Abstract
Limitation of bacterioplankton production by nutrients and temperature was investigated in eight temperate lakes in summer. Six of the lakes were resampled in autumn. The lakes differ in nutrient content, water color, and concentration of dissolved organic carbon. Nutrients (phosphorus, nitrogen, and organic carbon) were added alone and in all possible combinations to filtered lake water inoculated with bacteria from the lake. After incubation for 36–40 h at in situ temperatures (ranging from 7 to 20°C), the response in bacterioplankton production was determined. The effect of increased temperature on bacterioplankton growth was also tested. Bacterioplankton production was often limited by phosphorus alone, organic carbon alone, or the two in combination. Phosphorus limitation of bacterioplankton production was more common in the summer, whereas limitation by organic carbon was more frequently observed in the autumn. There was a close balance between limitation by phosphorus and organic carbon in the epilimnion in the summer. In the hypolimnion in the summer, bacterioplankton growth was primarily phosphorus-limited. The effect of phosphorus additions decreased with increasing phosphorus concentrations in the lakes. However, there were no correlations between the effect of added organic carbon and water color, dissolved organic carbon concentration, or phosphorus concentration. When temperature was low (in the hypolimnion in the summer, and throughout the water column in the autumn) temperature also limited bacterioplankton production. Thus, temperature and inorganic nutrients or organic compounds can limit bacterioplankton growth both alone and simultaneously. However, at low temperatures, temperature is the most important factor influencing bacterioplankton growth.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The importance of the microbial food web in aquatic systems is now generally recognized. Bacterioplankton biomass can constitute a large fraction of the total plankton biomass, especially in oligotrophic and dystrophic environments [4, 23, 38]. Therefore, bacteria may be important competitors with phytoplankton for inorganic nutrients [6]. Bacteria also provide important food resources for micro- and metazooplankton [36]. Limitation of bacterioplankton production by different elements can affect both the shape and size of bacteria [49, 53], thereby influencing grazing patterns on bacteria and thus the bacterial community structure [28]. Therefore, factors affecting bacterioplankton growth and biomass can have major effects on the composition of the bacterioplankton community and other constituents of the planktonic food web, and thus the structure of the entire ecosystem.
Limitation of bacterioplankton growth by inorganic nutrients and organic carbon (C) has been found in both lacustrine and marine systems [7, 15, 29, 48]. During the past decade, the importance of phosphorus (P) limitation has been stressed [10, 25, 42, 43, 50]. Bacterioplankton growth has been reported to be limited by inorganic nutrients and organic C, alone or in combination, in oligotrophic [8, 48], mesotrophic [37, 50], and eutrophic [17, 51] lakes. Thus, published data do not indicate that the trophic status of the lakes determines which of the various nutrients will be the limiting element for bacterioplankton growth.
Differences in limiting nutrients over the season have been found in numerous lakes. In the mesotrophic L. Dillon, P seems to be the primary limiting nutrient during most of the year [29]. In the mesotrophic L. Constance, P and C most often limited bacterioplankton production in experiments carried out between February and August[37]. In experiments in the mesotrophic L. Erken conducted from May to September it was shown that P alone, C alone, or the combination of P and nitrogen (N) mainly limited bacterioplankton production [50]. Although all these studies indicate that P is important in limiting bacterioplankton production, other elements also seem to be important. However, the data reveal no general seasonal pattern of changes in nutrient limitations.
Nutrient limitation of bacterioplankton growth may affect the turnover of the dissolved organic carbon (DOC) pool, since limitation of bacterioplankton production by inorganic nutrients can result in accumulations of labile DOC [46]. In a review by Søndergaard and Middelboe [41], it was found that the pool of labile DOC increases with increasing DOC concentrations. Thus, it can be hypothesized that limitation by inorganic nutrients should be stronger in systems with high concentrations of DOC, i.e., systems with high inputs of terrestrial material and/or high nutrient concentrations. Limitation of bacterioplankton production by inorganic nutrients has also been consistently reported in investigations of humic lakes [25, 32]. Hessen et al. [24] found in a study of two humic lakes that inorganic nutrients limited bacterioplankton growth when the external carbon supply was high. However, there was a close colimitation by C and inorganic nutrients at low loads of allochthonous matter. Thus, limitation of bacterioplankton production by C alone appears to be rare in humic lakes.
Bacterioplankton growth is also affected by temperature. Correlations between temperature and bacterioplankton growth have been found in seasonal studies in several lakes [12, 50]. It has been suggested that bacteria in temperate lakes are temperature dependent up to a certain threshold temperature, above which other factors regulate their growth [17, 31]. In mesotrophic Lake Constance [39], it was found that during most of the year the bacterial community was well adapted to in situ temperatures (ranging from 4 to 23°C) in the upper water column. However, in the deeper strata the bacterial growth was limited by temperature (ranging between 4 and 10°C). Together, these studies indicate that the coupling between temperature and growth can differ according to both temperature and strata. In a review of temperature–substrate interactions as limiting factors for bacteria, Pomeroy and Wiebe [33] conclude that substrate concentrations and temperature interact at all temperatures and substrate concentrations, and their interactive effects can differ with temperature regime. Thus, it is important to consider inorganic nutrients, organic substrates, and temperature simultaneously when studying growth limitation of bacterioplankton.
In this study, limitation of bacterioplankton growth by P, N, C, and temperature was investigated in lakes with differing nutrient and humic contents during both summer and autumn. It was hypothesized that P would be the most important limiting nutrient factor, and that temperature would primarily limit growth at low temperatures.
Methods
Nutrient addition experiments were conducted to investigate whether P, N, or C limits bacterioplankton growth. The effect of increasing temperature on bacterioplankton growth was also examined. Water from eight lakes was collected in July 2000, and six of these lakes were resampled in October 2000. A temperature profile was obtained for each lake by measuring temperatures throughout the water column at the deepest part of the lake, and if the lake was stratified the depths of the epi- and hypolimnion were determined. If the lake was stratified, one composite sample of water was taken from the epilimnion and one from the hypolimnion. Otherwise, one composite sample from the whole water column was taken. Samples for analyses of dissolved oxygen were taken over a depth profile. Water samples for the experiments and analyses of concentrations of total P, phosphate (PO4), total N, nitrate (NO3), ammonium (NH4), DOC, absorbance at 436 nm (abs436), and chlorophyll a (chl a) were kept at in situ temperature until arrival at the laboratory. Samples for analyses of dissolved oxygen were preserved in the field and analyzed immediately upon arrival at the laboratory. The incubations to measure bacterioplankton production and CTC+ bacteria in the lakes were started immediately after sampling (see below for details).
Experiments
Water for the assays was first filtered through a GF/D glass fiber filter (2.7 μm, Whatman), then through a Whatman GF/C glass fiber filter (1.2-μm pore size, Whatman) or a 0.2-μm polycarbonate filter (Nuclepore). The resulting filtrates were mixed in the proportions one part 1.2-μm filtrate to nine parts 0.2-μm filtrate. Portions (100 mL) of these mixtures were poured into 250-mL cell culture bottles (Nunclon). P (Na2HPO4), N (NH4NO3), and C as glucose (C6H12O6) were added alone and in all possible combinations (P, N, C, PN, PC, CN, CNP) to the culture bottles. Each treatment was carried out in triplicate. Three additional bottles received no nutrient additions. P, N, and C were added to final added concentrations of 40 μg P L−1, 180 μg N L1, and 1550 μg C L−1, respectively (molar C:N:P ratio of 100:10:1). The C:N:P ratio of the added nutrients was chosen to be similar to an average C:N:P ratio of bacterioplankton [16]. However, the amount of C was doubled to compensate for respiration. The bottles were incubated at in situ temperatures, i.e., ±3°C of the average temperature of the stratum. An additional set of bottles of water to which either P alone (40 μg P L−1 final added concentration) or no nutrients were added was incubated at 25°C, to test the effect of increased temperature (within the range that can naturally occur in these lakes) on all samples. These treatments were also carried out in triplicate. All samples were incubated in the dark and bacterioplankton production was measured after 36–40 h.
Analyses
Bacterioplankton production was measured in terms of [3H]thymidine incorporation rates (TdRinc) [1] with modifications described in Vrede [48] and as below. Thirty nM (final concentration) [3H]thymidine was added to 5 mL water (L. Vallentunasjön and L. Norrviken) or 10 mL water (all other lakes) and incubated in the dark, at the same temperature as in the lakes (in situ measurements) or the experimental bottles. Triplicates and one blank (prepared by adding formaldehyde before the isotope) were used for the in situ measurements. In the experiments, one sample was incubated from each experimental bottle. The samples were incubated for 45–80 min depending on temperature (i.e., the lower the temperature, the longer the incubation). To obtain an estimate of the number of active bacteria, the abundance of CTC+ bacteria was determined. CTC+ bacteria were defined as those capable of reducing the tetrazolium salt 5-cyano-2,3-ditolyltetrazolium chloride (CTC, Polysciences) to its fluorescent formazan derivative, and counted using a modified version of the method described by del Giorgio and Scarborough [14]. For this, CTC was added to 5 mL of lake water to a final concentration of 6 mM (chosen according to Haglund et al. [22]). The samples were incubated for 6 h in the dark at in situ temperature and the reaction was stopped by adding filtered (0.2 μm) formaldehyde (4% final concentration). The selected incubation time was based on the results of a time course experiment (data not shown). A subsample (1–2 mL) was counterstained with the nucleic acid stain SYTO 13 (Molecular Probes) for 15 min [22]. The bacteria were then filtered onto a 0.22-μm black membrane filter and the collected material was air-dried, mounted onto slides, and examined under an epifluoresence microscope. CTC+ bacteria were counted in red excitation light and nonactive bacteria in blue excitation light. At least 200 bacterial cells were counted and the bacteria that were visible in red light (CTC+ bacteria) were not recounted in blue light. At least 20 fields per sample were counted in each excitation light. The total bacterial abundance (BA) was calculated by adding the counts obtained in blue light and red light. Abs436, used here as a measure of water color, was measured in a 5-cm cuvette immediately upon arrival at the laboratory. NO3−N, after reduction to NO2−N, was analyzed in an Alpkem RFA 300 autoanalyzer according to Bendschneider and Robinson [3]. Total N concentrations were analyzed with an Antek 9000 Nitrogen analyzer and DOC concentrations with a Shimadzu TOC-5000. All other water chemistry parameters and chl a concentrations were analyzed with standard methods described in Goedkoop and Sonesten [19]. PO4, NH4, and chl a concentrations were determined immediately upon arrival at the laboratory. Water for the other chemical analyses was kept frozen until analysis, either untreated (for total P and total N contents) or filtered (GF/F filter, Whatman) prior to freezing (for DOC and NO3−N determinations).
Statistical Analyses
The results from the nutrient and temperature assays were analyzed separately with factorial analyses of variance (ANOVA) with the factors P, C, and N, and temperature (T) and P, respectively [52]. In these analyses, the factor assay (A) was also added. In a factorial ANOVA the responses of one of the interaction between several factors are calculated using all data and taking into account whether the factor/factors are present in the treatments or not. Thus, for example, a probability of the interaction P × C is calculated not only from the P + C treatment but from the results from all incubation units. The effect size of P, C, N, and T, i.e., the magnitude of the bacterioplankton production response to each of these factors, was calculated with Yates’s algorithm [5]. Yates’s algorithm is a simple way of calculating the size of both main and interaction effects in a factorial design. As with the factorial ANOVA the effect size of one factor is calculated using all data and taking into account whether the factor is present in the treatments or not. Each single experiment was regarded as a separate set of assays, i.e., tests on epilimnion and hypolimnion samples from the same lake were regarded as two different sets of assays, as were assays of summer and autumn samples from the same lake. The data set was also divided into subsets, in which results from summer, summer epilimnion, summer hypolimnion, and autumn water column samples were analyzed separately. Each set of assays was also analyzed individually. All data were log10−transformed to obtain equal variances in the ANOVA. The correlations between the effect size of P, C, and N, total P concentrations, oxygen concentrations, DOC concentrations, abs436, BA, CTC+ bacteria, TdRinc, and chl a concentrations were examined by calculating Spearman’s rank correlation coefficients [9]. The correlation between the effect size of T and increased temperature was examined in a similar fashion.
Results
Lake Descriptions
The lakes showed large variations in water chemistry (Table 1). The total P content varied between 7 and 458 μg P L−1 and the total N concentration between 200 and 2160 μg N L−1. Total P and total N concentrations were correlated (Spearman’s rho: 0.758, P = 0.0002), and total P will therefore be used in the following text as the measure of the lakes’ nutrient status. The DOC concentrations varied between 2.3 and 16.4 mg C L−1 and were correlated with abs436 (Tables 1 and 2). The PO4−P concentrations were often below the detection limit (<1 μg P L−1, Table 1). The NH4−N concentrations varied between 3 and 1500 μg NH4−N L−1 and the NO3−N concentrations between 2 and 150 μg NO3−N L−1, except in the summer in L. Vallentunasjön and L. Norrviken (in the epilimnion), where the NO3−N concentrations were below detection limit (<1 μg N L−1). There were also large differences in the oxygen concentration (Table 1). The oxygen concentrations were always high in the epilimnion during summer and in the autumn water column. In the hypolimnion, oxygen concentrations varied from high to anoxic. In the summer, the temperature varied between 16 and 20°C and between 7 and 14°C in the epilimnion and hypolimnion, respectively (Table 1). The temperature varied between 8 and 12°C in the autumn. Thus, the temperatures were similar in the summer hypolimnic water and in the autumn water column.
Chl a concentrations and TdRinc values were positively correlated to total P, and all three of these parameters varied widely between lakes, indicating that the lakes differed widely in character (Tables 1 and 2). BA varied between 0.5 109 L−1 and 9.5 109 L−1 and was also positively correlated with total P concentrations (Tables 1 and 2). In contrast, the abundance of CTC+ bacteria was positively correlated with abs436, but not with total P concentrations (Table 2). None of the biological parameters measured in the lakes was correlated with temperature (results not shown).
Nutrient Assays
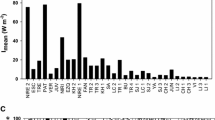
The bacterioplankton production response to nutrient additions varied between lakes, strata, and seasons (Fig. 1). The largest increase in TdRinc due to the nutrient manipulations was observed in the epilimnion of L. Fisklösen following the CNP treatment, in which TdRinc increased up to 39-fold compared to the corresponding controls. However, the increase in TdRinc was usually much smaller, seldom more than 10-fold compared with the respective control. In the summer, P stimulated TdRinc most in both epilimnion and hypolimnion (Table 3). In the epilimnion, C alone and PC together also had strong effects on TdRinc. In contrast, in the autumn, only C alone had a strong effect on TdRinc. Thus, there were clear differences in limiting nutrients between the seasons.
The effects of phosphorus, nitrogen, and organic carbon added alone and in all possible combinations on thymidine incorporation rates in the epilimnion and hypolimnion in the summer and the mixed water column in autumn. —: No addition; P: phosphorus; C: organic carbon; N: nitrogen. Error bars show standard deviations (n=3).
P had the strongest effect on TdRinc (highest sum of squares of the nutrient treatments) when data from all of the experiments were analyzed together (Table 3). However, more experiments were conducted in the summer (in which P had the strongest effect) than the autumn (in which C had the strongest effect). If only data from the lakes that were sampled in both the summer and autumn were used in the ANOVA, the effects of P and C were approximately the same (results not shown). Thus, overall P alone, C alone, and the combination of P and C were the treatments that stimulated TdRinc most strongly.
The analyses of individual sets of assays (i.e., data for each combination of stratum/season and lake) show that P, C and N alone, and in all possible combinations can stimulate TdRinc (Table 4). TdRinc was significantly stimulated by additions of both P alone and C alone in 10 cases. P and C added together also significantly stimulated TdRinc in seven cases. Additions of N stimulated bacterioplankton production in five cases and combinations with N in at most four cases. Thus, TdRinc was stimulated by additions of P alone, C alone, and the combination of P and C much more frequently than by the addition of N alone or in combination with P and C. The effect size of P was negatively correlated with total P concentrations and TdRinc (Table 2). Thus the responses of bacterioplankton production to added P were generally larger in unproductive than in productive lakes. The effect sizes of C and N were not significantly correlated to any of the parameters examined.
Temperature Assays
As indicated by the results from the ANOVA, including data from all of the experiments, both temperature and P had significant effects on TdRinc (Table 5). Increased temperature had no effect on TdRinc in the epilimnion, in which the temperature varied between 16 and 20°C (Table 5, Fig. 2). However, temperature had a strong effect on bacterioplankton production in the hypolimnion during the summer and in the autumn water column, where the in situ temperatures were lower than in epilimnion in summer (Table 5, Fig. 2). The stimulation by P was much weaker or insignificant in these experiments. The effect size of increased temperature was negatively correlated with in situ temperatures (Spearman’s rho −0.705, P = 0.0008).
The effects of increased temperature alone, addition of phosphorus alone, and the combination of the two on thymidine incorporation in the epilimnion and hypolimnion in the summer and the mixed water column in autumn. —: No treatment; P: phosphorus; T: increased temperature. Error bars show standard deviations (n= 3).
Discussion
In the nutrient assays in this study, P alone and C alone or in combination limited bacterioplankton production most often. Also, the study shows that the limiting nutrient differed between seasons and between strata. Additions of P stimulated bacterioplankton production most in the epilimnion and hypolimnion in the summer. P has been found to be the primary limiting resource for bacterioplankton growth in other lakes not only during the summer, but also at other times of the year [21, 29, 43]. Bacterioplankton have high P:C ratios [16] and thus high P requirements. It has been suggested that bacteria may not be such efficient competitors with phytoplankton for P as previously believed [45]. Also, it has been shown that N may be the primary limiting nutrient for phytoplankton growth at the same time as P limits bacterioplankton growth [25, 50]. Thus, bacteria may not always be able meet their high P demands because of competition for P both within the bacterioplankton community and with phytoplankton. The water chemistry data also indicate that the supply of P was deficient, since the PO4 concentrations were low in most lakes during summer. Additions of P and C in combination and C alone also stimulated bacterioplankton production in the epilimnion, indicating that there is a relatively close balance between P and C limitation in the upper strata in the summer. A close balance between limitation by P and other elements appears to be relatively common in lakes [15, 17, 44, 48].
In the autumn, additions of C had the largest positive effect on bacterioplankton production. Additions of P alone had no significant effect on bacterioplankton production when results of the autumn experiments were analyzed together. The investigated lakes are located in the temperate zone and have strong seasonal variations in temperature and biological activity. The maximum summer temperatures are normally around 20–25°C, and during the winter the lakes are covered by ice. With the exception of L. Vallentunasjön and L. Skotttjärn, which are shallow and therefore polymictic, the lakes are dimictic. When the water circulates, nutrient-rich water from the deeper parts of the lakes is mixed with the upper layer, and nutrients become available throughout the whole water column. The importance of P as limiting nutrient should therefore be less pronounced in autumn when the water column is mixed. An alternative explanation for the greater responses to C than P in the autumn is that bacterioplankton are dependent on C released from phytoplankton and therefore become C-limited when the phytoplankton biomass declines in late autumn. However, there was no correlation between the effect size of P, N, or C and chl a concentrations, indicating that the phytoplankton biomass per se does not directly determine nutrient limitations of bacterioplankton.
The effect size of P was negatively correlated with TdRinc and total P concentrations. Thus the effect of added P was generally stronger in the oligotrophic than in the eutrophic systems. In a comparison of four lakes, Smith and Prairie [40] found that additions of P stimulated bacterioplankton growth in all lakes except the lake with the highest total phosphorus concentration. These results together indicate that bacterioplankton production seems to be strongly limited by P more often in nutrient-poor than in nutrient-rich systems. However, some aspects of the experimental design in the present study have to be considered in this context. The amount of nutrients added was the same in all experiments, so the percentage increase in nutrient concentration was higher in the nutrient-poor than in the nutrient-rich systems. Thus, it could be argued that the effect size of P was not correlated to the nutrient status of the lake, but to the percentage increase in P concentration. The amount of P taken up by the bacteria can be calculated, since no organisms other than bacterioplankton were present. The calculated rate of uptake of P at the highest bacterial production in any replicate in the assays was 26.4 μg P L−1 day−1 (255 pmol thymidine L−1 h−1). The conversion factor 2 × 1018 cells mole thymidine−1 [2] and the atomic C:P ratio of 35 (exponentially growing cells [49]) were used in these calculations. A cell volume (V) of 0.14 (μm3 was chosen, which is the maximum cell volume measured in 1991 to 1992 in L. Vallentunasjön and L. Norrviken (unpublished data). The allometric formulas 120 × V0.72 was used to convert cell volume to C content [35]. None of the conversion factors used in the calculations are in the lower range of reported conversion factors, but still the amount of P added in the assays (40 μg P L−1) would meet the P requirements for almost twice the maximum production rates measured in the experiments. Furthermore, the maximum measured bacterial production in any of the investigated lakes was much lower (59 pmol thymidine L−1 h−1) than the maximum production in the experiments. Thus, the amount of P added should have been high enough to stimulate bacterioplankton production even in the most productive lakes if the bacterioplankton community had been P-limited there. It can also be argued that the negative correlation between effect size of P and total P concentrations in the lakes could be due to enzyme or growth kinetics, i.e., higher uptake or growth rates at lower concentrations than at higher concentrations. It may not be accurate to use total P concentration when discussing enzyme or growth kinetics since these are dependent on the concentrations of the available substrates. Bacteria can utilize both PO4 and organic bound P. The in situ concentrations of PO4 were below detection limit (<1 μg P L−1) or low in most cases. The concentration of dissolved organic P (DOP) could be calculated by subtracting particulate P from total P concentrations (data not shown). However, effect size of P was only significantly correlated with total P concentrations in the lakes and not with the concentrations of PO4, DOP, or the total concentration of dissolved P (PO4 plus DOP). Thus, it is unlikely that the correlation between effect size of P and total P would only be due to kinetics. It can therefore be suggested that increased P concentrations stimulate bacterioplankton production more in the oligotrophic than in the eutrophic systems.
Additions of C stimulated bacterioplankton growth in the humic lakes L. Gäddtjärn in the summer and L. Siggeforasjön in both summer and autumn. In contrast, additions of C had a negative effect on bacterioplankton growth in some cases, but only in lakes with high absorbance and DOC, i.e., humic lakes. In other humic systems it has been found that C alone does not limit bacterioplankton production, although a close balance between C and inorganic nutrient limitation has been found [24]. Thus, the effects of adding C to humic lake samples have not been consistent. In the present study, there was no correlation between the effect sizes of P, N, or C and abs436 or DOC. Thus, this study indicates that there are no general differences in C, P, or N limitation across the humic gradient spanned by the studied lakes.
It has been suggested that limitation of bacterioplankton production by inorganic nutrients can result in an accumulation of labile C [41, 46]. In the studied lakes, labile C may have accumulated during the summer when the systems were primarily P limited. However, during the autumn when concentrations of inorganic nutrients were high because of circulation, the accumulated C must have been utilized. This conclusion is supported by the observation that bacterioplankton production was C-limited in autumn. Thus, due to the seasonal variation in temperate lakes, with alternating periods of stratification and mixed water columns, it is unlikely that these lakes have a net accumulation of labile C.
Glucose was used as C source in the experiments. Utilization of different C compounds may differ between bacterial groups [11]. Also, Grover and Chrzanowski [20] found that the utilization of different groups of organic substrates varied over the season, which probably reflects changes in the bacterial community and their capability to use different substrates. In this study, the addition of organic C as glucose stimulated bacterial growth very often. Thus, it is reasonable to believe that glucose was an appropriate substrate in these experiments, although the choice of substrate may have affected the size of the response.
Increased temperature stimulated bacterioplankton production when the in situ temperatures were low, i.e., in the hypolimnion in summer and in the mixed water column in autumn. Also, there was a strong negative correlation between the effect of increased temperature on bacterioplankton production and in situ temperatures. It has previously been shown that the affinities for both organic substrates and inorganic nutrients are reduced at low temperatures (reviewed by [30]). In the present study, the interaction between P and temperature was only statistically significant in the epilimnion in summer. At the same time, temperature alone had no effect, while P alone had a strong effect on bacterioplankton production. This suggests that bacteria may utilize the added P more easily at the higher temperature. An alternative explanation is that the bacterioplankton growth rate rose as the temperature increased, so their P demand also increased. However, regardless of the mechanism involved, it can be concluded that a relatively small increase in temperature did not have a strong effect on bacterioplankton production, when the bacterial community was strongly nutrient limited.
At low temperatures, both increased temperature and additions of P (in the hypolimnion in summer) or C (in autumn) had strong effects on bacterioplankton production. Thus, in these cases bacterial growth was limited both by temperature and inorganic nutrients or organic substrates. Similar results have been found in Conception Bay, Newfoundland, where it was found that the growth of bacteria that live at low temperatures was stimulated both by increases in temperature and by additions of organic substrates [34].
The nutrient assays were conducted in dilution cultures in the present study. This experimental setup may have some limitations. It has been shown that the bacterial community composition can change during incubation in both untreated and manipulated dilution cultures [18, 27]. Since bacterial community composition was not investigated in the present study, it is difficult to make any conclusion as to whether this can have affected the results. However, Fuchs et al. [18] found in dilution culture experiments with bacterial inocula from different environments that the total bacterial cell densities in the end of the experiments were more dependent on the water quality than on the inoculum. Thus, irrespective of the community composition, the total bacterial growth response was similar. It has been shown that at least some bacterial taxa are favored by increased nutrient availability [26]. However, these bacterial taxa are probably favored under natural conditions as well [26]. Thus, this change in the bacterial community composition in the experiment would only reflect a response in the community that also would occur in nature.
The responses to the manipulations were measured after 24–36 h. This incubation time was chosen to reduce the risk of changes in the bacterial community composition or other changes due to “bottle effects” [27] that are likely to occur with time. The length of the incubation time is a trade-off between such “bottle effects” and the fact that the bacterial community must have time to respond to the manipulations. Previous studies show that bacteria respond to manipulation within the chosen incubation time in this type of experiment [17]. At lower temperatures, the incubation time could probably have been increased without large problems with “bottle effects.” However, to avoid problems in the interpretation of the results due to different incubation time in different assays, the same incubation time was chosen in all experiments. Since the bacterial community responded to the manipulations at lower temperatures as well, this is interpreted as if the incubation time was sufficient also in these experiments.
In simplified laboratory systems, like the one in this study, many naturally occurring processes are excluded. This gives the possibility of studying direct effects of different factors without interaction with other organisms (e.g., competition or effects of nutrient regeneration) in the system that could make the interpretation of the results more difficult. However, many of the excluded factors are of great importance in natural systems, and one can therefore raise the question whether it is possible to extrapolate the results to whole ecosystems as lakes. Other experiments in lakes that have been conducted on larger scales, both in time and volume, as well as whole lake investigations support the results that bacterioplankton growth is often regulated by P during summer [13, 25, 32, 47]. Seasonal trends that are consistent with the results in the present study have also been found previously in experiments conducted in larger enclosures [50]. Thus, together these studies indicate that results from small-scale experiments can be extrapolated to larger scales and time frames.
Collectively, the results in the present study show that additions of P alone, C alone, and P and C in combination often stimulate bacterioplankton production in temperate lakes. P limitation is more important during the summer and C limitation in autumn. Additions of P generally stimulated bacterioplankton production more in oligotrophic systems than in eutrophic systems. However, the results indicate that there are no general differences in P, C, or N limitation across the humic gradient spanned in the study. It can also be concluded that temperature and inorganic nutrients or organic compounds can limit bacterioplankton growth simultaneously, and that each of these factors alone can stimulate bacterioplankton production. However, at low temperatures, temperature is the most important factor influencing bacterioplankton growth.
References
RT Bell (1993) Estimating production of heterotrophic bacterioplankton via incorporation of tritiated thymidine PF Kemp BF Sherry JJ Cole (Eds) Handbook of Methods in Aquatic Microbial Ecology Lewis Boca Raton, FL 495–503
RT Bell (1990) ArticleTitleAn explanation for the variability in the conversion factor deriving bacterial cell production from incorporation of [3H] thymidine Limnol Oceanogr 35 910–915
K Bendschneider RJ Robinson (1952) ArticleTitleA new spectrophotometric determination of nitrite in sea water J Mar Res 11 87–96
P Blomqvist RT Bell H Olofssor U Stensdotter K Vrede (1995) ArticleTitlePlankton and water chemistry in Lake Njupfatet before and after liming Can J Fish Aquat Sci 52 551–565
H Box WG Hunter JS Hunter (1978) Statistics for Experimenters John Wiley & Sons New York
G Bratbak TF Thingstad (1985) ArticleTitlePhytoplankton–bacteria interactions: an apparent paradox? Analysis of a model system with both competition and commensalism Mar Ecol Prog Ser 25 23–30
DA Caron E Lin Lim RW Sanders MR Dennet U-G Berninger (2000) ArticleTitleResponses of bacterioplankton and phytoplankton to organic carbon and inorganic nutrient additions in contrasting oceanic ecosystems Aquat Microb Ecol 22 175–184
TH Chrzanowski RW Sterner JJ Elser (1995) ArticleTitleNutrient enrichment and nutrient regeneration stimulate bacterioplankton growth Microb Ecol 29 221–230 Occurrence Handle10.1007/BF00164886
WJ Conover (1980) Practical Nonparametric Statistics EditionNumber2 John Wiley & Sons New York
JB Cotner JW Ammerman ER Peele E Bentzer (1997) ArticleTitlePhosphorus-limited bacterioplankton growth in the Sargasso Sea Aquat Microb Ecol 13 141–149
MT Cottrell DL Kirchman (2000) ArticleTitleNatural assemblages of marine proteobacteria and members of the Cytophaga–Flavobacter cluster consuming low- and high-molecular-weight dissolved organic matter Appl Environ Microbiol 66 1692–1697 Occurrence Handle10.1128/AEM.66.4.1692-1697.2000 Occurrence Handle10742262
MF Coveney RG Wetzel (1995) ArticleTitleBiomass, production, and specific growth rate of bacterioplankton and coupling to phytoplankton in an oligotrophic lake Limnol Oceanogr 40 1187–1200
DJ Currie (1990) ArticleTitleLarge-scale variability and interactions among phytoplankton, bacterioplankton, and phosphorus Limnol Oceanogr 35 1437–1455
PA Giorgio Particledel G Scarborough (1995) ArticleTitleIncrease of the proportion of metabolically active bacteria along gradients of enrichment in freshwater and marine plankton: implications for estimates of bacterial growth and production J Plankton Res 17 1905–1924
JJ Elser BL Stable PR Hassett (1995) ArticleTitleNutrient limitation of bacterial growth and rates of bacterivory in lakes and oceans: a comparative study Aquat Microb Ecol 9 105–110
KM Fagerbakke M Heldal S Norland (1996) ArticleTitleContent of carbon, nitrogen, oxygen, sulphur and phosphorus in native aquatic and cultured bacteria Aquat Microb Ecol 10 15–27
M Felip ML Pace JJ Cole (1996) ArticleTitleRegulation of planktonic bacterial growth rates: the effects of temperature and resources Microb Ecol 31 15–28 Occurrence Handle10.1007/BF00175072
BM Fuchs MV Zubkoy K Saham PH Burkill R Amann (2000) ArticleTitleChanges in community composition during dilution cultures of maine bacterioplankton as assessed by flow cytometric and molecular biological techniques Environ Microbiol 2 191–201 Occurrence Handle10.1046/j.1462-2920.2000.00092.x Occurrence Handle11220305
W Goedkoop L Sonesten (1995) ArticleTitleLaborationsmanual för kemiska analyser av inlandsvatten och sediment [in Swedish] Scripta Limnologica Upsaliensia 1995 B 12
JP Grover TH Chrzanowski (2000) ArticleTitleSeasonal patterns of substrate utilization by bacterioplankton: case studies in four temperate lakes of different latitudes Aquat Microb Ecol 23 41–54
TB Gurung J Urabe (1999) ArticleTitleTemporal and vertical differences in factors limiting growth rate of heterotrophic bacterial in Lake Biwa Microb Ecol 38 IssueID2 136–145 Occurrence Handle10.1007/s002489900167 Occurrence Handle10441706
A-L Haglund E Törnblom B Boström L Tranvik (2002) ArticleTitleLarge differences in the fraction of active bacteria in plankton, sediments, and biofilms Microb Ecol 43 232–241 Occurrence Handle10.1007/s00248-002-2005-0 Occurrence Handle12023730
DO Hessen T Andersen A Lyche (1990) ArticleTitleCarbon metabolism in a humic lake: pool sizes and cycling through zooplankton Limnol Oceanogr 35 84–99
DO Hessen K Nygaard K Saloner A Vähätalo (1994) ArticleTitleThe effect of substrate stoichiometry on microbial activity and carbon degradation in humic lakes Environ Int 20 67–76 Occurrence Handle10.1016/0160-4120(94)90068-X
M Jansson P Blomqvist A Jonsson A-K Bergström (1996) ArticleTitleNutrient limitation of bacterioplankton, autotrophic and mixotrophic phytoplankton, and heterotrophic nanoflagellates in Lake Örträsk Limnol Oceanogr 41 1552–1559
ES Lindström K Vrede E Leskinen (2004) ArticleTitleResponse of a member of the Verrucomicrobia, among the dominating bacteria in a hypolimnion, to increased phosphorus availability J Plankton Res 26 IssueID2 241–246 Occurrence Handle10.1093/plankt/fbh010
R Massana P-A Carlos EO Casamayor JM Gasol (2001) ArticleTitleChanges in marine bacterioplankton phylogenetic composition during incubations designed to measure biogeochemically significant parameters Limnol Oceanogr 46 1181–1188
C Matz K Jürgens (2003) ArticleTitleInteraction of nutrient limitation and protozoan grazing determines the phenotypic structure of a bacterial community Microb Ecol 45 384–398 Occurrence Handle10.1007/s00248-003-2000-0 Occurrence Handle12704556
DP Morris WM Lewis SuffixJr (1992) ArticleTitleNutrient limitation of bacterioplankton growth in Lake Dillon, Colorado Limnol Oceanogr 37 1179–1192
DB Nedwell (1999) ArticleTitleEffect of low temperature on microbial growth: lowered affinity for substrates limits growth at low temperature FEMS Microb Ecol 30 101–111 Occurrence Handle10.1016/S0168-6496(99)00030-6
CA Ochs JJ Cole GE Likens (1995) ArticleTitlePopulation dynamics of bacterioplankton in an oligotrophic lake J Plankton Res 17 365–391
ML Pace JJ Cole (1996) ArticleTitleRegulation of bacteria by resources and predation tested in whole-lake experiments Limnol Oceanogr 41 1448–1460
LR Pomeroy WJ Wiebe (2001) ArticleTitleTemperature and substrates as interactive limiting factors for marine heterotrophic bacteria Aquat Microb Ecol 23 187–204
LR Pomeroy WJ Wiebe D Deibel JT Thompson GT Rowe JD Pakulaski (1991) ArticleTitleBacterial responses to temperature and substrate concentration during the Newfoundland spring bloom Mar Ecol Prog Ser 75 143–159
T Posch M Loferer-Krößbacher G Gad A Alfreider J Pernthaler R Psenner (2001) ArticleTitlePrecision of bacterioplankton biomass determination: a comparison of two fluorescent dyes, and of allomteric and linear volume-to-carbon conversion factors Aquat Microb Ecol 25 55–63
RW Sanders KG Porter SJ Bennett AE DeBiase (1989) ArticleTitleSeasonal patterns of bacterivory by flagellates, ciliates, rotifers, and cladocerans in a freshwater planktonic community Limnol Oceanogr 34 673–687
B Schweitzer M Simon (1995) ArticleTitleGrowth limitation of planktonic bacteria in a large mesotrophic lake Microb Ecol 30 89–104 Occurrence Handle10.1007/BF00184516
M Simon F Azam (1992) ArticleTitleSignificance of bacterial biomass in lakes and the ocean: comparison to phytoplankton biomass and biogeochemical implications Mar Ecol Prog Ser 86 103–110
M Simon C Wünsch (1998) ArticleTitleTemperature control of bacterioplankton growth in a temperate large lake Aquat Microb Ecol 16 119–130
EM Smith YT Prairie (2004) ArticleTitleBacterial metabolism and growth efficiency in lakes: the importance of phosphorus availability Limnol Oceanogr 49 137–147
M Søndergaard M Middelboe (1995) ArticleTitleA cross-system analysis of labile dissolved organic carbon Mar Ecol Prog Ser 118 283–294
TF Thingstad UL Zweife F Rassoulzadegan (1998) ArticleTitleP limitation of heterotrophic bacteria and phytoplankton in the northwest Mediterranean Limnol Oceanogr 43 88–94
T Toolan JD Weh S Findlay (1991) ArticleTitleInorganic phosphorus stimulation of bacterioplankton production in a meso-eutrophic lake Appl Environ Microbiol 57 2074–2078
T Tulonen P Kankaala L Arvola A Ojala (2000) ArticleTitleGrowth and production of bacterioplankton in a deep mesotrophic boreal lake Arch Hydrobiol 147 311–325
O Vadstein (2000) ArticleTitleHeterotrophic, planktonic bacteria and cycling of phosphorus Phosphorus requirements, competitive ability, and food web interactions. Adv Microb Ecol 16 115–167
O Vadstein LM Olsen A Busch T Andersen HR Reinertsen (2003) ArticleTitleIs phosphorus limitation of planktonic heterotrophic bacteria and accumulation of degradable DOC a normal phenomenon in phosphorus-limited systems? A microcosm study FEMS Microb Ecol 1556 1–10
K Vrede (1999) ArticleTitleEffects of inorganic nutrients and zooplankton on the growth of heterotrophic bacterioplankton—enclosure experiments in an oligotrophic clearwater lake Aquat Microb Ecol 18 133–144
K Vrede (1996) ArticleTitleRegulation of bacterioplankton production and biomass in an oligotrophic clearwater lake—the importance of the phytoplankton community J Plankton Res 18 1009–1032
K Vrede M Heldal S Norland G Bratbak (2002) ArticleTitleThe elemental composition (C, N, P) and cell volume of exponentially growing and nutrient limited bacterioplankton Appl Environ Microbiol 68 2965–2971 Occurrence Handle10.1128/AEM.68.6.2965-2971.2002 Occurrence Handle12039756
K Vrede T Vrede A Isaksson A Karlsson (1999) ArticleTitleEffects of nutrients (P, N, C) and zooplankton on bacterioplankton and phytoplankton—a seasonal study Limnol Oceanogr 44 1616–1624
L Wang TD Miller JC Priscu (1992) ArticleTitleBacterioplankton nutrient deficiency in a eutrophic lake Arch Hydrobiol 125 423–439
JH Zar (1984) Biostatistical Analysis EditionNumber2 Prentice Hall Upper Saddle River, NJ
T Östling L Holqvist K Flärdh B Svenblad Å Jouper-Jaan S Kjelleberg (1993) Starvation and recovery of Vibrio. S Kjelleberg (Eds) Starvation in Bacteria Plenum Press New York 103–127
Acknowledgments
Ulrika Stensdotter-Blomberg, Jan Johansson, Sandra Lindahl, Isabell Olevall, and Björn Vrede are acknowledged for help with fieldwork and/or analyses in the laboratory. Eva Lindström and Tobias Vrede and two anonymous reviewers are acknowledged for constructive comments on the manuscript. This study was financed by the Knut and Alice Wallenbergs Foundation, the Helge Ax:son Johnssons Foundation, the Foundation of Oscar and Lili Lamms Minne, and the Royal Swedish Academy of Science.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vrede, K. Nutrient and Temperature Limitation of Bacterioplankton Growth in Temperate Lakes. Microb Ecol 49, 245–256 (2005). https://doi.org/10.1007/s00248-004-0259-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-004-0259-4