Abstract
The pulp and paper industry largely depends on the biodegradation activities of heterotrophic bacteria to remove organic contaminants in wastewater prior to discharge. Our recent discovery of extensive cyanobacterial communities in pulp and paper waste treatment systems led us to investigate the potential impacts of cyanobacterial exudates on growth and biodegradation efficiency of three bacterial heterotrophs. Each of the three assessed bacteria represented different taxa commonly found in pulp and paper waste treatment systems: a fluorescent Pseudomonad, an Ancylobacter aquaticus strain, and a Ralstonia eutropha strain. They were capable of utilizing phenol, dichloroacetate (DCA), or 2,4-dichlorophenoxyacetic acid (2,4-D), respectively. Exudates from all 12 cyanobacterial strains studied supported the growth of each bacterial strain to varying degrees. Maximum biomass of two bacterial strains positively correlated with the total organic carbon content of exudate treatments. The combined availability of exudate and a known growth substrate (i.e., phenol, DCA, or 2,4-D) generally had a synergistic affect on the growth of the Ancylobacter strain, whereas mixed effects were seen on the other two strains. Exudates from four representative cyanobacterial strains were assessed for their impacts on phenol and DCA biodegradation by the Pseudomonas and Ancylobacter strains, respectively. Exudates from three of the four cyanobacterial taxa repressed phenol biodegradation, but enhanced DCA biodegradation. These dissimilar impacts of cyanobacterial exudates on bacterial degradation of contaminants suggest a species-specific association, as well as a significant role for cyanobacteria during the biological treatment of wastewaters.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The cyanobacteria are a cosmopolitan group of photosynthetic prokaryotes that play an important role in aquatic microbial communities. Similar to eukaryotic algae and other microorganisms, cyanobacteria release a variety of organic molecules such as organic acids, polypeptides, and polysaccharides [2, 13, 25]. The exudates of cyanobacteria can stimulate or inhibit other members of the microbial community, including heterotrophic bacteria [3, 9, 20, 24, 26]. Antimicrobial compounds found in cyanobacterial exudates include polyphenols, fatty acids, glycolipids, terpenoids, alkaloids, and a variety of yet to be described bacteriocins [4]. In a few studies, the authors suggest that the release of antimicrobials has a major affect on the community structure of heterotrophic bacteria [6, 27]. Other studies have found a significant coupling of cyanobacterial primary production and bacterial secondary production, particularly during the development and cessation of blooms [8, 16, 28]. According to the authors, the cyanobacterial exudates represented a striking proportion of bacterial organic carbon supply.
Although much is now known about cyanobacterial exudates and their impacts on natural aquatic microbial communities, little is known about their potential impacts on wastewater treatment performance. All secondary wastewater treatment systems depend on the biodegradative activities of heterotrophic bacteria. In secondary waste treatment systems used by the pulp and paper industry, high densities of heterotrophs are maintained to remove biological oxygen demand (BOD), suspended solids, and toxic organic compounds such as resin acids and organochlorines prior to discharge. Likely because of high toxicity levels, virtually no photosynthetic eukaryotes exist in these systems. However, we have recently shown that cyanobacteria can thrive in these systems despite the presence of toxicants and low light conditions [17, 18]. In some cases, cyanobacterial biomass can equal or exceed the biomass of heterotrophic bacteria [17].
Given these findings, we hypothesized that cyanobacterial exudates would have direct impacts on the metabolic/catabolic efficiency of bacterial degraders and by extension, on the structure of these particular bacterial communities. One of the objectives of this study was to test the first part of this hypothesis by assessing whether the exudates from 12 cyanobacterial taxa from pulp and paper waste treatment systems could affect the growth and the catabolic abilities of bacterial degraders. Experiments were conducted to determine if exudates could serve as bacterial growth substrates, as well as influence contaminant utilization. In addition, we examined the impact of exudates on the rates of phenol and dichloroacetate (DCA) degradation (i.e., removal) by pulp and paper bacteria.
Materials and Methods
Cyanobacterial strains were isolated from bleached kraft pulp and paper mills where cyanobacterial community composition and abundance has been previously documented [17]. All strains have been deposited at the University of Toronto Culture Collection (UTCC), Toronto, Ontario, Canada. Based on our previous analyses [17], we have determined that most of the isolated species are dominant members of the cyanobacterial communities found in pulp and paper wastewater (Table 1). After a series of plating and subculturing, each cyanobacterial strain was tested for its axenic status by both streak plating onto R2A enriched agar plates, and microscopic examination.
Cyanobacterial species were grown in sterile 500-mL flasks with organic-free BG-11 medium [1] on light banks (46.3 μmol m−2s−1) at 25°C until midstationary phase (3–4 weeks). Modified BG-11 medium did not contain citric acid, ferric ammonium citrate, and sodium EDTA, but did include 3.15 mg L−1 ferric chloride. To reduce the risk of bacterial contamination of axenic exudates, a rapid collection process using 10-min centrifugation (8000 ×g; HN-S Centrifuge, International Equipment Co., Needham Heights, MA USA) rather than filtration of hundreds of milliliters of cyanobacterial cultures was employed. To ensure that ample amounts of inorganic nutrients were available for bacterial growth, 20-mL subsamples of exudate supernatant was enriched with 50-μL aliquots of inorganic nutrients to produce final concentrations equivalent to those found in the original BG-11 medium [NaNO3 = 150 mg L−1, K2HPO4 = 30 mg L−1, FeCl3 = 3.15 mg L−1, H3BO3 = 2.86 mg L−1, MnCl2 = 1.15 mg L−1, ZnSO4 = 0.12 mg L−1, NaMoO4 = 0.26 mg L−1, CuSO4 = 0.05 mg L−1, Co(NO3)2 = 0.03 mg L−1].
The total organic carbon (TOC) of each exudate treatment was measured by using a Model 1010 TOC analyzer (OI Corporation, College Station, TX, USA) and the standard persulfate oxidation/infrared detection method. The molecular weight distribution of each exudate treatment was estimated by size exclusion chromatography using an aqueous ultrahydrogel linear GPC column (7.8 × 300 mm, 6–13 μm particle size; Waters Corp., Milford, MA, USA) with a flow rate of 0.4 mL min−1 and a UV–V photodiode detector (230–280 nm). The mobile phase was Tris/hydrochloric acid buffer (0.05 M, pH 7.6), amended with 0.1 M lithium chloride and 5 mg L−1 PEG. A protein-based standard pack (Sigma-Aldrich, St Louis, MA, USA) was used for molecular weight calibration.
We used three heterotrophic bacterial species for this study. A phenol degrader (Pseudomonas sp. P1) and a dichloroacetic acid degrader (Ancylobacter aquaticus CN13) were originally isolated from bleached kraft pulp and paper waste treatment systems [14, 15]. A. aquaticus (formerly Microcyclus sp.), in particular, is a common inhabitant of pulp and paper wastewater communities [29]. A third bacterial strain, Ralstonia eutropha (strain JMP134), was previously isolated from soil and can degrade 2,4-dichlorophenoxyacetic acid (2,4-D), 2,4-dichlorophenol, and chlorobenzoate via a commonly encountered pathway [10]. Cryopreserved bacterial isolates were revived by streaking onto R2A™ agar plates and incubated at 25°C. Individual colonies were transferred to 50 mL of organic-free BG-11 medium (pH 7.5) with their specific contaminant substrate: 1 mM phenol for P1, 2.5 mM dichloroacetate (DCA) for CN13, and 1 mM 2,4-D for JMP134. All bacterial cultures were grown to stationary phase prior to use in the growth assays.
Bacterial growth assays were performed by monitoring culture turbidity in sterile 96-well (200 μL) Nunclon™ microtitre plates. Treatments were designed to assess the growth of the three bacterial species on nutrient-amended exudates and their respective contaminant substrates. Treatment wells consisted of exudates, exudates + substrate, and medium + substrate (positive control). Negative controls consisted of exudate and medium-only wells where bacteria were not inoculated. After 150 μL of exudates and medium was pipetted into their respective wells, 20 μL of each substrate (phenol, DCA, and 2,4-D) was pipetted into their designated treatment wells for a final concentration of 1 mM (2.5 mM for DCA). Finally, 20 μL of each bacterial strain was inoculated into their respective treatment wells (eight replicates). The microplates were wrapped with parafilm to minimize evaporation, but were not airtight. The plates were shaken and incubated in the dark at 25°C for 7 days to reflect the typical retention time of a waste treatment system. Bacterial growth was measured by taking daily optical density (OD) readings with a Bio-Rad Model 3550 UV microplate reader. All OD measurements were converted to biomass values (cells mL−1−1) using species-specific standard curves.
To assess the effects of exudates on the bacterial degradation of phenol and DCA, biodegradation rates were measured in the presence and absence of exudates of three cyanobacterial strains representing different genera: Aphanocapsa, Pseudanabaena, and Phormidium. A fourth species, Phormidium insigne, was added because of its distinctive geosmin production (i.e., musty odour). Phenol biodegradation studies monitored phenol biodegradation with and without exudate amendment. Controls consisted of triplicate flasks containing 20 mL inorganic BG-11 medium + 1 mM phenol. Treatments were 20 mL triplicate flasks of nutrient-amended exudate plus 1 mM phenol. Both were inoculated with 100 μL of a P1 culture grown in phenol to stationary phase. Cultures were incubated and agitated in the dark at 25°C.
For each sampling period, two 1-mL subsamples were taken from each replicate for phenol and cell density determination. Subsamples for phenol analysis were centrifuged in microcentrifuge tubes at 13,000 × g for 10 min (Micro Biofuge; Heraeus Instruments, USA). The supernatants were transferred into 1-mL borosilicate autosampler vials, capped, and immediately frozen at −20°C. For each experiment, all samples were thawed at the same time and analyzed by high-pressure liquid chromatography (Waters Corp.) with a 70:30 mixture of analytical-grade acetonitrile/1% phosphoric acid run at 1 mL min−1 using a NovaPak C18 reverse-phase column. Bacterial growth in each treatment was measured by OD using a Spectronic (Genesys 5) spectrophotometer (Milton Roy, Rochester, NY, USA). All OD measurements were converted to cells mL−1 using a standard curve for P1.
To account for a potential inorganic nutrient imbalance between the controls and exudate treatments, it was important to rule out inorganic nutrients as a confounding variable in contaminant biodegradation. Preliminary biodegradation studies compared phenol biodegradation in standard medium and with 2× inorganic nutrient addition. These treatments encompassed the minimum and maximum nutrient concentrations that would possibly be available to P1 in exudate treatments. It was confirmed that inorganic nutrient amendment to the exudate treatments would not be a confounding variable as both phenol biodegradation and bacterial growth did not significantly differ (ANOVA, p > 0.1) between treatments.
DCA biodegradation studies with strain CN13 involved some modification to the above experimental design. DCA biodegradation in 2× nutrient-enriched BG-11 was significantly greater (ANOVA p < 0.05) than in BG-11 alone. Therefore, the media controls in the DCA biodegradation experiments were supplemented with 2× inorganic nutrients to discount an inorganic nutrient advantage in the exudate treatments. Changes to the media included the replacement of ferric chloride with ferric ammonium sulfate (6 mg L−1) to minimize background chloride levels. Both mineral medium and exudate treatments consisted of triplicate 2.5 mM DCA solutions inoculated with 100 μL of stationary phase DCA grown culture of CN13, for a final volume of 25 mL. All DCA biodegradation experiments were incubated at 25°C in the dark for 7 days.
DCA biodegradation was monitored via chloride release. Chloride concentrations in 5-mL subsamples were measured using an Accumet 1003 ion probe (Fisher Scientific, Fair Lawn, NJ, USA). CN13 was found to have a long lag phase (∼4 days); thus initial chloride measurements were taken on day 4. Final chloride measurements were taken on day 7. CN13 tended to form small flocs and settle in solution, hindering horizontal OD readings in standard cuvettes. As an alternative, CN13 turbidities were determined vertically in microplates with a microplate reader (as described above).
Results
All 12 of the cyanobacterial strains produced exudates that could support the growth of each of the three bacterial strains. However, the mean maximum biomass of each bacterium differed widely among exudate sources (Table 2). When grown only on exudates, each bacterium achieved average cell densities of 18 × 107 cells mL−1−1 (P1), 9.5 × 107 cells mL−1 (CN13), and 16 × 107 cells mL−1−1 (JMP134). Much of the variability in maximum biomass that CN13 and JMP134 reached on each exudate could be explained by TOC (Table 3) (66% and 75%, respectively) (Fig. 1). The regression of P1 maximum biomass with TOC was not significant. Most notably, growth of P1 on the exudates of Tychonema bourrellyi (exudate 17) and Phormidium rimosum (exudate 3) was unusually low, whereas growth on the exudate of P. autumnale (exudate2) was unusually high (Fig. 1). Multiple regression analyses using TOC and average molecular weight (AMW) as model predictors of bacterial biomass show that much of the variation in CN13 and JMP134 cell densities could be mainly explained by TOC (Table 4). P1 cell densities did not significantly correlate with either TOC or AMW.
Linear regression plots of mean maximum biomass as a function of the total organic carbon (TOC) of cyanobacterial exudates in the medium. NS: not statistically significant. Each number label represents the individual exudate number allocated to cyanobacterial isolates in Table 1.
Cluster analyses based on the molecular weight profile of the UV-absorbing compounds indicated that most cyanobacterial exudates fell into two major groups of greater than 80% dissimilarity (Fig. 2). Aphanothece floccosa, Phormidium animale, and P. insigne gave chromatograms that were <1% dissimilar, which suggests that although they may be assigned to different species, their metabolisms may be highly similar. The other group was composed of all the Geitlerinema species, plus P. rimosum, and T. bourrellyi. More loosely associated with this group were A. rivularis and P. autumnale. Pseudanabaena PP16 stood out as being significantly dissimilar from all the others. There was no obvious correspondence between molecular weight profiles and growth parameters.
When exudate and the substrates phenol, DCA, or 2,4-D were available in combination, the affect on growth efficiency was also variable among strains (Table 2). Most notably, when phenol was available with exudates from P. animale and T. bourrellyi, P1 biomass values were not only less than additive, but less than biomass values for growth on phenol alone. In contrast, CN13 experienced a synergistic effect in most cases when exudates and DCA were both available. JMP134 generally had less than additive growth efficiencies when grown on exudates with 2,4-D.
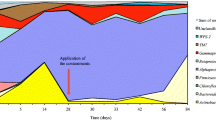
The results of P1 growth and phenol biodegradation in media and in the presence of exudates of four cyanobacteria are shown in Fig. 3. Exudates from A. rivularis, P. animale, and P. insigne significantly (repeated-measures ANOVA, p < 0.01) decreased phenol biodegradation and P1 growth. In contrast, Pseudanabaena PP16 had a positive affect on P1 and phenol degradation. Repeated-measures ANOVA indicated that the exudates of Pseudanabaena PP16 had a significantly (p < 0.01) positive affect on P1 growth and phenol biodegradation. Growth of CN13 and biodegradation of DCA for each treatment are presented in Fig. 4. Cell densities were all significantly (repeated-measures ANOVA, p < 0.001) greater in exudate treatments than in media controls. In the presence of exudates from Pseudanabaena PP16, P. animale, and P. insigne, DCA removed by day 7 was significantly (ANOVA, p < 0.001) greater than in the controls. In contrast, the presence of exudates from A. rivularis significantly (ANOVA, p < 0.001) decreased DCA biodegradation when compared to the medium control.
Discussion
The results from this study demonstrate the potential for cyanobacterial exudates to affect the metabolism of heterotrophic bacterial degraders, particularly in pulp and paper waste treatment systems. Cyanobacterial exudates would provide both an endogenous source of growth substrates to bacteria, as well as influence the rate of degradation of key aromatic and aliphatic contaminants. The differential response of the three bacterial species to exudates indicates that, as in natural aquatic systems, cyanobacteria have the potential to alter microbial community structure in pulp and paper waste treatment systems. Although we were not able to fully characterize the compounds in the exudates of our 12 cyanobacterial strains, cluster analysis shows that in terms of the molecular weight distribution of the UV absorbing fraction (i.e., aromatic and quinone structures), there was sufficient variability between species to allow them to separate into distinct groupings. In effect, our results not only demonstrate that diverse communities of cyanobacteria would likely contribute variable quantities of dissolved organic carbon to wastewater, but also exudates of variable quality for bacterial utilization. Moreover, these results also emphasize the importance of not lumping cyanobacteria, a diverse group of photosynthetic microorganisms, into one functional category.
What the cyanobacterial exudates did have in common was a relatively high AMW. As with other dissolved organic compounds, the bioavailability of exudates to bacteria would likely be related to the molecular size of its constituents. The larger the organic compound, the less available it would be for bacterial uptake and utilization. Cyanobacterial exudates are composed of numerous organic constituents that can either be predominantly high molecular weight (HMW > 1000 Da) [7, 23] or low molecular weight (LMW < 1000 Da) [12, 30]. The HMW substances detected in our analyses were likely aromatic compounds (e.g., polyphenols) and proteins. LMW compounds such as simple sugars would not have been detected.
Based on literature reports on the fate of HMW compounds in aquatic [7, 12] and pulp and paper waste treatment systems [11, 19], the HMW fraction in cyanobacterial exudates would not be expected to mineralize, but rather accumulate during biological treatment. As two of our heterotrophic bacteria seemed to metabolize exudate organic carbon independent of its molecular weight, these earlier findings might be more a reflection of the difference between the constituents of persistent HMW material in these systems (e.g., lignin), relative to those of cyanobacterial exudates (e.g., proteins).
A number of studies have shown that cyanobacterial exudates usually contain a host of sugars [2, 5, 7, 13, 25]. Nicolaus et al. [25] characterized the exopolysaccharides of 16 filamentous cyanobacteria, detecting an abundance of carbohydrates, such as glucose and galactose, which were universally produced. The Phormidium sp. assessed in their study was found to generate the most exopolysaccharide (30 mg L−1−1), the majority (>80%) of which was soluble. This is of particular relevance to our study as Phormidium and its subgenus Geitlerinema are among our test species and are typical members of cyanobacterial communities in pulp and paper waste treatment systems [17]. In addition to carbohydrates, other compounds known to be present in cyanobacterial exudates such as amino sugars, amino acids and other nitrogenous organics [21, 22, 31] would be available.
When exudate and contaminant substrate were provided together, bacterial growth responses were quite variable among strains and exudate sources (Table 2). When compared to the wastewater bacteria P1 and CN13, the soil bacterium JMP134 generally had less than additive growth on exudates and 2,4-D, which may indicate some substrate competition or catabolite repression. Unlike the wastewater bacteria, the soil bacterium JMP134 has been strongly selected for 2,4-D degradation, rather than for utilization of mixed substrates. However, not all exudates produced additive or greater than additive growth of the wastewater bacterium P1. Exudates from P. animale and T. bourrellyi clearly had an inhibitory affect on P1 in the presence of phenol. In contrast, CN13 greatly benefited from the presence of most exudates when grown with DCA. This was likely due to the readily metabolizable nature of the exudates, as well as the presence of additional growth factors (i.e., vitamins and cofactors) that ameliorated the catabolism of DCA.
Consistent with the growth data, exudate from P. animale also inhibited phenol biodegradation (Fig. 3). This suggests a toxic rather than competitive effect from this exudate on P1. In general, our biodegradation results showed that phenol removal and P1 growth were significantly lower in the presence of exudates from three of the four exudate sources, but were slightly stimulated by exudates from Pseudanabaena PP16. We suspect that this contrasting effect of Pseudanabaena PP16 is related to its unique exudate composition evident from the analyses of UV-absorbing compounds (Fig. 2).
In contrast, DCA biodegradation by CN13 was more efficient in three of the four exudate treatments tested (Fig. 4). Apparently, CN13 was using both cyanobacterial exudates and DCA for growth, and the increased DCA removed may have occurred as a consequence of the higher cell densities achieved by growth on exudates. However, DCA biodegradation remained unchanged from day 4 to day 7 in the presence of exudates from A. rivularis, even though CN13 growth was significantly greater on this exudate source compared to the medium control. This shows that CN13 was preferentially utilizing exudates for growth at the expense of DCA utilization, a scenario that would be undesirable during waste treatment.
Contrary to some literature reports [6, 26, 27], the cyanobacterial exudates assessed in our study do not completely inhibit bacterial growth, but rather can serve as growth substrates and growth supplements. The lack of complete growth inhibition by cyanobacterial exudates, particularly to those bacteria isolated from waste treatment systems, may simply be the result of bacterial selection by, or adaptation to, exudates in pulp and paper wastewaters. The influence of cyanobacterial exudates on bacterial community composition has been documented in other systems [6, 27], showing that dominant members of the bacterial community could tolerate or benefit from cyanobacterial exudates. The absence of other bacterial taxa was found to be related to their intolerance to exudates.
The production of exudates by cyanobacteria in pulp and paper secondary waste treatment systems has important implications for microbial community metabolism and structure. Our results suggest that exudates present in wastewater would likely be metabolized to varying degrees by the heterotrophic community. Those bacteria that can utilize exudates as a growth substrate would have an advantage over those bacteria that cannot, particularly during cyanobacterial bloom events. This could be a desirable occurrence in pulp and paper secondary waste treatment systems, as the maintenance of high bacterial densities is a primary objective. However, the maintenance of high bacterial densities on cyanobacterial exudates would also have to promote the catabolism of organic contaminants for this to be an effective waste treatment strategy. Based on our findings, there were significant, yet variable impacts on biodegradation efficiency by cyanobacterial exudates. Not only do these impacts vary by exudate source, but also between bacterial taxa. As such, final wastewater quality may be dependent on the abundance and diversity of cyanobacteria in the wastewater community, and the proportion of beneficial and/or antagonistic exudates that are produced.
References
MM Allen (1968) ArticleTitleSimple conditions for the growth of unicellular blue-green algae on plates J Phycol 4 1–4 Occurrence Handle1:CAS:528:DyaF1MXotVKl
Y Amemiya K Kato T Okino O Nakayama (1990) ArticleTitleChanges in chemical composition of carbohydrates and proteins in surface water during a bloom of Microcystis in Lake Suwa Ecol Res 5 153–162 Occurrence Handle1:CAS:528:DyaK3MXitlCltrc%3D
SB Baines ML Pace (1991) ArticleTitleThe production of dissolved organic matter by phytoplankton and its importance to bacteria: pattern across marine and freshwater systems Limnol Oceanogr 36 1078–1090
MA Borowitzka (1995) ArticleTitleMicroalgae as sources of pharmaceuticals and other biologically active compounds J Appl Phycol 7 3–15 Occurrence Handle1:CAS:528:DyaK2MXkvF2ju78%3D
RO Canizares-Villanueva AR Dominguez MS Cruz E Rios Leal (1995) ArticleTitleChemical composition of cyanobacteria grown in diluted, aerated swine wastewater Bioresour Technol 51 111–116 Occurrence Handle1:CAS:528:DyaK2MXls1Srsbw%3D
DA Casamatta CE Wickstrom (2000) ArticleTitleSensitivity of two disjunct bacterioplankton communities to EOCs from the cyanobacterium Microcystis aeruginosa Kutzing Microb Ecol 40 64–73 Occurrence Handle1:CAS:528:DC%2BD3cXnt12mu7o%3D Occurrence Handle10977878
JC Cho MW Kim DH Lee SJ Kim (1997) ArticleTitleResponse of bacterial communities to changes in composition of extracellular organic carbon from phytoplankton in Daechung Reservoir (Korea) Arch Hydrobiol 138 559–576 Occurrence Handle1:CAS:528:DyaK2sXit1ektL0%3D
KB Christoffersen B Riemann LR Hansen A Klysner HB Sorenson (1990) ArticleTitleQualitative importance of the microbial loop and plankton community structure in a eutrophic lake during a bloom of cyanobacteria Microb Ecol 20 253–272
JJ Cole (1982) ArticleTitleInteractions between bacteria and algae in aquatic ecosystems Annu Rev Ecol Syst 13 291–314 Occurrence Handle10.1146/annurev.es.13.110182.001451
RH Don AJ Weightman H-J Knackmuss KN Timmis (1985) ArticleTitleTransposon mutagensis and cloning analysis of the pathways for degradation of 2,4-dichlorophenoxyacetic acid and 3-chlorobenzoate in Alcaligenes eutrophus JMP134(pJP4) J Bacteriol 161 85–90 Occurrence Handle1:CAS:528:DyaL2MXhs1Wntrg%3D Occurrence Handle2981813
K Eriksson M Kolar (1985) ArticleTitleMicrobial degradation of chlorolignins Environ Sci Technol 19 1086–1089 Occurrence Handle1:CAS:528:DyaL2MXlsVemurg%3D
PM Fedorak PM Huck (1988) ArticleTitleMicrobial metabolism of cyanobacterial products: batch culture studies with applications to drinking water treatment Water Res 22 1267–1278 Occurrence Handle10.1016/0043-1354(88)90114-5 Occurrence Handle1:CAS:528:DyaL1MXot1WksA%3D%3D
GE Fogg C Nalewajko WD Watt (1965) ArticleTitleExtracellular products of phytoplankton photosynthesis Proc R Soc B162 517–534
RR Fulthorpe SN Liss DG Allen (1993) ArticleTitleCharacterization of bacteria isolated from a bleached kraft pulp mill wastewater treatment system Can J Microbiol 39 13–24 Occurrence Handle1:CAS:528:DyaK3sXhvVOksLk%3D Occurrence Handle8439869 Occurrence Handle10.1139/m93-003
RR Fulthorpe DG Allen (1995) ArticleTitleA comparison of organochlorine removal from bleached kraft pulp and paper-mill effluents by dehalogenating Pseudomonas, Anclyobacter and Methylobacterium strains Appl Microbiol Biotechnol 42 782–789 Occurrence Handle1:CAS:528:DyaK2MXjvF2qtbg%3D Occurrence Handle7765918
N Kamjunke W Boeing H Voight (1997) ArticleTitleBacterial and primary production under hypertrophic conditions Aquat Microb Ecol 13 IssueID1 29–35
AE Kirkwood C Nalewajko RR Fulthorpe (2001) ArticleTitleThe occurrence of cyanobacteria in pulp and paper waste-treatment systems Can J Microbiol 47 761–766 Occurrence Handle10.1139/cjm-47-8-761 Occurrence Handle1:CAS:528:DC%2BD3MXnt12gu78%3D Occurrence Handle11575503
AE Kirkwood C Nalewajko RR Fulthorpe (2003) ArticleTitlePhysiological characteristics of cyanobacteria from pulp and paper waste-treatment systems J Appl Phycol 15 325–335 Occurrence Handle10.1023/A:1025181116714 Occurrence Handle1:CAS:528:DC%2BD3sXmtFKksbc%3D
KP Kringstad K Lindstrom (1984) ArticleTitleSpent liquors from pulp bleaching Environ Sci Technol 18 236A–248A Occurrence Handle1:CAS:528:DyaL2cXkslyhs7k%3D Occurrence Handle10.1021/es00121a012
W Lange (1967) ArticleTitleEffect of carbohydrates on the symbiotic growth of planktonic blue-green algae with bacteria Nature 215 1277–1278 Occurrence Handle1:CAS:528:DyaF2sXkvFOktbo%3D Occurrence Handle6052729
ME Meffert (1993) ArticleTitleThe relations between Limnothrix redekei (Cyanophyta) and the accompanying heterotrophic bacteria Arch Hydrobiol Suppl 100 57–73
HV Morvan LJF Gloaguen L Hoffmann (1997) ArticleTitleStructure–function investigations on capsular polymers as a necessary step for new biotechnological applications: the case of the cyanobacterium Mastigocladus laminosus Plant Physiol Biochem 35 671–683 Occurrence Handle1:CAS:528:DyaK2sXmt1ymsrk%3D
C Nalewajko DRS Lean (1972) ArticleTitleGrowth and excretion in planktonic algae and bacteria J Phycol 12 1–5
C Nalewajko TG Dunstall H Shear (1976) ArticleTitleKinetics of extracellular release in axenic algae and in mixed algal–bacterial cultures: significance in estimation of total (gross) phytoplankton excretion rates J Phycol 12 1–5
BA Nicolaus A Panico L Lama I Romano MC Manca A DeGiulio A Gambacorta (1999) ArticleTitleChemical composition and production of exopolysaccharides from representative members of heterocystous and non-heterocystous cyanobacteria Phytochemistry 52 639–647 Occurrence Handle10.1016/S0031-9422(99)00202-2 Occurrence Handle1:CAS:528:DyaK1MXntlOjtr8%3D
O Ostensvik OM Skulberg B Underal V Hormazabal (1998) ArticleTitleAntibacterial properties of extracts from selected planktonic freshwater cyanobacteria: a comparative study of bacterial bioassays J Appl Microbiol 84 1117–1124 Occurrence Handle10.1046/j.1365-2672.1998.00449.x Occurrence Handle1:STN:280:DyaK1czotlShtA%3D%3D Occurrence Handle9717298
K Oufdou N Mezrioui B Oudra M Barakate M Loudiki (1998) ArticleTitleEffect of extracellular and endocellular products from cyanobacterium, Synechocystis sp., on the growth of some irrigation sanitation system bacteria Arch Hydrobiol, Suppl 125 139–148 Occurrence Handle1:CAS:528:DyaK1cXnvVaitb4%3D
RD Robarts T Zohary (1986) ArticleTitleInfluence of cyanobacterial hyperscum on heterotrophic activity of planktonic bacteria in a hypertrophic lake Appl Environ Microbiol 51 609–613 Occurrence Handle1:CAS:528:DyaL28XhsFGns7o%3D Occurrence Handle16347022
JJ Staley AE Konopka (1984) Genus Microcyclus NR Krieg JG Holt (Eds) Bergey's Manual of Systematic Bacteriology, Vol. 1 Williams and Wilkins Baltimore 133–134
I Sundh (1992) ArticleTitleBiochemical composition of dissolved organic carbon released from phytoplankton and used by heterotrophic bacteria Appl Environ Microbiol 58 2930–2947
BA Whitton (1965) ArticleTitleExtracellular products of blue-green algae J Gen Microbiol 40 1–11 Occurrence Handle1:CAS:528:DyaF28Xjsl2gtQ%3D%3D Occurrence Handle4285448
Acknowledgments
The authors wish to thank the University of Toronto Pulp and Paper Industrial Consortium members Aracruz, Domtar, Georgia-Pacific, Repap, Tasman, and the Pulp and Paper Centre, University of Toronto for financial assistance to R. Fulthorpe. The National Science and Engineering Research Council of Canada also provided funding to R. Fulthorpe and C. Nalewajko. We also extend our appreciation to T. Stuthridge at Forest Research Institute for acquiring access to pulp and paper waste treatment systems in New Zealand. Special thanks to H. Kling for her expertise in identifying the Chroococcales in our study, and C. Milestone for his assistance with molecular weight analyses.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kirkwood, A.E., Nalewajko, C. & Fulthorpe, R.R. The Effects of Cyanobacterial Exudates on Bacterial Growth and Biodegradation of Organic Contaminants. Microb Ecol 51, 4–12 (2006). https://doi.org/10.1007/s00248-004-0058-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-004-0058-y