Abstract
Most patients presenting cystinuria require multiple urological procedures during their lifetime. In this kind of patients the availability of minimally invasive procedure represents an advantage of minimizing the cumulative morbidity of several repeated treatments. Herein we report our experience using ureterorenoscopy (URS) for the treatment of recurrent renal cystine stones. From 2003 to 2007, 10 patients (4 males and 6 females) with one or multiple recurrent renal cystine stones underwent URS. Overall, 21 procedures have been performed. Mean maximum diameter of stones was 11.2 mm (range 5–30 mm). Either 8–9.5 F semirigid or 7.9 F flexible ureteroscopes were used. In 6 cases, stones were removed using a basket; in 9 procedures laser lithotripsy with flexible scope was performed; in 6 cases renal calculi were pulled down in the ureter using flexible instrument and then shattered with laser introduced by semirigid instrument. Stone-free status was defined as the absence of any residual fragment. A complete stone clearance was obtained in 15 out of 21 procedures (71%). In 5 cases (24%) significant residual fragments occurred; in the remaining case (5%) URS was ineffective. In 5 out of these unsuccessful procedures, stone clearance was obtained with auxiliary treatments. The last patient has not been treated yet. No major complications occurred as a result of the procedures. URS offers excellent advantages in case of recurrent hard calculi such as cystine stones. Minimally invasive procedures allow satisfactory outcomes, improving patients’ quality of life.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the past 25 years, the treatment of urolithiasis showed radical changes due to the development of less invasive treatment options like ureterorenoscopy (URS), percutaneous nephrolithotomy (PNL) and extracorporeal shock wave lithotripsy (ESWL). Currently open surgery is the modality of treatment in <5% of the cases [1–3].
Due to its apparent low morbidity, ESWL is the preferred treatment in the management of ureteral and renal stones with a maximum diameter up to 1.5 and 2 cm, respectively [2, 4].
From several surveys, a wide range of stone free rates is achieved following ESWL. The low-invasive nature and morbidity related to ESWL probably contributed to extending the indications of this therapeutic option. At the same time they explain the discordant results and frequently determine a large percentage of re-treatment [5–7]. Furthermore, treatment efficacy changes in relation to some parameters such as chemical stone composition [8–10], surface area [11, 12], localization [11] and morphology of the upper urinary tract [13–16], influence both the disintegration and the subsequent stone clearance.
Referring to chemical composition, calcium oxalate dehydrate, uric acid and magnesium ammonium phosphate stones are well susceptible to shock-wave treatment; other stones, such as cystine or calcium oxalate monohydrate calculi may be variably resistant [8–10].
For these reasons, retrograde approach to the upper urinary tract has become a widely diffused procedure aimed at removing both ureteral and renal stones. With the development of new small rigid and flexible instruments, in association with the availability of small laser fibers for stone fragmentation, URS may be considered quite a safe and effective procedure in the treatment of the upper urinary calculi, even in paediatric population [17]. Moreover, without doubt this modality of treatment appears to be less invasive than PNL and more effective than ESWL, especially in the management of hard stones [17]. Indeed, in the last decade there was an increased use of holmium laser, which is highly effective in stone fragmentation, independent of stone composition [18]. The availability of this minimally invasive procedure to retreat patients with recurrent stones could be an advantage to minimize the cumulative morbidity of several repeated re-treatments. Maximum stone size for URS depends on the available instruments and lithotripsy probes. Usually, even for ESWL. The upper limit for a renal stone may be considered 2 cm.
There is a wide consensus that cystine stone represents a serious problem due to its high recurrence rates. Although cystine calculi may occur at any age, they may frequently be evident in young people (usually within the first two decades) and tend to be recurrent over time, even within short periods. Despite preventive medical management, patients presenting cystinuria often require multiple urological procedures during their lifetime [17]. Therefore, endourological conservative treatments seem to improve the quality of life of these patients, where medical therapy may not be effective enough to prevent stone recurrences.
Herein is reported our experience relating to patients where a retrograde transureteral approach for recurrent cystine renal stones uses performed.
Materials and methods
From 2003 to 2007, 10 patients (4 males and 6 females) with one or multiple renal cystine stones have been treated through a retrograde transureteral approach. Patients’ mean age was 28.7 years (ranged from 6 to 44 years). Overall, 21 ureteropyeloscopic procedures have been performed. In one patient, a second URS was performed a few days after the primary treatment because of significant residual fragments. Later on, six patients underwent another URS: 5 of them required a third retrograde treatment subsequently, because of recurrent stones. Demographic characteristics of the study population are summarized in Table 1.
Stone localization and size were generally obtained by an abdominal ultrasound (US), combined with a plain film of the abdomen (KUB), or by a computed tomography (CT); intravenous pyelography (IVP) was rarely used. The maximum stone diameter ranged from 5 to 30 mm (average 11.2 mm). Most cases presented with a single stone (67%); the others had multiple cystine stones (33%). Table 2 shows the localization of both single and multiple stones.
According to stone location, ureteroscopic approach was performed using either 8 F or 9.5 F semirigid scopes, with 4 or 5 F operative working channel respectively, or the 2-way actively deflectable (270°/270°) Storz Flex-X2 flexible ureteroscope, with a 3.6 F working channel.
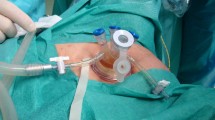
Stone lithotripsy was carried out using 230 μm diameter holmium-YAG laser fibers for flexible instrument; 365 μm fibers were used with the semirigid instruments. The laser energy and frequency settings were 0.8 J and 6–8 Hz, respectively. In 6 cases, stones were removed by simple basketing. In 9 cases, laser lithotripsy was performed through a flexible ureteroscope. In 6 other cases, renal calculi have been pulled down in the ureter using the flexible instrument and then fragmented with the laser using a semirigid ureteroscope. For removal of stone fragments either 1.9 F or 2.4 F Nitinol baskets were employed. Postoperatively, patients were followed with KUB and US of the abdomen: 1 month after URS and then repeated every 6 months subsequently. CT scans of the abdomen were seldom performed. Stone-free status was defined as the absence of any residual fragment. As a technical consideration, a 13 F ureteral access sheath was used in performing the flexible URS. In this way insertion and removal of the flexible endoscopes were far easier (Fig. 1).
Clinical data such as operative times, maximum voltage, localization system, types of anaesthesia and eventual auxiliary manoeuvres have been obtained from a database elaborated in our department.
Results
A complete stone clearance was obtained in 15 out of the 21 procedures (71%). In 5 cases (24%) residual significant fragments occurred: the stone clearance was obtained by means of auxiliary treatments: two cases underwent ESWL; one, a second URS; in 2 cases, a PNL was performed 1 month after URS. In 1 case (5%) URS was ineffective and the patient was no longer treated.
At the end of the ureteroscopic procedures, according to the different outcomes, either a double J stent (8 procedures) or a single J catheter (12 procedures) was left in situ from 2 days up to 1 month. In one case no ureteral stent was applied, as the basketing of a small stone was totally successful and the trauma on the ureteral wall was considered minimal.
Minor complications were observed in 5 cases: fever, <38°C (2 cases); transitory flank pain (2 cases); gross haematuria (1 case). None of these patients required additional manoeuvres or treatments, apart from symptomatic therapy. No major complications occurred as a result of the procedures.
Discussion
Cystinuria is a main cause of urolithiasis because of the high recurrence rate: in up to 60% of these patients, tends to recur [17]. Nowadays, it is the cause of about 1–2% of urinary stones in adults and 10% in children [17, 19]. Cystinuria is an autosomal recessive genetic disorder due to chromosomal mutations on chromosome 2 or/and 19 (respectively type A, type B and type AB). It is characterized by an impaired transport in the proximal renal tubule of the aminoacids cystine, ornithine, lysine and arginine but only cystine is insoluble enough to form urinary stones: cystine is poorly soluble at physiological urine pH values between 5 and 7 [17, 19].
Stone recurrences are highly likely for these patients even if preventive medical treatments are used, due to insufficient effectiveness of the therapy and low patients’ compliance. None, a specific medical regimen (hyper-hydratation, urine alkalinization and administration of cystine binders such as d-penicillamine and Tiopronin) may significantly increase stone free intervals [17, 19, 20].
Therefore, successful management of cystinuria requires a high lifelong patient compliance, regular follow-up examinations and, if necessary, state-of-the-art medical therapy. In our opinion biannual follow-up with ultrasound scan, urinalysis and preventive alkalinization are advised. In this way it is possible to detect small cystine stones still susceptible to minimal invasive treatments, such as ESWL and/or URS.
ESWL monotherapy provides satisfactory results only in patients with <1 cm pelvic cystine stones. For those with >1.5 cm diameter, the reported stone-free rate is about 71%; this percentage drops to 40% when the diameter exceeded 2 cm [12]. Moreover, patients with larger calculi up to 66% need repeated ESWL sections to reach good results [21].
Instead of multiple ESWL sessions, URS might represent a safe and effective treatment for all patients with ≥1 cm renal cystine stones and this procedure, despite being more invasive than ESWL, could resolve nephrolithiasis in only one session, even in prepubertal children [22]. Actually, in order to achieve good outcomes with this minimally invasive procedure, it is advisable that stones’ volume and number are limited. Usually the upper limit is 2 cm in maximum diameter [2] while there is no unanimous consensus concerning the maximum number of renal stones successfully treatable with URS. Concerning that, it is usually accepted that for stones with a diameter more than 2 cm, as far as for multiple calculi, the best modality of treatment is represented by the percutaneous approach that guarantees an high success rate, even if with more invasiveness and intra- and post-operative complications rate.
Moreover, in the debate between ESWL and URS, a significant role in the evaluation of a successful outcome is played by the definition of “stone free” status. In literature, there is a particular entity named “CIRF” standing for “Clinical Insignificant Residual Fragment” (≤4 mm fragment maximum diameter) aiming at defining the “stone free” condition after ESWL [23]. On the other side, provided that there is no clear definition to identify the stone free condition after URSs, most authors define the stone-free status as the complete absence of any residual fragment.
According to this statement, in the present study we codify the absence of residual fragments after single procedure as stone-free status, obtaining an overall success rate of 71% without significant complications. Even if our stone free rate appears to be similar to that reported in literature performing ESWL, it is important to emphasize that it has been obtained by single URS procedures while the outcomes reported using ESWL were obtained with repeated sessions in a high percentage of cases and only treating stones with a maximum diameter <20 mm [12, 21]. In contrast, our overall success rate included retrograde treatments of stones up to 30 mm, even if our limited series of patients did not permit a stratification analysis.
Conclusions
Retrograde transureteral removal of renal stones has to be considered a valid approach. It offers excellent advantages particularly in case of recurrent stone disease as it happens when we deal with cystine stones. These patients often require several treatments, sometimes within short periods of time, despite an accurate medical regimen. Minimally invasive procedures allow satisfactory outcomes and improve patients’ quality of life.
It is mandatory to recognize stones when their volume is still treatable with URS. In our experience, stone volume, and not the number of stones, seems to be the most limiting factor, having excluded anatomical abnormalities conditioning instruments passage. Therefore, patients at risk of stone recurrences must be suggested to perform a complete abdominal US twice a year throughout their life aimed at minimizing the occurrence of asymptomatic large renal stones that could require more invasive approaches.
Ethical considerations
The study was conducted in accordance with the ethical principles laid down in the 1964 Declaration of Helsinki and its later amendments. All persons gave their informed consent prior to their inclusion in the study.
References
Tiselius HG (2005) Removal of ureteral stones with extracorporeal shock wave lithotripsy and ureteroscopic procedures. What can we learn from the literature in terms of results and treatment efforts? Urol Res 33:185–190
Tiselius HG, Ackermann D, Alken P, Buck C, Conort P, Gallucci M (2001) Guidelines on urolithiasis. Eur Urol 40:362–371
Paik ML, Wainstein MA, Spirnak JP, Hampel N, Resnick MI (1998) Current indications for open stone surgery in the treatment of renal and ureteral calculi. J Urol 159:374–379
Lingeman JE (1996) Lithotripsy and surgery. Semin Nephrol 16:487–498
Lingeman JE, Coury TA, Newman DM, Kahnoski RJ, Mertz JH, Mosbaugh PG, Steele RE, Woods JR (1987) Comparison of results and morbidity of percutaneous nephrostolithotomy and extracorporeal shock wave lithotripsy. J Urol 138:485–490
Havel D, Saussine C, Fath C, Lang H, Faure F, Jacqmin D (1998) Single stones of the lower pole of the kidney. Comparative results of extracorporeal shock wave lithotripsy and percutaneous nephrostolithotomy. Eur Urol 33:396–400
May DJ, Chandhoke PS (1998) Efficacy and cost-effectiveness of extracorporeal shock wave lithotripsy for solitary lower pole renal calculi. J Urol 159:24–27
Graff J, Diederichs W, Schulze H (1988) Long-term follow-up in 1003 extracorporeal shock wave lithotripsy patients. J Urol 140:479–483
Schmeller NT, Kersting H, Schuller J, Chaussy C, Schmiedt E (1984) Combination of chemolysis and shock wave lithotripsy in the treatment of cystine renal calculi. J Urol 131:434–438
Wang LP, Wong HY, Griffith DP (1997) Treatment options in struvite stones. Urol Clin North Am 24:149–162
Politis G, Griffith DP (1987) ESWL: stone-free efficacy based upon stone size and location. World J Urol 5:255–258
Netto NR Jr, Claro JFA, Lemos GC, Cortado PL (1991) Renal calculi in lower pole calices: what is the best method of treatment? J Urol 146:721–723
Locke DR, Newman RC, Steinbock GS, Finlayson B (1990) Extracorporeal shock wave lithotripsy in horseshoe kidneys. Urology 35:407–411
Sampaio FJB, Aragao AHM (1994) Limitations of extracorporeal shockwave lithotripsy for lower caliceal stones: anatomic insight. J Endourol 8:241–247
Elbahnasy AM, Clayman RV, Shalhav AL, Hoenig DM, Chandhoke P, Lingeman JE, Denstedt JD, Kahn R, Assimos DG, Nakada SY (1998) Lower-pole caliceal stone clearance after shockwave lithotripsy, percutaneous nephrostolithotomy, and flexible ureteroscopy: impact of radiographic spatial anatomy. J Endourol 12:113–119
Elbahnasy AM, Shalhav AL, Hoenig DM, Elashry OM, Smith DS, Mcdougall EM, Clayman RV (1998) Lower caliceal stone clearance after shock wave lithotripsy or ureteroscopy: the impact of lower pole radiographic anatomy. J Urol 159:676–682
Knoll T, Zöllner A, Wendt-Nordahl G, Michel MS, Alken P (2005) Cystinuria in childhood and adolescence: recommendations for diagnosis, treatment, and follow up. Pediatr Nephrol 20:19–24
Yiu MK, Liu PL, Yiu TF, Chan AYT (1996) Clinical experience with holmium:YAG laser lithotripsy of ureteral calculi. Lasers Surg Med 19:103–106
Coe FL, Evan A, Worcester E (2005) Kidney stone disease. J Clin Invest 115:2598–2608
Straub M, Strohmaier WL, Berg W, Beck B, Hoppe B, Laube N, Lahme S, Schmidt M, Hesse A, Koehrmann KU (2005) Diagnosis and metaphylaxis of stone disease Consensus concept of the National Working Committee on Stone Disease for the Upcoming German Urolithiasis Guideline. World J Urol 23:309–323
Katz G, Lencovsky Z, Pode D, Shapiro A, Caine M (1990) Place of extracorporeal shock-wave lithotripsy (ESWL) in the management of cystine calculi. Urology 36:124–128
Schuster TG, Russell KY, Bloom DA, Koo HP, Faerber GJ (2002) Ureteroscopy for the treatment of urolithiasis in children. J Urol 167:1813
Streem SB, Yost A, Mascha E (1996) Clinical implications of clinically insignificant stone fragments after extracorporeal shock wave lithotripsy. J Urol 155:1186–1190
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ruggera, L., Zanin, M., Beltrami, P. et al. Retrograde transureteral approach: a safe and efficient treatment for recurrent cystine renal stones. Urol Res 39, 411–415 (2011). https://doi.org/10.1007/s00240-010-0358-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00240-010-0358-2