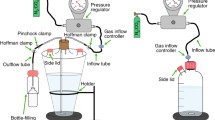
Abstract
The impact of the oxygen supply rate (OSR) on the metabolic activity and on the composition of hexadecane-degrading bacterial communities in a quasi-anoxic milieu (nominal DOT=0%) was studied in continuous cultures containing intertidal sediment. The dilution rate was kept constant at 0.035 h−1. The OSR was stepwise reduced from 3.5 mmol O2L−1 h−1 to 0.06 mmol O2L−1 h−1. Activity was determined by analyzing the respiration quotient (RQ) and the rates of hexadecane degradation (QHex), of hexadecane mineralization, and of protein production (PPR). The community composition and size were investigated by fluorescence in situ hybridization (FISH), by dilution plating (colony forming units or CFU), and by most probable number (MPN). The culture showed an aerobic hexadecane metabolism down to an OSR of 0.35 mmol O2L−1 h−1. Below this OSR, anaerobic metabolism was initiated. The relationship among the RQ, PPR, QHex, and the OSR can be approximated by hyperbola (Michaelis-Menten kinetics). We suggest that the metabolic adaptation of the culture to low OSRs is due to regulation of protein expression and enzyme activity. Reducing the OSR resulted in minor but significant changes in the concentration of different physiological and phylogenetic groups. This means that, in addition to protein expression and activity regulation, the adaptation of the population to low OSRs is due to changes in the community composition.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- QHEX :
-
Hexadecane consumption rate (mg L−1 h−1)
- QO2 :
-
Oxygen consumption rate (mg L−1 h−1)
- QCO2 :
-
Carbon dioxide consumption rate (mg L−1 h−1)
- PPR:
-
Protein production rate (mg L−1 h−1)
- OSR:
-
Oxygen supply rate (mmol L−1 h−1)
- RQ:
-
Respiration quotient (mol of produced CO2 per mol of consumed O2)
- Suboxic:
-
Refers to a milieu with an oxygen concentration ranging between 9 and 0.8 μmol O2 L−1
- Quasi-anoxic:
-
Refers to a habitat in which oxygen is not detectable by analytical methods (e.g., oxygen electrodes), although the habitat is supplied with small amounts of oxygen
- DOT:
-
Dissolved oxygen tension
- K AOSR :
-
Refers to the oxygen supply rate of a culture that permits the half-maximum rate of a specific metabolic activity
- K HOSR :
-
Refers to the oxygen supply rate of a culture that permits a half-maximum hexadecane consumption rate
- K POSR :
-
Refers to the oxygen supply rate of a culture that permits a half-maximum protein production rate
References
Aeckersberg F, Back F, Widdel F (1991) Anaerobic oxidation of saturated hydrocarbons to CO2 by a new type of sulfatereducing bacterium. Arch Microb 155:5–14
Alef K (1991) Methodenhandbuch Bodenmikrobiologie: Aktivitäten, Biomasse, Differenzierung. Ecomed, Landsberg/Lech, pp
Andersen FO, Helder W (1987) Comparison of oxygen microgradients, oxygen flux rates and electron transport sytem activity in coastal marine sediments. Mar Eco Progr Ser 37:259–264
In: Atlas RM (1984) Petroleum Microbiology. Macmillan, New York, pp
Berthe-Corti L, Bruns A (1999) The impact of oxygen tension on cell density and metabolic diversity of microbial communities in alkane degrading continuous-flow cultures. Microb Ecol 37:70–77
Berthe-Corti L, Bruns A, Hulsch R (1997) Semi-continuous-flow cultures with marine sediment suspensions containing non-polar carbon sources—culture control by a pneumatic sediment suspension dosage system. J Microbiol Methods 29:129–137
Berthe-Corti L, Ebenhöh W (1999) A mathematical model of cell growth and alkane degradation in Wadden Sea sediment suspensions. BioSystems 49:161–189
Borowski JD, Johnson MJ (1967) Experimental evaluation of liquid film resistance in oxygen transport to microbial cells. Appl Microbiol 15:1483–1488
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bruns A, Berthe-Corti L (1998) In situ detection of bacteria in continuous-flow cultures of seawater sediment suspensions with fluorescently labelled rRNA-directed oligonucleotide probes. Microbiology 144:2783–2790
Bruns A, Berthe-Corti L, Muricauda ruestringensis gen. nov., sp. nov., a facultative anaerobic species from German North Sea intertidal sediment. Submitted 2001
Chen J, Tannahill AL, Shuler ML (1985) Design of a system for the control of low dissolved oxygen concentrations: Critical oxygen concentrations for Azotobacter vinelandii and Escherichia coli. Biotech Bioeng 27:151–155
Colby J, Stirling DI, Dalton H (1977) The soluble methane mono-oxygenase of Methylococcus capsulatus (Bath): Its ability to oxygenate n-alkanes, n-alkenes, ethers, and alicyclic, aromatic and heterocyclic compounds. Biochem J 165:395–402
Dilling W, Cypionka H (1990) Aerobic respiration in sulfatereducing bacteria. FEMS Microbiol Ecol Lett 71:123–128
Dixon WJ (1992) BMDP Statistical Software Manual (version 7.0). University of California Press, Berkeley, pp
Ehrenreich P, Behrends A, Helder J, Widdel F (2000) Anaerobic oxidation of alkanes by newly isolated denitrifying bacteria. Arch Microbiol 173:58–64
Fritzsche Ch (1994) Degradation of pyrene at low defined oxygen concentrations by a Mycobacterium sp. Appl Environ Microbiol 60:1687–1689
Hulsch R, Berthe-Corti L (1994) Methoden zur Prozeßkontrolle bei geringer Sauerstoffzehrung im mikroaeroben Milieu. BioEngineering 1:11–14
Kukor JJ, Olsen RH (1996) Catechol 2,3-dioxygenases functional in oxygen-limited (hypoxic) environments. Appl Environ Microbiol 62:1728–1740
Leahy JG, Colwell RR (1990) Microbial degradation of hydrocarbons in the environment. Microbiol Rev 54:305–315
Le Gall J, Xavier AV (1996) Anaerobes response to oxygen: The sulfate-reducing bacteria. Anaerobe 9:1–9
Levring T (1946) Some culture experiments with Ulva and artificial sea water. K Fysiogr Sallsk Lund Forh 16:45–56
Llobet-Brossa E, Rosselló-Mora R, Amann R (1998) Microbial community composition of Wadden Sea sediments as revealed by fluorescence in situ hybridization. Appl Environ Microbiol 64:2691–2696
Marshall Ch, Frenzel P, Cypionka H (1993) Influence of oxygen on sulfate reduction and growth of sulfate-reducing bacteria. Arch Microbiol 159:168–173
Michaelsen M, Hulsch R, Höpner Th, Berthe-Corti L (1992) Hexadecane mineralization in oxygen-controlled sedimentseawater cultivations with autochthonous microorganisms. Appl Environ Microbiol 58:3072–3077
Minz D, Fishbain S, Green SJ, Muyzer G, Cohen Y, Rittmann BE, Stahl DA (1999) Unexpected population distribution in a microbial mat community: Sulfate-reducing bacteria localized to the highly oxic chemocline in contrast to a eucaryotic preference for anoxia. Appl Environ Microbiol 65:4659–4665
Oberzill W (1967) Mikrobiologische Analytik. Verlag Carl Hanser, Nürnberg, pp
Pearl HW, Pinckney JL (1996) A mini-review of microbial consortia: Their roles in aquatic production and biogeochemical cycling. Microb Ecol 31:225–247
Poole RK, Ingledew WJ (1987) Pathways of electrons to oxygen. Neidhardt FC (ed) Escherichia coli and Salmonella typhimurium—Cellular and Molecular Biology. American Society for Microbiology, Washington, DC, pp 170–200
Reichenbach H (1989) Genus I. Cytophaga Winogradsky 1929, 577,AL emend. In: Krieg NR, Holt JG (eds) Bergey's Manual of Systematic Bacteriology, vol 3. Williams & Wilkins, Baltimore, pp 2015–2050
Rice CW, Hempfling WP (1978) Oxygen-limited continuous culture and respiratory energy conservation in Escherichia coli. J Bacteriol 134:115–124
Rueter P, Rabus R, Wilkes H, Aeckersberg F, Rainey FA, Jannasch HW, Widdel F (1994) Anaerobic oxidation of hydrocarbons in crude oil by new types of sulphate-reducing bacteria. Nature 372:455–458
Sass H, Cypionka H, Babenzien D-H (1997) Vertical distribution of sulfate-reducing bacteria at the oxic-anoxic interface in sediments of the oligotrophic Lake Stechlin. FEMS Microbiol Ecol 22:245–255
Shaler TA, Klecka GM (1986) Effects of dissolved oxygen concentration on biodegradation of 2,4-dichlorphenoxyacetic acid. Appl Environ Microbiol 51:950–955
Sinclair CG, Ryder DN (1975) Models for the continuous culture of microorganisms under both oxygen and carbon limiting conditions. Biotech Bioeng 17:375–398
So CM, Young LY (1999) Initial reactions in anaerobic alkane degradation by a sulfate reducer, strain AK-01. Appl Environ Microbiol 65:5532–5540
Tyson RV, Pearson TH (1991) Modern and ancient continental shelf anoxia: an overview. In: Tyson RV, Pearson TH (eds) Modern and Ancient Shelf Anoxia, Geological Society Special publication no 58. Bulington House, London, pp 1–24
Vannelli T, Logan M, Arciero DM, Hooper AB (1990) Degradation of halogenated aliphatic compounds by the ammonia-oxidizing bacterium Nitrosomonas europaea. Appl Environ Microbiol 56:1169–1171
Vollbrecht D (1982) Oxygen-dependent switch-over from respiratory to fermentative metabolism in the strictly aerobic Alcaligenes eutrophus. Eur J Appl Microbiol Biotechnol 15:117–122
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Berthe-Corti, L., Bruns, A. Composition and activity of marine alkane-degrading bacterial communities in the transition from suboxic to anoxic conditions. Microb Ecol 42, 46–55 (2001). https://doi.org/10.1007/s002480000082
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002480000082