); ratfish II (L25549); skate II (L25566); horn shark II (L25560); sandbar shark II (M81314); salmon L3 (AF406956); fugu L1 (AB126061); carp L1b (AB073332); human κ (AAH73764); catfish F-type (U25705); X. laevis ρ (V and J (L15570), C (AAA49880)); mouse κ (V and J (M34739), C (CAA47650); horse κ (X75612); zebrafish L3 (AB246193); carp L3 (AB073335); zebrafish L1 (AF246185); catfish G-type (L25533); rainbow trout L1 (X65260); Salmon L1 (F273012); horn shark III (L25561); nurse shark NS4 (GSU15144); nurse shark σ (EF114759); dogfish shark σ (CX662702); X. laevis σ (S78544); rainbow trout L2 (AAB41310); carp L2 (AB091113); fugu L2 (DQ471453); catfish σ (EU872021); zebrafish L2 (AF246183); horn shark σ (EF114760); Atlantic cod L1 (X68515), and catfish β2-microglobulin (AAC67230)
Catfish Igσ and Igλ expression
At the message level functionally rearranged catfish Igσ transcripts were readily detected by RT-PCR in all catfish tissues analyzed, except for muscle by RT-PCR (Fig. 7a). Comparatively, two PCR products were consistently amplified using Igλ specific primers O-657F and O-650R (sTable 1). The larger product represented the 916-bp partially spliced unrearranged Igλ transcript and the smaller 646 bp product represented the functional rearranged catfish Igλ transcript. Both PCR products were observed in all tissues examined except brain, muscle and fin; however, the functional rearranged transcript product appeared to be more prevalent in spleen, head kidney (pronephros), trunk kidney (mesonephros), heart, and liver. As shown in Fig. 7b, message for catfish F and G light chains as well as Igσ and Igλ were detectable in freshly isolated PBL, as well as in surface (s) IgM+ cells, enriched by MACS-sorting. However, no G, or Igλ and negligible amounts of F message was detected in the sIgM- cell population (IgM depleted), which suggest that IgM- B cells preferentially express Igσ. Also, as with the catfish tissues, Igλ PCR products representing partially spliced unrearranged transcripts and rearranged transcripts were amplified from PBL and sIgM+ cells along with a truncated partially spliced unrearranged form due to mispriming of the V forward primer O-657F.
RT-PCR analyses of Igλ and Igσ expression in catfish tissues and sorted PBL. a Total RNA was obtained from various tissues and RT-PCR was performed using VL and CL specific primers for catfish Igσ, Igλ and EF1-α. Schematics of each IgL transcript are shown next to their respective panels. A partially spliced transcript and a fully rearranged transcript was amplified using the Vλ and Cλ primers. b Total RNA was obtained from unfractionated PBL, MACS sorted sIgM+ PBL, and MACS sorted sIgM− PBL from an individual catfish. RT-PCR was performed using primers specific for catfish mIgM, mIgD, G, F, Igλ, Igσ, and EF1-α. Schematics of the IGL transcripts are next to their respective panels. Base pair markers are indicated on the left. The resulting PCR products were verified by sequencing
The initial anti-catfish F and G mAb characterization studies demonstrated that individually, these reagents react with different lymphocyte populations in catfish PBL and different immunoglobulin populations in catfish serum. Also, when they were used in combination, they immunoprecipitated >90% of purified catfish anti-DNP IgM (Lobb and Clem 1982; Lobb et al. 1984). Therefore, in order to determine if catfish PBL contained IgM+ cells that did not express the F or G isotypes, a negative sort with anti-F and anti-G mAbs was performed. After depleting PBL of F and G positive cells a small population of the cells was still sIgM+ (~2%; Fig. 8a). As analyzed by flow cytometry, cells displaying the IgM+/F−/G− phenotype are small, agranular, and fall within the traditionally defined catfish lymphocyte gate (Fig. 8b). At the message level the F-/G- population expressed message for Igλ and Igσ, as well as membrane IgM. Currently, it is unknown whether the F message present in the F-/G- cell population is due to transcription of nonproductively rearranged F genes. These results combined with the RT-PCR data showing that a sIgM− cDNA pool expresses message for IgD and Igσ, but not Igλ, suggest Igσ potentially associates with both IgM and the IgD molecules, and that a small IgM+ B cell population within catfish PBL expresses the Igλ isotype. Thus, a logical assumption is that the apparent low expression levels of the Igσ and Igλ isotypes are because of their limited repertoire, of two to four V genes. Currently, it is not known if the Igσ+ and Igλ+ B cell populations can be expanded by antigen driven selection and/or if they represent populations with specialized functions. For example, it was previously shown that the ratio of catfish F and G chains changes during an immune response to dinitrophenyl (DNP): in pre-immune serum the ratio of F:G was 60:40, 1 to 2 weeks after primary immunization the F:G ratio was 80:20, and 3 weeks to 1 year later the ratio was 90:10, indicating a preferential usage of the F isotype in the anti-DNP response (Lobb 1986). However, while it is tempting to speculate that teleost Igλ or Igσ chains may have a specialized function, positive proof will only come after anti-teleost Igλ and Igσ chain antibodies become available, which will allow these respective cell populations to be isolated and functional studies to be performed.
IgM and IgL expression in catfish PBL. a Freshly isolated PBL and PBL depleted of surface F and G positive cells (F−/G−) by MACS were analyzed by flow cytometry. Aliquots from the unsorted (top panel) and sorted (bottom panel) populations were stained with anti-catfish IgM 9E1, anti-catfish F, and anti-catfish G mAbs. Anti-rainbow trout IgM 1.14 was the isotype control. The total and negatively sorted PBL populations were from the same fish. b IgM positive cells (~2%) from the depleted population (F−/G−) were gated and examined side and forward scatter. c Total RNA was obtained from PBL and the F and G depleted fraction (F−/G−) and RT-PCR was performed using primers specific for IgM, G, F, Igλ, Igσ, and EF1-α. Schematics of the IgL transcripts are next to their respective panels. Base pair size markers are indicated on the left. The resulting PCR products were verified by sequencing
The presence of four IgL isotypes in teleosts also raises questions concerning regulation of their expression. For example, with IgL loci organized in multi-copy clusters, how is allelic exclusion possible and do the different isotypes undergo gene rearrangements in a preferential order? More than two decades ago, two models for regulation of mammalian Igκ and Igλ gene rearrangements were proposed, the stochastic model and the regulated/ordered model (reviewed in Mostoslavsky et al. 2004). In the stochastic model, it was assumed that both alleles of the Igκ and Igλ loci were accessible for gene rearrangements simultaneously and that due to junctional diversity, productive rearrangements on more than one allele was unlikely (Coleclough et al. 1981). Also, it was predicted that Igκ genes were more likely to produce successful rearrangements due to the greater number of available V and J sequences and the quality of the Igκ RSS (Arakawa et al. 1996; Ramsden and Wu 1991). In contrast, the regulated/ordered model proposed that Igκ and Igλ undergo gene rearrangements through a sequential order of events, where changes in the chromatin structure first occurred on one of the Igκ alleles allowing it to attempt V–J recombination, while the other Igκ allele and the Igλ alleles remained inaccessible (Storb et al. 1981). More recently, support for the regulated/ordered model comes from the identification of several B cell developmentally controlled mechanisms, including remodeling of the chromatin structure, demethylation of distinct alleles, and selective accessibility of recombination machinery through differential expression of transcription factors (Asenbauer et al. 1999; and reviewed in Corcoran 2005; Geier and Schlissel 2006). However, this process is not absolutely strict, while Igλ expression is upregulated after deletion of the Igκ locus, Igλ rearrangement is not dependent on this deletion and can precede rearrangement of Igκ (Nadel et al. 1990; Oberdoerffer et al. 2003; Zou et al. 1993). That Igκ and Igλ rearrangement can occur independently is also supported by the presence of individual B cells expressing both functionally rearranged Igκ and Igλ chains (Sirac et al. 2006; van der Burg et al. 2001). The regulated/ordered model also implies that different subsets of B cells can preferentially use one or another IGL isotype, since different developmental programs could apply to different B cell subsets. Examples of preferential usage of IGL chains have been observed in Xenopus, where the majority IgY is associated with Igκ chains and almost never with Igσ. In contrast, Igσ chains are found associated with IgM and IgX (Hsu et al. 1991; Lee et al. 2000). Another example of preferential IGL chain usage occurs in humans, where a population of IgM−/IgD+ B cells present in respiratory mucosa expresses almost exclusively Igλ chains (Chen et al. 2008; Liu et al. 1996). To date, regulation of IGL chain rearrangements has not been studied in any non-mammalian vertebrate. Therefore, based on the findings presented here and as has been proposed for shark (Fleurant et al. 2004), it is tempting to speculate that the expression in catfish is stochastically driven since the IgF and IgG isotype, which are encoded by loci that combined contain 60–80 V segments combined, constitute ≥90 of the expressed IGL found in PBL and in serum (Ghaffari and Lobb 1993, 1997; Lobb and Clem 1982; Lobb et al. 1984). In comparison, the Igσ and Igλ loci possess two to four V segments, and these isotypes appear to be expressed only in 2–10% of PBL. However, based upon what is now known about IGL regulation in mice and humans, the regulation of the teleost light-chain rearrangements and expression is likely more complex.
In summary, catfish Igσ and Atlantic cod, catfish and rainbow trout Igλ cDNAs and genes have been identified. While catfish Igσ clearly shows strong homology with previously identified Igσ sequences, the teleost Igλ sequences are less conserved. Nevertheless, together with the “Igλ-type” RSS, the conserved CDR lengths, and phylogenetic analyses support the teleost Igλ classification. In contrast to the teleost Igκ type L1 and type L3 loci, the teleost Igσ and Igλ loci contain only a few VL genes suggesting that Igσ and Igλ do not contribute greatly to the overall available repertoire. Even so, studies of preferential IGL chain usage in catfish IgM+/IgD+, IgM+/IgD− and IgM−/IgD+ B cell populations are currently being pursued and it may be that these isotypes provide unique paratopes by combining with certain IGHV chains. Development of mAbs will allow for the functional characterization of catfish B cell Igσ+ and Igλ+ populations, as well as determinations concerning if preferential Igσ/Igλ chain usage occurs between IgM and IgD isotypes and if Igσ/Igλ expression can be expanded by antigen stimulation.
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Arakawa H, Shimizu T, Takeda S (1996) Re-evaluation of the probabilities for productive arrangements on the kappa and lambda loci. Int Immunol 8:91–99. doi:10.1093/intimm/8.1.91
Asenbauer H, Combriato G, Klobeck HG (1999) The immunoglobulin lambda light chain enhancer consists of three modules which synergize in activation of transcription. Eur J Immunol 29:713–724. doi:10.1002/(SICI)1521-4141(199902)29:02<713::AID-IMMU713>;3.0.CO;2-M
Bengtén E, Wilson M, Miller N, Clem LW, Pilström L, Warr GW (2000) Immunoglobulin isotypes: structure, function, and genetics. Curr Top Microbiol Immunol 248:189–219
Chen K, He B, Santini P, Chiu A, Chadburn A, Xu W, Cerutti A, Cattalini M, Plebani A (2008) Cmu to Cdelta class switch recombination and IgD production contribute to mucosal immunity. FASEB J 22:Abstract 854.7
Church GM, Gilbert W (1984) Genomic sequencing. Proc Natl Acad Sci USA 81:1991–1995. doi:10.1073/pnas.81.7.1991
Coleclough C, Perry RP, Karjalainen K, Weigert M (1981) Aberrant rearrangements contribute significantly to the allelic exclusion of immunoglobulin gene expression. Nature 290:372–378. doi:10.1038/290372a0
Corcoran AE (2005) Immunoglobulin locus silencing and allelic exclusion. Semin Immunol 17:141–154. doi:10.1016/j.smim.2005.01.002
Coscia MR, Giacomelli S, De Santi C, Varriale S, Oreste U (2008) Immunoglobulin light chain isotypes in the teleost Trematomus bernacchii. Mol Immunol 45:3096–3106. doi:10.1016/j.molimm.2008.03.006
Criscitiello MF, Flajnik MF (2007) Four primordial immunoglobulin light chain isotypes, including lambda and kappa, identified in the most primitive living jawed vertebrates. Eur J Immunol 37:2683–2694. doi:10.1002/eji.200737263
Daggfeldt A, Bengtén E, Pilström L (1993) A cluster type organization of the loci of the immunoglobulin light chain in Atlantic cod (Gadus morhua L.) and rainbow trout (Oncorhynchus mykiss Walbaum) indicated by nucleotide sequences of cDNA and hybridization analysis. Immunogenetics 38:199–209. doi:10.1007/BF00211520
Danilova N, Bussmann J, Jekosch K, Steiner LA (2005) The immunoglobulin heavy-chain locus in zebrafish: identification and expression of a previously unknown isotype, immunoglobulin Z. Nat Immunol 6:295–302. doi:10.1038/ni1166
Das S, Nikolaidis N, Klein J, Nei M (2008) Evolutionary redefinition of immunoglobulin light chain isotypes in tetrapods using molecular markers. Proc Natl Acad Sci USA 105:16647–16652. doi:10.1073/pnas.0808800105
DeLuca D, Wilson M, Warr GW (1983) Lymphocyte heterogeneity in the trout, Salmo gairdneri, defined with monoclonal antibodies to IgM. Eur J Immunol 13:546–551. doi:10.1002/eji.1830130706
Edelman GM, Gally JA (1962) The nature of Bence-Jones proteins. Chemical similarities to polypetide chains of myeloma globulins and normal gamma-globulins. J Exp Med 116:207–227. doi:10.1084/jem.116.2.207
Fleurant M, Changchien L, Chen CT, Flajnik MF, Hsu E (2004) Shark Ig light chain junctions are as diverse as in heavy chains. J Immunol 173:5574–5582
Gadol N, Waldman RH, Clem LW (1976) Inhibition of macrophage migration by normal guinea pig intestinal secretions. Proc Soc Exp Biol Med 151:654–658
Geier JK, Schlissel MS (2006) Pre-BCR signals and the control of Ig gene rearrangements. Semin Immunol 18:31–39. doi:10.1016/j.smim.2005.11.001
Ghaffari SH, Lobb CJ (1993) Structure and genomic organization of immunoglobulin light chain in the channel catfish. An unusual genomic organizational pattern of segmental genes. J Immunol 151:6900–6912
Ghaffari SH, Lobb CJ (1997) Structure and genomic organization of a second class of immunoglobulin light chain genes in the channel catfish. J Immunol 159:250–258
Giudicelli V, Lefranc MP (1999) Ontology for immunogenetics: the IMGT-ONTOLOGY. Bioinformatics 15:1047–1054
Greenberg AS, Steiner L, Kasahara M, Flajnik MF (1993) Isolation of a shark immnoglobulin light chain cDNA clone encoding a protein resembling mammalian k light chains: implications for the evolution of light chains. Proc Natl Acad Sci USA 90:10603–10607. doi:10.1073/pnas.90.22.10603
Haire RN, Ota T, Rast JP, Litman RT, Chan FY, Zon LI, Litman GW (1996) A third Ig light chain gene isotype in Xenopus laevis consists of six distinct VL families and is related to mammalian lambda genes. J Immunol 157:1544–1550
Haire RN, Rast JP, Litman RT, Litman GW (2000) Characterization of three isotypes of immunoglobulin light chains and T-cell antigen receptor alpha in zebrafish. Immunogenetics 51:915–923. doi:10.1007/s002510000229
Hansen JD, Landis ED, Phillips RB (2005) Discovery of a unique Ig heavy chain isotype in rainbow trout: implications for a novel B-cell developmental pathway in teleost fish. Proc Natl Acad Sci USA 102:6919–6924. doi:10.1073/pnas.0500027102
Heery DM, Gannon F, Powell R (1990) A simple method for subcloning DNA fragments from gel slices. Trends Genet 6:173. doi:10.1016/0168-9525(90)90158-3
Hogan RJ, Waldbieser GC, Goudie CA, Antao A, Godwin UB, Wilson MR, Miller NW, Clem LW, McConnell TJ, Wolters WR, Chinchar VG (1999) Molecular and immunologic characterization of gynogenetic channel catfish (Ictalurus punctatus). Mar Biotechnol 1:317–327. doi:10.1007/PL00011781
Hohman VS, Schluter SF, Marchalonis JJ (1992) Complete sequence of a cDNA clone specifying sandbar shark immunoglobulin light chain: gene organisation and implications for the evolution of light chains. Proc Natl Acad Sci USA 89:276–280. doi:10.1073/pnas.89.1.276
Hsu E, Criscitiello MF (2006) Diverse immunoglobulin light chain organizations in fish retain potential to revise B cell receptor specificities. J Immunol 177:2452–2462
Hsu E, Lefkovits I, Flajnik M, Du Pasquier L (1991) Light chain heterogeneity in the amphibian Xenopus. Mol Immunol 28:985–994. doi:10.1016/0161-5890(91)90184-L
Ishikawa J, Imai E, Moritomo T, Nakao M, Yano T, Tomana M (2004) Characterisation of a fourth immunoglobulin light chain isotype in the common carp. Fish Shellfish Immunol 16:369–379. doi:10.1016/j.fsi.2003.06.002
Kubo RT, Rosenblum IY, Benedict AA (1970) The unblocked N-terminal sequence of chicken IgG lambda-like light chains. J Immunol 105:534–536
Lee SS, Greenberg A, Hsu E (2000) Evolution and somatic diversification of immunoglobulin light chains. Curr Top Microbiol Immunol 248:285–300
Lefranc MP, Pommie C, Kaas Q, Duprat E, Bosc N, Guiraudou D, Jean C, Ruiz M, Da Piedade I, Rouard M, Foulquier E, Thouvenin V, Lefranc G (2005) IMGT unique numbering for immunoglobulin and T cell receptor constant domains and Ig superfamily C-like domains. Dev Comp Immunol 29:185–203. doi:10.1016/j.dci.2004.07.003
Liu YJ, de Bouteiller O, Arpin C, Briere F, Galibert L, Ho S, Martinez-Valdez H, Banchereau J, Lebecque S (1996) Normal human IgD+IgM− germinal center B cells can express up to 80 mutations in the variable region of their IgD transcripts. Immunity 4:603–613. doi:10.1016/S1074-7613(00)80486-0
Lobb CJ (1986) Preferential expression of catfish light chain immunoglobulin isotypes in anti-dinitrophenyl antibodies. J Immunogenet 13:19–28. doi:10.1111/j.1744-313X.1986.tb01079.x
Lobb CJ, Clem LW (1982) Fish lymphocytes differ in the expression of surface immunoglobulin. Dev Comp Immunol 6:473–479
Lobb CJ, Olson MO, Clem LW (1984) Immunoglobulin light chain classes in a teleost fish. J Immunol 132:1917–1923
Lundqvist ML, Middleton DL, Radford C, Warr GW, Magor KE (2006) Immunoglobulins of the non-galliform birds: antibody expression and repertoire in the duck. Dev Comp Immunol 30:93–100. doi:10.1016/j.dci.2005.06.019
Magor KE, Higgins DA, Middleton DL, Warr GW (1994) cDNA sequence and organization of the immunoglobulin light chain gene of the duck, Anas platyrhynchos. Dev Comp Immunol 18:523–531. doi:10.1016/S0145-305X(06)80006-6
McCormack WT, Carlson LM, Tjoelker LW, Thompson CB (1989) Evolutionary comparison of the avian IgL locus: combinatorial diversity plays a role in the generation of the antibody repertoire in some avian species. Int Immunol 1:332–341. doi:10.1093/intimm/1.4.332
Miller NW, Bly JE, van Ginkel F, Ellsaesser CF, Clem LW (1987) Phylogeny of lymphocyte heterogeneity: identification and separation of functionally distinct subpopulations of channel catfish lymphocytes with monoclonal antibodies. Dev Comp Immunol 11:739–747. doi:10.1016/0145-305X(87)90061-9
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215. doi:10.1093/nar/16.3.1215
Miller NW, Rycyzyn MA, Wilson MR, Warr GW, Naftel JP, Clem LW (1994) Development and characterization of channel catfish long term B cell lines. J Immunol 152:2180–2189
Mostoslavsky R, Alt FW, Rajewsky K (2004) The lingering enigma of the allelic exclusion mechanism. Cell 118:539–544. doi:10.1016/j.cell.2004.08.023
Nadel B, Cazenave PA, Sanchez P (1990) Murine lambda gene rearrangements: the stochastic model prevails over the ordered model. EMBO J 9:435–440
Oberdoerffer P, Novobrantseva TI, Rajewsky K (2003) Expression of a targeted lambda 1 light chain gene is developmentally regulated and independent of Ig kappa rearrangements. J Exp Med 197:1165–1172. doi:10.1084/jem.20030402
Ota T, Sitnikova T, Nei M (2000) Evolution of vertebrate immunoglobulin variable gene segments. Curr Top Microbiol Immunol 248:221–245
Partula S, Schwager J, Timmusk S, Pilström L, Charlemagne J (1996) A second immunoglobulin light chain isotype in the rainbow trout. Immunogenetics 45:44–51. doi:10.1007/s002510050165
Qin T, Ren L, Hu X, Guo Y, Fei J, Zhu Q, Butler JE, Wu C, Li N, Hammarstrom L, Zhao Y (2008) Genomic organization of the immunoglobulin light chain gene loci in Xenopus tropicalis: evolutionary implications. Dev Comp Immunol 32:156–165
Ramsden DA, Wu GE (1991) Mouse kappa light-chain recombination signal sequences mediate recombination more frequently than do those of lambda light chain. Proc Natl Acad Sci USA 88:10721–10725. doi:10.1073/pnas.88.23.10721
Rast JP, Anderson MK, Ota T, Litman RT, Margittai M, Shamblott MJ, Litman GW (1994) Immunoglobulin light chain class multiplicity and alternative organizational forms in early vertebrate phylogeny. Immunogenetics 40:83–99. doi:10.1007/BF00188170
Reynaud C-A, Dahan A, Weill J-C (1983) Complete sequence of a chicken λ light chain immunoglobulin derived from the nucleotide sequence of its mRNA. Proc Natl Acad Sci USA 80:4099–4103. doi:10.1073/pnas.80.13.4099
Reynaud C-A, Anquez V, Grimal H, Weill J-C (1987) A hyperconversion mechanism generates the chicken light chain preimmune repertoire. Cell 48:379–388. doi:10.1016/0092-8674(87)90189-9
Saha NR, Suetake H, Suzuki Y (2004) Characterization and expression of the immunoglobulin light chain in the fugu: evidence of a solitaire type. Immunogenetics 56:47–55. doi:10.1007/s00251-004-0662-5
Schwager J, Burckert N, Schwager M, Wilson M (1991) Evolution of immunoglobulin light chain genes: analysis of Xenopus IgL isotypes and their contribution to antibody diversity. EMBO J 10:505–511
Shen L, Stuge TB, Evenhuis JP, Bengten E, Wilson M, Chinchar VG, Clem LW, Miller NW (2003) Channel catfish NK-like cells are armed with IgM via a putative FcmicroR. Dev Comp Immunol 27:699–714. doi:10.1016/S0145-305X(03)00042-9
Sirac C, Carrion C, Duchez S, Comte I, Cogne M (2006) Light chain inclusion permits terminal B cell differentiation and does not necessarily result in autoreactivity. Proc Natl Acad Sci USA 103:7747–7752. doi:10.1073/pnas.0509121103
Solem ST, Stenvik J (2006) Antibody repertoire development in teleosts—a review with emphasis on salmonids and Gadus morhua L. Dev Comp Immunol 30:57–76. doi:10.1016/j.dci.2005.06.007
Storb U, Wilson R, Selsing E, Walfield A (1981) Rearranged and germline immunoglobulin kappa genes: different states of DNase I sensitivity of constant kappa genes in immunocompetent and nonimmune cells. Biochemistry 20:990–996. doi:10.1021/bi00507a053
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882. doi:10.1093/nar/25.24.4876
Timmusk S, Partula S, Pilstrom L (2000) Different genomic organization and expression of immunoglobulin light-chain isotypes in the rainbow trout. Immunogenetics 51:905–914. doi:10.1007/s002510000221
van der Burg M, Tumkaya T, Boerma M, de Bruin-Versteeg S, Langerak AW, van Dongen JJ (2001) Ordered recombination of immunoglobulin light chain genes occurs at the IGK locus but seems less strict at the IGL locus. Blood 97:1001–1008. doi:10.1182/blood.V97.4.1001
van Ginkel FW, Miller NW, Lobb CJ, Clem LW (1992) Characterization of anti-hapten antibodies generated in vitro by channel catfish peripheral blood lymphocytes. Dev Comp Immunol 16:139–151. doi:10.1016/0145-305X(92)90014-4
Wu TT, Kabat EA (1970) An analysis of the sequences of the variable regions of Bence Jones proteins and myeloma light chains and their implications for antibody complementarity. J Exp Med 132:211–250. doi:10.1084/jem.132.2.211
Zezza DJ, Stewart SE, Steiner LA (1992) Genes encoding Xenopus laevis Ig L chains. Implications for the evolution of k and l chains. J Immunol 149:3968–3977
Zimmerman AM, Yeo G, Howe K, Maddox BJ, Steiner LA (2008) Immunoglobulin light chain (IgL) genes in zebrafish: genomic configurations and inversional rearrangements between (V(L)-J(L)-C(L)) gene clusters. Dev Comp Immunol 32:421–434. doi:10.1016/j.dci.2007.08.005
Zou YR, Takeda S, Rajewsky K (1993) Gene targeting in the Ig kappa locus: efficient generation of lambda chain-expressing B cells, independent of gene rearrangements in Ig kappa. EMBO J 12:811–820
Acknowledgments
This work was supported by grants from the National Institutes of Health (RO1AI-19530), US Department of Agriculture (2002-35204-12211, 2003-35205-12829, and 2006-35204-16880), UMMC IRSP (59908) awarded to M.W., and UMMC IRSP (55901) awarded to N.W.M. We also thank the Drs Ellen Hsu and Mike Criscitiello for sharing their IGL sequence information pre-publication.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
sTable 1
Channel catfish primers used for PCR, probes and sequencing (PDF 18 kb)
sTable 2
Molecular markers distinguishing κ, λ and σ isotypes (PDF 12 kb)
sFig. 1
Catfish Igσ and Igλ and Atlantic cod Igλ chains. Nucleotide and predicted amino acid sequences of a catfish Igσ, b catfish Igλ and c Atlantic cod Igλ. The predicted leader sequence, VL, JL and C regions are labeled above the sequence. The stop (TGA) codons are marked with (*) and nucleotide and amino acid numbers are at left. GenBank accession numbers are: Catfish Igσ (IpIgσ, EU872021), catfish Igλ (IpIgLλ, EU872022), Atlantic cod Ig λ (GmIgLλ, CAC03754). (PDF 25 kb)
sFig. 2
Amino acid alignment of catfish Vσ sequences. IGSV1-1*01, IGSV1-2*01, and IGSV1-3P are genomic sequences obtained from IgLσ gene clusters 3C and 5A, respectively. IGSV1-2*02 was obtained by RT-PCR and IGSV1-1*02 and IGSV1-1*03 by 5'-RACE. The conserved cysteine residues that form the intrachain disulfide bonds are labeled (*). Residues identical to the catfish sequence are shown as (.) and (–) represents gaps in the alignment, an arrow denotes the primer used for 5′ RACE. GenBank accession numbers are: IGSV1-1*01 (EU872024), IGSV1-2*01 and IGSV1-3P (EU872023), IGSV-2*02 (EU925377), IGSV1-1*02 (EU925378), IGSV1-1*03 (EU925379). (PDF 48 kb)
sFig. 3
Amino acid alignment of catfish Cσ sequences. IGSC1*01 is a genomic sequence obtained from the IgLσ gene cluster 3C. IGSC1*02, IGSC1*03 and IGSC1*04 are representative sequences obtained by 5'-RACE. In total, 23 RACE products were amplified from PBL cDNA made from a single catfish: three IGSC1*03, five IGSC1*02, and 15 IGSC1*04 products were sequenced. The conserved cysteine residues that form the intrachain disulfide bonds are labeled (*) and the cysteine that form the interchain disulfide bond is marked by (↓). The number of amino acids is on the right. Residues identical to the catfish sequence are shown as (.). GenBank accession numbers are: IGSC1*01 (EU872024), IGSC1*02 (EU925380), IGSC1*03 (EU925381), IGSC1*04 (EU925382). (PDF 16 kb)
sFig. 4
Comparison of nucleotide sequence identities of teleost Vσ and Vλ sequences. Pairwise alignments were performed using CLUSTAL W and yellow highlights indicate >75% identity. VS and Vl subgroup membership is indicated above the sequence names and Genbank accession numbers are listed in the figure. Accession numbers referring to genomic sequences containing more than 1 V are labeled −1, −2, or −3, depending on the order of the V in the gene fragment. Species abbreviations are: Cc carp, Dr zebrafish, Om rainbow trout, Ip channel catfish, If blue catfish, Gm Atlantic cod. (PDF 7 kb)
sFig. 5
Amino acid alignment of catfish Vλ sequences. IGLV1-1*01 is a genomic sequence obtained from the IgLλ gene cluster 2A. IGLV1-1*02 and IGLV1-1*03 were obtained by RT-PCR and IGLV1-2*01 by 5′-RACE. The conserved cysteine residues that form the intrachain disulfide bonds are labeled (*) and the two cysteines found at the 3' end are labeled (^). Residues identical to the catfish sequence are shown as (.) and (–) represents gaps in the alignment, arrows denotes the primers used for 5′ RACE and genomic PCR. GenBank accession numbers are: IGLV1-1*01 (EU872025), IGLV1-1*02 (EU925383), IGLV1-1*03 (EU925384), IGLV1-2*01 (EU925385), IGLV1-2*02 (FJ716623). (PDF 49 kb)
sFig. 6
Amino acid alignment of representative IGL sequences. Sequences are aligned as recommended by Das et al. 2008 and molecular markers that distinguish κ, λ, and σ isotypes are boxed in blue (or red when not valid for teleosts). The amino acids conforming to the criteria proposed by Das et al. 2008 are highlighted in yellow and additional markers not defined by Das are highlighted in green. CDR regions are removed to optimize the alignment and their respective locations are marked by triangles. Amino acid numbers are according to the standardized rules of the IMGT unique numbering and are shown above the sequence (Lefranc et al. 2005). Accession numbers are catfish G-type (IpG, L25533); Carp L1 (CcL1, V and J (AB073328), C (AB015904)); zebrafish L1 (DrL1, AF246185); rainbow trout L1 (OmL1, X65260); Atlantic cod L1 (GmL1, V and J (AF104898.3), C (X68514)); catfish F-type (IpF, U25705); carp L3 (CcL3, V and J (BAB91006), C (BAB90987)); zebrafish L3 (DrL3, AB246193); X. laevis κ (Xlκ, V (L15570), J and C (M94392)); green anole κ (Acκ, FG748778); human κ (Hsκ, AAH73764); channel catfish λ (Ipλ, EU872022); blue catfish λ (Ifλ, CK403484); rainbow trout λ (Omλ, BX861350); Atlantic cod λ (Gmλ, AJ293807); X. laevis III (XlIII, L76579); green anole λ (Acλ, FG746006); chicken λ (Ggλ, K00678); human λ (Ηsλ, ABU90549); catfish σ (Ιpσ, EU872023); carp L2 (CcL2, V and J (AB091113), C (BAC81202)); zebrafish L2 (DrL2, AAG31698); rainbow trout L2 (OmL2, AAB41310); Atlantic cod σ (Gmσ, V (EX732557), J and C (EC907978)); and X. laevis σ (Xlσ, AAH82413). (PDF 69 kb)
Rights and permissions
About this article
Cite this article
Edholm, ES., Wilson, M., Sahoo, M. et al. Identification of Igσ and Igλ in channel catfish, Ictalurus punctatus, and Igλ in Atlantic cod, Gadus morhua . Immunogenetics 61, 353–370 (2009). https://doi.org/10.1007/s00251-009-0365-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-009-0365-z