Abstract
We report 14 consecutive children with 23 posterior cranial fossa arteriovenous fistula (AVF); six had multifocal lesions, involving the supratentorial brain in three and the spinal cord in one. There were two boys and four girls with a family history compatible with hereditary haemorrhagic telangiectasia. The diagnosis was made in infancy in eight cases and in a further six before the age of 12 years; mean age at diagnosis was 3.5 years. The male-to-female ratio was 1.8:1. Presenting features were macrocrania in four cases, haemorrhage or headache in three and nonhaemorrhagic neurological deficits or and cardiac overload in two. Dominant supply to the symptomatic fistula arose from the posterior inferior cerebellar artery in five cases, anterior inferior cerebellar artery in two and the upper basilar artery system in seven. All children were primarily treated by transarterial embolisation. We treated thirteen children (93%) by transarterial embolisation alone; one older child with a history of haemorrhage also underwent radiosurgery. We obtained 100% exclusion of the fistula(e) in six children, 95-80% in five, 80-50% in one and <50% in one. Of the incompletely treated cases, three had conservative management, and two with 80% and one with 60% reduction of their lesion are scheduled for elective treatment; two partially treated case died. There was no morbidity due to the endovascular procedures. Follow-up since referral is 6 months–10 years (mean 4.5 years). Ten children are neurologically normal, two have persistent (pre-existing) neurological deficits and two are dead.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Arteriovenous fistulas (AVF) are characterised by between one and three dilated feeding arteries opening into a dilated vein, often associated with a large venous pouch. Intradural AVF are always on the surface of the supra- or infratentorial cortex. Posterior cranial fossa AVF are rare; there are few reports in the literature [1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15].
Management of intracranial arteriovenous malformations (AVMs) in children has undergone significant changes in the last 20 years. It has evolved from neurosurgical to interventional neuroradiological management [16]. The children are assessed by a paediatric team consisting of a neurointerventionist, neurologist and neurosurgeon. The natural history of these lesions is not well known. Posterior cranial fossa AVF are rare in children (as in adults) and represent 15.5% of our cases. They do, however, represent a quarter of intradural AVF diagnosed in children which make them a distinct type. Many are seen at early age, before major clinical complications have occurred. It is therefore necessary to review the presentation, treatment strategy and outcome. We review our series of 14 consecutive children managed over 18 years.
Material and methods
Since 1989, our clinical information is prospectively entered in a dedicated data-bank. All children reported here had angiographically confirmed AVF; other intradural AVMs were excluded. The data related to these patients were reviewed and the cases grouped according to age at onset of symptoms, of first consultation and when first treated. There were three age groups: neonates (birth–1 month), infants (1 month–2 years), and children (2–15 years) (Table 1).
The clinical status of each child was assessed at admission and during follow-up by a paediatric neurologist. The examination included Brunet Leizine and Denver neurocognitive testing and incorporated a modified Glasgow outcome score [16]. All patients underwent CT and/or MRI/MRA and had digital angiography before treatment.
All children reported were treated. Surgery was not considered the treatment of choice in any of these children. There was, however, no contraindication to active intervention and embolisation was chosen because of its high risk/benefit ratio. Transarterial superselective embolisation was performed via femoral puncture under general anaesthesia, with N-butyl cyanoacrylate (NBCA) in 10 children (72%), platinum coils in two (14%) and both in another two (14%). The femoral approach was successful in all cases. The 14 patients had a median age of 3.5 years (range 5 months–12 years) when first treated and 23 AVF were embolised. Thirteen children (93%) were treated by embolisation alone, one older child (7%) by a combination of embolisation and radiosurgery. Eleven children (79%) were treated with glue alone, 2 cases (14%) by a combination of coils and glue, and 1 (7%) by coils alone.
The children were kept ventilated in the intensive care unit for 24 h after closure of the shunt, with normal blood pressure, to facilitate the establishment of new cerebral haemodynamics, in accordance with a protocol established previously (Table 2).
All children were followed clinically, using the same scoring system as before, and with MRI/MRA, CT and angiography. The last of these is routinely performed a year after complete anatomic disconnection of the lesions, to confirm the stability of treatment and to assess postnatal venous maturation and remodelling.
Results
There were no patients diagnosed antenatally or as neonates. Symptoms started in eight infants (57%) and six children (43%). There were two boys and four girls with a family history compatible with hereditary haemorrhagic telangiectasia (HHT). The male-to-female ratio was 1.8:1 (nine boys, five girls), and 7:1 (seven boys, one girl) excluding children with HHT.
Presentation and clinical features are detailed in Table 3, reflecting the predominance of infants. Haemorrhage was diagnosed on CT, some patients also having MRI. In the three affected patients the haemorrhage was subarachnoid, intracerebral and intracerebral and intraventricular.
Angiographic data are summarised in Tables 4 and 5. More than one separate lesion was found in six children (43%), three of whom we suspected of having HHT: three had both posterior cranial fossa and supratentorial AVF, two had two lesions in the posterior cranial fossa and one had AVF in both the posterior cranial fossa and spinal cord. We found no correlation between site, numbers of feeding vessels or fistulae and the age of onset of symptoms or signs. The most remarkable angioarchitectural features involved the venous side of the lesions (Table 5).
Treatment was considered complete following at most three embolisation sessions in 11 children (78%) (Table 6) which included those with multifocal lesions. The average number of sessions was 2.3 per child, each lasting between 30–90 min. Morphological results are summarised in Table 6. Of the eight incompletely treated cases, three are scheduled for further treatment, their clinical status having already significantly improved, with satisfactory angiographic remodelling and brain maturation. We managed three patients conservatively after targeted embolisation of identifiable risk areas. A 5–10% residual AVF was not felt to justify the risk of complementary treatment although two partially treated children died despite treatment (Figs. 1, 2).
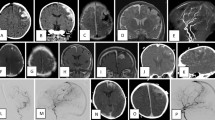
A 20-month-old boy presented with macrocrania and moderate asymptomatic cardiomegaly since the age of 12 months. a, b MRI and c–e angiography demonstrated two arteriovenous fistulae (AVF) in the posterior cranial fossa, fed from the basilar artery and draining into the tectal vein, the vein of Galen and the straight sinus. We carried out four embolisation sessions, occluding six feeding vessels with pure N-butyl cyanoacrylate (NBCA) f, and flow through the fistulae fell to 30–50% g, h over 2 years. The child was doing very well and was scheduled for further treatment after 6 months; no other treatment was felt possible at that time due to the eloquence of the brain involved. Just before his fourth birthday, he died due from haemorrhage into the brain stem while diving into a swimming pool
A 23-month-old girl presented with congestive heart failure, mental retardation and a progressive left hemiparesis. a, b MRI and c, d angiography demonstrated AVF in the right parietal and temporal cortex and brain stem, the last of these fed from the tip of the basilar artery. The child had no significant family history, but was suspected of having hereditary haemorrhagic telangiectasia 1 on the basis of the multifocality and fistulous architecture. We carried out three embolisation sessions, occluding four feeding vessels with NBCA, and flow through the fistulae reduced to 50% e–j over 1 year. Another session was offered a year later, but the family refused further treatment. The child had an ischaemic stroke related to a pulmonary AVF, and died 14 months after the last embolisation
Of the six children with multiple lesions, three had all lesions completely treated successfully. One, who presented at 6 months. with cardiac overload requiring embolisation of supratentorial and posterior cranial fossa AVF is still under going treatment; another who presented at 13 months with a posterior cranial fossa haemorrhage (Fig. 3) leading to embolisation of two AVF, rebled 1 month later from a spinal cord AVF, subsequently 100% excluded by the endovascular approach [17]; the last patient is shown in Fig.1.
A 6-year-old boy presented with headache and macrocrania for 1 year. a, b MRI demonstrated hydrocephalus related to a huge ectatic vein in the posterior cranial fossa. c, d Angiography demonstrated a right cerebellar AVF, fed by the right posterior inferior cerebellar artery and draining into the vein of Galen. The straight sinus was occluded and there was major supratentorial venous congestion. e–g Curative embolisation of the fistula was performed in one session with pure NBCA. Immediate angiography showed partial filling of the fistula, but the cast of glue was expected to provoke rapid secondary thrombosis of the lesion. At 6 weeks h, i and j–n angiography showed complete cure and satisfactory vascular remodelling. At 1 year the boy is neurologically normal and his macrocrania is stable
There was one non-neurological complication during the 31 embolisation sessions. Following deposition of pure glue, a small droplet remained attached to the catheter, precluding its complete removal. The catheter tip was then surgically removed by aortotomy, with full clinical recovery.
Outcome according to our clinical grading system is shown in Table 7. Ten children (71%) are neurologically normal (grades 5, 4 and 3) (Fig. 4). Follow-up ranges from 6 months to 10 years (mean 4.5 years).
A 3-year-old girl presented with severe headache, a right 3rd nerve palsy, left facial weakness and weakness of the left leg. a, b MRI showed an upper brain-stem haemorrhage and acute hydrocephalus. An emergency ventriculoperitoneal shunt was performed. c, d 3 weeks later angiography demonstrated a right mesencephalic AVF, fed from the right superior cerebellar artery and draining into the vein of Galen. We thought the feeding vessel too short for safe injection of glue with no risk of reflux into the parent artery. e, f During a single session, Guglielmi detachable coils and fibred coils were placed in the fistula, reducing flow though it by 90%. g–i Follow up MRI showed complete closure of the lesion, with resolution of the neurological signs. Follow-up showed complete clinical recovery
Discussion
In our experience the incidence of multifocal arteriovenous lesions in children is twice that of adults: 17.2 vs 9% [16]. The characteristics of each shunt vary, but in the same individual the same type of architecture, i.e., multiple fistulae or multiple AVMs with a nidus, is often found. The lesions are usually bilateral as well as supra- and infratentorially. Some are grouped together in a limited space. Most arteries feeding a pial AVF open into a single ectatic draining vein. Depending on the age at which the AVF is discovered, the tolerance of young children to these lesions can be striking when one considers their size and flow.
Since none of these lesions was diagnosed before or soon after birth, cardiac failure was not a prominent presentation. The infants presented with hydrodynamic disorders, and older children with headaches and occasionally haemorrhage. There were few cases of macrocrania and none of hydrocephalus, despite some giant venous pouches in the posterior cranial fossa. Some high-flow lesions in infants did lead to macrocrania but none developed a melting brain syndrome unless jugular vein occlusion occurred. As the indication for early treatment was to allow satisfactory maturation of the venous system [16], we did not observe secondary jugular vein occlusion. No significant cerebellar atrophy was seen because of the rapid subarachnoid drainage of the shunt. Mechanical effects on the brain stem were symptomatic in only one case. The clinical findings reflect the higher mean age at diagnosis (3.5 years), than with supratentorial AVF (23 months) or vein of Galen aneurysmal malformations, 60% of which are diagnosed before or shortly after birth. Male dominance is similar to that reported in dural sinus malformations, arterial aneurysms or vein of Galen aneurysmal malformations. It was not apparent in the subgroup with HHT.
Haemorrhage was uncommon, probably because of the relative thickness of the wall of the venous varix in response to increased flow, turbulence and pressure [15]. Intra-axial bleeding, the most common manifestation of childhood AVMs, occurred in only three children. However the real incidence could be higher, as death might result from rupture of an unrecognised posterior cranial fossa AVF.
Complete angioarchitectural analysis is crucial to decision-making for the therapeutic strategy and end-points. The natural history of these lesions depends on the age at which they become active and interfere with postnatal brain development and posterior cranial fossa vasculature. Neurological deficits are caused by venous ischaemia. Mass effect and steal, although classically suggested to explain neurological deficits, are not demonstrated on MRI or angiography. In symptomatic intranidal partial venous thrombosis, the high signal on T1- and T2-weighted images indicates slow flow rather than haemorrhage. Mass effect on the brain by venous pouches can result from partial thrombosis, with an extravascular response. On CT one must be careful not to misinterpret intravascular blood (in a venous pouch) as haemorrhage; contrast-enhanced sections confirm the intraluminal nature of flowing blood.
The frequency of HHT is 1 in 5–10000 [18]. The typical features are unusual before puberty; telangiectases and epistaxis are not expected in young children. The presence of central nervous system or visceral AVMs in patients with HHT is well documented. It is suggested suggest that 2% of brain AVMs are associated with HHT [19] and that 5–10% of patients with HHT will have a brain AVMs [20, 21]; the numbers vary depending on whether presentation is with head and neck, pulmonary or alimentary lesions. We have not seen a new cerebral AVM develop on follow-up of any patient with HHT; the brain AVMs in adults resemble the usual congenital types and can be distinguished from telangiectasia. Willinsky et al. reported that 28% of patients with multiple cerebral AVMs had HHT and that the presence of multiple cerebral AVMs should lead one to suspect HHT1 [19].
García-Mónaco et al. [6] reported four cases of pial AVF in children as the presentation of HHT; three were supratentorial (and had multiple lesions) and one was in the posterior cranial fossa. Matsubara et al. [22] reported that multiple small cortical AVM or AVF are highly suggestive of HHT. Mazighi et al. [17] reported a rare case of HHT1 with brain and spinal AVMs in an infant.
The fistulous nature of the lesions and their multifocality are typical of HHT in children, suggesting a phenotypic difference between the AVMs in adults and AVF in children. One can postulate a different effect of the same genotype depending on the degree of vascular maturation rather than different genotypes as the family history of all these children is similar. The lungs must be screened for pulmonary AVF, which in one of our cases led to a lethal embolic stroke.
Batista et al. [3] reported a child with cephalocraniocutaneous lipomatosis with posterior cranial fossa AVF. The child had two supra- and infratentorial high-flow fistulae, successfully treated, and nonvascular intracranial malformations: lipoma, arachnoid cysts and cortical dysplasia.
All the patients referred to us had been assessed by other specialists. It is therefore predictable that they were all considered poor surgical candidates. Treatment of AVF in young children aims to preserve normal neurocognitive maturation and function as well as to provide stable protection from haemorrhage or progressive neurological deficits. Treatment of an asymptomatic pial AVF is controversial [11] since the natural history has not been established. Treatment decisions mostly depend upon the morbidity and mortality in similar patient groups of a given team. Most of these children were treated on an urgent or emergency basis. Empirical and literature experience suggest that symptomatic or preventive treatment is imperative in children with posterior cranial fossa AVF.
In most instances, a morphological target can be assigned to the clinical objective, but complete eradication in one sitting is often considered hazardous in these babies. Although complete exclusion of the lesion is desirable with the endovascular approach, partial targeted and staged treatment can be performed. Therapeutic decision-making is complex in high-flow lesions in children, in particular with multifocal AVF, which must be discussed case-by-case. Since most are constituted by several large arteries opening into a single vein, the sequence and order of feeding vessels to be embolised is crucial for ensure safe haemodynamic and morphological remodelling. The smallest lesion will often be treated first, followed by the largest, to protect against the recirculation effects on a smaller AVF following sudden occlusion of a high-flow shunt. No postembolisation “breakthrough” was been seen in any of these cases.
Except when the posterior cranial fossa AVF is treated completely in one session (30–40%), partial, progressive occlusion is achieved during several sessions or ends up being partial. Sessions are usually staged at 3–6 month intervals depending on the urgency of restoring normal haemodynamics for the maturing and growing brain. The aim of partial targeted endovascular treatment of cerebral AVF is the orderly elimination of dangerous angioarchitectural features. It combines two judgements: target and timing, a strategy whose efficacy has been shown in reducing the risk of haemorrhagic in adult AVMs. Our results support this strategy with no neurological morbidity, and good clinical outcome in 71% of cases.
Endovascular embolisation of pial AVF is best accomplished by the transarterial approach with pure NBCA mixed with tantalum powder; we have also used detachable balloons or coils. We favour NBCA as the first agent. Once stable, total obliteration was demonstrated on follow-up angiography; no further studies were indicated; and no new AVMs developed. No reopening of the AVF has been seen in any case.
References
Aoki N, Sakai T, Oikawa A (1991) Intracranial arteriovenous fistula manifesting as progressive neurological deterioration in an infant: case report. Neurosurgery 28: 619–623
Barnwell SL, Ciricillo SF, Halbach VV, Edwards MSB, Cogen PH (1990) Intracranial arteriovenous fistulas associated with intraparenchymal varix in childhood: case reports. Neurosurgery 26: 122–125
Batista LL, Mahadevan J, Sachet M, et al (2002) Encephalocraniocutaneous lipomatosis syndrome in a child: association with multiple high flow cerebral arteriovenous fistulae: case report and review. Intervent Neuroradiol 8: 273–283
Carrillo R, Carreria LM, Prada J, Rosas C, Egas G (1984) Giant aneurysm arising from a single arteriovenous fistula in a child. J Neurosurg 60: 1085–1088
Chul Suh D, Alvarez H, Bhattacharya JJ, Godesch G, Lasjaunias P (2001) Intracranial haemorrhage within the first two years of life. Acta Neurochir (Wien) 143: 997–1004
García Mónaco R, Alvarez H, Goulão A, Pruvost P, Lasjaunias P (1990) Posterior fossa arteriovenous malformations: angioarchitecture in relation to their hemorrhagic episodes. Neuroradiology 31: 471–475
García Mónaco R, Taylor W, Rodesch G, et al (1995) Pial arteriovenous fistula in children as presenting manifestation of Rendu-Osler-Weber disease. Neuroradiology 37: 60–64
Hung PC, Wang HS (2002) Successful endovascular treatment of cerebral arteriovenous fistula. Paediatr Neurol 27: 300–302
Iizuka Y, Rodesch G, García-Mónaco R, et al (1992) Multiple cerebral arteriovenous shunts in children: report of 13 cases. Child’s Nerv Syst 8: 437–444
Lasjaunias P, Hui F, García-Mónaco R, et al (1995) Cerebral arteriovenous malformations in children: management of 179 consecutive cases and review of the literature. Child’s Nerv Syst 11: 66–79
Nelson PK, Niimi Y, Lasjaunias P, Berenstein A (1992) Endovascular embolization of congenital arteriovenous fistulas. Neuroimaging Clin North Am 2: 309–331
Rodesch G, Malherbe V, Alvarez H, Zerah M, Devictor D, Lasjaunias P (1995) Nongalenic cerebral arteriovenous malformation in neonates and infants: review of 26 consecutive cases (1982–1992). Child’s Nerv Syst 11: 231–241
Talamonti G, Versari PP, D’Aliberti G, Villa F, Fontana RA, Collice M (1997) Complex arteriovenous fistula of the brain in an infant: case report. J Neurosurg Sci 41: 337–341
Tomlinson FH, Rüfenacht DA, Sundt TM Jr, Nichols DA, Fode NC (1993) Arteriovenous fistula of the brain and spinal cord. J Neurosurg 79: 16–27
Viñuela F, Drake CG, Fox AJ, Pelz DM (1987) Giant intracranial varices secondary to high-flow arteriovenous fistulae. J Neurosurg 66: 198–203
Lasjaunias P, terBrugge K (1997) Vascular diseases in neonates, infants, and children. Springer-Verlag, Berlin, pp 203–219
Mazighi M, Porter P, Alvarez H, Rodesch G, Meisel J (2000) Associated cerebral and spinal AVM in infant and adult—report of two cases treated by endovascular approach. Intervent Neuroradiol 6: 321–326
Guttmacher AE, Marchuk DA, White RIJ (1995) Hereditary hemorrhagic telangiectasia: N Eng J Med 333: 918–924
Willinsky RA, Lasjaunias P, Ter Brugge K, Burrows P (1990) Multiple cerebral arteriovenous malformations (AVMs): review of our experience from 203 patients with cerebral vascular lesions. Neuroradiology 32: 207–210
Ference BA, Shannon TM, White RIJ, Zawin M, Burdge CM (1994) Life-threatening pulmonary hemorrhage with pulmonary arteriovenous malformations and hereditary hemorrhagic telangiectasia. Chest 106: 1387–1390
Wirth JA, Pollak JS, White RIJ (1996) Pulmonary arteriovenous malformations: Curr Pulm Circ Care Med 17: 272–274
Matsubara S, Manzia JL, Ter Brugge K, Willinsky RA, Montanera W, Faughnan ME (2000) Angiographic and clinical characteristics of patients with cerebral arteriovenous malformations associated with hereditary hemorrhagic telangiectasia: AJNR 21: 1016–1020
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshida, Y., Weon, Y.C., Sachet, M. et al. Posterior cranial fossa single-hole arteriovenous fistulae in children: 14 consecutive cases. Neuroradiology 46, 474–481 (2004). https://doi.org/10.1007/s00234-004-1176-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-004-1176-4