Abstract
Diet studies are an essential component of ecosystem-based approaches to fisheries management. In the eastern Pacific Ocean (EPO), the silky shark (Carcharhinus falciformis) is the most common species of shark in the bycatch of the tuna purse-seine fishery. A rare, comprehensive dataset of stomach contents of 786 silky sharks sampled in mostly tropical regions of the EPO (25°N–15°S; 79°W–162°W) during 1992–1994 and 2003–2005 was analyzed via classification tree and quantile regression methodologies to gain insight into its ecosystem role. Results suggest that the silky shark is an opportunistic predator that forages on a variety of prey. Broad-scale spatial and shark size covariates explained the feeding habits of silky sharks captured in sets on floating objects, primarily drifting fish-aggregating devices (FADs). A strong spatial shift in diet was identified by the tree analysis, with different foraging patterns in the eastern (inshore) and western (offshore) regions. Greater proportions of FAD-associated prey than non-FAD-associated prey were observed in the diet throughout the EPO, with the greatest proportion in the offshore region. Thus, silky sharks appear to take advantage of the associative behavior of prey fishes to increase their probability of encountering and capturing prey. Evaluation of prey–predator size relationships showed that maximum prey size increased with increasing silky shark size, but minimum prey size remained relatively constant across the range of shark sizes. Results such as these from spatially oriented analyses of predator feeding habits are essential for populating ecosystem models with space-based food webs, which otherwise suffer from generic representations of food webs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Underlying an increasing worldwide interest in ecologically based approaches to fisheries management (Pikitch et al. 2004; Marasco et al. 2007) is a requirement to thoroughly understand the dynamics of food webs. Large pelagic sharks play an important role as top predators in the ecosystem, with the capacity to influence community structure (Heithaus et al. 2008; Ferretti et al. 2010), and as keystone predators (Hinman 1998; Stevens et al. 2000), essential to the maintenance and stability of food webs (Power et al. 1996). Data on trophic ecology are a prerequisite for understanding the ecological role of top predators and the consequences of changes in their abundance (Cortés 1999; Matich et al. 2011). Food habits studies are necessary for investigating pathways of energy flow in exploited ecosystems (Olson et al. 2014), and knowledge of trophic position and trophic linkages is essential for informing ecosystem models (e.g., Ecopath with Ecosim, Christensen and Pauly 1992). In particular, space-based ecosystem models, e.g., Ecospace (Walters et al. 1999; Pauly et al. 2000) and Atlantis (Fulton et al. 2004), can suffer from generic depictions of food webs, and spatially oriented analyses of diet composition are useful for populating such models.
The silky shark (Carcharhinus falciformis) is the most common shark incidentally caught by tuna purse-seine fisheries (Román-Verdesoto and Orozco-Zӧller 2005; Bonfil 2008) and high-seas tuna longline fisheries (Bonfil 2008) and is targeted by coastal longline and gillnet fisheries (Sánchez-de Ita et al. 2011) in the eastern Pacific Ocean (EPO). It is one of the most common epipelagic sharks in tropical and subtropical waters worldwide, occurring from inshore and island areas to the open ocean (Compagno 1984). Despite the importance of the silky shark as both top predator and component of the bycatch, little is known about its trophic dynamics in the open ocean. Food habits studies have been restricted in space to subtropical and tropical inshore areas off Baja California, Mexico (Galván-Magaña et al. 1989; Cabrera-Chávez-Costa et al. 2010), and the Gulf of Tehuantepec, Mexico (Cabrera 2000; Barranco 2008), where the diet consisted primarily of invertebrates.
Much of the incidental fishing mortality of silky sharks in the tropical EPO occurs when tuna purse-seine vessels set their nets on tunas associated with floating objects, in particular man-made fish-aggregating devices (FADs) (Gerrodette et al. 2012; IATTC 2013: p. 132, Table 3). Numerous pelagic fishes commonly aggregate around floating objects in the tropical and subtropical EPO (Hunter and Mitchell 1966; Arenas et al. 1999), but the basis of the association remains unresolved. FADs are thought to modify the habitat of pelagic fishes (e.g., Marsac et al. 2000; Dagorn et al. 2013), and the deployment of massive numbers of FADs may alter the trophic interactions of associated pelagic fishes (Deudero 2001; Hunsicker et al. 2012). Specifically, pelagic fishes may be more vulnerable to predation by top predators when aggregated at FADs (or other flotsam) than when unassociated.
Identifying ontogenetic patterns in the diet is important because the functional role of a predator can change with growth (Karpouzi and Stergiou 2003; Pinnegar et al. 2003), which can, in turn, affect the species and size composition of a prey community (Heupel et al. 2014). Prey size and predator size are important factors influencing trophic interactions and foraging success in marine fish predators (Scharf et al. 2000). Size-related variability in diet composition is common in the aquatic environment. Larger predators eat progressively larger prey, while minimum prey size often remains relatively constant over a range of predator sizes (Scharf et al. 1998, 2000; Juanes 2003; Ménard et al. 2006; Young et al. 2010). Smaller predators are limited by gape size and thus tend to be restricted to small prey (Magnuson and Heitz 1971). Prey–predator size ratios provide information about the breadth of a predator’s trophic niche as a function of size (Scharf et al. 2000; Juanes 2003; Ménard et al. 2006; Young et al. 2010).
We analyzed the foraging ecology, based on the stomach contents, of silky sharks captured opportunistically as bycatch by the tuna purse-seine fishery in mostly tropical regions of the EPO. Our dataset is novel because stomach samples from open-ocean carcharhinid sharks are rare, and it includes silky sharks caught over a broad region of the tropical EPO. Our objectives were to describe silky-shark-feeding habits in the pelagic tropical EPO, to quantify the incidence of flotsam-related feeding (largely on FADs), and to identify size-specific predation patterns. Our approach was twofold: (1) to explore broad-scale spatial and size relationships in silky shark diet using classification tree methodology and (2) to assess finer-scale prey–predator size relationships in foraging behavior, using quantile regression techniques. This research provides a comprehensive description of silky shark predation in the tropical EPO and will facilitate improvement in future ecosystem models.
Materials and methods
Stomach sampling
Silky shark stomachs were collected at sea by Inter-American Tropical Tuna Commission (IATTC) observers, who recorded the date, time, location, set type, and sea surface temperature (SST) for each set that yielded samples. Observers measured total length (TL) to the nearest millimeter, excised the stomach, and recorded the sex of each silky shark. Stomach samples were frozen and saved for analysis in the laboratory.
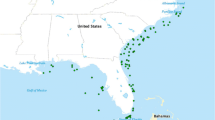
We analyzed the stomach contents from specimens originating from 47 purse-seine sets during 41 fishing trips between December 1992 and July 1994 and from 97 purse-seine sets during 29 fishing trips between August 2003 and February 2005. Three types of purse-seine sets have incidental catch of silky sharks in the EPO: (1) “dolphin sets,” whereby the net is deployed around a school of tuna associated with a dolphin aggregation (Scott et al. 2012), (2) “unassociated sets,” whereby the net is deployed around a free-swimming school of tuna (Hall 1998), and (3) “floating-object sets,” whereby the net is deployed around a school of tuna associated with a floating object (Hall 1998; Dagorn and Fréon 1999). Floating-object sets are usually made in the early morning, while dolphin sets and unassociated sets typically are made throughout the day. Sample locations are displayed by set type, stomach condition, and sampling period (1992–1994 and 2003–2005) in Fig. 1. Numbers of stomach samples by fishing method and stomach condition are presented in Table 1.
Diet composition
Laboratory procedures consisted of thawing the stomachs, identifying the prey to the lowest possible taxon, classifying the prey by digestion state, weighing the prey to the nearest gram, and enumerating the prey when individuals were recognizable. We used the following keys to identify fish prey: Jordan and Evermann (1896), Meek and Hildebrand (1923), Clothier (1950), Parin (1961), Monod (1968), Miller and Lea (1972), Miller and Jorgenson (1973), Thomson et al. (1979), Allen and Robertson (1994), and Fischer et al. (1995b, c). We identified crustacean prey from exoskeleton remains using the keys of Garth and Stephenson (1966), Brusca (1980), and Fischer et al. (1995a). We identified cephalopod prey from mandible remains (Clarke 1962, 1986; Iverson and Pinkas 1971; Wolff 1982). We also relied on our (F. G-M.’s) reference collections, the fish collections at Scripps Institution of Oceanography and the Natural History Museum of Los Angeles County, and the cephalopod collection at the Santa Barbara Museum of Natural History to compare and validate prey identifications. For each prey item, we recorded a numeric digestion state that ranged from 1 for fresh or nearly intact prey to 5 for digestion-resistant hard parts (primarily fish otoliths and cephalopod mandibles, i.e., no undigested soft tissue remaining) (Olson and Galván-Magaña 2002; Bocanegra-Castillo 2007). We measured prey lengths whenever possible, to the nearest millimeter for fishes (fork length), cephalopods (mantle length), and crustaceans (carapace length). Counts of residual hard parts were divided by two to estimate numbers of individual organisms ingested.
Silky sharks are known to feed occasionally while encircled in purse-seine nets. Since it is more difficult for corralled prey to escape a predator inside the net, we considered net feeding as artificial predation and omitted it from the analysis. Food items in the freshest digestion state were considered to have been eaten inside the net during a set if the same species was reported in the catch data of the same set. We therefore omitted data for fresh dolphin (Delphinidae) remains in the stomach contents of silky sharks captured in dolphin sets, fresh tuna remains (Scombridae: Thunnus albacares and Katsuwonus pelamis) found in silky sharks captured in unassociated sets, and fresh remains of several fishes that aggregate at floating objects (Coryphaenidae, Carangidae: Decapterus macarellus, Elagatis bipinnulata; Scombridae: Acanthocybium solandri, Auxis spp., Auxis thazard, Euthynnus lineatus, K. pelamis, T. albacares, Thunnus obesus, Thunnus spp.; Istiophoridae: Makaira spp.; Balistidae: Xanthichthys mento) found in silky sharks from floating-object sets. For data analysis, we combined FAD and flotsam sets under the category floating-object sets, and the majority of these sets were made on FADs. We thus, hereafter, refer to floating objects as FADs.
We used gravimetric, numeric, and occurrence indices of diet importance to analyze the stomach contents data. We calculated proportional compositions by weight and by number of each prey type eaten by each individual silky shark and then averaged the proportions for each prey type over all silky sharks with prey remains in the stomachs (Chipps and Garvey 2007). For prey weights,
where \(\bar{W}_{i}\) is mean proportion by weight for prey type i, W ij is the weight of prey type i in silky shark j, P is the number of silky sharks with food in their stomachs, and Q is the number of prey types in all the samples. For prey counts,
where \(\bar{N}_{i}\) is mean proportion by number for prey type i, N ij is the number of individuals of prey type i in silky shark j, and P and Q are as defined for Eq. 1. For frequency of occurrence (O i ),
where J i is the number of silky sharks containing prey i, and P is as defined for Eqs. 1 and 2. We omitted prey data based on residual hard parts and stomachs that contained only residual hard parts from the \(\bar{W}_{i}\) and \(\bar{N}_{i}\) computations because hard parts can accumulate in the stomachs from feeding over an unknown number of previous days. Relatively few prey taxa in the EPO have prominent hard parts that resist digestion (Olson and Galván-Magaña 2002), and treating hard parts the same as undigested soft tissue would over-represent the dietary importance of taxa with digestion-resistant hard parts, especially based on numeric and gravimetric indices (Olson and Galván-Magaña 2002; Chipps and Garvey 2007; Olson et al. 2014). We included the contents of stomachs that contained residual hard parts in the O i computation because this index represents how frequently a particular prey item was eaten, but does not indicate relative importance to the overall diet.
Classification tree analysis
We applied classification tree methodologies (Breiman et al. 1984) to the silky shark stomach contents data to explore spatial and shark size structure in the diet composition, using the modified approach for diet data outlined in Kuhnert et al. (2012) and applied in Olson et al. (2014). We used only data from floating-object sets because the majority of stomachs sampled were from sets on floating objects (see “Stomach sampling” in “Results” section). Unlike previous approaches for analyzing diet data, this tree methodology provides both an exploratory and predictive framework for identifying complex relationships between predictor variables and diet composition (Kuhnert et al. 2012). The vector of prey proportions eaten by an individual predator is represented as a univariate categorical response variable of prey type (class), with observation (case) weights equal to the proportion of the prey type eaten by the predator (Kuhnert et al. 2012). The data are recursively partitioned, using a greedy algorithm, forming homogenous groups. The split criterion used is the Gini index of diversity (D) (Breiman et al. 1984) that ranges between 0 and 1, where values near 0 indicate low diet diversity and values near 1 represent a highly diverse diet. A large tree is grown and then pruned back using tenfold cross-validation. The “1 standard error” (“1 SE”) rule, that aims to identify a parsimonious tree within 1 SE of the tree yielding the minimum error, is then applied. Variable importance rankings are computed to identify important predictor variables (Breiman et al. 1984; Kuhnert et al. 2012). A spatial bootstrap technique is implemented to obtain diet composition predictions and associated uncertainty, for each node in the tree (Kuhnert et al. 2012). The classification tree approach is implemented in R (R Development Core Team 2013), using the ‘rpart’ package (Therneau et al. 2013); further details can be found in Kuhnert et al. (2012). To explore potential spatial and size influences on the diet of silky sharks, we used latitude, longitude, and silky shark total length as covariates in the best classification tree analysis.
We included 17 principal prey groups in the classification tree analysis, based on their gravimetric (\(\bar{W}_{i}\)) importance in the overall diet. We examined silky shark predation data in terms of gravimetric (\(\bar{W}_{i}\)), rather than numeric (\(\bar{N}_{i}\)) or occurrence (O i ) importance, because prey weights are appropriate for comparing the bioenergetics importance of a variety of prey to a predator (Chipps and Garvey 2007) and pertinent for delineating food web dynamics. The 17 groups consisted of two groups of cephalopods: Dosidicus gigas and Sthenoteuthis oualaniensis; one group of crustaceans: Portunidae, and 14 groups of fishes (Osteichthyes): Exocoetidae, Oxyporhamphus micropterus, Coryphaenidae, Carangidae, Decapterus spp., unidentified scombrids, other identified scombrids, A. solandri, Auxis spp., K. pelamis, T. albacares, Thunnus spp., Cubiceps pauciradiatus, and Tetraodontiformes (see Table 2 for phylogenetic affiliations of the prey groups). These groups ranged in taxonomic level from species to family because the taxonomic resolution of stomach contents identifications varied due to the digestion state of the prey and because some rare prey were combined into broader taxa. We categorized each prey group as (1) animals that associate with FADs, according to at-sea observations during 1993–2005, and (2) animals that do not commonly associate with FADs, to evaluate the importance of FAD-associated prey in the diet of silky sharks (Table 2). We excluded unidentified fishes, cephalopods, and crustacea from the tree analysis. We also omitted prey taxa that did not constitute at least 1 % wet weight of the overall diet for either sampling period. We analyzed the stomach contents from 289 silky sharks, which resulted in 341 predator–prey observations.
Because the stomach samples from multiple silky sharks caught in the same set (Fig. 2) were not statistically independent and a limitation of the classification tree method is its inability to handle dependent observations (Kuhnert et al. 2012), we performed a sensitivity analysis to determine the effect of these dependent samples on the tree structure resulting from the full dataset. In this sensitivity analysis, we built a classification tree for each of 1,000 randomly selected datasets created by subsampling the data for one silky shark from each purse-seine set in the full dataset. The set locations represented in both the full dataset and the subsampled datasets were the same. Each tree based on the subsampled datasets was pruned to match the size of the tree for the full dataset, and the resulting tree partition structure was compared against that of the full dataset.
FAD- and non-FAD-associated prey
Given the variety of animals that commonly associate with FADs and the apparent importance of FADs in the predation behavior of silky sharks, we quantified the incidence of FAD- versus non-FAD-associated prey in the silky shark diet (see Table 2 for prey assigned to these two categories). To determine whether the diet proportions of FAD- and non-FAD-associated prey differed, we implemented a beta regression using the concepts of Figueroa-Zúñiga et al. (2012). Using this approach, we modeled the proportion of FAD-associated prey to determine whether higher proportions of FAD-associated prey were eaten overall when compared with non-FAD-associated prey and whether there were spatial differences observed within regions of the tropical EPO identified by the classification tree analysis. The model can be represented as,
where \(y_{ij}\) represents the proportion of FAD-associated prey eaten by silky shark \(i\) in purse-seine set \(j\), \(x_{ij}\) represents a binary variable that indicates the location of the sample (east: 0 or west: 1; see “Tree partitions: the eastern versus western region” in “Results” section for definitions of the eastern and western boundaries), and \(b_{j}\) represents the random effect for \(j\) to account for multiple silky sharks sampled in some purse-seine sets. The intercept, \(\alpha\), in the model examines whether high proportions of FAD-associated prey are eaten compared to non-FAD-associated prey, while the regression coefficient, \(\beta\), examines the effect of western regions versus eastern regions. Note that \(y_{ij}^{*}\) represents a transformation of \(y_{ij}\), proposed by Smithson and Verkuilen (2006) suitable for proportions that lie at the extremes. The model was implemented in WinBUGS (Lunn et al. 2000) with non-informative priors assigned to the regression coefficients, \(\alpha\) and \(\beta\), and the precision parameter, \(\phi\), for the purse-seine set random effect. As well as considering a precision parameter that is constant across observations, we considered a more general formulation of the model where a separate precision parameter, \(\phi_{ij}\) is estimated for each response, \(y_{ij} .\) In this scenario, we assigned a non-informative gamma distribution to each precision parameter.
Prey–predator size relationships
We used quantile regression techniques (Koenker and Basset 1978; Geraci 2013; Geraci and Bottai 2013) to assess fine-scale prey–silky shark size relationships within regions of the tropical EPO identified by the classification tree analysis, because the classification tree-based approach is not suitable for fine-scale analyses of any single dietary predictor (Kuhnert et al. 2012). We estimated the rates of change in minimum and maximum prey sizes with increasing silky shark size (Scharf et al. 1998, 2000; Juanes 2003; Bethea et al. 2004; Ménard et al. 2006; Chipps and Garvey 2007; Costa 2009; Lucifora et al. 2009; Young et al. 2010). Scatterplots of prey versus predator lengths frequently show polygonal patterns, suggesting that upper and lower size limits of prey use commonly change at different rates over predator ontogeny. Our approach for silky sharks was, therefore, to examine the upper and lower bounds of the scatterplots, rather than changes in mean prey sizes (Scharf et al. 1998; Cade et al. 1999). We implemented quantile regression in R, using the ‘lqmm’ package (Geraci 2013; Geraci and Bottai 2013). We fitted a mixed-effects model to the prey–silky shark data, with purse-seine set as a random effect, and a fixed-effect slope. The selection of which quantiles to use to best represent upper and lower bounds was based on sample size (Scharf et al. 1998). We obtained standard errors and approximate confidence intervals for the intercept and slope with a bootstrap procedure (Geraci 2013; Geraci and Bottai 2013). Among regions of the tropical EPO, we compared slope estimates for the prey–silky shark length relationship of the same quantile using a t test, assuming unequal variance.
We examined ontogenetic changes in the lower and upper bounds of trophic-niche breadth on a ratio scale (prey length/silky shark length) across the range of silky shark sizes, using quantile regression (Scharf et al. 2000; Juanes 2003; Bethea et al. 2004; Ménard et al. 2006; Young et al. 2010). Within regions of the tropical EPO, we compared slope estimates for the trophic-niche breadth of quantile pairs (e.g., 0.10 and 0.90) by constructing bootstrap confidence intervals of the differences in slopes (Geraci 2013). We compared slope estimates for the upper quantile, among regions, using the same methods as those used for the prey–silky shark length analysis. Significant differences correspond to ontogenetic increases (diverging slopes) or decreases (converging slopes) in trophic-niche breadth in relation to silky shark size (Scharf et al. 2000; Juanes 2003; Bethea et al. 2004).
Results
Stomach sampling
Numbers of silky sharks collected for stomach contents analysis by sampling period, set type, and stomach condition, i.e., whether or not the stomach contained food remains, residual hard parts, or only food consumed in the purse-seine net, are presented in Table 1. Overall, 786 silky sharks were sampled, 56 % of the stomachs contained partially digested food remains, 30 % were empty or contained residual hard parts only, and 14 % contained only prey determined to have been eaten in the net during the purse-seine set (see “Diet composition” in “Methods” section). The majority of silky sharks (86 %) were captured in FAD sets. Small numbers of silky sharks were captured in dolphin sets during each sampling period (n = 38: 1992–1994 and n = 5: 2003–2005) and in unassociated sets during the 1990s sampling period (n = 70). Numbers of silky sharks sampled from individual purse-seine sets varied from 1 to 18 (Fig. 2), with 47 % of sets represented by only one silky shark.
Diet composition
We summarized percent prey composition for the three diet indices (Eqs. 1–3), by several levels of taxonomic resolution for the silky sharks captured only in FAD sets (Table 2). We present the prey composition for the few sharks captured in unassociated and dolphin sets in Online Resource Table S1. The overall diet for silky sharks captured in FAD sets was diverse, consisting of 26 families: 10 cephalopod families, 2 crustacean families, and 14 fish families. The relative importance of each taxon within each diet index did not vary much among the three indices, e.g., when a prey taxon was dominant by weight, it was also dominant by number and occurrence. Silky sharks were primarily piscivorous (Osteichthyes: \(\bar{W}_{i}\) > 89 %, \(\bar{N}_{i}\) > 73 %, and O i > 83 %, Table 2), and fishes in the Scombridae family were the main component of the diet (>50 %) by all three diet indices. Of the scombrids, K. pelamis (skipjack tuna) was the dominant prey species, followed by T. albacares (yellowfin tuna), unidentified Thunnus spp. (likely yellowfin tuna), and Auxis spp. (bullet and frigate tunas). The remaining scombrids contributed little to the overall diet (Table 2). Other fishes, including exocoetids (flyingfishes), hemiramphids (halfbeaks), and carangids (jacks and pompanos), were also common in the diet. Molluscs, predominantly ommastrephid squids, were important prey items, especially by frequency of occurrence, due primarily to the apparent retention and accumulation of cephalopod beaks.
Exploratory analysis
A thorough exploratory analysis is recommended prior to any classification tree modeling because data collection via balanced sampling designs is often not possible (Kuhnert et al. 2012). We began by fitting preliminary classification trees to the silky shark food habits data to identify important variables that would be included in the best model. We considered additional covariates, including year, quarter of the year, SST, and set type. Our best tree analysis included only spatial and size covariates due to a high level of confounding with other covariates. For example, the purse-seine sets yielding the samples were spatially segregated by sampling period and set type (Fig. 1). Although the first split of the preliminary tree separated silky sharks sampled in 1992–1993 from those sampled in 1994 and 2003–2005, examination of the summary output indicated that the spatial variables were masked by the year variable; longitude was a strong competitor variable to year, and the spatial variables were ranked higher than year in variable importance. SSTs were also segregated in space.
The main covariates of the preliminary trees were, as for the best tree, spatial factors (latitude and longitude), and the same spatial structure was observed in the preliminary and best trees. We revisit some of the covariates omitted from the best tree analysis in the “Discussion” section because they may have partly influenced the predation characteristics of silky sharks.
Classification tree analyses
The best classification tree analysis, based on the continuous covariates longitude, latitude, and silky shark size (cross-validated error rate = 0.888, SE = 0.036), showed strong spatial trends in the diet composition (Fig. 3), suggesting that silky shark diet varied with abundance and zoogeography of prey fauna and much less so due to silky shark ontogenetic trends. Silky shark length did not appear as a split variable in the tree analysis based on the full dataset and was not found to be a strong surrogate for the spatial variables using either the full dataset (Fig. 3) or the datasets of the sensitivity analysis. From the 1,000 trees fitted during the sensitivity analysis, median variable importance values for latitude and longitude were 0.88 (inter-quartile range, IQR 0.65–1.0) and 1.0 (IQR 0.80–1.0), respectively, compared with 0.3 (IQR 0.0–0.52) for total length.
The 1 SE classification tree for silky shark diet composition in the tropical eastern Pacific Ocean during 1992–1994 and 2003–2005 for floating-object (primarily fish-aggregating device: FAD) sets, yielding a cross-validated error rate of 0.888 (SE = 0.036). The prey category with the highest proportion weight, among a suite of prey in the diet, is displayed at each terminal node. Broad prey groups are characterized as: Sq squids, Cr crabs, N-Sc non-scombrid fishes, and Sc scombrid fishes. Variable importance rankings for each covariate, split variables and their values, and node numbers are shown. Node numbers are labeled according to the naming convention of Breiman et al. (1984). Competitor split variables for those nodes in which the improvement in the first competitor was 95 % of the best are shown in italics and shaded gray
The initial split of the full dataset partitioned 267 silky sharks south of 10.9°N (node 2) from 22 silky sharks north of that latitude (terminal node 3) (Figs. 3, 4a). These 22 silky sharks had an uncharacteristically low diet diversity (D = 0.216) because they had eaten large proportions (82 %) of unidentified Thunnus spp., which is atypically specialized foraging for silky sharks in the tropical EPO. Eighteen of the 22 silky sharks at node 3 were sampled from the same purse-seine set, and therefore, the first partition was not supported by the sensitivity analysis.
Details of the splits defined in the 1 SE classification tree (Fig. 3), showing sample locations, numbers of silky sharks, diet diversity (D), and prey composition in proportion weight for each node in the tree: a 289 sharks partitioned by latitude into nodes 2 and 3, b 267 sharks partitioned by longitude into nodes 4 and 5 (sharks at node 4 were further divided into four areas in the eastern region), c 124 sharks partitioned by longitude into nodes 8 (area 1, shaded) and 9, d 96 sharks partitioned by longitude into nodes 18 (area 2, shaded) and 19, and e 81 sharks partitioned by longitude into nodes 38 (area 4, offshore-shaded region) and 39 (area 3, inshore-shaded region). Light gray filled circles in b–e show sample locations that are not included in the nodes for which prey composition is displayed. Broad prey groups are characterized as: Sq squids, Cr crabs, N-Sc non-scombrid fishes and Sc scombrid fishes. Prey are categorized as FAD-associated (fish-aggregating device) or non-FAD-associated
Tree partitions: the eastern versus western region
There is strong support for a pronounced east–west diet shift based on both the full and sensitivity analyses. The second split of the full dataset was based on a major difference in the diet composition. One hundred and twenty-four silky sharks sampled east of 118.5°W (node 4, hereafter the “eastern region”) were separated from 143 silky sharks in the west (node 5, hereafter the “western region”) (Figs. 3, 4b) based on their diets. The tree indicated greater diet diversity (D = 0.877) for the silky sharks in the eastern region, where important components of the prey community included squids (e.g., D. gigas), crabs (Portunidae), and fishes, e.g., frigate and bullet tunas (Auxis spp.), yellowfin (T. albacares) and skipjack (K. pelamis) tunas, jacks and pompanos (Carangidae), and flyingfishes (Exocoetidae), than the silky sharks in the western region (Fig. 4b, node 4). Silky sharks from the western region foraged primarily on scombrid fishes, with skipjack and yellowfin tunas dominating the diet; diet diversity for these silky sharks was moderate (D = 0.618) (Fig. 4b, node 5). The same large-scale, longitudinal, spatial structure was also prominent in the sensitivity analysis. Of the 1,000 trees, 859 had the first (top) partition at either 118.5°W (232) or 120.6°W (627), and longitude 120.6°W was a strong competitor split for the first partition of the tree based on the full dataset (Fig. 3). Hereafter, we refer to longitudes 118.5°W and 120.6°W as ~120°W, i.e., the partition into eastern and western regions.
To determine which prey were driving the east–west diet partition, the prey were aggregated into four broad categories that comprised the dominant prey predicted using the full dataset: squids, crabs, non-scombrid fishes, and scombrid fishes and compared to the diet obtained from the sensitivity analysis. The broad-scale pattern in the diet identified by the full-dataset tree was supported by the sensitivity analysis (Fig. 5). Squids and non-scombrid fishes were dominant prey items in the eastern region, and scombrid fishes were important prey in the western region.
Boxplots showing prey composition estimates by proportion weight obtained from the sensitivity analysis for 859 trees with a top partition at either 118.5°W or 120.6°W. Prey taxa were combined into four dominant groups identified by the classification tree analysis of the full dataset (Fig. 3): squids (D. gigas, S. oualaniensis), crabs (Portunidae), non-scombrid fishes (Carangidae, Coryphaenidae, C. pauciradiatus, Decapterus spp., Exocoetidae, O. micropterus, Tetraodontiformes), and scombrid fishes (A. solandri, Auxis spp., K. pelamis, other identified scombrids (E. lineatus, Sarda orientalis, and T. obesus), T. albacares, Thunnus spp., and unidentified scombrids
Tree partitions: the eastern region
In the analysis of the full dataset, the eastern region was further partitioned by longitude into four areas (Figs. 3, 4c–e), reflecting a spatial transition in diet. Invertebrates, especially D. gigas (Humboldt squid) and portunid swimming crabs, were prominent components of the diet in the eastern inshore region, and both non-scombrid and scombrid fishes dominated the diet in the offshore eastern region. The most inshore area was east of 85°W along the coast of South America, predominantly off Peru (Fig. 4c: node 8). This region is highly productive due to upwelling induced by the Humboldt Current. The diet diversity of silky sharks in this region was moderate (D = 0.529), reflecting a short, simple food chain characteristic of a wasp-waist ecosystem (Cury et al. 2000). Humboldt squid was the principal diet component of the silky sharks in this inshore area (Fig. 4c, node 8). The sensitivity analysis supported an area encompassing the Humboldt Current region, although the two dominant tree splits partitioned the data latitudinally versus longitudinally. Of the 859 trees from the sensitivity analysis that had a first partition at ~120°W, the most common (51 % of the trees) first partition of the eastern region was on latitude, at either 3°S (319 trees) or 2°N (123 trees), instead of 85°W longitude, as occurred for the full dataset (Fig. 3, node 8). These two latitudinal partitions resulted in areas similar to those observed for the full dataset, however, given the sparseness of data around the equator and in the inshore area. In addition, 2°N latitude was a strong competitor split for the first partition of the eastern region based on the full dataset (Fig. 3). For the following two splits, at nodes 9 and 19 (Fig. 3), the sensitivity analysis did not produce a tree structure similar to that of the full dataset, and areal boundaries in the eastern area are uncertain. Thus, the diet composition in the eastern region is heterogeneous, and alternative representations of the diet exist.
Best tree representation of silky shark diet
We conclude that the tree for the full dataset (Fig. 3) gives the best representation of the diet of the silky sharks in our sample because the sensitivity analysis identified the same large-scale diet trends that were revealed by the full dataset. Both analyses identified a marked difference in the prey composition between the eastern and western regions of the tropical EPO, with invertebrates more important in the inshore eastern region and scombrid fishes more important in the western region (Figs. 4b, 5). Also, both analyses identified diet differences in the area encompassing the Humboldt Current, where the Humboldt squid was a prominent prey. The uncertainty as to the boundaries of the smaller-scale spatial areas within the eastern region is the result of heterogeneity in diet composition.
Spatial trends in diet diversity
Spatial trends in diet diversity, based on the full-dataset tree, are presented by set location and smoothed with a generalized additive model (GAM, Wood 2006) in Fig. 6. Overall diet diversity was high zonally across the sample region (D = 0.7–1.0), with the exception of the Humboldt Current region where diet diversity was moderate (D ≈ 0.5). As indicated above, diet diversity was low (D ≈ 0.2) in the north, because multiple silky sharks from the same purse-seine set were feeding nearly exclusively on Thunnus spp.
Spatial trends in the Gini index of diet diversity for silky sharks, predicted by the 1 SE classification tree (Fig. 3) and smoothed with a generalized additive model (GAM). Black points represent silky shark sample locations, and white lines denote standard error contours
Influence of FADs on feeding behavior
FAD-associated prey dominated the diet of silky sharks in our sample. Our results showed greater proportions of FAD-associated than non-FAD-associated prey in the diet over the entire sampling region (\(\alpha = 1.16 \, (\text{CI} = 1.11,2.05)\)). In addition, higher proportions of FAD-associated prey appeared in the western region compared with the eastern region (\(\beta = 2.12 \, ({\text{CI}} = 2.11,3.40)\)). The prey taxa included in each category are shown by species in Table 2 and by group in Fig. 4a–e.
Prey–predator size relationships
We examined the size compositions of the silky sharks, prey items, and prey/silky shark size ratios to identify potential spatial patterns. First, we found that the silky sharks sampled from the same purse-seine sets were fairly uniform in size. However, a strong spatial component in the size composition of the silky shark bycatch is well known in the EPO (Román-Verdesoto and Orozco-Zӧller 2005; Watson et al. 2009). Shark size classes are defined by the IATTC observer program (Román-Verdesoto and Orozco-Zӧller 2005), with large sharks >1,500 mm and medium sharks 900–1,500 mm caught throughout the EPO and small sharks <900 mm caught primarily north of the equator. We found, in general, that the size distributions for medium and small silky sharks in our samples were mostly consistent with the reported patterns, whereas our large silky sharks were sampled primarily inshore (Fig. 7). Lastly, when we stratified the continuous size data according to the eastern and western regions identified by the classification tree (Fig. 3, nodes 4 and 5), there were proportionally more silky sharks greater than about 1500 mm TL in the east (Fig. 8). On the other hand, prey in the east and west were fairly similar in size, although there were proportionally more prey larger than approximately 300 mm in the west (Fig. 8). Prey/silky shark size ratios (i.e., actual prey–silky shark pairs) were smaller in the east than those in the west (Fig. 8), demonstrating that size ratios alone are insufficient for determining whether differences are due to prey sizes, silky shark sizes, or both. To further examine diet diversity by the three size classes used in the IATTC observer program (small, medium, and large), we created preliminary maps of the spatial trends in the Gini index of diversity for silky sharks in each size class and smoothed with a generalized additive model. Prey diversity was moderate to high for each size class in both the east and the west.
Proportions of silky sharks sampled from floating-object (primarily fish-aggregating device: FAD) sets in three total length (TL) intervals as defined by the Inter-American Tropical Tuna Commission (IATTC) observer program (Román-Verdesoto and Orozco-Zӧller 2005) for each 5-degree area in the tropical eastern Pacific Ocean during this study. Small sharks are <900 mm TL, medium sharks 900–1,500 mm, and large sharks >1,500 mm
Q–Q (quantile) plots of size distributions of silky sharks, prey, and prey/silky shark ratios in the eastern (east of 118.5°W) and western (west of 118.5°W) regions identified by the classification tree (Fig. 3: nodes 4 and 5)
Our results suggest that silky sharks, especially the large sharks, are opportunistic predators, and size-selective predation is relatively unimportant. The quantile regression results showed that the size range of prey eaten increased with silky shark size. Large silky sharks ate progressively larger prey, likely due to increasing gape size, but large sharks, particularly those captured in the eastern region, also ate small prey including squids, non-scombrid fishes, and scombrid fishes (Fig. 9a). The quantile regression parameters and summary statistics from the mixed-effects model fitted to the prey–silky shark data are presented by region in Table 3. The slopes were significantly different from zero for the median and upper bound (90th) quantiles. For each region, maximum prey size significantly increased (P < 0.01) with silky shark size, while no significant trend was observed in minimum prey size (10th quantile) (east: P = 0.541; west: P = 0.724) (Table 3). A broader range of prey sizes was eaten in the west than in the east for a given size silky shark. Upper bound slopes among regions were not significantly different (t 170 = −1.140, P = 0.872). Prey/silky shark ratio-based trophic-niche breadth estimated from quantile regressions versus silky shark size (10th and 90th quantiles) did not change with ontogeny in either region (bootstrap confidence intervals included zero at the α = 0.05 level) (Fig. 9b) further confirming opportunistic predation based on size.
Quantile regression scatterplots showing a prey size versus silky shark total length and b prey/silky shark size ratios (trophic-niche breadth) versus silky shark total length for sharks west and east of 118.5°W, as defined by the classification tree (Fig. 3). Prey taxa are combined into broad categories. “Squids” (D. gigas and S. oualaniensis), “crabs” (Portunidae), “non-scombrid fishes” (Carangidae, Coryphaenidae, C. pauciradiatus, Decapterus spp., Exocoetidae, O. micropterus, and Tetraodontiformes), and “scombrid fishes” (E. lineatus, Sarda orientalis, T. obesus, T. albacares, Thunnus spp., and unidentified scombrids). The lower and upper bounds of prey size are represented by the 10th and 90th quantiles (gray lines), respectively. The median, 50th quantile, is represented by the dark solid line, and the dashed line represents the least squares estimate of the conditional mean
Discussion
We provided a detailed account of the predation habits of silky sharks collected from the bycatch of the tropical EPO tuna purse-seine fishery based on a comprehensive dataset of stomach contents, and using classification tree and quantile regression methods. General synopses of the biology and ecology of silky sharks have described them as opportunistic predators that feed primarily on fishes, but also eat molluscs and crustaceans (Compagno 1984; Bonfil 2008). Researchers working on the feeding habits of coastal silky sharks off Mexico, caught by longline gear, however, surmised that the sharks they studied were selective, specialist predators. In near-shore areas off Mexico, Cabrera-Chávez-Costa et al. (2010) reported that silky sharks ate mainly red crabs (Pleuroncodes planipes), chub mackerel (Scomber japonicus), and Humboldt squid (D. gigas). Silky shark diets were dominated by the swimming crab Portunus xantusii affinis in the southern Gulf of Tehauntepec, Mexico (Cabrera 2000; Barranco 2008), and the red crab in the subtropical areas of Baja California Sur, most likely due to the local abundance and availability of these prey (Cabrera-Chávez-Costa et al. 2010). Our results for silky sharks sampled over a large region showed a broader diet than those revealed by these smaller, near-shore studies, suggesting that silky sharks indeed fulfill the role of opportunistic predators in the pelagic EPO.
Classification tree analysis
Our classification tree analysis identified a strong spatial shift in the diet composition, and silky shark length was not an important variable for explaining diet trends. We found that the diet composition of silky sharks changes little with ontogeny. If specialization through selection of certain prey taxa increased with predator size, then the silky shark size covariate would have either appeared in the tree or would have been masked by the spatial variables. If this were the case, then silky shark size would have been more important in the variable importance plot (Fig. 3) for explaining diet composition and a different species composition of the diet would have been identified based on silky shark size. Instead, we found that foraging patterns were different in the eastern and western regions of the tropical EPO, likely due to variability in the distribution, abundance, and zoogeography of the prey.
We used preliminary tree models to explore temporal covariates, i.e., year and quarter, in conjunction with spatial and size covariates for explaining the predation variability in silky sharks. Temporal trends in the diet are potentially important because our silky shark samples were from two 2-year periods separated by a decade, and because Olson et al. (2014) found important changes in the forage communities of yellowfin tuna taken from the same purse-seine sets over the same decade. Moreover, yellowfin tuna and silky sharks shared some of the same prey resources during this time interval, e.g., Humboldt squid, flyingfishes, jacks and pompanos, and Tetraodontiformes. The preliminary tree models that included temporal covariates, however, did not provide conclusive results because of the confounding of space and time. Specifically, the distribution of the purse-seine fishery has changed considerably over the decade, and the sets from which samples were taken were spatially segregated differentially by sampling period and set type. We, therefore, omitted temporal covariates from the best tree analysis.
We suspect that spatial and temporal factors both have a role in determining silky shark predation habits, but the samples are inadequate to test whether the diet has changed over time. We attempted to examine whether the spatial structure observed in the tree analysis of the full dataset was due to a temporal change in the diet by fitting a separate classification tree to each dataset (i.e., the 1990s vs. 2000s datasets), using the spatial and silky shark size covariates. The top split of both trees indicated an inshore/offshore difference in silky shark diet. We cannot, however, rule out the possibility that the diet has changed over time. Widespread climate-induced habitat compression (Stramma et al. 2010, 2012), reductions in biological production (Behrenfeld et al. 2006; Polovina et al. 2008; Stramma et al. 2008), and changes in phytoplankton community and size composition (Barnes et al. 2010; Polovina and Woodworth 2012) may be altering food webs in the subtropical and tropical Pacific Ocean (Olson et al. 2014).
Influence of FADs on feeding behavior
According to Marsac et al. (2000) and Dagorn et al. (2013), for example, the large-scale deployment of FADs has modified the natural habitat of pelagic fishes in tropical and subtropical regions. Our work supports a hypothesis that FADs can alter trophic interactions and predation patterns of silky sharks by increasing predation pressure on tunas and other fishes that aggregate at FADs (see also Hunsicker et al. 2012). In the tropical EPO (Román-Verdesoto and Orozco-Zӧller 2005) and other oceans (Ménard et al. 2000; Filmalter et al. 2011), silky sharks commonly aggregate at FADs that are placed in the ocean by fishermen to facilitate the capture of tunas. Our study is focused on silky sharks that were caught while associated with FADs in the tropical EPO. Our results showed greater proportions of FAD-associated prey (especially skipjack and yellowfin tunas) than non-FAD-associated prey (e.g., squids, crabs, and flyingfishes) in the silky shark diet. These results indicate that pelagic fishes associated with FADs may be more vulnerable to predation by silky sharks than prey that do not associate. Thus, silky sharks appear to take advantage of the associative behavior of fishes to increase their probability of encountering and capturing prey.
Prey–predator size relationships
Prey–silky shark length relationships determined based on quantile regression analysis illustrated that maximum prey size increased with increasing silky shark size, while minimum prey size remained relatively constant across the size range. An expanding size range of prey indicates increasing foraging success for larger silky sharks, presumably due to increasing swimming speeds, visual acuity, and gape size (Scharf et al. 2000). Larger silky sharks, therefore, appear to have a competitive advantage over smaller silky sharks. Asymmetric patterns of prey use are widespread in aquatic ecosystems (Scharf et al. 2000), and our results are supported by similar studies of other large pelagic predators. Young et al. (2010) used quantile regression to evaluate prey–predator length relationships for the ten most abundant fishes caught by longline gear off eastern Australia, and found similar asymmetric prey–predator size distributions. The exceptions were lancetfish (Alepisaurus spp.) and swordfish (Xiphias gladius), which had positive lower-bound slopes. Ménard et al. (2006) used quantile regression analysis for yellowfin and bigeye (T. obesus) tunas in the French Polynesian Exclusive Economic Zone and also found a similar pattern of prey–predator size distributions. The upper bound slopes for bigeye and yellowfin tunas (0.127 and 0.091, respectively), however, were much lower than those we reported for silky sharks in the tropical EPO, indicating that silky sharks affect a wider range of prey sizes per unit of body length throughout their ontogeny than do tunas. Lucifora et al. (2009) reported that minimum and median prey size (mass) did not significantly increase with predator size for copper sharks, Carcharhinus brachyurus, captured by recreational fishermen in Anegada Bay, Argentina, while maximum prey size increased significantly. The upper bound (90th quantile) slope for copper sharks was much higher (0.8303) (Lucifora et al. 2009) than we observed for silky sharks in the tropical EPO. In contrast to our results, and to those of Young et al. (2010), Lucifora et al. (2009), and Ménard et al. (2006), Costa (2009) found that, among a suite of marine predators, both maximum and minimum prey sizes increased for larger individuals. Bethea et al. (2004) used quantile regression analysis for Atlantic sharpnose (Rhizoprionodon terraenovae), and blacktip (Carcharhinus limbatus) sharks taken from fishery-independent surveys in Apalachicola Bay, Florida, and found, like Costa (2009), that both maximum and minimum prey sizes increased as predator size increased, although the minimum prey size showed only a marginal increase for blacktip sharks. These contradictory results suggest that different shark species display different ontogenetic shifts in diet (Costa 2009), further emphasizing the opportunistic nature of shark predation.
We observed no size-related trends in trophic-niche breadth, i.e., no significant differences between the slopes of the upper and lower bounds of the relative prey-length distributions versus silky shark length. Young et al. (2010) also found no significant trends in trophic-niche breadth among ten species, with the exception of bigeye tuna, southern bluefin tuna (Thunnus maccoyi), and swordfish, and no consistent ontogenetic changes in trophic-niche breadth, i.e., diverging slopes for one species and converging slopes for two species. Ménard et al. (2006) found that neither yellowfin nor bigeye tunas exhibited significant ontogenetic changes in trophic-niche breadth. Bethea et al. (2004), however, observed ontogenetic changes in trophic-niche breadth, with the slopes of the quantiles converging, i.e., a decrease in the range of relative prey sizes taken with increasing predator size for Atlantic sharpnose sharks, and no change with increasing predator size for blacktip sharks. Scharf et al. (2000) pointed out that no ontogenetic change in trophic-niche breadth was a consistent result in several previous studies of larval and juvenile fishes, but in their study of 18 marine fish predators including flatfishes, groundfishes, sculpins, anglerfishes, fast-swimming pelagics, and elasmobranchs, it held only for the smallest predators. For several of the largest predators (>500 mm average length), they found a decrease in the breadth of relative prey sizes over ontogeny. It is clear from our results, in conjunction with those of Young et al. (2010) and Ménard et al. (2006), that Scharf et al.’s (2000) observations do not hold for all large marine top predators.
Implications
Our study offers potentially important implications on the role of a ubiquitous, generalist, apex predator in the regulation of prey populations and on evaluating food web effects of environmental changes.
Selective removal of large predatory fishes from marine food webs can impart top-down changes in trophic structure and stability via trophic cascades (Carpenter et al. 1985; Pace et al. 1999; McClanahan and Arthur 2001; Worm and Myers 2003; Essington and Hansson 2004; Frank et al. 2005). Trophic cascades that follow reductions in upper-trophic-level predators can cause increases in invertebrate predator and mesopredator populations, the latter termed mesopredator release (Baum and Worm 2009; Hunsicker et al. 2012). Our analysis of silky shark predation provides rigorous support for Hunsicker et al.’s (2012) conclusion that yellowfin and skipjack tunas act as mesopredators of a variety of sharks, billfishes, and large-bodied tunas in the tropical EPO. Moreover, yellowfin and skipjack tunas of a large size range were consumed by the sharks (Fig. 9a). Hunsicker et al. (2012) determined that tuna prey of sharks ranged from early life stages to subadults, including individuals that had important reproductive potential values (i.e., the expected number of eggs that an individual of a particular age would produce over its remaining lifetime, given that it already survived to that age).
Silky sharks have declined in the EPO (Minami et al. 2007), potentially triggering mesopredator release of the tunas. Catch per unit effort (CPUE) for silky sharks caught in purse-seine sets on floating objects showed a decline (1994–1998), followed by a period of relative stability (1998–2006), possible increase (2006–2010), and decline (2010–2013) for the northern stock while those for the southern stock showed a decline (1994–2004) followed by a period of stability (Aires-da-Silva et al. 2014). Direct evidence of mesopredator release in tunas is not available for the EPO. Worm and Tittensor (2011) suggested, however, that increases in the number and range of skipjack tuna in the tropical EPO could be attributed to the depletion of large-bodied tunas, sharks, and marlins. A food web model of the north Central Pacific Ocean (CPO) showed inconclusive evidence of mesopredator release. Some CPO model scenarios did not reveal evidence of mesopredator release in response to longline catches of apex predators (Kitchell et al. 2002), while others suggested increased biomass of small tropical tunas (i.e., yellowfin and skipjack tunas) resulted from reduced predation by sharks and billfishes (Cox et al. 2002). Given the strong evidence provided by our data for mesopredation of small tropical tunas by sharks, and by other apex predators (Hunsicker et al. 2012), we advocate that stock assessments of yellowfin and skipjack tunas should take into account the implications of variable natural mortality rates congruent with reductions in shark and billfish populations.
We conclude that silky sharks in the tropical EPO are opportunistic predators, and as such may be adaptable to changes in food webs and prey communities over time. Food web changes can result from changes in physical factors induced by climate perturbations from the bottom up (Pace et al. 1999; Hays et al. 2005; Doney et al. 2012; Polovina and Woodworth 2012; Caron and Hutchins 2013), and the potential exists for physical factors to alter the predation habits of opportunistic predators. Examples of contemporary changes in ecosystems are plentiful. (1) Primary production has declined over vast oceanic regions in the recent decade(s) (Behrenfeld et al. 2006; Polovina et al. 2008; Stramma et al. 2008; Polovina and Woodworth 2012), presumably due to increased upper-ocean temperature and vertical stratification, which influences the availability of nutrients for phytoplankton growth (Behrenfeld et al. 2006; Polovina et al. 2008). (2) Evidence is strong that the community composition and size structure of primary producers have changed in recent decades (Barnes et al. 2010, 2011). Phytoplankton cell size has declined in the subtropical oceans (Polovina and Woodworth 2012), and long-term model projections are for this decline to continue with ongoing ocean warming and intensification of stratification in the euphotic zone (Polovina et al. 2011). Phytoplankton cell size is relevant to predation dynamics because food webs that have small picophytoplankton at their base require more trophic steps to reach predators of a given size than do food webs that begin with larger phytoplankton (e.g., diatoms) (Seki and Polovina 2001). (3) Widespread climate-induced habitat compression resulting from a vertical expansion and intensification of the oxygen minimum zone (OMZ) is evident in the central and eastern tropical Pacific and Atlantic Oceans (Stramma et al. 2008, 2010, 2012). Shoaling of the OMZ can restrict the depth distribution of epipelagic predators, thus narrowing the foraging habitat and potentially altering forage communities. (4) The results of Olson et al.’s (2014) study of the predation habits of a sympatric opportunistic predator, yellowfin tuna, caught by purse-seine indicated a major decadal-scale diet shift had transpired over a broad region of the EPO. The data showed a decline in predation on larger epipelagic species and an increase in predation on smaller mesopelagic species over the decade, suggesting that broad-scale food web changes had occurred, and yellowfin tuna were feeding at lower trophic levels in the 2000s than in the 1990s sampling period.
In our present study of silky shark predation habits, we were not able to analyze the data for changes in the prey communities over time due to the confounding of space and time in our dataset. We recognize that our dataset was deficient in some aspects, and we conclude with a discussion of sampling considerations.
Sampling considerations
Silky sharks sampled for this study were collected opportunistically by observers onboard purse-seine vessels targeting tunas in mostly the tropical EPO. Fisheries-independent surveys are not available for these vast open-ocean regions in any ocean. Because fisheries-dependent sampling precludes implementing purposeful sampling designs, we observed confounding issues with space and time in our silky shark food habits dataset. Some areas of the tropical EPO were not thoroughly covered by our silky shark samples due to the opportunistic sampling design. There were large gaps in the eastern sampling region, e.g., no samples were collected from approximately 105°W to the coast of the Americas and south of the equator to about 2°S. We noticed inconsistent results from the analysis of the full dataset and the sensitivity analysis in the eastern region. More samples from this region are needed to better define the boundaries of the silky shark diet composition from this inshore region.
Pseudo-replication is a concern when multiple samples are collected from the same sampling event and treated as statistically independent samples (Hurlbert 1984). In some instances, multiple silky sharks were sampled from the same purse-seine set. Although many of the modeling assumptions of classification and regression tree analysis are well-suited for diet data and trees are a good tool for visualizing interactions, the technique is nonparametric and cannot deal with random effects (e.g., a purse-seine set level effect to examine the influence of pseudo-replication). Results from our classification tree analysis, using the full dataset, and from the sensitivity analysis were equally biologically interpretable. Both methods showed a pronounced difference in the diet in the eastern and western regions of the tropical EPO and also an influence of the Humboldt Current where the diet consisted of primarily Humboldt squid. Some areas identified by the analysis of the full dataset, however, were likely influenced by the sampling design. For example, the first split of the classification tree using the full dataset was due to multiple silky sharks from the same purse-seine set foraging nearly exclusively on unidentified Thunnus spp., indicating that pseudo-replication influenced this tree partition. Selecting multiple animals from the same sampling event may be of little use if animals are opportunistically foraging on aggregated prey. We advocate that future stomach sampling programs implement the collection of fewer samples per set, but more samples from a wider range of sets in space and time to effectively identify heterogeneous predation patterns. By increasing sampling coverage over space and time and collecting fewer samples per sampling event, scientists can investigate potential temporal and spatial influences on predator diets and use this knowledge to inform ecosystem models. Ecosystem modeling depends upon well-designed diet analyses for examining the effects of fisheries and climate variation on ecosystems over time.
References
Aires-da-Silva A, Lennert-Cody C, Maunder M, Román-Verdesoto M (2014) Stock status indicators for silky sharks in the eastern Pacific Ocean: Document SAC-05-11a Inter-American Tropical Tuna Commission Scientific Advisory Committee Fifth Meeting. Inter-Amer Trop Tuna Comm, La Jolla, CA
Allen GR, Robertson DR (1994) Fishes of the tropical eastern Pacific. University of Hawaii Press, Honolulu
Arenas P, Hall M, Garcia M (1999) Association of fauna with floating objects in the eastern Pacific Ocean. In: Scott MD, Bayliff WH, Lennert-Cody CE, Schaefer KM (eds) Proceedings of the international workshop on the ecology and fisheries for tunas associated with floating objects. Inter-Amer Trop Tuna Comm Special report 11, pp 285–326
Barnes C, Maxwell D, Reuman DC, Jennings S (2010) Global patterns in predator–prey size relationships reveal size dependency of trophic transfer efficiency. Ecology 91:222–232. doi:10.1890/08-2061.1
Barnes C, Irigoien X, De Oliveira JAA, Maxwell D, Jennings S (2011) Predicting marine phytoplankton community size structure from empirical relationships with remotely sensed variables. J Plankton Res 33:13–24. doi:10.1093/plankt/fbq088
Barranco SLM (2008) Food habits and trophic level from the silky shark, Carcharhinus falciformis, Mûller & Henle 1841 (Elasmobranchii: Carcharhinidae) in the Gulf of Tehuantepec, México using stomach contents and stable isotopes of δ13C and δ15N. Master thesis, Universidad del Mar, Oaxaca
Baum JK, Worm B (2009) Cascading top-down effects of changing oceanic predator abundances. J Anim Ecol 78:699–714. doi:10.1111/j.1365-2656.2009.01531.x
Behrenfeld MJ, O’Malley RT, Siegel DA, McClain CR, Sarmiento JL, Feldman GC, Milligan AJ, Falkowski PG, Letelier RM, Boss ES (2006) Climate-driven trends in contemporary ocean productivity. Nature 444:752–755. doi:10.1038/nature05317
Bethea DM, Buckel JA, Carlson JK (2004) Foraging ecology of the early life stages of four sympatric shark species. Mar Ecol Prog Ser 268:245–264. doi:10.3354/meps268245
Bocanegra-Castillo N (2007) Relaciones tróficas de los peces pelágicos asociados a la pesquería del atún en el Océano Pacífico oriental. Dissertation, Instituto Politécnico Nacional, Centro Interdisciplinario de Ciencias Marinas, La Paz
Bonfil R (2008) The biology and ecology of the silky shark, Carcharhinus falciformis. In: Camhi MD, Pikitch EK, Babcock EA (eds) Sharks of the open ocean. Blackwell, Oxford, pp 114–127. doi:10.1002/9781444302516.ch10
Breiman L, Friedman JH, Olshen RA, Stone CJ (1984) Classification and regression trees. Wadsworth, California
Brusca R (1980) Common intertidal invertebrates of the Gulf of California. University of Arizona Press, Tuscon
Cabrera CCA (2000) Determination of the food habits of Carcharhinus falciformis, Sphyrna lewini and Nasolamia velox (Carcharhiniformes: Carcharhinidae) during spring and summer season, through stomach content analysis in the Gulf of Tehuantepec, Mexico. Bachelor Thesis, Universidad Nacional Autónoma México (UNAM), Mexico
Cabrera-Chávez-Costa AA, Galván-Magaña F, Escobar-Sánchez O (2010) Food habits of the silky shark Carcharhinus falciformis (Müller & Henle, 1839) off the western coast of Baja California Sur, Mexico. J Appl Ichthyol 26:499–503. doi:10.1111/j.1439-0426.2010.01482.x
Cade BS, Terrell JW, Schroeder RL (1999) Estimating effects of limiting factors with regression quantiles. Ecology 80:311–323. doi:10.1890/0012-9658(1999)080[0311:EEOLFW]2.0.CO;2
Caron DA, Hutchins DA (2013) The effects of changing climate on microzooplankton grazing and community structure: drivers, predictions and knowledge gaps. J Plankton Res 35:235–252. doi:10.1093/plankt/fbs091
Carpenter SR, Kitchell JF, Hodgson JR (1985) Cascading trophic interactions and lake productivity. Bioscience 35:634–639. doi:10.2307/1309989
Chipps SR, Garvey JE (2007) Assessment of diets and feeding patterns. In: Guy CS, Brown ML (eds) Analysis and interpretation of freshwater fisheries data. American Fisheries Society, Maryland, pp 473–514
Christensen V, Pauly D (1992) Ecopath II: a software for balancing steady-state ecosystem models and calculating network characteristics. Ecol Model 61:169–185. doi:10.1016/0304-3800(92)90016-8
Clarke MR (1962) The identification of cephalopod “beaks” and the relationship between beak size and total body weight. Bull Br Mus (Nat Hist) Zool 8:419–480
Clarke MR (1986) A handbook for the identification of cephalopod beaks. Oxford University Press, Oxford
Clothier CR (1950) A key to some southern California fishes based on vertebral characters. Calif Dep Fish Game Fish Bull 79:1–83
Compagno L (1984) FAO species catalogue, vol 4. Sharks of the world. An annotated and illustrated catalogue of shark species known to date. FAO, Rome
Cortés E (1999) Standardized diet compositions and trophic levels of sharks. ICES J Mar Sci 56:707–717. doi:10.1006/jmsc.1999.0489
Costa GC (2009) Predator size, prey size, and dietary niche breadth relationships in marine predators. Ecology 90:2014–2019. doi:10.1890/08-1150.1
Cox SP, Essington TE, Kitchell JF, Martell SJD, Walters CJ, Boggs C, Kaplan I (2002) Reconstructing ecosystem dynamics in the central Pacific Ocean, 1952–1998. II. A preliminary assessment of the trophic impacts of fishing and effects on tuna dynamics. Can J Fish Aquat Sci 59:1736–1747. doi:10.1139/f02-138
Cury P, Bakun A, Crawford RJM, Jarre A, Quiñones RA, Shannon LJ, Verheye HM (2000) Small pelagics in upwelling systems: patterns of interaction and structural changes in “wasp-waist” ecosystems. ICES J Mar Sci 57:603–618. doi:10.1006/jmsc.2000.0712
Dagorn L, Fréon P (1999) Tropical tuna associated with floating objects: a simulation study of the meeting point hypothesis. Can J Fish Aquat Sci 56:984–993. doi:10.1139/f98-209
Dagorn L, Bez N, Fauvel T, Walker E (2013) How much do fish aggregating devices (FADs) modify the floating object environment in the ocean? Fish Oceanogr 22:147–153. doi:10.1111/fog.12014
Deudero S (2001) Interspecific trophic relationships among pelagic fish species underneath FADs. J Fish Biol 58:53–67. doi:10.1111/j.1095-8649.2001.tb00498.x
Doney SC, Ruckelshaus M, Duffy JE, Barry JP, Chan F, English CA, Galindo HM, Grebmeier JM, Hollowed AB, Knowlton N, Polovina J, Rabalais NN, Sydeman WJ, Talley LD (2012) Climate change impacts on marine ecosystems. Ann Rev Mar Sci 4(4):11–37. doi:10.1146/annurev-marine-041911-111611
Essington TE, Hansson S (2004) Predator-dependent functional responses and interaction strengths in a natural food web. Can J Fish Aquat Sci 61:2215–2226. doi:10.1139/f04-146
Ferretti F, Worm B, Britten GL, Heithaus MR, Lotze HK (2010) Patterns and ecosystem consequences of shark declines in the ocean. Ecol Lett 13:1055–1071. doi:10.1111/j.1461-0248.2010.01489.x
Figueroa-Zúñiga JI, Arellano-Valle RB, Ferrari SLP (2012) Mixed beta regression: a bayesian perspective. Comput Stat Data Anal 61:137–147. doi:10.1016/j.csda.2012.12.002
Filmalter JD, Dagorn L, Cowley PD, Taquet M (2011) First descriptions of the behavior of silky sharks, Carcharhinus falciformis, around drifting fish aggregating devices in the Indian Ocean. Bull Mar Sci 87:325–337. doi:10.5343/bms.2010.1057
Fischer W, Krupp F, Schneider W, Sommer C, Carpenter KE, Niem VH (1995a) Guía FAO para la identificación de especies para los fines de la pesca. Pacífico centro-oriental. Volumen I. Plantas e invertebrados. FAO (UN Food and Agriculture Organization), Rome
Fischer W, Krupp F, Schneider W, Sommer C, Carpenter KE, Niem VH (1995b) Guía FAO para la identificación de especies para los fines de la pesca. Pacífico centro-oriental. Volumen II. Vertebrados - Parte 1. FAO (UN Food and Agriculture Organization), Rome
Fischer W, Krupp F, Schneider W, Sommer C, Carpenter KE, Niem VH (1995c) Guía FAO para la identificación de especies para los fines de la pesca. Pacífico centro-oriental. Volumen III. Vertebrados - Parte 2. FAO (UN Food and Agriculture Organization), Rome
Frank KT, Petrie B, Choi JS, Leggett WC (2005) Trophic cascades in a formerly cod-dominated ecosystem. Science 308:1621–1623. doi:10.1126/science.1113075
Fulton EA, CSIRO, Australian Fisheries Management Authority (2004) Ecological indicators of the ecosystem effects of fishing: final report. Australian Fisheries Management Authority, Hobart, CSIRO, Canberra
Galván-Magaña F, Nienhuis H, Klimley P (1989) Seasonal abundance and feeding habits of sharks of the lower Gulf of California, Mexico. Calif Fish Game 75:74–84
Garth J, Stephenson W (1966) Brachyura of the Pacific coast of America, Brachyrhyncha: Portunidae. Allan Hancock Monographs in Marine Biology, vol 1. Allan Hancock Foundation, University of Southern California
Geraci M (2013) lqmm: linear quantile mixed models. R package version 103. http://CRANR-project.org/package=lqmm
Geraci M, Bottai M (2013) Linear quantile mixed models. Stat Comput. doi:10.1007/s11222-013-9381-9
Gerrodette T, Olson R, Reilly S, Watters G, Perrin W (2012) Ecological metrics of biomass removed by three methods of purse-seine fishing for tunas in the eastern tropical Pacific Ocean. Conserv Biol 26:248–256. doi:10.1111/j.1523-1739.2011.01817.x
Hall MA (1998) An ecological view of the tuna-dolphin problem: impacts and trade-offs. Rev Fish Biol Fish 8:1–34. doi:10.1023/A:1008854816580
Hays GC, Richardson AJ, Robinson C (2005) Climate change and marine plankton. Trends Ecol Evol. doi:10.1016/j.tree.2005.03.004
Heithaus MR, Frid A, Wirsing AJ, Worm B (2008) Predicting ecological consequences of marine top predator declines. Trends Ecol Evol 23:202–210. doi:10.1016/j.tree.2008.01.003
Heupel MR, Knip DM, Simpfendorfer CA, Dulvy NK (2014) Sizing up the ecological role of sharks as predators. Mar Ecol Prog Ser 495:291–298. doi:10.3354/meps10597
Hinman K (1998) Ocean roulette: conserving swordfish, sharks and other threatened pelagic fish in longline-infested waters. National Coalition for Marine Conservation, Virginia
Hunsicker M, Olson R, Essington T, Maunder M, Duffy L, Kitchell J (2012) The potential for top-down control on tropical tunas based on size structure of predator–prey interactions. Mar Ecol Prog Ser 445:263–277. doi:10.3354/meps09494
Hunter JR, Mitchell CHT (1966) Associations of fishes with flotsam in the offshore waters of Central America. US Natl Mar Fish Serv Fish Bull 66:13–29
Hurlbert SH (1984) Pseudoreplication and the design of ecological field experiments. Ecol Monogr 54:187–211. doi:10.2307/1942661
IATTC (2013) Tunas and billfishes in the eastern Pacific Ocean in 2012. Inter-Amer Trop Tuna Comm Fishery status report 11, p 171
Iverson ILK, Pinkas L (1971) A pictorial guide to beaks of certain eastern Pacific cephalopods. Calif Dep Fish Game Fish Bull 152:83–105
Jordan DS, Evermann BW (1896) The fishes of North and Middle America: a descriptive catalogue of the species of fish-like vertebrates found in the waters of North America, north of the Isthmus of Panama. Smithsonian Institution Bull US Natl Mus, Washington (Reprinted 1963, TFH Publications, Jersey City)
Juanes F (2003) The allometry of cannibalism in piscivorous fishes. Can J Fish Aquat Sci 60:594–602. doi:10.1139/f03-051
Karpouzi VS, Stergiou KI (2003) The relationships between mouth size and shape and body length for 18 species of marine fishes and their trophic implications. J Fish Biol 62:1353–1365. doi:10.1046/j.1095-8649.2003.00118.x
Kitchell JF, Essington TE, Boggs CH, Schindler DE, Walters CJ (2002) The role of sharks and longline fisheries in a pelagic ecosystem of the central Pacific. Ecosystems 5:202–216. doi:10.1007/s10021-001-0065-5
Koenker R, Basset G (1978) Regression quantiles. Econometrica 46:33–50. doi:10.2307/1913643
Kuhnert P, Duffy L, Young J, Olson R (2012) Predicting fish diet composition using a bagged classification tree approach: a case study using yellowfin tuna (Thunnus albacares). Mar Biol. doi:10.1007/s00227-011-1792-6
Lucifora L, García V, Menni R, Escalante A, Hozbor N (2009) Effects of body size, age and maturity stage on diet in a large shark: ecological and applied implications. Ecol Res 24:109–118. doi:10.1007/s11284-008-0487-z
Lunn DJ, Thomas A, Best N, Spiegelhalter D (2000) WinBUGS—a Bayesian modelling framework: concepts, structure and extensibility. Stat Comput 10:325–337. doi:10.1023/A:1008929526011
Magnuson JJ, Heitz JG (1971) Gill raker apparatus and food selectivity among mackerels, tunas and dolphins. US Natl Mar Fish Serv Fish Bull 69:361–370
Marasco RJ, Goodman D, Grimes CB, Lawson PW, Punt AE, Quinn TJI (2007) Ecosystem-based fisheries management: some practical suggestions. Can J Fish Aquat Sci 64:928–939. doi:10.1139/f07-062
Marsac F, Fonteneau A, Ménard F (2000) Drifting FADs used in tuna fisheries: an ecological trap? In: Le Gall JY, Cayré P, Taquet M (eds) Pêche thonière et dispositifs de concentration de poissons, Ed. Ifremer, Actes Colloq., vol 28, pp 537–552
Matich P, Heithaus MR, Layman CA (2011) Contrasting patterns of individual specialization and trophic coupling in two marine apex predators. J Anim Ecol 80:294–305. doi:10.1111/j.1365-2656.2010.01753.x
McClanahan TR, Arthur R (2001) The effect of marine reserves and habitat on populations of East African coral reef fishes. Ecol Appl 11:559–569. doi:10.1890/1051-0761(2001)011[0559:TEOMRA]2.0.CO;2
Meek SE, Hildebrand SF (1923) The marine fishes of Panama. Part I. Field Mus (Natl Hist) Zool Ser Chic. doi:10.5962/bhl.title.2887
Ménard F, Fonteneau A, Gaertner D, Nordstrom V, Stéquert B, Marchal E (2000) Exploitation of small tunas by a purse-seine fishery with fish aggregating devices and their feeding ecology in an eastern tropical Atlantic ecosystem. ICES J Mar Sci 57:525–530. doi:10.1006/jmsc.2000.0717
Ménard F, Labrune C, Shin Y-J, Asine A-S, Bard F-X (2006) Opportunistic predation in tuna: a size-based approach. Mar Ecol Prog Ser 323:223–231. doi:10.3354/meps323223
Miller DL, Jorgenson SC (1973) Meristic characters of some marine fishes of the western Atlantic Ocean. US Fish Wildl Serv Fish Bull 71:301–312
Miller DJ, Lea RN (1972) Guide to the coastal marine fishes of California. Calif Dep Fish Game Fish Bull 157:1–249
Minami M, Lennert-Cody CE, Gao W, Román-Verdesoto M (2007) Modeling shark bycatch: the zero-inflated negative binomial regression model with smoothing. Fish Res 84:210–221. doi:10.1016/j.fishres.2006.10.019
Monod J (1968) Le complexe urophore des poisons teleosteens. Mem Inst Fond Afr Noire 81:1–70
Olson RJ, Galván-Magaña F (2002) Food habits and consumption rates of common dolphinfish (Coryphaena hippurus) in the eastern Pacific Ocean. US Natl Mar Fish Serv Fish Bull 100:279–298
Olson RJ, Duffy LM, Kuhnert PM, Galván-Magaña F, Bocanegra-Castillo N, Alatorre-Ramirez V (2014) Decadal diet shift in yellowfin tuna (Thunnus albacares) suggests broad-scale food web changes in the eastern tropical Pacific Ocean. Mar Ecol Prog Ser 497:157–178. doi:10.3354/meps10609
Pace ML, Cole JJ, Carpenter SR, Kitchell JF (1999) Trophic cascades revealed in diverse ecosystems. Trends Ecol Evol 14:483–488. doi:10.1016/S0169-5347(99)01723-1
Parin N (1961) The basis for the classification of the flying fishes (family Oxyporhamphidae and Exocoetidae). US Natl Mus NMFS Transl 67:104
Pauly D, Christensen V, Walters C (2000) Ecopath, Ecosim, and Ecospace as tools for evaluating ecosystem impact of fisheries. ICES J Mar Sci 57:697–706. doi:10.1006/jmsc.2000.0726
Pikitch EK, Santora C, Babcock EA, Bakun A, Bonfil R, Conover DO, Dayton P, Doukakis P, Fluharty D, Heneman B, Houde ED, Link J, Livingston PA, Mangel M, McAllister MK, Pope J, Sainsbury KJ (2004) Ecosystem-based fishery management. Science 305:346–347. doi:10.1126/science.1098222
Pinnegar JK, Trenkel VM, Tidd AN, Dawson WA, Du Buit MH (2003) Does diet in Celtic Sea fishes reflect prey availability? J Fish Biol 63:197–212. doi:10.1111/j.1095-8649.2003.00204.x
Polovina JJ, Woodworth PA (2012) Declines in phytoplankton cell size in the subtropical oceans estimated from satellite remotely-sensed temperature and chlorophyll, 1998–2007. Deep Sea Res Part II 77–80:82–88. doi:10.1016/j.dsr2.2012.04.006
Polovina JJ, Howell EA, Abecassis M (2008) Ocean’s least productive waters are expanding. Geophys Res Lett 35:L03618. doi:10.1029/2007gl031745
Polovina J, Dunne J, Woodworth P, Howell E (2011) Projected expansion of the subtropical biome and contraction of the temperate and equatorial upwelling biomes in the North Pacific under global warming. ICES J Mar Sci 68:986–995. doi:10.1093/icesjms/fsq198
Power ME, Tilman D, Estes JA, Menge BA, Bond WJ, Mills LS, Daily G, Castilla JC, Lubchenco J, Paine RT (1996) Challenges in the quest for keystones. Bioscience 46:609–620. doi:10.2307/1312990
R Development Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/
Román-Verdesoto M, Orozco-Zӧller M (2005) Bycatches of sharks in the tuna purse-seine fishery of the eastern Pacific Ocean reported by observers of the Inter-American Tropical Tuna Commission, 1993–2004. Inter Am Trop Tuna Comm Data Rep 11:67
Sánchez-de Ita JA, Quiñónez-Velázquez C, Galván-Magaña F, Bocanegra-Castillo N, Félix-Uraga R (2011) Age and growth of the silky shark Carcharhinus falciformis from the west coast of Baja California Sur, Mexico. J Appl Ichthyol 27:20–24. doi:10.1111/j.1439-0426.2010.01569.x
Scharf FS, Juanes F, Sutherland M (1998) Inferring ecological relationships from the edges of scatter diagrams: comparison of regression techniques. Ecology 79:448–460. doi:10.1890/0012-9658(1998)079[0448:ierfte]2.0.co;2
Scharf FS, Juanes F, Rountree RA (2000) Predator size–prey size relationships of marine fish predators: interspecific variation and effects of ontogeny and body size on trophic-niche breadth. Mar Ecol Prog Ser 208:229–248. doi:10.3354/meps208229
Scott MD, Chivers SJ, Olson RJ, Fiedler PC, Holland K (2012) Pelagic predator associations: tuna and dolphins in the eastern tropical Pacific Ocean. Mar Ecol Prog Ser 458:283–302. doi:10.3354/meps09740
Seki MP, Polovina J (2001) Ocean gyre ecosystems. In: Steele JH, Turekian K, Thorpe S (eds) Encyclopedia of Ocean Sciences. Academic Press, London, pp 1959–1965
Smithson M, Verkuilen J (2006) A better lemon squeezer? Maximum likelihood regression with beta-distributed dependent variables. Psychol Methods 11:54–71. doi:10.1037/1082-989X.11.1.54
Stevens JD, Bonfil R, Dulvy NK, Walker PA (2000) The effects of fishing on sharks, rays, and chimaeras (chondrichthyans), and the implications for marine ecosystems. ICES J Mar Sci 57:476–494. doi:10.1006/jmsc.2000.0724
Stramma L, Johnson GC, Sprintall J, Mohrholz V (2008) Expanding oxygen-minimum zones in the tropical oceans. Science 320:655–658. doi:10.1126/science.1153847
Stramma L, Schmidtko S, Levin LA, Johnson GC (2010) Ocean oxygen minima expansions and their biological impacts. Deep Sea Res Part I 57:587–595. doi:10.1016/j.dsr.2010.01.005
Stramma L, Prince ED, Schmidtko S, Luo J, Hoolihan JP, Visbeck M, Wallace DWR, Brandt P, Kortzinger A (2012) Expansion of oxygen minimum zones may reduce available habitat for tropical pelagic fishes. Nature 2:33–37. doi:10.1038/nclimate1304
Therneau T, Atkinson B, Ripley B (2013) rpart: recursive partitioning. R package version 4.1-3. http://CRAN.R-project.org/package=rpart
Thomson DA, Findley LT, Kerstitch AN (1979) Reef fishes of the Sea of Cortez. Wiley, New York
Walters C, Pauly D, Christensen V (1999) Ecospace: prediction of mesoscale spatial patterns in trophic relationships of exploited ecosystems, with emphasis on the impacts of marine protected areas. Ecosystems 2:539–554. doi:10.1007/s100219900101
Watson JT, Essington TE, Lennert-Cody CE, Hall MA (2009) Trade-offs in the design of fishery closures: management of silky shark bycatch in the eastern Pacific Ocean tuna fishery. Conserv Biol 23:626–635. doi:10.1111/j.1523-1739.2008.01121.x
Wolff CA (1982) A beak key for eight eastern tropical Pacific cephalopods species, with relationship between their beak dimensions and size. US Natl Mar Fish Serv Fish Bull 80:357–370
Wood SN (2006) Generalized additive models: an introduction with R. Chapman and Hall/CRC Press, New York
Worm B, Myers RA (2003) Meta-analysis of cod-shrimp interactions reveals top-down control in oceanic food webs. Ecology 84:162–173. doi:10.1890/0012-9658(2003)084[0162:MAOCSI]2.0.CO;2
Worm B, Tittensor DP (2011) Range contraction in large pelagic predators. Proc Natl Acad Sci USA 108:11942–11947. doi:10.1073/pnas.1102353108
Young JW, Lansdell MJ, Campbell RA, Cooper SP, Juanes F, Guest MA (2010) Feeding ecology and niche segregation in oceanic top predators off eastern Australia. Mar Biol 157:2347–2368. doi:10.1007/s00227-010-1500-y
Acknowledgments
This project was partly funded by Cooperative Agreement NA17RJ1230 between the Joint Institute for Marine and Atmospheric Research (JIMAR) and the US National Oceanic and Atmospheric Administration (NOAA). The views expressed herein are those of the authors and do not necessarily reflect the views of NOAA or any of its subdivisions. This project was also partly supported by a special appropriation of the US Congress in 1992 for research leading to new methods of catching tuna without the incidental capture of dolphins. We also acknowledge the funding support from Commonwealth Scientific and Industrial Research Organization (CSIRO) through the Julius Award that was granted to P. Kuhnert. F. Galván-Magaña was supported by the Instituto Politécnico Nacional [Comisión de Operación y Fomento de Actividades Académicas (COFAA) and Estímulos al Desempeño de los Investigadores (EDI)], and N. Bocanegra-Castillo was supported by the Consejo Nacional de Ciencia y Tecnología (CONACYT). We are grateful to many observers in Ecuador, Venezuela, and Mexico for collecting stomach samples at sea, with the valuable assistance of Inter-American Tropical Tuna Commission (IATTC) staff E. Largacha, H. Pérez, K. Loor, V. Fuentes, C. de la A.-Florencia, A. Basante, W. Paladines, F. Cruz, and C. Maldonado and the captains and crews of numerous purse-seine vessels. Assistance with stomach-content analysis was provided in Ecuador by L. Cedeño, J. Morales, and M. Loor, and in Venezuela by J. Martinez. We also thank IATTC staff members A. Aires-da-Silva, for making the R code and resources available for producing the maps, C. Patnode, for modifying and improving the graphics, and D. Fuller, for assistance with figures. We thank W. Bayliff (IATTC) and J. Young (CSIRO) for their thoughtful, constructive reviews of this manuscript. We are grateful to the Integrated Marine Biogeochemistry and Ecosystem Research (IMBER, formerly GLOBEC) regional program Climate Impacts on Oceanic Top Predators (CLIOTOP) for sponsoring a workshop to develop Classification and Regression Tree (CART) methodologies for analyzing diet data for top predators on a global scale, with special thanks to F. Ménard and J. Young for their leadership.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. K. Carlson.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Duffy, L.M., Olson, R.J., Lennert-Cody, C.E. et al. Foraging ecology of silky sharks, Carcharhinus falciformis, captured by the tuna purse-seine fishery in the eastern Pacific Ocean. Mar Biol 162, 571–593 (2015). https://doi.org/10.1007/s00227-014-2606-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-014-2606-4