Abstract
We tested the hypothesis that nonspecific matrix metalloproteinase (MMP) inhibition with doxycycline would decrease the incidence of intracranial aneurysm formation in a rat aneurysm model. We performed common carotid artery ligation on 96 Long-Evans rats. A treatment group of 48 animals was chosen at random to receive oral doxycycline (3 mg/kg) in addition to standard rat chow, and the control group of 48 animals received standard rat chow only. The major circle of Willis arteries was dissected at 1 year following carotid ligation, and the proportions of animals with aneurysms were compared between groups using Fisher’s exact test. Four animals given oral doxycycline and ten control animals expired before 1 year. Of the examined animals, eight saccular intracranial aneurysms were found in 8 of 45 animals which had received doxycycline (17.8%) and seven saccular intracranial aneurysms were found in 7 of 37 control animals (18.9%). There was no significant difference in aneurysm formation between the doxycycline-treated and control groups (P=0.894). Nonspecific MMP inhibition with doxycycline is not effective in preventing intracranial aneurysm formation in a rat model.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Matrix metalloproteinases (MMPs) comprise a family of at least 23 enzymes that, through their collective ability to degrade most classes of extracellular matrix proteins, are instrumental in extracellular matrix remodeling. Their activity and regulation have been postulated to be a fundamental part of the biochemistry underlying many vascular pathological processes. Specifically, the proteolytic activity of certain MMPs has been associated with abdominal aortic aneurysm (AAA) formation and rupture in animals and humans [1, 8, 11, 17–19, 21, 23, 29]. Furthermore, some investigators have proposed an association between locally increased MMP-2 and MMP-9 activity and intracranial aneurysm formation in humans, despite some presumed differences in the etiologies of intracranial and abdominal aortic aneurysm formation, perhaps because of a shared involvement of extracellular matrix remodeling [6, 13, 14, 16, 22, 27].
Because of the apparent role of MMPs in AAA formation and rupture, pharmacological MMP inhibition has been proposed as a therapy for the inhibition of AAA growth [26]. Tetracyclines and other pharmaceuticals have been shown to inhibit MMP activity and aneurysm growth in animal models [3–5, 9, 15, 20, 24, 28], and experimental AAAs have been suppressed through the targeted gene disruption of MMP-9 in mice [25]. It has been shown that tetracyclines are taken up into the walls of human AAAs where they inhibit MMP activity [10, 12]. A human trial using doxycycline as a nonspecific inhibitor of MMPs for the therapy of small AAAs is currently underway [2].
We hypothesized that if MMPs are involved in the etiology of intracranial aneurysms, their pharmacological inhibition may decrease the incidence of intracranial aneurysm. Using a rat model of intracranial aneurysms, we compared the incidence of aneurysm formation between control animals and animals treated with doxycycline, a systemic inhibitor of MMPs.
Methods
Aneurysm model
Our institution’s Animal Care and Use Committee approved our protocol. Using aseptic technique, we performed unilateral (right) common carotid artery (CCA) ligation on 96 female 150 g Long-Evans rats, a strain susceptible to aneurysm formation with this technique [7]. Intraperitoneal ketamine (70 mg/kg; Phoenix Scientific, St. Joseph, Mo.) and xylazine (8 mg/kg; Phoenix Scientific) anesthesia was administered for the procedure according to weight. A longitudinal incision was made in the right lower neck and blunt dissection isolated the right CCA. The artery was divided by silk sutures at its mid-cervical portion. Absorbable suture was used for skin closure. Postoperative care included subcutaneous buprenorphine analgesia (0.4 mg/kg; Reckitt Benckiser Pharmaceuticals, Hull, UK).
As a secondary specific aim, we tested the hypothesis that bilateral CCA ligation would be more successful than unilateral CCA ligation in inducing intracranial aneurysm formation by performing bilateral CCA ligations in a subset of animals in two stages, 4 weeks apart.
Experimental groups
Animals were randomly assigned to one of four groups. Groups 1 and 2, considered the test animals, underwent unilateral and staged bilateral CCA ligation followed by long-term administration of doxycycline. Group 1, consisting of 38 animals, received doxycycline 3 mg/kg of standard rat chow (Dox Diet, Bio-Serv, Frenchtown, N.J.), mixed at the supply company. We had previously demonstrated serum levels of doxycycline (approximately 1.5–2.5 μg/ml) in the therapeutic range in rats receiving this dose (unpublished data). Group 2, a second treatment group of ten animals, also received doxycycline 3 mg/kg of standard rat chow, but additionally underwent surgical ligation of the contralateral (left) CCA at 4 weeks following right CCA ligation.
Groups 3 and 4, considered the control animals, underwent unilateral and staged bilateral CCA ligation without subsequent doxycycline administration. Group 3, consisting of 38 animals, received standard rat chow without doxycycline. Group 4, a second control group of ten animals, also received standard rat chow without doxycycline, but additionally underwent surgical ligation of the contralateral (left) CCA at 4 weeks following right CCA ligation.
Follow-up
Animals were housed for 1 year following carotid ligation and continued their respective diets. At the time they were killed, all surviving animals were given ketamine and xylazine anesthesia by weight, and perfusion-fixation was performed. Phosphate-buffered saline (PBS) was mechanically infused through a butterfly needle into the left ventricle for 10 min, followed by a 10-min infusion of 4% paraformaldehyde in PBS with 5% sucrose. A diluted solution of methylene blue in PBS was then infused to stain the intracranial vasculature. The animals’ brains were removed for analysis, with the intracranial vasculature intact.
Gross aneurysm examination
The ventral surfaces of the animals’ brains were examined under a dissecting microscope. A reader blinded to treatment group recorded the presence or absence of intracranial aneurysms in all animals.
Statistical analysis
The proportion of aneurysms found in the treatment group was compared to the proportion of aneurysms found in the control group using Pearson’s chi-squared test without Yates’ continuity correction. The proportion of aneurysms found in those animals which had undergone unilateral CCA ligation was compared to the proportion of aneurysms found in those which had received bilateral CCA ligation using Fisher’s exact test. Mortality among control animals was compared with mortality among those treated with doxycycline using Pearson’s chi-squared test without Yates’ continuity correction. Power calculations were made using the binomial distribution. All statistical analyses were performed using S-Plus software (Insightful Corporation, Seattle, Wash.).
Results
A total of 14 animals died before 1 year: two from group 1, two from group 2, eight from group 3, and two from group 4. However, we were able to examine grossly one doxycycline-treated animal which died before 1 year; an intracranial aneurysm was found in this animal. We were not able to examine the remainder of the animals which expired before 1 year. Therefore, 45 of 48 total doxycycline-treated animals were able to be examined. One of the control animals, though surviving 1 year after carotid ligation, was rendered unexaminable by its cage mates. Therefore, we examined 37 of 48 total control animals. The majority of deaths were associated with the spread of a respiratory illness in the vivarium at approximately 10 months into the protocol, and a few deaths were of unknown cause. The difference in mortality between control animals and those treated with doxycycline did not reach statistical significance (χ2=3.033, P=0.082).
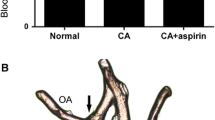
All animals were asymptomatic when they were killed except one, which was lethargic and stumbling. This particular animal was found to have acute subarachnoid hemorrhage and a large intracranial aneurysm at dissection. All animals which had received unilateral (right) CCA ligation had dolichoectasia of the first segment of the right posterior cerebral artery (P1) and of the right posterior communicating artery (Fig. 1). All animals which had received bilateral CCA ligation had dolichoectasia of the basilar, bilateral P1, and bilateral posterior communicating arteries (Fig. 2).
All aneurysms except three were quite large, and most or all of these large aneurysms would be considered giant. These large aneurysms generally did not have large patent lumens and only partially filled with methylene blue dye following fixation-perfusion (Fig. 3). Of the 12 large aneurysms, 11 were of the posterior circulation, although their origins were usually difficult to ascertain precisely. The one large anterior circulation aneurysm appeared to arise from the first segment of the right anterior cerebral artery (A1).
A small aneurysm in one animal originated from the middle portion of P1, and the contralateral P1 and posterior communicating arteries were very small in caliber. Small aneurysms were seen arising from the proximal end of a left A1 fenestration in two animals. The three small aneurysms had completely patent lumens and filled completely with methylene blue dye (Fig. 4).
Of the 45 animals from groups 1 and 2 (doxycycline-treated), 8 (17.8%) had developed an aneurysm by 1 year following carotid ligation. Of the 37 animals from groups 3 and 4 (control), 7 (18.9%) had developed an aneurysm. The proportion of aneurysm formation from all four groups combined was 15 aneurysms per 82 animals for the 1-year interval or 18.3%. There was no statistically significant difference in the proportion of aneurysms between the doxycycline-treated and control groups (χ2=0.0177, P=0.894). Power to detect a 20% relative difference in aneurysm formation between the two groups was calculated at 77%.
Among those animals which had received unilateral CCA ligation, 7 (18.9%) of the 37 surviving doxycycline-treated (group 1) animals examined had developed one saccular intracranial aneurysm by 1 year following carotid ligation, and 5 (17.2%) of the 29 control (group 3) animals examined had developed one saccular intracranial aneurysm. Among those which had received bilateral CCA ligation, 1 (12.5%) of the 8 doxycycline-treated (group 2) animals examined had developed one saccular intracranial aneurysm and 2 (25.0%) of the 8 control (group 4) animals examined had developed one saccular intracranial aneurysm. The proportion of aneurysms was not significantly different between those which had received unilateral CCA ligation and those which had received bilateral CCA ligation (P=0.631).
Discussion
In this study we tested the hypothesis that systemic administration of an inhibitor of MMPs would protect against aneurysm formation in rats. We did not observe a protective effect of doxycycline, a nonspecific MMP inhibitor, against the formation of intracranial aneurysms in a rat carotid ligation model. The incidence of aneurysm formation was similar in both doxycycline-treated and control animals. This result suggests that intracranial aneurysm formation may not be directly dependent upon MMP-mediated weakening of the vessel wall. This may represent a general failure of pharmaceutical MMP inhibition to inhibit intracranial aneurysm formation; alternatively, our results may only apply to the aneurysm model and MMP inhibitor chosen.
We did observe predictable dolichoectatic remodeling of intracranial arteries with common carotid ligation, presumably in response to increased blood flow from the posterior circulation to the anterior circulation. Such remodeling was seen in both doxycycline-treated and control animals.
The incidence of intracranial saccular aneurysm formation in our Long-Evans rat model was approximately 18%, which is considerably less than what has previously been published with a similar model [7]. However, we performed gross analysis of the circle of Willis arteries under a dissecting microscope only and did not evaluate for microaneurysmal formations. We believe that an 18% incidence of aneurysm formation is a more reasonable expectation of this model, if one counts only true saccular aneurysms and not dolichoectasia or microaneurysms. We also did not observe a greater incidence of intracranial aneurysm formation in rats that had received bilateral CCA ligation compared to those that had received unilateral CCA ligation. We conclude that bilateral CCA ligation does not improve the efficiency of intracranial aneurysm induction over unilateral CCA ligation.
Pharmaceutical MMP inhibition has been shown to suppress AAA growth in animal models [4, 9, 20] and is now being evaluated in human clinical trials for AAA [2]. It remains possible that there are important differences in the role of MMPs in the formation and growth of AAAs as compared to that of intracranial aneurysms. Likewise, it is possible that there are important distinctions between MMP activity during aneurysm initiation and during various stages of growth and rupture; our model involved aneurysm initiation as well as growth.
It is also possible that doxycycline may have an effect size on aneurysm growth that was too small to detect with the power we had in our experiment. Larger studies or studies using aneurysm models with a higher incidence of aneurysm formation might be able to detect such a potentially small effect size. We do not know if arterial wall doxycycline concentrations in our model were sufficient for the degree of MMP inhibition that might be needed for aneurysm prevention, despite our achieving what are considered “therapeutic” serum levels. Finally, we do not know if another MMP inhibitor than doxycycline would be more successful.
We noted 14 premature deaths out of 96 animals; most deaths were secondary to respiratory illness. However, a greater number of premature deaths (ten) were seen in the control group than in the treatment group receiving doxycycline (four). Since we failed to determine whether aneurysms were present in 13 of the 14 animals that died prematurely, we cannot exclude the possibility that aneurysms were, in fact, present. If there were aneurysms in these animals, and disproportionately in those not receiving doxycycline, the results may have been provocative.
As many treated or untreated human intracranial aneurysms are followed with imaging to ensure stability or to evaluate for growth, a safe and tolerable pharmaceutical product that could decrease growth and the chance of rupture would be highly desirable for these patients. As we learn more about the pathogenesis of intracranial aneurysm formation, growth and rupture, it may be possible to find such a pharmaceutical.
References
Annabi B, Shedid D, Ghosn P, Kenigsberg RL, Desrosiers RR, Bojanowski MW, Beaulieu E, Nassif E, Moumdjian R, Beliveau R (2002) Differential regulation of matrix metalloproteinase activities in abdominal aortic aneurysms. J Vasc Surg 35:539–546
Baxter BR, Pearce WH, Waltke EA, Littooy FN, Hallett JW, Kent KC, Upchurch GR, Chaikof EL, Mills JL, Fleckten B, Longo GM, Lee JK, Thompson RW (2002) Prolonged administration of doxycycline in patients with small asymptomatic abdominal aortic aneurysms: report of a prospective (phase II) multicenter study. J Vasc Surg 36:1–12
Bendeck MP, Conte M, Zhang M, Nili N, Strauss BH, Farwell SM (2002) Doxycycline modulates smooth muscle cell growth, migration, and matrix remodeling after arterial injury. Am J Pathol 160:1089–1095
Bigatel DA, Elmore JR, Carey DJ, Cizmeci-Smith G, Franklin DP, Youkey JR (1999) The matrix metalloproteinase inhibitor BB-94 limits expansion of experimental abdominal aortic aneurysms. J Vasc Surg 29:130–138
Boyle JR, McDermott E, Crowther M, Wills AD, Bell PR, Thompson MM (1998) Doxycycline inhibits elastin degradation and reduces metalloproteinase activity in a model of aneurysmal disease. J Vasc Surg 27:354–361
Bruno G, Todor R, Lewis I, Chyatte D (1998) Vascular extracellular matrix remodeling in cerebral aneurysms. J Neurosurg 89:431–440
Coutard M, Osborn-Pellegrin M (1997) Genetic susceptibility to experimental cerebral aneurysm formation in the rat. Stroke 28:1035–1042
Crowther M, Goodall S, Jones JL, Bell PRF, Thompson MM (2000) Localization of matrix metalloproteinase 2 within the aneurysmal and normal aortic wall. Br J Surg 87:1391–1400
Curci JA, Petrinec D, Liao S, Golub LM, Thompson RW (1998) Pharmacologic suppression of experimental abdominal aortic aneurysms: a comparison of doxycycline and four chemically modified tetracyclines. J Vasc Surg 28:1082–1093
Curci JA, Mao D, Bohner DG, Allen BT, Rubin BG, Reilly JM, Sicard GA, Thompson RW (2000) Preoperative treatment with doxycycline reduces aortic wall expression and activation of matrix metalloproteinases in patients with abdominal aortic aneurysms. J Vasc Surg 31:325–342
Davis V, Persidskaia R, Baca-Regen L, Itoh Y, Nagase H, Persidsky Y, Ghorpade A, Baxter BT (1998) Matrix metalloproteinase-2 production and its binding to the matrix are increased in abdominal aortic aneurysms. Arterioscler Thromb Vasc Biol 18:1625–1633
Franklin IJ, Harley SL, Greenhalgh RM, Powell JT (1999) Uptake of tetracycline by aortic aneurysm wall and its effect on inflammation and proteolysis. Br J Surg 86:771–775
Gaetani P, Tartara F, Grazioli V, Tancioni F, Infuso L, Rodriguez y Baena R (1998) Collagen cross-linkage, elastolytic and collagenolytic activities in cerebral aneurysms: a preliminary investigation. Life Sci 63:285–292
Gaetani P, Rodriguez y Baena R, Tartara F, Messina AL, Tancioni F, Schiavo R, Grazioli V (1999) Metalloproteases and intracranial vascular lesions. Neurol Res 21:385–390
Kaito K, Urayama H, Watanabe G (2003) Doxycycline treatment in a model of early abdominal aortic aneurysm. Surg Today 33:426–433
Kim SC, Singh M, Huang J, Prestigiacomo CJ, Winfree CJ, Solomon RA, Connolly ES (1997) Matrix metalloproteinase-9 in cerebral aneurysms. Neurosurgery 41:642–646
Knox JB, Sukhova GK, Whittemore AD, Libby P (1997) Evidence for altered balance between matrix metalloproteinases and their inhibitors in human aortic diseases. Circulation 95:205–212
Longo GM, Xiong W, Greiner TC, Zhao Y, Fiotti N, Baxter BT (2002) Matrix metalloproteinases 2 and 9 work in concert to produce aortic aneurysms. J Clin Invest 110:625–632
McMillan WD, Patterson BK, Keen RR, Pearce WH (1995) In situ localization and quantification of seventy-two-kilodalton type IV collagenase in aneurysmal, occlusive, and normal aorta. J Vasc Surg 22:295–305
Moore G, Liao S, Curci JA, Starcher BC, Martin RL, Hendricks RT, Chen JJ, Thompson RW (1999) Suppression of experimental abdominal aortic aneurysms by systemic treatment with a hydroxamate-based matrix metalloproteinase inhibitor (RS 132908). J Vasc Surg 29:522–532
Newman KM, Jean-Claude J, Li H, Scholes JV, Ogata Y, Nagase H, Tilson MD (1994) Cellular localization of matrix metalloproteinases in the abdominal aortic aneurysm wall. J Vasc Surg 20:814–820
Peters DG, Kassam A, St Jean PL, Yonas H, Ferrell RE (1999) Functional polymorphism in the matrix metalloproteinase-9 promoter as a potential risk factor for intracranial aneurysm. Stroke 30:2612–2616
Petersen E, Wagberg F, Angquist KA (2002) Proteolysis of the abdominal aortic aneurysm wall and the association with rupture. Eur J Vasc Endovasc Surg 23:153–157
Petrinec D, Liao S, Holmes DR, Reilly JM, Parks WC, Thompson RW (1996) Doxycycline inhibition of aneurysmal degeneration in an elastase-induced rat model of abdominal aortic aneurysm: preservation of aortic elastin associated with suppressed production of 92 kD gelatinase. J Vasc Surg 23:336–346
Pyo R, Lee JK, Shipley JM, Curci JA, Mao D, Ziporin SJ, Ennis TL, Shapiro SD, Senior RM, Thompson RW (2000) Targeted gene disruption of matrix metalloproteinase-9 (gelatinase B) suppresses development of experimental abdominal aortic aneurysms. J Clin Invest 105:1641–1649
Thompson RW, Baxter BT (1999) MMP inhibition in abdominal aortic aneurysms—rationale for a prospective randomized clinical trial. Ann N Y Acad Sci 878:159–178
Todor DR, Lewis I, Bruno G, Chyatte D (1998) Identification of a serum gelatinase associated with the occurrence of cerebral aneurysms as pro-matrix metalloproteinase-2. Stroke 29:1580–1583
Treharne GD, Boyle JR, Goodall S, Loftus IM, Bell PR, Thompson MM (1999) Marimastat inhibits elastin degradation and matrix metalloproteinase 2 activity in a model of aneurysm disease. Br J Surg 86:1053–1058
Yamashita A, Noma T, Nakazawa A, Saito S, Fujioka K, Zempo N, Esato K (2001) Enhanced expression of matrix metalloproteinase-9 in abdominal aortic aneurysms. World J Surg 25:259–265
Acknowledgements
A Radiological Society of North America Research Resident Grant supported this work. This work was performed in the Department of Radiology, University of Virginia, Charlottesville, VA, USA. We sincerely thank Kevin S. Lee, Ph.D., University of Virginia, for invaluable advice and assistance in perfusion-fixation.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors have no personal or institutional financial interest in drugs, materials or devices described in this submission
Rights and permissions
About this article
Cite this article
Kaufmann, T.J., Marx, W.F. & Kallmes, D.F. A failure of matrix metalloproteinase inhibition in the prevention of rat intracranial aneurysm formation. Neuroradiology 48, 190–195 (2006). https://doi.org/10.1007/s00234-005-0025-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-005-0025-4