Abstract
Gait impairments are a common and consequential motor symptom in Parkinson’s disease (PD). A cognitive strategy that incorporates instructions to concentrate on specific parameters of walking is an effective approach to gait rehabilitation for persons with PD during single-task and simple dual-task walking conditions. This study examined the ability to modify dual-task walking in response to instructions during a complex walking task in people with PD compared to healthy older adults (HOA). Eleven people with PD and twelve HOA performed a cognitive task while walking with either a usual base or a narrow base of support. Dual-task walking and cognitive task performance were characterized under two conditions—when participants were instructed focus on walking and when they were instructed to focus on the cognitive task. During both usual base and narrow base walking, instructions affected cognitive task response latency, with slower performance when instructed to focus on walking compared to the cognitive task. Regardless of task or instructions, cognitive task performance was slower in participants with PD compared to HOA. During usual base walking, instructions influenced gait speed for both people with PD and HOA, with faster gait speed when instructed to focus on walking compared to the cognitive task. In contrast, during the narrow base walking, instructions affected gait speed only for HOA, but not for people with PD. This suggests that among people with PD the ability to modify walking in response to instructions depends on the complexity of the walking task.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Difficulty walking is one of the most consequential motor symptoms of Parkinson’s disease (PD), contributing to disability (Post et al. 2007), falls (Dennison et al. 2007; Kerr et al. 2010), and reduced quality of life (Rahman et al. 2008; Soh et al. 2011). Although gait impairments are not typically a presenting sign of PD, over 85 % of people with clinically probable PD develop gait problems within 3 years of diagnosis (Kang et al. 2005). Gait impairments in PD such as reduced speed and step length (Morris et al. 1994) are exacerbated during complex walking tasks, including dual-task walking conditions that require concurrent performance of a cognitive or motor task (Bond and Morris 2000; Hausdorff et al. 2003; Galletly and Brauer 2005).
People with PD report the use of increased concentration to monitor and improve walking (Jones et al. 2008; Lamont et al. 2012). This type of cognitive strategy, in which attention or concentration is focused on walking, is also a common and effective physical therapy approach for people with PD (Morris et al. 1996; Canning 2005). For example, verbal instructions to focus on walking increase gait speed and stride length in persons with PD during single-task walking (Lehman et al. 2005; Fok et al. 2011). Instructions can also improve walking under simple dual-task walking conditions, such as walking in a straight line with a usual base of support (Kelly et al. 2012; Yogev-Seligmann et al. 2012). However, the degree to which people with PD can modify walking in response to instructions under dual-task conditions may depend on walking task complexity (Canning 2005; Lohnes and Earhart 2011).
One factor that may limit the efficacy of a cognitive strategy is the need for increased cognitive resources to modify gait during complex walking tasks. The control of walking under simple conditions is characterized by two invariant features—progression, defined as the ability to move in the desired direction, and stability, defined as the ability to control the body’s center of mass with respect to the base of support (Das and McCollum 1988). While progression and stability sufficiently describe the task requirements for walking in very simple environments, mobility in the home and community requires adaptation, defined as the ability to continuously modify the gait pattern in response to varied task demands and environmental circumstances (Patla and Shumway-Cook 1999). Complex walking tasks require sensorimotor processing to guide appropriate gait pattern modifications, resulting in increased demands on cognitive processes and resources. For people with PD, the beneficial effects of a cognitive strategy under simple walking conditions suggest that cognitive processes become important to monitor and improve the basic task requirement of progression (i.e., step length). Consistent with a limited ability to adapt walking, people with PD report mobility challenges in situations that demand concentration, such as walking while performing concurrent tasks, and when walking in varied environments, such as crowded environments or over uneven or slippery terrain (Jones et al. 2008; Lamont et al. 2012).
The aim of this study was to examine the ability to modify dual-task walking in response to instructions during simple and complex walking tasks in people with PD compared to healthy older adults (HOA). We characterized walking and cognitive task performance under simple (usual base of support) and complex (narrow base of support) conditions. The ability to modify dual-task performance in response to instructions to focus on the cognitive task or focus on walking was assessed using absolute and relative measures of performance. We hypothesized that both people with PD and HOA would demonstrate a reduced ability to modify walking in response to instructions during a complex walking task compared to a simple task; however, we expected that this reduction would be greater in people with PD compared to HOA.
Methods
Participants
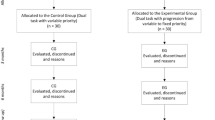
We recruited HOA from local exercise classes and from the community and people with PD from the community and from a state registry program. Participants with PD were included if they had a clinical diagnosis of PD and were excluded if they had a history of surgery for PD. Exclusion criteria for all participants were as follows: (1) any uncorrected visual or auditory impairments and (2) a diagnosis of any neurologic or orthopedic condition that affected walking (200 feet without assistance), cognition, or the ability to complete the protocol. Prior to data collection, written informed consent was obtained in accordance with approved institutional review board procedures.
Experimental procedures
Participants attended a single testing session at a university-based motion analysis laboratory. People with PD were tested in the medication-on condition, with the walking assessment beginning within 1–2 h after taking anti-Parkinson’s medications. Age, number of medical conditions, medications, and falls were assessed in a structured interview. Motor symptoms of PD were characterized using the Unified Parkinson’s Disease Rating Scale (UPDRS) Motor Examination and Hoehn and Yahr staging. Cognitive function was assessed using the Montreal Cognitive Assessment (MoCA) (Nasreddine et al. 2005).
Cognitive task
Participants performed a cognitive task while seated (single-task condition) and while walking (dual-task conditions). The cognitive task was an auditory analog of the Stroop task in which the words “high” and “low” were presented in either a high or low pitch. Participants were instructed to verbally identify the pitch of the word “as quickly and as accurately as possible.” Each trial of the cognitive task was 3 s in length, with a variable 0–1 s delay before stimulus presentation. After orientation to the task, participants performed three seated training blocks consisting of 20 stimuli per block to minimize learning effects. For the remainder of testing, blocks consisted of 8–12 stimuli. Block length was consistent for a given participant but was adjusted between participants in order to capture a similar number of strides per condition while minimizing fatigue. Within a block, each of the four stimuli was presented an equal number of times in random order. Seated single-task blocks of the cognitive task were performed at the beginning and at the end of the testing session. Two blocks were performed in each dual-task walking condition. Stimuli were presented and responses were recorded using a wireless headset and microphone system (Plantronics, Inc., Santa Cruz, USA; Jabra Corporation, Nashua, USA) that was integrated with custom hardware and software. The primary outcome measures were response latency and response accuracy. Response latency was measured as the time from stimulus onset to response onset. Response accuracy was the number of correct responses divided by the total number of stimuli, expressed as a percentage. Use of both measures allowed characterization of any trade-offs between speed and accuracy in performance of the cognitive task.
Walking tasks
Participants walked on a level surface along an 8.8 m pathway under two conditions: usual base (UB) walking and narrow base (NB) walking. Participants walked continuously back and forth across the walkway for approximately 30 s per block. Walking was recorded only when participants were walking in a straight line in the middle 4 m of the walkway. Participants were asked to walk with arms crossed for all conditions to eliminate the use of arms for balance and to ensure adequate data capture for whole-body modeling. The UB path (60 cm wide) and the NB path (20 cm wide) were displayed on the floor using tape. For UB walking, instructions were to “walk as quickly as possible.” For NB walking, instructions were to “walk as quickly and as accurately as possible.” Single-task walking was performed in separate blocks at the beginning and end of each walking condition. A Qualisys Motion Capture System (Qualisys, Gothenburg, Sweden) recorded the position of markers placed bilaterally on the feet (heel, 3rd metatarsal–phalangeal joint, lateral malleolus), legs (tibial tuberosity, lateral knee joint, patella, mid-thigh, greater trochanter), pelvis (anterior superior iliac spine, iliac crest), and trunk (acromion, sternum, and thorax). Whole-body center of mass (CoM) was calculated as the weighted sum of an 8-segment model (trunk, pelvis, and bilateral thighs, shanks, and feet).
Primary outcome measures for walking were gait speed and, for NB walking only, step accuracy. For both UB and NB walking, gait speed was measured as the distance traveled by the ankle joint center during each stride (i.e., one heel strike to the next heel strike of the same foot) divided by the stride time (i.e., time between one heel strike to the next heel strike of the same foot). For NB walking, step accuracy was measured as the lateral ankle joint position at heel strike relative to the NB path. An accurate step was one in which the ankle marker fell on or within the path boundary. Step accuracy was the number of accurate steps divided by the total number of steps, expressed as a percentage. For inaccurate steps, the step error magnitude was calculated as the distance between the ankle marker and the path boundary.
In addition to these primary outcome measures, several spatiotemporal measures were used as secondary variables to further characterize changes in walking. Stride length was defined as the distance (length) between the ankle joint center at one heel strike and the ankle joint center at the next heel strike of the same foot. Step width was calculated as the distance (width) between the ankle joint center of one foot at heel strike and the ankle joint center of the opposite foot at heel strike. Cadence, or the rate of stepping, was defined as the number of steps per minute. Step width variability and stride time variability were expressed as the coefficient of variation to quantify the spatial and temporal consistency of the gait pattern.
Control of the CoM in the frontal plane was used to assess biomechanical stability during walking. The frontal plane inclination angle quantifies the position of the center of mass relative to the base of support (approximated by the ankle joint). The inclination angle was calculated as the angle between a line connecting the CoM and the lateral malleolus marker and a vertical line through the CoM. The peak frontal plane inclination angle value was then determined during single limb stance (Chen and Chou 2010).
Dual-task conditions
Under dual-task walking conditions, participants performed the cognitive task while walking. In each block, instructions were given to focus on either the cognitive task or walking. In the cognitive focus (DTcog) condition, instructions were “focus on the cognitive task, and perform it as quickly and as accurately as you did when you were sitting.” For the walking focus (DTwalk) condition, instructions were “focus on walking, and walk as quickly (and as accurately, in the NB condition) as you did when you were only walking.” The order of UB versus NB walking was randomized, and within each walking condition, the order of instructed prioritization was randomized.
The effect of instructions on dual-task walking and cognitive task performance was assessed by comparing (1) absolute measures of performance and (2) the dual-task effect (DTE) in the DTcog and DTwalk conditions. The DTE is a relative measure of dual-task compared to single-task performance and was calculated for each of the primary outcome measures as the difference between single-task and dual-task performance normalized to single-task performance and expressed as a percentage (Bock 2008; Kelly et al. 2010). All DTEs were operationally defined such that a negative value represents a dual-task cost or decrement and a positive value represents a dual-task benefit (Kelly et al. 2010).
Statistical analysis
Descriptive analysis was performed for all variables (IBM SPSS Statistics version 19.0, Armonk, USA). Potential group differences in age and cognitive function were assessed using t tests. The effects of instructions and walking task were examined for each outcome measure using repeated-measures ANOVA with two within-subject factors, instructions (DTwalk, DTcog) and walking task (UB, NB), and one between-subject factor, group (HOA, PD). Step accuracy was assessed only during NB walking, so the effect of instructions was assessed using a repeated-measures ANOVA with one within-subject factor, instructions, and one between-subject factor, group. The level of significance for all tests was set at α = .05. When ANOVAs were statistically significant, post hoc comparisons were performed using the Scheffé test.
Results
Participants
Participants were 11 people with PD and 12 HOA (Table 1). Groups were similar with respect to age (P = .49), cognitive function assessed by the MoCA (P = .39), number of comorbidities (P = .43), and number of medications (P = .07). Two of 12 HOA and four of 11 people with PD reported falls in the previous 3 months. Under single-task conditions, people with PD walked more slowly than HOA (P = .002) but had similar NB step accuracy (P = .73). For both groups, UB walking was faster than NB walking (P < .001). For single-task cognitive task performance, people with PD had similar response latency (P = .09) but lower response accuracy (P = .04) than HOA.
Cognitive task performance
During dual-task performance, participants with PD performed the cognitive task more slowly than HOA (P = .05) but with similar accuracy (P = .15; Table 2; Fig. 1). Participants with PD and HOA had similar DTEs for response latency (P = .41) and response accuracy (P = .52), reflecting similar declines in dual-task relative to single-task performance of the cognitive task. Instructions impacted response latency but not response accuracy, regardless of the walking task. A main effect of instructions indicated that instructions to focus on walking resulted in longer response latencies (P = .03) and greater response latency dual-task costs (P = .03), with no interactions. Response accuracy and response accuracy dual-task costs were not affected by instructions or the walking task (all P > .14).
Dual-task performance of the cognitive task, showing a response latency and b response accuracy for both groups during UB and NB conditions. Symbols represent the group mean, and bars represent the standard error. Open symbols ( ,
, ) represent people with PD; filled symbols (
) represent people with PD; filled symbols ( ,
, ) represent healthy older adults. Squares (
) represent healthy older adults. Squares ( ,
, ) represent the UB condition; triangles (
) represent the UB condition; triangles ( ,
, ) represent the NB condition. DTwalk indicates the dual-task condition with instructions to focus on walking; DTcog indicates the dual-task condition with instructions to focus on the cognitive task
) represent the NB condition. DTwalk indicates the dual-task condition with instructions to focus on walking; DTcog indicates the dual-task condition with instructions to focus on the cognitive task
Walking performance
Approximately 19 strides per condition were analyzed for each participant, and the number of strides per condition was not impacted by group, task, or instructions (all P > .29). As shown in Table 2 and Fig. 2, a three-way interaction between group, walking task, and instructions for gait speed (P = .05) and gait speed DTE (P = .02) indicated that both groups modified gait speed in response to instructions during simple UB walking conditions, but only HOA modified walking in response to instructions during complex NB walking. During UB walking, both groups had faster gait speeds when instructed to focus on walking than when instructed to focus on the cognitive task (main effect of instructions: P = .01). In contrast, during NB walking, only HOA walked faster when instructed to focus on walking. People with PD did not modify gait speed in response to instructions during NB walking (group × instruction interaction P = .008). In addition, an interaction (P = .01) between group and walking task indicated that NB gait speed was slower than UB gait speed only for people with PD. Narrow base step accuracy and step accuracy DTE did not differ between groups (both P > .68) and were not influenced by instructions (both P > .35).
Dual-task performance of walking demonstrating a gait speed and b step accuracy for both groups during UB and NB conditions. Symbols represent the group mean, and bars represent the standard error. Open symbols ( ,
, ) represent people with PD; filled symbols (
) represent people with PD; filled symbols ( ,
, ) represent healthy older adults. Squares (
) represent healthy older adults. Squares ( ,
, ) represent the UB condition; triangles (
) represent the UB condition; triangles ( ,
, ) represent the NB condition. DTwalk indicates the dual-task condition with instructions to focus on walking; DTcog indicates the dual-task condition with instructions to focus on the cognitive task
) represent the NB condition. DTwalk indicates the dual-task condition with instructions to focus on walking; DTcog indicates the dual-task condition with instructions to focus on the cognitive task
Secondary spatiotemporal and stability measures
Table 3 shows secondary spatiotemporal and stability measures. Similar to gait speed, a three-way interaction for stride length (P = .002) indicated that both HOA and people with PD modified stride length in response to instructions under UB conditions. In contrast, only HOA modified stride length in response to instructions during NB walking. Cadence was slower in the PD group compared to HOA (P = .002) and was higher when participants were instructed to focus on walking compared to instructions to focus on the cognitive task (P = .007). Cadence did not differ between UB and NB walking tasks and there were no interactions. During NB compared to UB walking, step width was smaller (P < .001) and both stride time variability (P = .01) and step width variability (P = .004) were greater. There were no effects of instructions or group and no interactions for step width or variability parameters. Similarly, CoM frontal plane inclination angle was reduced during NB compared to UB walking (P < .001), but did not differ between groups or in response to instructions.
Discussion
The main finding of this research was that during simple, UB walking, both people with PD and HOA demonstrated faster gait speed when instructed to focus on walking and slower gait speed when instructed to focus on the cognitive task. In contrast, during the more complex, NB walking task, instructions influenced gait speed for HOA, but not for people with PD, despite the fact that both groups maintained NB step accuracy. This suggests that among people with PD, the ability to modify walking in response to instructions depends on the complexity of the walking task. These results support the hypothesis that people with PD have a diminished ability to modify walking in response to instructions when performing a complex walking task compared to a simple walking task.
This study is consistent with previous research in showing that people with PD are able to modify dual-task walking performance in response to instructions under simple, UB conditions (Kelly et al. 2013; Yogev-Seligmann et al. 2012). The current study expands this research by demonstrating that the ability to modify walking in response to instructions may be reduced among people with PD during the performance of more complex walking tasks. Modification of the basic gait pattern to accommodate more complex task and environmental challenges requires increased sensorimotor processing, contributing to greater cognitive demands during complex tasks, such as stair climbing (Ojha et al. 2009) and stepping onto a curb (Wellmon et al. 2013), compared to simple walking tasks. The need for increased cognitive resources when performing complex walking tasks may limit the ability to modify walking in response to instructions, particularly in people with PD. Mobility in daily life requires frequent modification of the gait pattern to accommodate changing task and environmental constraints, yet people with PD report difficulty walking while performing concurrent tasks and walking in attention demanding environments, such as at road crossings, on ramps, or in crowded environments (Jones et al. 2008; Lamont et al. 2012). Thus, the efficacy of a cognitive strategy may be attenuated during functional mobility in complex home and community environments compared to simple walking tasks in controlled environments.
A second finding of interest was that changes in speed among people with PD did not come at the expense of biomechanical stability. While people with PD walked more slowly than HOA, regardless of instructions or the walking task, the groups did not differ with respect to frontal plane inclination angle. This finding is consistent with recent research by Galna et al. (2013), demonstrating that people with PD and healthy controls had similar inclination angles during single-task, level-ground walking. However, in that study (Galna et al. 2013), participants with PD did demonstrate a greater increase in the frontal plane inclination angle from level-ground walking to obstacle crossing compared to control participants. In contrast, participants with PD in the current study did not demonstrate increased inclination angle with the more challenging NB walking task. Together, these studies suggest that biomechanical stability is not uniformly compromised in people with PD, but may vary with individual characteristics, such as disease severity (Galna et al. 2013), or walking task demands.
In the current study, stability did not differ regardless of how it was measured. Groups were similar in terms of both biomechanical stability, assessed using the frontal plane inclination angle, and stride-to-stride variability, which is sometimes considered a proxy for stability. These findings suggest that neither slower gait speed nor the ability to modify gait speed in response to instructions necessarily come at the expense of reduced stability. People with PD also maintained similar NB step accuracy compared to HOA. Thus, differences in walking between groups and changes in response to instructions were isolated to gait speed. The reduction in gait speed along with preserved NB step accuracy and stability may reflect a safe and functional strategy for modifying walking in response to increased task complexity. It is important to note that these findings may not generalize to people with PD who have cognitive dysfunction, as the participants in this study had relatively preserved cognition.
During dual-task walking, changes in performance are often used to infer whether walking or the concurrent task is prioritized. It has been proposed that postural control should be prioritized during dual-task standing or walking in order to maintain stability and prevent falls. However, some research suggests that people with PD may prioritize concurrent tasks over postural control (Bloem et al. 2006), contributing to dual-task walking deficits, reduced safety, and increased risk for falls. In many studies, however, postural stability is not measured directly but is inferred from subjective assessments of walking (Bloem et al. 2001) or measures of spatiotemporal variability (Yogev-Seligmann et al. 2012). A recent study by Lord and colleagues examined domains of gait in older adults and demonstrated that variability parameters loaded on pace and variability domains of gait performance, but not postural control domains (Lord et al. 2013). Thus, the relationship between measures of variability and a direct, biomechanical measure of postural stability during walking is not well understood. In the current study, both the direct measure of biomechanical stability and measurements of stride-to-stride variability indicated that postural control was comparable between people with PD and HOA during both the simple UB task and the more challenging NB walking tasks.
Several limitations of this study should be noted. First, aspects of the walking tasks were driven by methodological considerations but may limit the generalizability of these findings to natural settings. Participants were asked to walk as quickly as they safely could in order to optimize the sensitivity to dual-task interference. This instruction, in and of itself, could be considered a form of instructed focus. However, instructions to focus on the cognitive task nevertheless resulted in declines in walking speed for HOA and people with PD during the UB walking condition. We asked all participants to walk with their arms crossed in order to standardize the protocol by eliminating use of the arms while optimizing the quality of marker position data necessary for center-of-mass calculation. The more complex task of NB walking was incorporated because tandem and NB walking are clinically useful in the assessment of postural control during walking and because these tasks require frontal plane control of the center of mass relative to the base of support. However, the degree to which this replicates the postural control challenge of daily mobility tasks, such as walking in narrow or crowded spaces, is not known. Another limitation was that the cognitive task chosen, an auditory analog of the Stroop test, may not optimally represent the challenges of dual- or multitask walking situations in daily life. This task was chosen because of the high demand it places on executive functioning and because it can be carefully quantified with respect to both the speed and accuracy of responses.
Results from this study suggest that the effectiveness of a cognitive strategy to modify walking in people with PD may be task specific. While concentration improves walking in people with PD during simple walking tasks, this cognitive strategy may not transfer to the full repertoire of complex walking tasks necessary for safe and effective mobility in the home and community. An inability to modify walking through the use of concentration under complex conditions may explain the observation by people with PD of difficulty walking while performing concurrent tasks or walking in busy or unpredictable environments (Jones et al. 2008; Lamont et al. 2012). The current study suggests that one reason for this self-reported observation is that increased complexity of walking tasks or environments limits the ability to improve walking through a cognitive strategy. Further research is need to identify additional factors, such as individual cognitive status, severity of motor symptoms, or medication-related fluctuations, that may impact the efficacy of a cognitive strategy to improve walking in people with PD.
The ability to modify walking in response to changing individual capacity, task demands, and environmental situations is critical to functional mobility in the home and community. In the current study, people with PD demonstrated a limited ability to flexibly modify walking performance in response to an instructed focus on walking, but only during complex walking tasks. Other individual, task, and environmental factors that impact the ability to modify walking should be examined to understand the limits of using a cognitive strategy to improve mobility in PD.
References
Bloem BR, Valkenburg VV, Slabbekoorn M, van Dijk JG (2001) The multiple tasks test. Strategies in Parkinson’s disease. Exp Brain Res 137:478–486
Bloem BR, Grimbergen YA, van Dijk JG, Munneke M (2006) The “posture second” strategy: a review of wrong priorities in Parkinson’s disease. J Neurol Sci 248:196–204
Bock O (2008) Dual-task costs while walking increase in old age for some, but not for other tasks: an experimental study of healthy young and elderly persons. J Neuroeng Rehabil 5:27
Bond JM, Morris M (2000) Goal-directed secondary motor tasks: their effects on gait in subjects with Parkinson disease. Arch Phys Med Rehabil 81:110–116
Canning CG (2005) The effect of directing attention during walking under dual-task conditions in Parkinson’s disease. Parkinsonism Relat Disord 11:95–99
Chen CJ, Chou LS (2010) Center of mass position relative to the ankle during walking: a clinically feasible detection method for gait imbalance. Gait Posture 31:391–393
Das P, McCollum G (1988) Invariant structure in locomotion. Neuroscience 25:1023–1034
Dennison AC, Noorigian JV, Robinson KM et al (2007) Falling in Parkinson disease: identifying and prioritizing risk factors in recurrent fallers. Am J Phys Med Rehabil 86:621–632
Fok P, Farrell M, McMeeken J, Kuo YL (2011) The effects of verbal instructions on gait in people with Parkinson’s disease: a systematic review of randomized and non-randomized trials. Clin Rehabil 25:396–407
Galletly R, Brauer SG (2005) Does the type of concurrent task affect preferred and cued gait in people with Parkinson’s disease? Aust J Physiother 51:175–180
Galna B, Murphy AT, Morris ME (2013) Obstacle crossing in Parkinson’s disease: mediolateral sway of the centre of mass during level-ground walking and obstacle crossing. Gait Posture. doi:10.1016/j.gaitpost.2013.1003.1024
Hausdorff JM, Balash J, Giladi N (2003) Effects of cognitive challenge on gait variability in patients with Parkinson’s disease. J Geriatr Psychiatry Neurol 16:53–58
Jones D, Rochester L, Birleson A et al (2008) Everyday walking with Parkinson’s disease: understanding personal challenges and strategies. Disabil Rehabil 30:1213–1221
Kang GA, Bronstein JM, Masterman DL, Redelings M, Crum JA, Ritz B (2005) Clinical characteristics in early Parkinson’s disease in a central California population-based study. Mov Disord 20:1133–1142
Kelly VE, Janke AA, Shumway-Cook A (2010) Effects of instructed focus and task difficulty on concurrent walking and cognitive task performance in healthy young adults. Exp Brain Res 207:65–73
Kelly VE, Eusterbrock AJ, Shumway-Cook A (2012) The effects of instructions on dual-task walking and cognitive task performance in people with Parkinson’s disease. Parkinsons Dis 2012:1–9
Kelly VE, Eusterbrock AJ, Shumway-Cook A (2013) Factors influencing dynamic prioritization during dual-task walking in healthy young adults. Gait Posture 37:131–134
Kerr GK, Worringham CJ, Cole MH, Lacherez PF, Wood JM, Silburn PA (2010) Predictors of future falls in Parkinson disease. Neurology 75:116–124
Lamont RM, Morris ME, Woollacott MH, Brauer SG (2012) Community walking in people with Parkinson’s disease. Parkinsons Dis 2012:856237
Lehman DA, Toole T, Lofald D, Hirsch MA (2005) Training with verbal instructional cues results in near-term improvement of gait in people with Parkinson’s disease. J Neurol Phys Therapy 29:2–8
Lohnes CA, Earhart GM (2011) The impact of attentional, auditory, and combined cues on walking during single and cognitive dual tasks in Parkinson disease. Gait Posture 33:478–483
Lord S, Galna B, Verghese J, Coleman S, Burn D, Rochester L (2013) Independent domains of gait in older adults and associated motor and nonmotor attributes: validation of a factor analysis approach. J Gerontol A Biol Sci Med Sci 68:820–827
Morris ME, Iansek R, Matyas TA, Summers JJ (1994) The pathogenesis of gait hypokinesia in Parkinson’s disease. Brain 117(Pt 5):1169–1181
Morris ME, Iansek R, Matyas TA, Summers JJ (1996) Stride length regulation in Parkinson’s disease. Normalization strategies and underlying mechanisms. Brain 119(Pt 2):551–568
Nasreddine ZS, Phillips NA, Bedirian V et al (2005) The Montreal cognitive assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53:695–699
Ojha HA, Kern RW, Lin CH, Winstein CJ (2009) Age affects the attentional demands of stair ambulation: evidence from a dual-task approach. Phys Ther 89:1080–1088
Patla A, Shumway-Cook A (1999) Dimensions of mobility: defining the complexity and difficulty associated with community mobility. J Aging Phys Act 7:7–19
Post B, Merkus MP, de Haan RJ, Speelman JD (2007) Prognostic factors for the progression of Parkinson’s disease: a systematic review. Mov Disord 22:1839–1851
Rahman S, Griffin HJ, Quinn NP, Jahanshahi M (2008) Quality of life in Parkinson’s disease: the relative importance of the symptoms. Mov Disord 23:1428–1434
Soh SE, Morris ME, McGinley JL (2011) Determinants of health-related quality of life in Parkinson’s disease: a systematic review. Parkinsonism Relat Disord 17:1–9
Wellmon R, Barr-Gillespie AE, Newton R, Ruchinskas RA, Stephens J (2013) The effects of aging on the attentional demands of walking toward and stepping up onto a curb. Gait Posture 38:198–202
Yogev-Seligmann G, Rotem-Galili Y, Dickstein R, Giladi N, Hausdorff JM (2012) Effects of explicit prioritization on dual task walking in patients with Parkinson’s disease. Gait Posture 35:641–646
Acknowledgments
Research reported in this publication was supported by the University of Washington Stolov Research Fund and the National Institute of Child Health and Human Development of the National Institutes of Health under award number K01HD052018. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The authors sincerely thank the participants for their involvement in this research. They thank the Washington State Parkinson Disease Registry for assistance with recruitment. The authors gratefully acknowledge R. Price for engineering support and L. Flexner for recording the stimuli. They thank S. Woldemariam, A. Gillespie, M. Durrant, M. Manges, A. Nova, N. Babitsky, K. Bermensolo for assistance with data collection. They gratefully acknowledge R. Archer and A. Eusterbrock for assistance with data collection and analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kelly, V.E., Shumway-Cook, A. The ability of people with Parkinson’s disease to modify dual-task performance in response to instructions during simple and complex walking tasks. Exp Brain Res 232, 263–271 (2014). https://doi.org/10.1007/s00221-013-3737-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-013-3737-4