Abstract
Introduction
The purpose of this prospective study was to compare 3T and 1.5T magnetic resonance angiography (MRA) with digital subtraction angiography (DSA) for the follow-up of endovascular treated intracranial aneurysms to assess the grade of occlusion.
Materials and methods
Thirty-seven patients with 41 aneurysms who had undergone endovascular treatment with detachable coils were included. MRA was performed on the same day using an eight-channel sensitivity encoding head-coil with 3D axial inflow technique. At 3T, a contrast-enhanced transverse 3D fast gradient echo acquisition was also performed. Most patients underwent DSA the following day. MRA scans and DSA were classified first independently by two neuroradiologists and an interventional neuroradiologist. Secondly, a consensus was done. Source images, maximum intensity projection, multiplanar reconstruction and volume rendering reconstructions were used for MRA evaluations. A modification of the Raymond classification, previously used for DSA evaluation of recanalization, was used.
Results
Statistical comparison of the consensus showed that 3T MRA with 3D axial inflow technique had better agreement with DSA (κ = 0.43) than 1.5T MRA(κ = 0.21) and contrast-enhanced MRA (CE-MRA) at 3T (κ = 0.17). The susceptibility artefacts from the coil mesh were significally smaller at 3T (p = 0.002–0.007) than at 1.5T.
Conclusion
3T MRA, using a sensitivity encoding head-coil, showed better agreement with DSA than 1.5T and CE-MRA at 3T for evaluation of aneurysms treated with endovascular coiling.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Endovascular treatment of intracranial aneurysms with coils is used in clinical practice since more than a decade and constitutes an excellent alternative to surgery [1]. The patients usually undergo follow-up examinations with digital subtraction angiography (DSA), although magnetic resonance angiography (MRA) is used more and more frequently because of the risk of aneurysm regrowth or recanalization due to coil compaction or enlargement of a remnant [2, 3]. DSA is an invasive technique with risk of neurological complications, although low (1.3%) [4]. Computed tomography angiography (CTA) is not appropriate for follow-up due to severe beam hardening artefacts from the platinum coils. A disadvantage with DSA as well as CTA is also the additional radiation dose to the patients who already have been exposed to ionizing radiation during the endovascular treatment. Magnetic resonance angiography is a fast and non-invasive technique without ionizing radiation for detection and characterization of intracranial aneurysms but has limitations especially for the evaluation of small aneurysms [5, 6]. Several groups have reported the usefulness of MRA for evaluation of coiled aneurysms using 3D inflow technique at 1.0T [7, 8], 1.5T [6, 9–13] and 3T [14]. Reports of contrast-enhanced MRA (CE-MRA) have also been published [15–17] including studies performed before and after injection of gadolinium-based contrast agents [18–22]. Comparison between the field strengths 1.5T and 3T for MRA have been reported in other patients than those with coiled aneurysms [23–25]. It was concluded that 3D inflow MRA at 3T offers an improvement and a superior image quality compared with 1.5T. For evaluation of coiled aneurysms, the artefacts from the coil mesh could potentially be larger at higher field strength [26]. The purpose of this study was to compare the agreement between 3T MRA (before and after contrast injection) and 1.5T MRA with DSA for the follow-up of intracranial aneurysms treated with detachable coils in order to select the best MRA method.
Materials and methods
Patients and aneurysms
This prospective study was approved by the local ethics committee, and informed consent was obtained from all patients.
From March 2004 to April 2005, 37 consecutive patients with 41 aneurysms (22 women, 15 men), age 24–69 years (median 51 years), who had undergone endovascular treatment with coils for intracranial aneurysms, were included. Due to limited capacity for magnetic resonance imaging, only patients scheduled for DSA follow-up on Fridays could be included.
The endovascular treatments had been performed 3–84 months (median 27 months) prior to the follow-up examinations.
In 35 patients, subarachnoid hemorrhage was present at the time of the treatment, and in two patients, the aneurysms had been discovered incidentally.
The localisation of the aneurysms were as follows: internal carotid artery (ICA) 7, anterior communicating artery (ACom) 16, pericallosal artery (PA) 5, middle cerebral artery (MCA) 5, basilar artery (BA) 4, superior cerebellar artery (SCA) 2 and posterior inferior cerebellar artery (PICA) 2 (Table 1). According to the pre-treatment DSA, the size of the aneurysms was 2–22 mm (median 8 mm).
Examinations
The pre-treatment and post-treatment DSA as well as follow-up DSA, 3D inflow MRA at 1.5T, 3T and CE-MRA at 3T were evaluated in this study. The follow-up MRA examinations were performed the same day or the day before the follow-up DSA in most cases. In three of the patients, there was a delay between MRA and DSA exceeding 1 day due to emergency patients in the angio suite. The DSA was performed 4, 26 and 39 days after the MRA in these patients. In the two latter cases, patient’s delay added to the delay.
The DSA was performed using a biplane unit (Philips Integris, Best, the Netherlands) with the contrast agent iohexol 240 mg I/ml (Omnipaque; GE Healthcare, Chalfont St. Giles, UK) and different 2D projections. MRA examinations were performed on a 1.5T Philips Intera and a 3T Philips Intera MR scanner with the parameters shown in Table 2. For contrast-enhanced MRA, a test injection of 2 ml gadolinium-based contrast agent (Magnevist, Schering AG, Germany) followed by 30 ml saline at 2 ml/s was performed for timing using a power injector (Spectris Solaris EP, Medrad, the Netherlands). The contrast bolus of 18 ml was injected, followed by 30 ml saline with the same injection rate.
Analysis
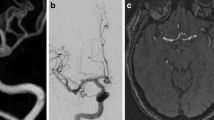
The MRA images were evaluated on a View Forum workstation (Philips, Best, the Netherlands) using source images, maximum intensity projection (MIP), multi-planar reconstruction (MPR) and volume rendering reconstructions (Fig. 1). The source images were transferred to the workstation, and reconstruction was performed interactively by the observers. They had the possibility to alter the thickness and the angulations of the MPR and MIP depending on the aneurysm location and configuration in order to get projections comparable to DSA. DSA images were evaluated on the same workstation (Figs. 2 and 3), and both frontal and lateral projections were included as well as the working projection of the aneurysm.
An anterior communicating aneurysm depicted with 3D inflow MRA at 3T (a), 3D inflow MRA at 1.5T (b) and contrast-enhanced 3T MRA (c). The first image is a source image, the second volume rendering, the third image maximum intensity projection and the fourth multiplanar reconstruction, at the workstation
The same aneurysm at the anterior communicating artery as in Fig. 1; pre-treatment (a), coilmesh (b) and post-treatment (c)
Two experienced neuroradiologists (numbers 1 and 2) and one interventional neuroradiologist (number 3) evaluated all images including the DSA. The different MRA sequences were evaluated separately and with at least one weak in between, by each neuroradiologist without knowledge of the other MRA sequences or the follow-up DSA. The pre- and post-treatment DSA were available, however, as in the clinical situation. The evaluation criteria below had been discussed and defined thoroughly before the first evaluation. The initial evaluation was done independently by each observer. Because of poor agreement between the observers even for the DSA, all cases with disagreement at the first assessment were graded in consensus. The DSA served as the gold standard in the comparisons with MRA.
The aneurysms were evaluated on both DSA and MRA according to the classification of recanalization shown in Fig. 4, which is a modification of the Raymond classification for DSA [27] Recanalization was divided into central or excentric and into different groups with regard to percentage of recanalization (Fig. 4). We also divided the aneurysms into two groups with regard to risk of rebleeding. Aneurysms with an excentric recanalization (10–49% recanalization, grade 2b in Fig. 4) and aneurysms with ≥50% recanalization (grade 3 in Fig. 4) were considered to have a potential risk of rebleeding and were therefore candidates for discussion of retreatment. Furthermore, the aneurysms were divided into two groups regarding no or any recanalization. Regrowth was defined as >20% enlargement of the recanalized aneurysm and coil mesh compared with the initial size of the aneurysm. The size of the aneurysms was measured on the pre-treatment angiogram as the longest distance from the neck to the dome of the aneurysm. On the same frame (working projection), either the ICA below the bifurcation of MCA and anterior cerebral artery was estimated to measure 4 mm, or the BA, midsection, was estimated to measure 3 mm for calibration of the measurements. The same method was used to measure the recanalization.
Classification of coiled aneurysms (modified J Raymond classification). 0 = Complete occlusion (100%) (without illustration), 1 = minor recanalization, at the base of the aneurysm (1–9% of the original size of the aneurysm) (a), 2a = moderate central recanalization, neck remnant (10–49%) (b), 2b = moderate excentric recanalization, neck remnant (10–49%) (c), 3 = major recanalization (>50% of the initial size of the aneurysm) (d)
On the MRA studies, the susceptibility artefacts from the coil mesh were measured as largest diameter in the transverse plane on the source images. Evaluation of visualization of the parent artery was done on the MRA, and finally, the general image quality was rated as good or poor.
The statistical comparison was done as a range of agreement by κ analysis, and the comparison of the coil-artefacts was evaluated with Wilcoxon signed rank test. For all statistical evaluations, the DSA was used as gold standard.
Results
In three of the patients, the CE-MRA 3T was not possible to evaluate because of bad timing of contrast. These examinations were in the statistical evaluation classified as disagreeing with the gold standard. In the first evaluation, when the observers made the analysis independently, a comparison between MRA (all three examinations) and the DSA (considered to be the gold standard) showed poor to fair (κ = 0.16–0.36) agreement using κ analysis. A comparison of the grading of DSA performed by observer number 3 (interventional neuroradiologist) with those of the other two observers showed only fair to moderate agreement (κ = 0.22 and 0.50). The evaluations with previous disagreement (80 of 164 evaluations) were graded in consensus. The statistical analysis of the total material after this consensus showed for 3D inflow MRA at 3T moderate agreement (κ = 0.43), MRA at 1.5T fair agreement (κ = 0.21) and CE-MRA at 3T poor (κ = 0.17) agreement with DSA (Table 3).
The distribution of the grade of recanalization according to the classification used is shown in Table 4. The classification was also divided into two groups aiming at separating no or minor (1, 2a) from major recanalization (2b, 3), the latter requiring consideration whether further intervention was needed or not. In this scenario, 3D inflow MRA at 3T, 1.5T MRA and CE- MRA 3T showed moderate agreement (κ = 0.50, κ = 0.42 and κ = 0.40) with DSA. The sensitivity for major recanalization (n = 26) was 50–61.5%, and there was a tendency of underestimating the grade at all the MRA examinations (Table 5). Regarding any recanalization (n = 35), the sensitivity was 82.8–91.4% (Table 6). Regrowth was seen in eight patients in the follow-up DSA, but only four of them were detected with 3D inflow MRA at 3T and 1.5T, and three were diagnosed with CE-MRA at 3T. Endovascular retreatment was performed in nine patients with moderate to major recanalization; six of them had aneurysm regrowth.
The parent artery was not visualized due to coil artefacts on MRA in one examination at 3T, in one at 1.5T and two at CE-MRA 3T, by observers 2 and 3. Observer 1 identified all the parent arteries.
The image quality was rated as good in all examinations at 1.5T, but at 3D inflow 3T, one observer considered the image quality poor in one examination. In CE-MRA 3T, all observers had marked the same three examinations as poor, but observer 1 assigned poor grading to six additional, observer 2 to two and observer 3 to seven additional CE-MRA 3T examinations.
All observers measured significantly larger susceptibility artefacts from the coil mesh at 3D inflow MRA 1.5T than at MRA at 3T (p = 0.002–0.007) and CE-MRA at 3T (p < 0.001).
Discussion
In this study, using the modified Raymond classification, 3D inflow MRA at 3T showed better agreement with DSA than 3D inflow MRA at 1.5T or CE-MRA at 3T with regard to recanalization of aneurysms after endovascular treatment with coils. In a recent study [25], it was demonstrated that the increased signal-to-noise ratio obtained at 3D inflow MRA at 3T made it possible to increase the spatial resolution, and if a high-resolution protocol with a true voxel size of 0.30 × 0.44 × 1.00 mm and a reconstructed voxel size 0.24 × 0.24 × 0.50 was used, the image quality was superior to standard resolution at 1.5T and 3T. In our study, the 3T examinations were done with sensitivity encoding (SENSE) which is a technique that enables scan time reduction, however, slightly decreasing the signal-to-noise ratio. We also obtained a reconstructed voxel size of 0.24 × 0.24 × 0.50 with a scan time of 5 min 58 s compared with 7 min 57 s in the study mentioned above. The image quality of our CE-MRA at 3T was, in some cases, poor because of inadequate contrast timing and made classification of three patients with three aneurysms impossible. A few additional CE-MRA examinations had inferior image quality but were possible to evaluate. The short time window between the arterial and the venous phase of contrast enhancement, also discussed in recent articles [17, 22], may lead to venous enhancement and vessel overlaps (Fig. 1c). Thus, incorrect timing of the contrast bolus may be a problem.
Some previous studies have compared 1.5T MRA before and after contrast injection. In one study, it was found that contrast enhancement was only beneficial in the evaluation of a giant aneurysm [19]. Another study showed that an auto-triggered elliptic-centric-ordered three-dimensional gadolinium-enhanced (ATECO) MRA is superior to 3D inflow MRA for the follow-up of coiled aneurysms [21]. In one study comparing CE-MRA with 3D inflow MRA at 1.5T at the follow-up of coiled aneurysms, no significant difference was shown [22].
Regrowth of aneurysms, in our study defined as >20% enlargement of the size compared to the initial aneurysm size, was difficult to detect on MRA. Less than half of the cases seen on DSA were detected on MRA. The reason was probably the difficulty to estimate the aneurysm size because of the coil artefacts. The metal artefacts from the coils may increase at higher field strength, but the coil artefacts in our study were smaller at 3T, probably due to higher bandwidth and shorter echo time (TE) than at 1.5T (Table 2). The opposed phase imaging of our MRA sequences required a TE at 1.5T which was twice as long as the TE used at 3T.
Recanalization was reliably detected with MRA, but there was a tendency to underestimate it. Only about half of cases with major recanalization observed on DSA were diagnosed on MRA. In ambiguous cases, it would therefore be recommended to perform a supplementary DSA. The rather poor agreement between observers (despite being experienced neuroradiologists) in the first part of our study shows that it is very important to use a grading system that is unambiguous and easy to apply. One study using examination with both DSA and 3D inflow MRA in a silicon aneurysm model filled with different percentage of coils showed poor agreement between two observers concerning the aneurysmal residue [28]. This is in agreement with the corresponding difficulties in our study.
Our grading system did get a low sensitivity of the different MRA techniques compared to DSA probably because the design of classification of recanalization was too detailed and mainly focused on comparing the different MR techniques. In a meta-analysis including 16 studies [29], sensitivity for detection of residual flow within the aneurysmal neck and/or coil mesh was 83–87%. We considered such criteria only differentiating recanalization from no recanalization in the treated aneurysm too simple in order to separate different MR techniques in our study, even if they may be useful in the clinical routine setting. In our clinical practise today, we use MRA for follow-up of endovascular treated intracranial aneurysms as the first choice because it is non-invasive and ionizing radiation is avoided. It is also important with experience and understanding of endovascular treatment when assessing coil treatment. In ambiguous cases, a supplementary DSA should be performed for evaluation of potential retreatment or not.
Conclusion
In this study, 3D inflow MRA at 3T showed better agreement with the gold standard DSA than 3D inflow MRA at 1.5T and CE-MRA 3T.
References
Molyneux A, Kerr R, Stratton I, Sandercock P, Clarke M, Shrimpton J, Holman R (2002) International Subarachnoid Aneurysm Trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised trial. Lancet 360:1267–1274
Cognard C, Weill A, Spelle L, Piotin M, Castaings L, Rey A, Moret J (1999) Long-term angiographic follow-up of 169 intracranial berry aneurysms occluded with detachable coils. Radiology 212:348–356
Raymond J, Guilbert F, Weill A, Georganos SA, Juravsky L, Lambert A, Lamoureux J, Chagnon M, Roy D (2003) Long-term angiographic recurrences after selective endovascular treatment of aneurysms with detachable coils. Stroke 34:1398–1403
Willinsky RA, Taylor SM, TerBrugge K, Farb RI, Tomlinson G, Montanera W (2003) Neurologic complications of cerebral angiography: prospective analysis of 2,899 procedures and review of the literature. Radiology 227:522–528
Papke K, Brassel F (2006) Modern cross-sectional imaging in the diagnosis and follow-up of intracranial aneurysms. Eur Radiol 16:2051–2066
Okahara M, Kiyosue H, Yamashita M, Nagatomi H, Hata H, Saginoya T, Sagara Y, Mori H (2002) Diagnostic accuracy of magnetic resonance angiography for cerebral aneurysms in correlation with 3D-digital subtraction angiographic images: a study of 133 aneurysms. Stroke 33:1803–1808
Weber W, Yousry TA, Felber SR, Henkes H, Nahser HC, Roer N, Kuhne D (2001) Noninvasive follow-up of GDC-treated saccular aneurysms by MR angiography. Eur Radiol 11:1792–1797
Nome T, Bakke SJ, Nakstad PH (2002) MR angiography in the follow-up of coiled cerebral aneurysms after treatment with Guglielmi detachable coils. Acta Radiol 43:10–14
Westerlaan HE, van der Vliet AM, Hew JM, Meiners LC, Metzemaekers JD, Mooij JJ, Oudkerk M (2005) Time-of-flight magnetic resonance angiography in the follow-up of intracranial aneurysms treated with Guglielmi detachable coils. Neuroradiology 47:622–629
Okahara M, Kiyosue H, Hori Y, Yamashita M, Nagatomi H, Mori H (2004) Three-dimensional time-of-flight MR angiography for evaluation of intracranial aneurysms after endosaccular packing with Guglielmi detachable coils: comparison with 3D digital subtraction angiography. Eur Radiol 14:1162–1168
Cottier JP, Bleuzen-Couthon A, Gallas S, Vinikoff-Sonier CB, Bertrand P, Domengie F, Barantin L, Herbreteau D (2003) Follow-up of intracranial aneurysms treated with detachable coils: comparison of plain radiographs, 3D time-of-flight MRA and digital subtraction angiography. Neuroradiology 45:818–824
Yamada N, Hayashi K, Murao K, Higashi M, Iihara K (2004) Time-of-flight MR angiography targeted to coiled intracranial aneurysms is more sensitive to residual flow than is digital subtraction angiography. Am J Neuroradiol 25:1154–1157
Kahara VJ, Seppanen SK, Ryymin PS, Mattila P, Kuurne T, Laasonen EM (1999) MR angiography with three-dimensional time-of-flight and targeted maximum-intensity-projection reconstructions in the follow-up of intracranial aneurysms embolized with Guglielmi detachable coils. Am J Neuroradiol 20:1470–1475
Majoie CB, Sprengers ME, van Rooij WJ, Lavini C, Sluzewski M, van Rijn JC, den Heeten GJ (2005) MR angiography at 3T versus digital subtraction angiography in the follow-up of intracranial aneurysms treated with detachable coils. Am J Neuroradiol 26:1349–1356
Gauvrit JY, Leclerc X, Caron S, Taschner CA, Lejeune JP, Pruvo JP (2006) Intracranial aneurysms treated with Guglielmi detachable coils: imaging follow-up with contrast-enhanced MR angiography. Stroke 37:1033–1037
Boulin A, Pierot L (2001) Follow-up of intracranial aneurysms treated with detachable coils: comparison of gadolinium-enhanced 3D time-of-flight MR angiography and digital subtraction angiography. Radiology 219:108–113
Gauvrit JY, Leclerc X, Pernodet M, Lubicz B, Lejeune JP, Leys D, Pruvo JP (2005) Intracranial aneurysms treated with Guglielmi detachable coils: usefulness of 6-month imaging follow-up with contrast-enhanced MR angiography. Am J Neuroradiol 26:515–521
Anzalone N, Righi C, Simionato F, Scomazzoni F, Pagani G, Calori G, Santino P, Scotti G (2000) Three-dimensional time-of-flight MR angiography in the evaluation of intracranial aneurysms treated with Guglielmi detachable coils. Am J Neuroradiol 21:746–752
Cottier JP, Bleuzen-Couthon A, Gallas S, Vinikoff-Sonier CB, Bertrand P, Domengie F, Barantin L, Herbreteau D (2003) Intracranial aneurysms treated with Guglielmi detachable coils: is contrast material necessary in the follow-up with 3D time-of-flight MR angiography? Am J Neuroradiol 24:1797–1803
Leclerc X, Navez JF, Gauvrit JY, Lejeune JP, Pruvo JP (2002) Aneurysms of the anterior communicating artery treated with Guglielmi detachable coils: follow-up with contrast-enhanced MR angiography. Am J Neuroradiol 23:1121–1127
Farb RI, Nag S, Scott JN, Willinsky RA, Marotta TR, Montanera WJ, Tomlinson G, Terbrugge KG (2005) Surveillance of intracranial aneurysms treated with detachable coils: a comparison of MRA techniques. Neuroradiology 47:507–515
Pierot L, Delcourt C, Bouquigny F, Breidt D, Feuillet B, Lanoix O, Gallas S (2006) Follow-up of intracranial aneurysms selectively treated with coils: Prospective evaluation of contrast-enhanced MR angiography. Am J Neuroradiol 27:744–749
Gibbs GF, Huston J 3rd, Bernstein MA, Riederer SJ, Brown RD Jr. (2004) Improved image quality of intracranial aneurysms: 3.0-T versus 1.5-T time-of-flight MR angiography. Am J Neuroradiol 25:84–87
Gaa J, Weidauer S, Requardt M, Kiefer B, Lanfermann H, Zanella FE (2004) Comparison of intracranial 3D-ToF-MRA with and without parallel acquisition techniques at 1.5T and 3.0T: preliminary results. Acta Radiol 45:327–332
Willinek WA, Born M, Simon B, Tschampa HJ, Krautmacher C, Gieseke J, Urbach H, Textor HJ, Schild HH (2003) Time-of-flight MR angiography: comparison of 3.0-T imaging and 1.5-T imaging—initial experience. Radiology 229:913–920
Walker MT, Tsai J, Parish T, Tzung B, Shaibani A, Krupinski E, Russell EJ (2005) MR angiographic evaluation of platinum coil packs at 1.5T and 3T: an in vitro assessment of artifact production: technical note. Am J Neuroradiol 26:848–853
Raymond J, Roy D, Bojanowski M, Moumdjian R, L’Esperance G (1997) Endovascular treatment of acutely ruptured and unruptured aneurysms of the basilar bifurcation. J Neurosurg 86:211–219
Costalat V, Lebars E, Sarry L, Defasque A, Barbotte E, Brunel H, Bourbotte G, Bonafe A (2006) In vitro evaluation of 2D-digital subtraction angiography versus 3D-time-of-flight in assessment of intracranial cerebral aneurysm filling after endovascular therapy. Am J Neuroradiol 27:177–184
Kwee TC, Kwee RM (2007) MR angiography in the follow-up of intracranial aneurysms treated with Guglielmi detachable coils: systematic review and meta-analysis. Neuroradiology 49:703–713
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ramgren, B., Siemund, R., Cronqvist, M. et al. Follow-up of intracranial aneurysms treated with detachable coils: comparison of 3D inflow MRA at 3T and 1.5T and contrast-enhanced MRA at 3T with DSA. Neuroradiology 50, 947–954 (2008). https://doi.org/10.1007/s00234-008-0429-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-008-0429-z