Abstract.
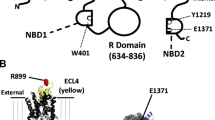
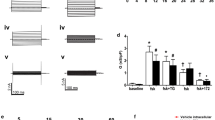
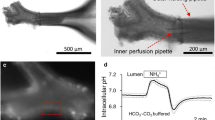
In cystic fibrosis airway epithelia, mutation of the CFTR protein causes a reduced response of Cl− secretion to secretagogues acting via cAMP. Using a Ca2+ imaging system, the hypothesis that CFTR activation may permit ATP release and regulate [Ca2+] i via a receptor-mediated mechanism, is tested in this study. Application of external nucleotides produced a significant increase in [Ca2+] i in normal (16HBE14o− cell line and primary lung culture) and in cystic fibrosis (CFTE29o− cell line) human airway epithelia. The potency order of nucleotides on [Ca2+] i variation was UTP ≫ ATP > UDP > ADP > AMP > adenosine in both cell types. The nucleotide [Ca2+] i response could be mimicked by activation of CFTR with forskolin (20 μm) in a temperature-dependent manner. In 16HBE14o− cells, the forskolin-induced [Ca2+] i response increased with increasing temperature. In CFTE29o− cells, forskolin had no effect on [Ca2+] i at body temperature-forskolin-induced [Ca2+] i response in CF cells could only be observed at low experimental temperature (14°C) or when cells were cultured at 26°C instead of 37°C. Pretreatment with CFTR channel blockers glibenclamide (100 μm) and DPC (100 μm), with hexokinase (0.5 U/mg), and with the purinoceptor antagonist suramin (100 μm), inhibited the forskolin [Ca2+] i response. Together, these results demonstrate that once activated, CFTR regulates [Ca2+] i by mediating nucleotide release and activating cell surface purinoceptors in normal and CF human airway epithelia.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 3 April 2000/Revised: 30 June 2000
Rights and permissions
About this article
Cite this article
Walsh, D., Harvey, B. & Urbach, V. CFTR Regulation of Intracellular Calcium in Normal and Cystic Fibrosis Human Airway Epithelia. J. Membrane Biol. 177, 209–219 (2000). https://doi.org/10.1007/s002320010004
Issue Date:
DOI: https://doi.org/10.1007/s002320010004