Abstract
Ixodes ticks transmit infectious agents and also harbor their own parasites and symbionts. The presumptive endosymbiont of Ixodes ricinus, ‘Candidatus Midichloria mitochondrii’, has a unique ability to invade mitochondria within tick ovarian cells and is transovarially transmitted with 100% efficiency. A closely related bacterium, provisionally named Montezuma (now ‘Candidatus Lariskella arthropodarum’), was isolated from the Ixodes persulcatus ticks and human blood in 2004 as well as from Ixodes pavlovskyi in 2015. These microorganisms belong to the family ‘Candidatus Midichloriaceae fam. nov.’ and were detected not only in tick salivary glands, but also in animal blood. Nevertheless, the relative importance of vertical and horizontal routes for their transmission or maintenance in natural tick populations remains unclear. We analyzed the prevalence of L. arthropodarum and M. mitochondrii in two sympatric zones, where I. persulcatus/I. ricinus and I. persulcatus/I. pavlovskyi cohabit and produce interspecific hybrids. A specificity of the associations of L. arthropodarum with I. persulcatus (100%) and M. mitochondrii with I. ricinus (96.2%) was observed in the sympatric zone in Estonia, possibly showing poor contribution of the horizontal route to the overall prevalence of endosymbionts. L. arthropodarum was observed probably multiplying in I. pavlovskyi and also subjected to transovarial transmission, but much less efficiently compared to I. persulcatus. We revealed two new genetic variants of the rrl-rrf intergenic spacer of L. arthropodarum isolated from I. pavlovskyi ticks that possibly could indicate an ongoing process of adaptation of the microorganism to a new host species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Ixodes ticks transmit multiple infectious agents and also harbor their own parasites and symbionts. Since the discovery of the alphaproteobacterium ‘Candidatus Midichloria mitochondrii’, which has a unique ability to invade the mitochondria within ovarian cells of Ixodes ricinus L. [1–3], many studies have been focused on its biological features, evolution, and potential pathogenicity. In particular, 100% of Ixodes ricinus females have been reported to be infected with M. mitochondrii, whereas the prevalence of this microorganism in males did not exceed 44% [3, 4]. Moreover, tick sex determines the relative abundance of the presumptive endosymbiont, with females harboring 1000-fold more copies of M. mitochondrii genome than males [4, 5]. The observed differences between sexes led to the hypothesis that M. mitochondrii, although being transmitted from an infected female to both female and male eggs transovarially, can efficiently multiply only in females while being somehow lost during specialization of nymphs to adult males [3–5]. It is worth noting that M. mitochondrii has been found in a number of hard ticks [6, 7], tick salivary glands as well as in the blood of mammals being parasitized by ticks [8–10]. Thus, both vertical and horizontal routes of transmission have been shown for M. mitochondrii; however, their relative importance in overall prevalence is still to be elucidated.
In 2004, Mediannikov et al. isolated a new bacterium, phylogenetically close to M. mitochondrii, from Ixodes persulcatus Schulze females, and human blood in the Russian Far East; its prevalence in adult I. persulcatus females was 97.1% [11]. This bacterium was provisionally named ‘Montezuma’. Later, Montezuma was detected in the European part of Russia where its prevalence in adult females was shown to be from 8.4 to 51.9%, and in adult males from 2.5 to 16% [12, 13]. Although the prevalence of Montezuma in I. persulcatus females was shown to be lower than those of M. mitochondrii in I. ricinus, the hypothesis about the loss of endosymbiont during male development was extended to Montezuma in I. persulcatus as well [12]. This microorganism has been scarcely studied, and little is known about its biological features. Recently, the DNA of Montezuma has been detected in Ixodes pavlovskyi Pomerantsev females in a tick microbiome analysis [14], as well as in the salivary glands of I. persulcatus females in Japan [15]. Sequence analysis of the 16S rRNA gene, the only genetic marker used, showed almost no genetic diversity in this bacterium from different territories (Arkhangelsk, Novgorod Regions and St. Petersburg in the North West Russia; Russian Far East) [13].
M. mitochondrii and Montezuma colonize closely related tick host species, yet they are not the closest relatives themselves. These two microorganisms are grouped as related alphaproteobacteria notably associated with a wide range of hosts, from amoebae to a variety of invertebrates [16–20]. Since M. mitochondrii was the first organism formally described for this phylogenetic group, it has been proposed to designate it as ‘Midichloria and like organisms (MALOs)’ [8], and then to elevate it to the family rank, ‘Candidatus Midichloriaceae fam. nov.’ within the order Rickettsiales [21]. Phylogenetic analysis revealed that the bacterium Montezuma is closer to endosymbionts isolated from stinkbugs of different families, as well as fleas, than it is to M. mitochondrii. It has been proposed that this clade, including Montezuma, forms a new species, ‘Candidatus Lariskella arthropodarum’ [18]. Lariskella arthropodarum in stinkbugs was shown to be concentrated in oocytes in the ovaries of adult females, indicating the vertical transmission of the symbiont through the host generations. No significant difference in L. arthropodarum prevalence between females and males was previously shown [18].
The sympatric zones of overlapping distribution of two species represent an opportunity for the study of microorganism-host associations. Recently, we have shown that sympatric zones of Ixodes ticks are also zones of hybridization between these tick populations [22, 23]. Analysis of hybrid populations, i.e., populations containing individuals resulting from the breeding of two tick species, accompanied by the detection of associated symbionts could give clues on how they are transmitted in natural host populations. For example, genotyping of Wolbachia, a well-known intracellular, maternally inherited endosymbiont of insects, accompanied by a parallel characterization of the associated host mitochondrial and nuclear DNA diversity, revealed a complex set of mechanisms of its transfer, including co-speciation, horizontal transfer, and hybrid introgression [24–26]. In the present work, we have estimated the prevalence and relative abundance of L. arthropodarum (previously Montezuma) in I. persulcatus, I. ricinus, and I. pavlovskyi ticks, as well as in their hybrids from the sympatric zones in Estonia and West Siberia. Discussion on the relative importance of vertical and horizontal routes of transmission for the establishment of L. arthropodarum and M. mitochondrii in natural populations of their Ixodes hosts is presented. Moreover, genetic analysis revealed new variants of the rrl-rrf intergenic spacer sequences of L. arthropodarum isolated from I. pavlovskyi ticks that could possibly indicate an ongoing process of adaptation of the symbiont to a new host species.
Materials and Methods
To determine the prevalence of L. arthropodarum, ticks were collected by flagging in the sympatric zone of I. persulcatus/I. ricinus in Estonia in 2006–2008 (n = 250) and in the sympatric zone of I. persulcatus/I. pavlovskyi in the Tomsk region (Russia) in 2013–2014 (n = 473), as well as in two regions in Russia where only I. persulcatus was found: the Komi Republic (Syktyvkar) in 2014 (n = 120) and the Sverdlovsk region (Yekaterinburg) in 2015 (n = 120). All ticks were analyzed individually. Detailed information on the methods of sample preparation, DNA extraction, and detection of tick hybrids is given in previous papers [22, 23]. Briefly, interspecific tick hybrids were revealed by carrying out two real-time PCRs with species-specific probes for both mitochondrial (cox1) and nuclear (ITS2) markers, with subsequent identification of three groups of specimen: (1) ‘pure’ species with both mtDNA and nucDNA belonging to the same species; (2) hybrids with a mix of ITS2 alleles of two species; and (3) hybrids with mtDNA and nucDNA indicating different species (mitochondrial introgression).
We developed new primers and TaqMan probes to detect and estimate the abundance of L. arthropodarum and M. mitochondrii DNA by real-time PCR (Table 1). Primers and probes were designed based on the alignment of the 16S rDNA sequences of L. arthropodarum (uncultured Rickettsiales bacterium Montezuma GenBank AF493952) and M. mitochondrii (GenBank CP002130). The mitochondrial 16S rRNA gene fragment, amplified for all three Ixodes species with the same primers and probe [27], was used as a reference gene to assess the quality of DNA extraction and amplification. Two PCR reactions were performed separately on the ABI 7500 thermocycler (Applied Biosystems™, USA). The PCR conditions were as follows: 95 °C for 15 min; 40 cycles at 95 °C for 15 s, 68 °C (for the bacterial target gene) or 60 °C (for the Ixodes target gene) for 1 min. The relative abundance of L. arthropodarum was estimated as a ratio between threshold cycles C t of an analyzed and a reference gene using the formula Lariskella/mitochondria ratio = 2^(C t tick 16S rDNA − C t Lariskella 16S rDNA). Statistical analysis and generation of a boxplot graph were carried out with Microsoft Excel (Microsoft™ Corp., USA) and Statistica software v.13. (StatSoft™, USA). The prevalences of L. arthropodarum and M. mitochondrii in different tick species were estimated using 95% confidence interval (95%CI) and compared with t test for the comparison of percentages (Tables 2 and 3). Lariskella/mitochondria ratios between different tick species, sexes, and geographical origins were compared using Mann-Whitney U test (Fig. 1). P values less than 0.05 were considered to be significant.
The L. arthropodarum 16S rDNA/Ixodes sp. mitochondrial 16S rDNA ratios between different sexes and species of ticks. The number of samples for each group is given in Table 3. The boxes represent the 25th and 75th percentiles of the values, with the line inside the boxes marking the median (Me). The whiskers indicate the maximum and minimum values. Statistically significant differences between tick species and geographical territories (Mann-Whitney test, p < 0.05) are shown by an asterisk
Primers for the amplification of rrl (23S rDNA) - rrf (5S rDNA) intergenic spacer (rrl-rrf IGS) of L. arthropodarum were designed in two stages. In the first stage, we designed the primers Midi-5S-F and Midi-23S-R2 based solely on the annotated whole-genome sequence of M. mitochondrii; they were used to amplify the corresponding genome fragment of L. arthropodarum isolated from I. persulcatus (Table 1). In the second stage, the obtained sequence, 405 bp in length, was used to design L. arthropodarum-specific primers Mtz-5S-F and Mtz-23S-R. They were used subsequently to amplify and sequence rrl-rrf IGS fragments for 62 samples of I. persulcatus and I. persulcatus/I. pavlovskyi hybrids (with mtDNA of I. persulcatus) from different geographical regions (25 samples from Tomsk region, 7—Estonia, 7—Krasnoyarsk, 8—Vladivostok, 5—Komi Republic, 4—Urals, 2—Altai, 2—Tyva Republic, 2—Arkhangelsk region), as well as for 23 I. pavlovskyi and I. persulcatus/I. pavlovskyi hybrids (with mtDNA of I. pavlovskyi). For the samples with low abundance of L. arthropodarum, where no amplification was detected, a nested PCR was performed using two pairs of primers (Table 1). PCR reactions were performed on the Veriti® 60-well Thermal Cycler (Applied Biosystems™, USA) under the thermocycling conditions as follows: initial denaturation at 94 °C for 1 min; 42 cycles at 94 °C for 20 s, 53 °C or 61 °C for 20 s, 72 °C for 40 s; final elongation at 72 °C for 5 min.
Nucleotide sequences of the rrl-rrf IGS were determined on the ABI PRIZM 3130 Genetic Analyzer (Applied Biosystems™, USA). Sequence alignment and analysis were performed using the software MEGA 6 [28]. Positions of the 23S and 5S rRNA genes flanking the intergenic spacer were determined compared to the corresponding bacterial rRNA sequences from the Rfam database (http://rfam.xfam.org).
Results
Prevalence of L. arthropodarum and M. mitochondrii in Ticks in the I. persulcatus/I. ricinus Sympatric Zone
Genetic features of the detected tick hybrids, where DNA analysis shows a mix of two species, and their proportion relative to ‘pure’ species in natural populations have been described previously [22, 23]. The prevalence of L. arthropodarum and M. mitochondrii in the ‘pure’ species I. persulcatus and I. ricinus as well as in their hybrids are presented in Table 2. Tick hybrids were grouped based on the species identified by the analysis of mtDNA, in other words, based on their maternal species; no hybrids with a mix of mitochondria were detected. Although I. persulcatus and I. ricinus in the sympatric zone probably feed on the same animals, a clear association between M. mitochondrii and I. ricinus and between L. arthropodarum and I. persulcatus was observed. Only two I. persulcatus males harbored the DNA of M. mitochondrii and no sample of I. ricinus infected with L. arthropodarum was detected. The prevalence of L. arthropodarum in I. persulcatus females and M. mitochondrii in I. ricinus females was 86–88% (Table 2); in males it was significantly lower (Table 2). Detection of both endosymbionts in nymphs revealed intermediate values, 77.8 and 54.5%, respectively. Association of L. arthropodarum and M. mitochondrii with their hosts was observed for the tick hybrids as well, but because of their small number statistical analysis was not performed.
Prevalence and Relative Abundance of L. arthropodarum in Ticks I. persulcatus, I. pavlovskyi, and their Hybrids
The prevalence of L. arthropodarum was determined for adult I. persulcatus and I. pavlovskyi ticks of both sexes collected in the Tomsk region, a sympatric and hybridization zone, as well as for I. persulcatus ticks from two regions where only I. persulcatus is found (Table 3). The prevalence of L. arthropodarum in I. persulcatus females did not depend on their origin and was about 90% (95%CI = 84.3–93.5%), 3-fold the observed in males with 30% on average (95%CI = 23.2–36.8%). The percentage of L. arthropodarum-infected ticks was significantly higher for I. persulcatus males than for both females and males of I. pavlovskyi (t test, p < 0.05). At the same time, no statistical difference between the prevalence of L. arthropodarum in females and males of I. pavlovskyi was found. I. persulcatus/I. pavlovskyi hybrids were divided into two groups as follows: (1) Hybrper—the hybrids with mtDNA of I. persulcatus and nuclear DNA of either a mix of both species or I. pavlovskyi (hybrids with mitochondrial introgression); (2) Hybrpav—the hybrids with mtDNA of I. pavlovskyi and nuclear DNA of either a mix of both species or of I. persulcatus. Statistical analysis of the two groups of hybrids revealed the same pattern as shown for the ‘pure’ species, namely, that L. arthropodarum is much more frequently found in Hybrper females than in males and less frequently in both females and males of Hybrpav than in Hybrper males (Table 3).
Relative abundance of L. arthropodarum in ticks was performed by qPCR determination based on the C t ratio for the Lariskella/tick target genes (Fig.1). The median Lariskella/mitochondria ratio in I. persulcatus females was 0.5, 250-fold greater than those in I. persulcatus males (0.002), which, in turn, was significantly greater than the median value in I. pavlovskyi, 1.5·10−5 in females and 6.1·10−5 in males (Fig. 1). It is interesting that the comparison of Lariskella/mitochondria ratios between I. persulcatus females originating from different regions showed statistically significant differences: the values are maximal in female ticks from Syktyvkar (North-West Russia), decreased in Yekaterinburg (Urals) and minimal in Tomsk (Fig. 1). No statistical difference between Lariskella/mitochondria ratios in I. persulcatus males from the same regions, as well as between females and males of I. pavlovskyi, was found (Fig. 1). Comparison between hybrids was not performed because of the small number of Hybrper samples.
Genetic Diversity of L. arthropodarum in I. persulcatus and I. pavlovskyi Ticks
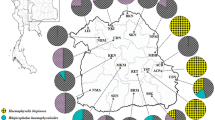
Since low genetic diversity of 16S rDNA sequences has been shown for L. arthropodarum isolated from I. persulcatus [13], we decided to use another, presumably more variable genetic marker, the rrl-rrf intergenic spacer. Design of the primers for its amplification was complicated by the absence of a whole-genome sequence of L. arthropodarum. In the present study, we obtained a new sequence of rrl-rrf IGS of L. arthropodarum isolated from Ixodes ticks (GenBank Acc. Nos. KX578087–KX578171). The length of the spacer was 120 bp, 21 bp longer than in M. mitochondrii with 99 bp. Alignment of the 62 rrl-rrf IGS sequences of L. arthropodarum from I. persulcatus, including those collected in the sympatric zone, and Hybrper showed 100% identity, i.e., all samples harbored the same IGS variant, designated here as Per1 (GenBank Acc. No. KX578110). However, analysis of I. pavlovskyi and Hybrpav ticks revealed two new IGS variants in 18 out of 23 samples; they were designated as Pav1 (KX578087) and Pav2 (KX578104). The former had a nucleotide substitution T200C (position numbers are given according to KX578087) and was found in 17 samples; the latter had two substitutions compared to Per1, T200C and T227C, and was observed in only one sample (Fig. 2).
Distribution of L. arthropodarum rrl-rrf IGS variants between different localities and tick species. The size of a circle corresponds to the number of the L. arthropodarum sequences obtained (85 in total). Tomsk region, a sympatry zone where both tick species were sampled together, is shadowed in gray; samples from other regions are presented by I. persulcatus only. The L. arthropodarum rrl-rrf IGS variants are indicated by colors; GenBank accession numbers and the sequences’ variation are indicated
Discussion
Two main routes of transmission of microorganisms from one tick to another are known: vertical (transovarial), from female to its progeny, and horizontal, either by feeding on an infected animal or by co-feeding with infected ticks simultaneously on the same host [29]. Once a tick acquires a microorganism, two scenarios are possible. The first one assumes that this microorganism is unable to multiply in tick tissues and is consequently digested in the tick gut, although even in this case the microorganism’s DNA can be detected by sensitive molecular genetics methods. For example, animal DNA, equally exposed to digestion, can be detected in ticks several months after the bloodmeal [30, 31]. An alternative scenario is that the microorganism multiplies in the tick tissues, thus undergoing natural selection imposed by a host.
Nowadays, it is well known that M. mitochondrii can be efficiently transmitted transovarially. However, its DNA has been detected in animal blood which suggests the possibility of horizontal transmission [6]. As an example, horizontal transmission of a phylogenetically related microorganism, Anaplasma phagocytophilum, by co-feeding has been demonstrated experimentally [32–34]. Since DNA of L. arthropodarum has been detected in human blood [11] and I. persulcatus salivary glands [15], we suggest horizontal transmission for this bacterium as well.
Since it is virtually impossible to distinguish between microorganisms of the same species acquired either transovarially or horizontally, we have analyzed as a model the sympatric zone of two tick species, each of them associated with its own endosymbiont, I. ricinus with M. mitochondrii and I. persulcatus with L. arthropodarum. The sympatric zone of these tick species is located in the East European Plain and the Baltic countries; ticks not only co-habit the same area but also produce interspecific hybrids [23]. Our assumption was that the horizontal route, which is generally accidental, would be seen as detection of endosymbionts in non-specific hosts. Nevertheless, analysis of 144 I. persulcatus and 86 I. ricinus ticks, as well as a small number of their hybrids from Estonia revealed almost complete specificity of tick-symbiont association. Only in two I. persulcatus males, which consist of 1.4% of the total number of ticks of this species, did we detect the non-specific endosymbiont M. mitochondrii; this value probably reflects the relative contribution of the horizontal transmission route in the overall prevalence. No I. ricinus tick harbored DNA of L. arthropodarum. Therefore, we are inclined to think that the vertical transmission route is dominant for M. mitochondrii and L. arthropodarum in natural tick populations, while the horizontal route does not contribute much to their prevalence in ticks or could not be detected by the methods used in this study.
Taken together, if a significant prevalence of M. mitochondrii or L. arthropodarum in ticks is detected, it is probably due to a specific vertical transmission route accompanied by multiplication of endosymbionts in host reproductive organs. It is a situation that was observed for the second sympatric zone, that of I. persulcatus and I. pavlovskyi. The distribution range of I. pavlovskyi is disjoined into Western Siberian and Far Eastern parts, which almost completely overlaps with that of I. persulcatus. In this paper, we studied only the Western Siberian part, where interspecific hybrids had been previously detected [22]. The prevalence of L. arthropodarum was evenly high, about 90%, in I. persulcatus females and about 30% in males. Both high prevalence and relative abundance of L. arthropodarum in females proves the hypothesis assuming that this bacterium efficiently multiplies in I. persulcatus females but is probably lost in males during ontogenesis [12]. DNA of L. arthropodarum has been previously detected in I. pavlovskyi females from the Tomsk region [14]; however, no systematic studies to estimate its prevalence or characterize its genetic diversity have been made. We showed that, unlike I. persulcatus, there is no statistical difference between the prevalence of L. arthropodarum in I. pavlovskyi females (19.3%) and males (13.7%); the relative abundance of this bacterium is also almost the same and about 100-fold smaller than those in I. persulcatus males. Moreover, tick hybrids grouped according to their maternal species showed a similar pattern. It could be suggested that the relatively low prevalence and quantity of L. arthropodarum in I. pavlovskyi is probably due to its accidental uptake with animal blood during tick feeding, but the results obtained for the I. ricinus/I. persulcatus sympatric zone do not support this hypothesis. Therefore, L. arthropodarum is most probably able to multiply in I. pavlovskyi and can be transmitted transovarially, but much less efficiently than in I. persulcatus, based on both the lower quantity and prevalence of the endosymbiont.
Since multiplying microorganisms necessarily undergo natural selection imposed by a host, it would be logical to assume that I. pavlovskyi ticks harbor an isolated population of L. arthropodarum, even at the genetic level. To test this assumption, we estimated genetic diversity of this microorganism isolated from I. persulcatus outside the sympatric zone, as a control, and from both species within the sympatric zone of the Tomsk region. The rrl-rrf intergenic spacer was used as a genetic marker. Although this marker is not used as widely as 16S rRNA gene, it has been shown to be twice as variable as rRNA genes [35]. It has also been used to study bacteria of the genus Rickettsia [36]. In this study, for the first time, we obtained the sequences of rrl-rrf IGS of L. arthropodarum, isolated from Ixodes ticks. They were 46.7% identical to the rrl-rrf IGS sequence of M. mitochondrii; the identity of the flanking 174 bp fragment of the 23S rDNA with the corresponding sequence of M. mitochondrii was 85.6%. The most interesting thing is that all studied I. persulcatus ticks collected in different regions in Russia and Estonia harbored the same variant of the rrl-rrf IGS (Fig. 2), while in 78% of I. pavlovskyi samples we found a specific nucleotide substitution, T200C. Another variant, with an additional substitution, T227C, was found in one I. pavlovskyi female. Formally, greater diversity of L. arthropodarum in I. pavlovskyi, yet minimal, could lead to the conclusion that I. pavlovskyi has been associated with this bacterium for longer time period, and that one particular strain then invaded I. persulcatus and spread to fixation. However, we think such a scenario to be unlikely because the prevalence and transmission efficiency of L. arthropodarum in I. persulcatus exhibit a strong symbiotic relationship, while its association with I. pavlovskyi is more likely to indicate an initial stage of adaptation. In fact, it has been shown previously that the sympatric zone between I. persulcatus and I. pavlovskyi in the Tomsk region is relatively recent and had been formed due to the spread of I. pavlovskyi northward and even its dominance near Tomsk city [37, 38]. The absence of new genetic variants of L. arthropodarum in I. persulcatus argues for its association with I. pavlovskyi and against the possibility of its accidental uptake with bloodmeal.
In conclusion, we can say that the biological features of L. arthropodarum in ticks, namely preferential multiplication in females, capacity for vertical transmission through the host generations and, probably, its high efficiency, resemble those of M. mitochondrii. Detection of new genetic variants of L. arthropodarum in I. pavlovskyi, even with a minor difference from the variant in I. persulcatus, could indicate an ongoing process of adaptation and formation of symbiotic relationships.
References
Sassera D, Beninati T, Bandi C, Bouman EA, Sacchi L, Fabbi M, Lo N (2006) ‘Candidatus Midichloria mitochondrii’, an endosymbiont of the tick Ixodes ricinus with a unique intramitochondrial lifestyle. Int. J. Syst. Evol. Microbiol. 56(Pt 11):2535–2540. doi:10.1099/ijs.0.64386-0
Lewis D (1979) The detection of rickettsia-like microorganisms within the ovaries of female Ixodes ricinus ticks. Z Parasitenkd 59(3):295–298. doi:10.1007/BF00927523
Beninati T, Lo N, Sacchi L, Genchi C, Noda H, Bandi C (2004) A novel alpha-Proteobacterium resides in the mitochondria of ovarian cells of the tick Ixodes ricinus. Appl. Environ. Microbiol. 70(5):2596–2602. doi:10.1128/AEM.70.5.2596-2602.2004
Lo N, Beninati T, Sassera D, Bouman EA, Santagati S, Gern L, Sambri V, Masuzawa T, Gray JS, Jaenson TG, Bouattour A, Kenny MJ, Guner ES, Kharitonenkov IG, Bitam I, Bandi C (2006) Widespread distribution and high prevalence of an alpha-proteobacterial symbiont in the tick Ixodes ricinus. Environ. Microbiol. 8(7):1280–1287. doi:10.1111/j.1462-2920.2006.01024.x
Sassera D, Lo N, Bouman EA, Epis S, Mortarino M, Bandi C (2008) "Candidatus Midichloria" endosymbionts bloom after the blood meal of the host, the hard tick Ixodes ricinus. Appl. Environ. Microbiol. 74(19):6138–6140. doi:10.1128/AEM.00248-08
Epis S, Sassera D, Beninati T, Lo N, Beati L, Piesman J, Rinaldi L, McCoy KD, Torina A, Sacchi L, Clementi E, Genchi M, Magnino S, Bandi C (2008) Midichloria mitochondrii is widespread in hard ticks (Ixodidae) and resides in the mitochondria of phylogenetically diverse species. Parasitology 135(4):485–494. doi:10.1017/S0031182007004052
Beninati T, Riegler M, Vilcins IM, Sacchi L, McFadyen R, Krockenberger M, Bandi C, O’Neill SL, Lo N (2009) Absence of the symbiont Candidatus Midichloria mitochondrii in the mitochondria of the tick Ixodes holocyclus. FEMS Microbiol. Lett. 299(2):241–247. doi:10.1111/j.1574-6968.2009.01757.x
Mariconti M, Epis S, Gaibani P, Dalla Valle C, Sassera D, Tomao P, Fabbi M, Castelli F, Marone P, Sambri V, Bazzocchi C, Bandi C (2012) Humans parasitized by the hard tick Ixodes ricinus are seropositive to Midichloria mitochondrii: is Midichloria a novel pathogen, or just a marker of tick bite? Pathogens and global health 106(7):391–396. doi:10.1179/2047773212Y.0000000050
Bazzocchi C, Mariconti M, Sassera D, Rinaldi L, Martin E, Cringoli G, Urbanelli S, Genchi C, Bandi C, Epis S (2013) Molecular and serological evidence for the circulation of the tick symbiont Midichloria (Rickettsiales: Midichloriaceae) in different mammalian species. Parasit. Vectors 6:350. doi:10.1186/1756-3305-6-350
Serra V, Cafiso A, Bazzocchi C (2016) Molecular and serological evidences of Midichloria mitochondrii transmission to vertebrate hosts during the tick bite. International Journal of Health, Animal Science and Food Safety 3 (1 s). doi:10.13130/2283-3927/7049
Mediannikov O, Ivanov LI, Nishikawa M, Saito R, Sidel’nikov Iu N, Zdanovskaia NI, Mokretsova EV, Tarasevich IV, Suzuki H (2004) Microorganism "Montezuma" of the order Rickettsiales: the potential causative agent of tick-borne disease in the Far East of Russia. Zh. Mikrobiol. Epidemiol. Immunobiol. 1:7–13 [in Russian]
Eremeeva ME, Oliveira A, Robinson JB, Ribakova N, Tokarevich NK, Dasch GA (2006) Prevalence of bacterial agents in Ixodes persulcatus ticks from the Vologda Province of Russia. Ann. N. Y. Acad. Sci. 1078:291–298. doi:10.1196/annals.1374.054
Eremeeva ME, Oliveira A, Moriarity J, Robinson JB, Tokarevich NK, Antyukova LP, Pyanyh VA, Emeljanova ON, Ignatjeva VN, Buzinov R, Pyankova V, Dasch GA (2007) Detection and identification of bacterial agents in Ixodes persulcatus Schulze ticks from the north western region of Russia. Vector Borne Zoonotic Dis 7(3):426–436. doi:10.1089/vbz.2007.0112
Kurilshikov A, Livanova NN, Fomenko NV, Tupikin AE, Rar VA, Kabilov MR, Livanov SG, Tikunova NV (2015) Comparative metagenomic profiling of symbiotic bacterial communities associated with Ixodes persulcatus, Ixodes pavlovskyi and Dermacentor reticulatus ticks. PLoS One 10(7):e0131413. doi:10.1371/journal.pone.0131413
Qiu Y, Nakao R, Ohnuma A, Kawamori F, Sugimoto C (2014) Microbial population analysis of the salivary glands of ticks; a possible strategy for the surveillance of bacterial pathogens. PLoS One 9(8):e103961. doi:10.1371/journal.pone.0103961
Erickson DL, Anderson NE, Cromar LM, Jolley A (2009) Bacterial communities associated with flea vectors of plague. J. Med. Entomol. 46(6):1532–1536
Richard S, Seng P, Parola P, Raoult D, Davoust B, Brouqui P (2009) Detection of a new bacterium related to 'Candidatus Midichloria mitochondrii' in bed bugs. Clin. Microbiol. Infect. 15(Suppl 2):84–85. doi:10.1111/j.1469-0691.2008.02244.x
Matsuura Y, Kikuchi Y, Meng XY, Koga R, Fukatsu T (2012) Novel clade of alphaproteobacterial endosymbionts associated with stinkbugs and other arthropods. Appl. Environ. Microbiol. 78(12):4149–4156. doi:10.1128/AEM.00673-12
Sunagawa S, Woodley CM, Medina M (2010) Threatened corals provide underexplored microbial habitats. PLoS One 5(3):e9554. doi:10.1371/journal.pone.0009554
Vannini C, Ferrantini F, Schleifer KH, Ludwig W, Verni F, Petroni G (2010) "Candidatus Anadelfobacter veles" and "Candidatus Cyrtobacter comes," two new rickettsiales species hosted by the protist ciliate Euplotes harpa (Ciliophora, Spirotrichea). Appl. Environ. Microbiol. 76(12):4047–4054. doi:10.1128/AEM.03105-09
Montagna M, Sassera D, Epis S, Bazzocchi C, Vannini C, Lo N, Sacchi L, Fukatsu T, Petroni G, Bandi C (2013) "Candidatus Midichloriaceae" fam. nov. (Rickettsiales), an ecologically widespread clade of intracellular alphaproteobacteria. Appl. Environ. Microbiol. 79(10):3241–3248. doi:10.1128/AEM.03971-12
Kovalev SY, Mikhaylishcheva MS, Mukhacheva TA (2015) Natural hybridization of the ticks Ixodes persulcatus and Ixodes pavlovskyi in their sympatric populations in Western Siberia. Infect. Genet. Evol. 32:388–395. doi:10.1016/j.meegid.2015.04.003
Kovalev SY, Golovljova IV, Mukhacheva TA (2016) Natural hybridization between Ixodes ricinus and Ixodes persulcatus ticks evidenced by molecular genetics methods. Ticks and tick-borne diseases 7(1):113–118. doi:10.1016/j.ttbdis.2015.09.005
Raychoudhury R, Baldo L, Oliveira DC, Werren JH (2009) Modes of acquisition of Wolbachia: horizontal transfer, hybrid introgression, and codivergence in the Nasonia species complex. Evolution 63(1):165–183. doi:10.1111/j.1558-5646.2008.00533.x
Schuler H, Bertheau C, Egan SP, Feder JL, Riegler M, Schlick-Steiner BC, Steiner FM, Johannesen J, Kern P, Tuba K, Lakatos F, Koppler K, Arthofer W, Stauffer C (2013) Evidence for a recent horizontal transmission and spatial spread of Wolbachia from endemic Rhagoletis cerasi (Diptera: Tephritidae) to invasive Rhagoletis cingulata in Europe. Mol. Ecol. 22(15):4101–4111. doi:10.1111/mec.12362
Reuter M, Pedersen JS, Keller L (2005) Loss of Wolbachia infection during colonisation in the invasive Argentine ant Linepithema humile. Heredity (Edinb) 94(3):364–369. doi:10.1038/sj.hdy.6800601
Schwaiger M, Cassinotti P (2003) Development of a quantitative real-time RT-PCR assay with internal control for the laboratory detection of tick borne encephalitis virus (TBEV) RNA. J. Clin. Virol. 27(2):136–145. doi:10.1016/S1386-6532(02)00168-3
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 30(12):2725–2729. doi:10.1093/molbev/mst197
Randolph SE, Gern L, Nuttall PA (1996) Co-feeding ticks: epidemiological significance for tick-borne pathogen transmission. Parasitol. Today 12(12):472–479. doi:10.1016/S0169-4758(96)10072-7
Kirstein F, Gray JS (1996) A molecular marker for the identification of the zoonotic reservoirs of Lyme borreliosis by analysis of the blood meal in its European vector Ixodes ricinus. Appl. Environ. Microbiol. 62(11):4060–4065
Moran Cadenas F, Rais O, Humair PF, Douet V, Moret J, Gern L (2007) Identification of host bloodmeal source and Borrelia burgdorferi sensu lato in field-collected Ixodes ricinus ticks in Chaumont (Switzerland). J. Med. Entomol. 44(6):1109–1117. doi:10.1093/jmedent/44.6.1109
Levin ML, Fish D (2000) Immunity reduces reservoir host competence of Peromyscus leucopus for Ehrlichia phagocytophila. Infect. Immun. 68(3):1514–1518. doi:10.1128/IAI.68.3.1514-1518.2000
Karpathy SE, Allerdice ME, Sheth M, Dasch GA, Levin ML (2016) Co-feeding transmission of the Ehrlichia muris-like agent to mice (Mus musculus). Vector Borne Zoonotic Dis 16(3):145–150. doi:10.1089/vbz.2015.1878
Ogden NH, Casey AN, Woldehiwet Z, French NP (2003) Transmission of Anaplasma phagocytophilum to Ixodes ricinus ticks from sheep in the acute and post-acute phases of infection. Infect. Immun. 71(4):2071–2078. doi:10.1128/IAI.71.4.2071-2078.2003
Massung RF, Lee K, Mauel M, Gusa A (2002) Characterization of the rRNA genes of Ehrlichia chaffeensis and Anaplasma phagocytophila. DNA Cell Biol. 21(8):587–596. doi:10.1089/104454902320308960
Vitorino L, Ze-Ze L, Sousa A, Bacellar F, Tenreiro R (2003) rRNA intergenic spacer regions for phylogenetic analysis of Rickettsia species. Ann. N. Y. Acad. Sci. 990:726–733. doi:10.1111/j.1749-6632.2003.tb07451.x
Romanenko V (2011) Long-term dynamics of population density and species composition of pasture ixodid ticks (Parasitiformes, Ixodidae) in anthropogenic and natural areas. Entomol Rev 91(9):1190–1195. doi:10.1134/S0013873811090132
Livanova N, Livanov S, Panov V (2011) Distribution of the ticks Ixodes persulcatus and Ixodes pavlovskyi on the boundary of the forest and forest-steppe zones in the Ob region. Entomol Rev 91(9):1184–1189. doi:10.1134/S0013873811090120
Acknowledgements
We are thankful to Dr. I. Golovljova (Department of Virology, National Institute for Health Development, Tallinn, Estonia) for the material collection and to Dr. K. Chamberlain (Rothamsted Research, UK) for his help in preparing the manuscript. This project was partly supported by Act 211 Government of the Russian Federation, agreement № 02.A03.21.0006, and partly by the Russian Foundation of Basic Research (No. 17-04-00709).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mukhacheva, T.A., Kovalev, S.Y. Bacteria of the Family ‘Candidatus Midichloriaceae’ in Sympatric Zones of Ixodes Ticks: Genetic Evidence for Vertical Transmission. Microb Ecol 74, 185–193 (2017). https://doi.org/10.1007/s00248-017-0932-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-017-0932-z