Abstract
Aquatic macroaggregates (flocs ≥0.5 mm) provide an important mechanism for vertical flux of nutrients and organic matter in aquatic ecosystems, yet their role in the transport and fate of zoonotic pathogens is largely unknown. Terrestrial pathogens that enter coastal waters through contaminated freshwater runoff may be especially prone to flocculation due to fluid dynamics and electrochemical changes that occur where fresh and marine waters mix. In this study, laboratory experiments were conducted to evaluate whether zoonotic pathogens (Cryptosporidium, Giardia, Salmonella) and a virus surrogate (PP7) are associated with aquatic macroaggregates and whether pathogen aggregation is enhanced in saline waters. Targeted microorganisms showed increased association with macroaggregates in estuarine and marine waters, as compared with an ultrapure water control and natural freshwater. Enrichment factor estimations demonstrated that pathogens are 2–4 orders of magnitude more concentrated in aggregates than in the estuarine and marine water surrounding the aggregates. Pathogen incorporation into aquatic macroaggregates may influence their transmission to susceptible hosts through settling and subsequent accumulation in zones where aggregation is greatest, as well as via enhanced uptake by invertebrates that serve as prey for marine animals or as seafood for humans.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Contamination of coastal waters with terrestrially derived fecal pathogens can result in morbidity and mortality in marine wildlife, as well as in humans who utilize nearshore waters for recreation or as a source of seafood [2, 5]. Yet, the precise waterborne transport mechanisms that govern pathogen distribution and disease transmission to susceptible hosts are largely unknown. Large (≥0.5 mm) aquatic aggregates or flocs are viewed as crucially important for vertical transport of nutrients in oceans [19] and may have a significant impact on the distribution of fecal pathogens. Whether pathogens are present in aquatic habitats as suspended particles or associated with aggregates is significant, because attached pathogens are likely to experience increased settling velocities with resulting accumulation in the benthos, as well as enhanced uptake by invertebrates that can act as disease vectors [12].
Pathogens that enter coastal waters through contaminated freshwater runoff may be especially prone to flocculation due to fluid dynamics and electrochemical changes that occur where fresh and marine waters mix [3, 8]. A recent investigation demonstrated that the zoonotic protozoan parasite, Toxoplasma gondii, can be incorporated into macroaggregates, especially in estuarine and marine waters [17]. To further investigate whether enhanced aggregation of pathogens in saline environmental waters is a process that can be generalized beyond T. gondii, this study examined whether fecal protozoa, bacteria, and a virus surrogate can incorporate into macroaggregates in fresh, estuarine, and marine waters. The protozoan parasites Cryptosporidium and Giardia were targeted because environmentally resistant (oo)cyst stages pose waterborne health risks to humans and animals and also because they have been previously detected in marine waters and shellfish [4, 13]. Salmonella was selected as a model fecal bacterium due to its known contamination of bivalves, as well as its potential to cause gastrointestinal infections [6, 20]. Viruses were also considered because they are a leading cause of waterborne and seafood-borne gastroenteritis [9, 10], and their smaller size may result in different aggregation dynamics. This study utilized the bacteriophage PP7 to examine virus aggregation behavior, as this virus is a validated surrogate for pathogenic enteric viruses in water filtration studies [14].
Materials and Methods
Freshwater was collected from San Lorenzo River (37.045 N, 122.072 W), estuarine water was collected at the mouth of San Lorenzo River at Monterey Bay (36.964 N, 122.013 W), and marine water was collected from the Santa Cruz pier (36.957 N, 122.017 W). The chemical properties of the different water types are provided as supplementary material (Table S1). Water samples (1,200 mL) were placed in glass bottles filled to the rim to reduce air space and spiked with 103 each of Cryptosporidium parvum oocysts and Giardia lamblia cysts (Waterborne™, Inc.), 104–105 cells of Salmonella enterica serovar Typhimurium labeled with a green fluorescent protein (GFP), and 109 PP7 virus particles. To simulate currents that enhance production of aggregates, a rolling apparatus was used to horizontally rotate bottles [15]. In each experiment, aggregation control bottles for each water type were spiked with the same numbers of organisms but instead placed on a stationary surface. In addition to environmental waters, pathogens were also added to bottles containing ultrapure water, to test for pathogen distribution in the absence of additional particles that could induce macroaggregate formation and to evaluate for potential of pathogens to aggregate upon themselves. Triplicate bottles were used for each treatment and water type (a total of 24 bottles).
After 24 h, rolled bottles were placed upright for 30 min to allow the readily visible aggregates to settle, and the top 1,100 mL operationally defined “aggregate-free” water was gently removed [12]. A photograph was obtained through the mouth of the open bottle to capture an image of particles in the remaining, aggregate-rich bottom water (100 mL). Photographs were processed using ImageJ image analysis software [11], and the volume of aggregates was estimated by applying a right cylinder equation using the measured long and short axis of aggregates [17]. Nonrolled bottles were gently inverted several times and then processed as described above. Spiked microorganisms were quantified in the top aggregate-free and the bottom aggregate-rich water fractions separately. Protozoa were enumerated using immunomagnetic separation followed by direct fluorescent antibody staining and microscopy [22]. Salmonella cells were quantified by membrane filtration and aerobic culture on Luria Bertani agar, followed by colony enumeration under UV illumination. The bacteriophage PP7 was quantified using real-time PCR [14].
A Kruskal–Wallis ANOVA test followed by a nonparametric post hoc pairwise comparison [18] was performed to evaluate whether the proportions of pathogens recovered from the aggregate-rich bottom water fractions differed among ultrapure, fresh, estuarine, and marine water samples. A Mann–Whitney U test was used to test whether the total volume of aggregates differed between rolled and nonrolled aggregation control bottles, within each water type. To approximate the numbers of microorganisms present in the aggregate-rich fraction that were due to association with macroaggregates, an aggregate enrichment factor (EF) was estimated for each targeted pathogen [19]. Pathogen EFs were calculated for rolled bottles by applying the following equation [17]:
where the numerator was calculated using the following equation:
The expected number of pathogens in the aggregate-rich water given no adherence to aggregates was approximated by multiplying the total numbers of recovered pathogens in each bottle by the proportion of water volume occupied by the bottom aggregate-rich fraction (100 mL in 1,200 mL total volume = 0.08). The number of pathogens in surrounding water was obtained from the concentration of pathogens in the top aggregate-free fraction.
Results and Discussion
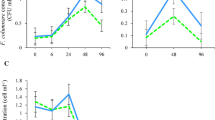
Significantly higher proportions of pathogens were recovered from the aggregate-rich fractions of rolled estuarine and marine water samples as compared with an ultrapure control or freshwater samples (Fig. 1). In nonrolled aggregation control bottles, the proportions of C. parvum, Salmonella, and PP7 recovered from the bottom water fractions did not significantly differ among water types. The observed increase in pathogen distribution within the aggregate-rich fractions of rolled estuarine and marine waters supports our hypothesis that diverse classes of harmful microorganisms display a greater degree of association with aquatic macroaggregates in saline waters, as compared with freshwater.
Proportions of microorganisms (median and range) recovered from the bottom (aggregate-rich) fractions of bottles containing ultrapure, fresh, estuarine, and marine water samples. Water types that do not share a superscript in common had significantly different proportions of organisms recovered from the bottom water fractions of rolled bottles. The asterisk denotes significantly higher proportions of pathogens recovered in the rolled marine (Giardia and PP7) and estuarine (Salmonella) bottles as compared with nonrolled bottles
For G. lamblia, the proportion of cysts recovered from the bottom water fractions of nonrolled estuarine bottles was significantly higher than in nonrolled freshwater bottles (P < 0.05, Fig. 1). These findings suggest that some incorporation of pathogens with sinking macroaggregates can occur in the absence of enhanced differential settling provided by the rolling apparatus. Image analysis of naturally present and/or experimentally formed macroaggregates showed that the total volume of aggregates in rolled bottles was significantly greater than the aggregate volume present in nonrolled bottles containing fresh, estuarine, and marine waters (P < 0.05, Fig. 2). In comparing pathogen aggregation between rolled and nonrolled samples in estuarine and marine waters, the proportions of all recovered pathogens appeared to be higher in the aggregate-rich fractions of rolled bottles but were only significantly higher for G. lamblia and PP7 in marine waters and for Salmonella in estuarine waters (P < 0.05, Fig. 1). Thus, while a significantly greater total volume of macroaggregates was present in rolled fresh, estuarine, and marine waters, it is noteworthy that pathogen association with the aggregate-rich fraction was not significantly greater in all rolled bottles, as compared with the nonrolled aggregation control bottles. In addition to the physicohemical properties of different water types that influence the density of suspended particles and turbulence and collision forces that can enhance aggregation, the surface properties of nonmotile microorganisms are also a key determinant of pathogen attachment with other particulate matter [1], including aquatic aggregates. Thus, while the abundance and total volume of macroaggregates present in the water column is one factor likely to impact the magnitude of pathogen–aggregate association, unique physical properties such as charge and hydrophobicity govern microscale forces that are exerted between pathogen surfaces and components of aggregates, thereby influencing their likelihood of association.
Total macroaggregate volume (median and range) estimated using image analyses in rolled and nonrolled bottles containing fresh, estuarine, and marine water samples. Aggregates were present in both rolled and nonrolled bottles but were significantly greater in total volume in rolled bottles as compared with nonrolled, aggregation control bottles (P < 0.05). Note log scale of y-axis
Obtaining the volume of aggregates in rolled bottles also allowed estimations of pathogen enrichment in aggregates, a measure that can approximate the factor of pathogen concentration within aggregates as compared with their concentration as suspended particles in surrounding bulk water. Enrichment factor calculations demonstrated that pathogens were 102–104 more concentrated in estuarine and marine aggregates than in surrounding water (Table 1). Enrichment factors observed in this study are similar to those reported previously for microorganisms in marine snow, including T. gondii oocysts [17, 19]. An aggregation enrichment factor for freshwater is reported as ~1, because pathogen concentrations in the bottom waters of rolled bottles were not consistently different than in surrounding, aggregate-free water.
We hypothesized that pathogen aggregation would be highest in saline waters, based on prior research that showed reduced surface charge and enhanced aggregation of T. gondii oocysts in estuarine and marine waters [16, 17]. Estuarine habitats in particular can facilitate flocculation processes due to salinity changes and presence of high levels of particulate and organic matter, resulting in higher likelihood of particle collisions and subsequent association [24]. While all organisms targeted in this study showed increased aggregation in estuarine and marine waters, the proportion of organisms associated with the aggregate-rich water fractions differed. The largest pathogen, G. lamblia, displayed the highest association with estuarine and marine aggregates, followed by the second largest pathogen, C. parvum. The proportions of the smaller sized Salmonella bacterial cells and PP7 virus particles recovered from the aggregate-rich fractions were lower than those of the protozoan parasites. While pathogen size may be one factor influencing aggregation, the concentration of added microorganisms may also influence experimental results. Spiking levels in this study were set at the lowest concentrations that would be measurable in environmental waters (Table 2). Thus, the higher spiking concentration of PP7 could have led to increased particle collision and subsequent aggregation in this study. Notably, Salmonella cells are approximately 50 times larger in diameter than PP7 particles, and both had similar aggregation patterns. Finally, a direct comparison of aggregation across pathogen types was hindered due to the inherent variability associated with the different detection methods required for optimal quantification of different classes of microorganism in environmental samples (Table 2).
These novel data confirm that terrestrially derived zoonotic pathogens associate with aquatic macroaggregates and provide insights into their waterborne transport and bioavailability to susceptible hosts, including marine mammals and humans. Results suggest that diverse classes of pathogens, including parasites, bacteria, and viruses, display a similar pattern of enhanced aggregation in estuarine and marine waters. Previous microorganisms reported to associate with aggregates include aquatic pathogens, halophilic bacteria, and fecal indicator bacteria [11, 12, 23] that can multiply in aquatic environments and/or are derived from a multitude of marine animals. In contrast to microbes that are adapted to aquatic systems, the distribution of zoonotic pathogens that cannot multiply in aquatic environments is solely a function of their transport dynamics. Pathogens that are associated with large aggregates may have increased settling velocities and are more likely to reach the benthos or become strained by vegetation. Once pathogens are removed from the water column, they may persist in the benthic environment for relatively long periods of time. Fecal protozoa in particular are environmentally robust, and C. parvum is known to survive in marine waters for at least 1 year [21]. An additional implication for disease transmission is that invertebrates, including those consumed by humans as seafood, are known to ingest and retain microscopic particles associated with aggregates more readily than particles freely suspended in the water column [7, 25]. Thus, the association of zoonotic pathogens with aquatic aggregates could influence disease transmission through enhanced bioavailability to invertebrates and subsequent incorporation into the marine food web.
References
Dumetre A, Aubert D, Puech PH, Hohweyer J, Azas N, Villena I (2012) Interaction forces drive the environmental transmission of pathogenic protozoa. Appl Environ Microbiol 78:905–912
Fayer R, Dubey JP, Lindsay DS (2004) Zoonotic protozoa: from land to sea. Trends Parasitol 20:531–6
Gebhardt AC, Schoster F, Gaye-Haake B, Beeskow B, Rachold V, Unger D, Ittekkot V (2005) The turbidity maximum zone of the Yenisei River (Siberia) and its impact on organic and inorganic proxies. Estuar Coast Shelf 65:61–73
Giangaspero A, Cirillo R, Lacasella V, Lonigro A, Marangi M, Cavallo P, Berrilli F, Di Cave D, Brandonisio O (2009) Giardia and Cryptosporidium in inflowing water and harvested shellfish in a lagoon in Southern Italy. Parasitol Int 58:12–7
Haile RW, Witte JS, Gold M, Cressey R, McGee C, Millikan RC, Glasser A, Harawa N, Ervin C, Harmon P, Harper J, Dermand J, Alamillo J, Barrett K, Nides M, Wang G (1999) The health effects of swimming in ocean water contaminated by storm drain runoff. Epidemiology 10:355–63
Heinitz ML, Ruble RD, Wagner DE, Tatini SR (2000) Incidence of Salmonella in fish and seafood. J Food Prot 63:579–592
Kach DJ, Ward JE (2008) The role of marine aggregates in the ingestion of picoplankton-size particles by suspension-feeding molluscs. Mar Biol 153:797–805
Kranck K (1984) The role of flocculation in the filtering of particulate matter in estuaries. In: Kennedy VS (ed) The estuary as a filter. Academic Press, Orlando
Leclerc H, Schwartzbrod L, Dei-Cas E (2002) Microbial agents associated with waterborne diseases. Crit Rev Microbiol 28:371–409
Lipp EK, Rose JB (1997) The role of seafood in foodborne diseases in the United States of America. Rev Sci Tech OIE 16:620–640
Lyons MM, Ward JE, Gaff H, Hicks RE, Drake JM, Dobbs FC (2010) Theory of island biogeography on a microscopic scale: organic aggregates as islands for aquatic pathogens. Aquat Microb Ecol 60:1–13
Lyons MM, Ward JE, Smolowitz R, Uhlinger KR, Gast RJ (2005) Lethal marine snow: pathogen of bivalve mollusc concealed in marine aggregates. Limnol Oceanogr 50:1983–1988
Miller WA, Atwill ER, Gardner IA, Miller MA, Fritz HM, Hedrick RP, Melli AC, Barnes NM, Conrad PA (2005) Clams (Corbicula fluminea) as bioindicators of fecal contamination with Cryptosporidium and Giardia spp. in freshwater ecosystems in California. Int J Parasit 35:673–684
Rajal VB, McSwain BS, Thompson DE, Leutenegger CM, Kildare BJ, Wuertz S (2007) Validation of hollow fiber ultrafiltration and real-time PCR using bacteriophage PP7 as surrogate for the quantification of viruses from water samples. Water Res 41:1411–22
Shanks AL, Edmondson EW (1989) Laboratory-made artificial marine snow—a biological model of the real thing. Mar Biol 101:463–470
Shapiro K, Largier J, Mazet JA, Bernt W, Ell JR, Melli AC, Conrad PA (2009) Surface properties of Toxoplasma gondii oocysts and surrogate microspheres. Appl Environ Microbiol 75:1185–91
Shapiro K, Silver MW, Largier J, Conrad PA, Mazet JAK (2012) Association of Toxoplasma gondii oocysts with fresh, estuarine, and marine macroaggregates. Limnol Oceanogr 57:449–456
Siegel S, Castellan NJ (1988) Nonparametric statistics for the behavioral sciences, 2nd edn. McGraw-Hill, New York
Silver MW, Shanks AL, Trent JD (1978) Marine snow—microplankton habitat and source of small-scale patchiness in pelagic populations. Science 201:371–373
Smith WA, Mazet JAK, Hirsh DC (2002) Salmonella in California wildlife species: prevalence in rehabilitation centers and characterization of isolates. J Zoo Wildl Med 33:228–235
Tamburrini A, Pozio E (1999) Long-term survival of Cryptosporidium parvum oocysts in seawater and in experimentally infected mussels (Mytilus galloprovincialis). Int J Parasit 29:711–715
U.S. EPA. (2001) Method 1623: Cryptosporidium and Giardia in water by filtration/IMS/FA EPA-821-R-01-025. Office of Water, U.S. Environmental Protection Agency
Venkateswaran K, Kiiyukia C, Nakanishi K, Nakano H, Matsuda O, Hashimoto H (1990) The role of sinking particles in the overwintering process of Vibrio parahaemolyticus in a marine environment. FEMS Microbiol Ecol 73:159–166
Verney R, Lafite R, Brun-Cottan JC (2009) Flocculation potential of estuarine particles: the importance of environmental factors and of the spatial and seasonal variability of suspended particulate matter. Estuar Coasts 32:678–693
Ward JE, Kach DJ (2009) Marine aggregates facilitate ingestion of nanoparticles by suspension-feeding bivalves. Mar Environ Res 68:137–142
Acknowledgments
We thank Timothy Doane for the water quality analyses; Ann Melli for the protozoal analyses; and Maha Abdulla, Terra Berardi, and Beatriz Aguilar for their assistance with the Salmonella analyses. Stefan Wuertz and Alexander Schriewer provided oversight and advice on quantitative molecular enumeration of the bacteriophage PP7. Andreas Bäumler’s laboratory kindly provided a culture of GFP-labeled Salmonella, and Robert Crawford provided valuable insight on the quantification of this bacterium. Funding for this research was provided by a National Science Foundation Ecology of Infectious Disease grant (Division of Ocean Sciences OCE-1065990) and by fellowship support from the National Oceanic and Atmospheric Administration Oceans and Human Health Initiative (S08-67884).
This publication was prepared by K. Shapiro under NOAA Grant # NA10OAR4170060, California Sea Grant College Program Project # R/CONT-216, through NOAA’S National Sea Grant College Program, U.S. Dept. of Commerce. The statements, findings, conclusions and recommendations are those of the author(s) and do not necessarily reflect the views of the aforementioned organizations.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
Water quality characteristics of the different water types used in aggregation experiments (DOCX 13 kb)
Rights and permissions
About this article
Cite this article
Shapiro, K., Miller, W.A., Silver, M.W. et al. Research Commentary: Association of Zoonotic Pathogens with Fresh, Estuarine, and Marine Macroaggregates. Microb Ecol 65, 928–933 (2013). https://doi.org/10.1007/s00248-012-0147-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-012-0147-2