Abstract
In order to reveal the mechanism of eye position coding, we measured the effect of eye position on assessing the position of a light viewed in a dark environment using a sound as a reference point, before and after adapting to prolonged periods of eccentric viewing (11° right or left). During testing, eye position was varied over ±22°. For each test position, a PEST procedure was used to align the perceived position of a light (with no visual reference points) and a fixed sound source. The perceived position of the light was veridical when looking straight ahead but large and consistent errors were found with eccentric viewing indicating that only about 77% of the eye eccentricity was taken into account, an eye position gain of 0.77, consistent with previous reports. The error was altered by prolonged eccentric viewing. There were two components to this effect: a direction bias and a gain change. The direction bias was symmetric: in a change rather like that induced by prism adaptation, the perceived straight ahead moved in the direction of the previous eccentric viewing. The gain change was asymmetric: after looking to the left, the gain of eye position increased to closer to one (0.77–0.81), in other words the subjects became more accurate. After looking right, the gain of eye position decreased (0.77–0.73): subjects consequently became less accurate. We model these systematic changes in terms of an asymmetric coding system.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Knowing where the eyes are in their orbits is central to establishing perception. Vision is only available in fine detail in the part of the image that falls on the fovea, so the eyes move around to foveate different points of interest. Knowledge of eye position may be necessary to integrate the resulting series of transient images. Equally importantly, knowing any visual direction relative to the head requires knowledge of eye position. The source of this eye position information is not the topic of this paper. It arises from some combination of efference copy and afferent sensory information (e.g., Bridgeman and Stark 1991). The question we address here is how eye position information is coded.
Several studies have reported the presence in macaque parietal cortex of cells tuned to particular eye positions (e.g., Mountcastle et al. 1975; Sakata et al. 1980; Andersen et al. 1990; Nakamura et al. 1999; Bremmer et al. 1999). In many cases, cells respond with a tonic discharge related to eye position even in darkness. Such behaviour distinguishes these cells from cells showing modulated sensory responses and suggests that they may be part of an eye position coding system that could subsequently be used to gain modulate cells elsewhere, including occipital cortex and other parietal areas. There are many reports that the magnitude of visual responses in occipital visual neurons with retinocentric receptive fields can be modified, sometimes strongly, by eye position (e.g., Galletti and Battaglini 1989; Trotter and Celebrini 1999; Rosenbluth and Allman 2002; Bremmer et al. 1997a, b). Occipital cells have been found to be affected by eye positions on both sides of straight-ahead. That is, each hemisphere appears to have access to eye positions in all directions, with no contralateral bias. This is important because, while the contralateral visual field is more completely represented in each hemisphere, all visual field locations need to be interpreted in the context of all eye positions.
Although the existence of eye-position neurons is well documented, the nature of the code for eye position is unknown. In order to investigate this code, we used a behavioural measure along with adaptation to eccentric eye positions. Adaptation is a tried and true psychophysical tool. A classic demonstration of how adaptation can reveal coding mechanisms is the tilt after effect (Campbell and Maffei 1970). After adaptation to a visual grating or bar of a particular orientation, the perceived orientation of slightly, but not greatly, different orientations is altered demonstrating a channel coding mechanism for orientation. This framework is supported by physiological evidence for habituation of responses of visual neurons during prolonged viewing of a stimulus at the preferred orientation (Maffei et al. 1973). In the case of parietal fixation neurons, evidence for such habituation is lacking. Over the 3s or so for which awake animals maintain eccentric fixation in the relevant experiments, the responses are often quite stable. But habituation may nonetheless occur over a longer time frame and, if so, perceptual errors of localization should occur. Thinking that eye position coding might also be by channels, we adapted to a constantly maintained eccentric eye position and looked for a distortion in the perceived visual direction of objects when, and only when, the eyes were close to the adapted direction. Such a distortion was not found. Instead, distortions of perceived object direction were induced by this adaptation technique for all eye positions during testing, in a complex pattern that was not consistent with a channel hypothesis.
We used an indirect method for measuring perceived eye position. Identifying the location of a light in space requires knowledge of eye position. So by getting subjects to judge the position of a light relative to an earth-fixed sound source while their eyes were held at various eccentricities, we were able to estimate the knowledge of eye position that the brain was using to deduce the spatial location of each light. We then attempted to alter the relationship between perceived and actual eye position by having subjects maintain an eccentric eye position and thus adapt or fatigue the neural mechanisms involved.
Methods
Participants
Eleven participants (five male, six female; age 25–55years) took part in these experiments. One (female) withdrew before completing the study, leaving ten. They had no known visual or oculomotor deficits apart from refractory errors. All experiments were approved by the York Ethics Board.
Visual display
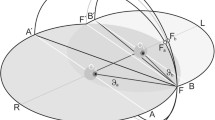
A schematic of the display is shown in Fig. 1. Visual stimuli were generated by an X/Y laser device (GSI Lumonics, model VM 500-T), controlled through the serial port of a PC. This projected a single light dot onto a white fabric-display screen, positioned 122cm in front of the subject. Apart from the dot, the visual field was in complete darkness.
Schematic of the stimulus arrangement. Two small spots were projected onto a screen from a laser source located above and behind the subject’s head. A speaker was positioned directly in front of the subject, hidden from view behind the screen. Subjects fixated the fixation point and judged whether the probe spot was to the left or right of the speaker
Auditory stimulus
The screen was transparent to sound. Behind the display screen, a small computer loudspeaker was positioned straight ahead of the subject. White noise stimuli were presented in 50-ms bursts at a decibel level of 67 dB SPL.
Procedure
Subjects sat on a chair in front of the screen in the dark and wore light-attenuating glasses that reduced light levels by 1.8 dB to remove any scattered light reflected from the laser. In trials in which the eye movements were recorded, they wore a helmet holding the Eye Scan cameras and calibration trials were performed prior to data collection.
To measure the influence of eye position on perceived straight ahead, participants were instructed to look at a point illuminated by a laser beam in order to control where they looked. The fixation light was present continuously and participants were instructed to fixate it at all times, making saccades to follow it when it moved. It moved only along the horizontal meridian. A second light appeared at a different point on the horizontal meridian and flashed two times, simultaneously with presentation of a noise burst. Subjects maintained fixation on the first point and judged whether the second light was to the left or right of the sound. They indicated their choice using the buttons of a mouse. There were 21 eye positions from 22° left to 22° right in 2.2° steps.
The horizontal position of the second light, the position of which was judged relative to the sound source, was controlled by a PEST staircase procedure (Taylor and Creelman 1967). The rules were as follows: on every reversal, the step size was reduced to half until a minimum step size was reached where it remained constant. If there were no reversals, the third step in the same direction caused a doubling of the step size following which each successive step in the same direction also doubled the step size until a reversal occurred. The minimum step size was 0.22°. Staircases were terminated after eight reversals. Separate, interleaved staircases were used for each eye position (42 staircases in all) and all trials were presented in a random order. For each staircase, the positions of the last two reversals were averaged to obtain the point where the locations of the light and sound were perceived as the same (the point of subject equality: PSE).
Experiments were of three kinds: control, adapting to eccentric viewing to the left and adapting to eccentric viewing to the right. These were conducted in separate sessions for each subject in a balanced design. Control sessions consisted of a series of trials as described above. During the adapt-left and adapt-right conditions, the fixation spot was first positioned 11° to either the left or right of straight ahead where it remained for 3 min, the adaptation period, during which time the participant maintained fixation. After this the experiment proceeded as in control sessions, except that after each three probe trials, an adaptation top-up period occurred in which the fixation dot resumed its adaptation position for a further 20s.
Adaptation sessions (left or right) lasted about 50 min, depending on the time taken for the PESTs to reach criterion. Control sessions lasted about 20 min. Each participant ran each session twice and the average result was taken (six sessions total per person).
Eye movement recording
Eye movements were recorded in eight subjects in all three conditions of a parallel session with five eye positions. Eye movements were recorded using an EyeScan monocular video-oculographic system. The participant’s head was stabilized using a bite bar. The system was calibrated using a manufacturer-provided procedure involving looking at a sequence of carefully positioned targets. Resolution was about 0.1°. Eye movements were monitored only to be sure that our procedures did not lead to systematic changes in the fixation of targets. Over the course of the experiment, eye position was maintained on the adapt target with a typical standard deviation of 0.3°. There was no systematic drift either in the eye fixation on the adapt stimulus or on the individual eye position control targets.
Results
The effect of horizontal eye position on comparing the location of a visual target with that of a sound
Figure 2a shows the effect of eye position on the estimation of the position of a light relative to a fixed sound. Each grey line represents one subject and the mean judgment is shown as a thick line. If eye position had no effect on the task, that is, if eye position were accurately taken into account, the function would be horizontal. However, there was a strong linear relationship (error/eye position: slope = 0.23; r 2 = 0.93) indicating systematic errors related to the eye position held while the judgments were made. For each degree of eye movement there was an apparent shift of straight ahead of about 0.23° in the direction of eye eccentricity. Thus, since error = actual − perceived eye position, the gain (perceived eye position/actual eye position) is given by (1 − slope), which is 0.77.
The effect of horizontal eye position on the perceived location of a light spot relative to an earth-fixed sound. Each point on each graph represents the horizontal location of a light that could not be distinguished from a speaker at position zero. This position is shown (ordinate) for each of the 21 eye positions (abscissa) for ten subjects. Individual subjects’ data are given as grey lines with the average curve shown as a thick black line. a Results with no adaptation, b after adapting to eccentric viewing, 11° right (indicated by solid vertical line), and c after adapting 11° left
It has been demonstrated that eye position has a small effect on perceived auditory location (Lewald 1997, 1998; Lewald and Ehrenstein 1996). This raises the question of whether the misperception we report may reflect an error of localization of the sound rather than the probe light. However, the reported effect of gaze on sound localization is small (1°–2°). Moreover, the shift in perceived sound location is in the opposite direction to the eye eccentricity, that is opposite to the effects shown here.
The effect of sustained horizontal eccentric viewing on comparing a visual target with a sound
Figure 2b, c shows the effect of adapting the eye position system by maintaining an eccentric eye position at 11° to the right and left, respectively. The light position that was matched with the auditory target was still linearly related to eye position but the function was shifted on the vertical axis (t = 1.95, P = 0.04, df = 9) by about 1.5° in the direction of previous eccentric viewing. Additionally the slope increased to 0.27 (gain = 0.73; r 2 = 0.97) after adapting right and decreased to 0.19 (gain = 0.81; r 2 = 0.94) after adapting left (t = 2.6, P = 0.03, df = 9). Table 1 shows, for all three conditions, the slope of the function for each participant and the mean slopes quoted above. A repeated measures analysis of variance (ANOVA) showed that there was a highly significant main effect of eccentricity on eye position [F (19,171) = 26.6, P < 0.0001] and using linear contrasts showed that eccentricity had a significant linear effect [F(1,9) = 52.6; P < 0.001]. There was a significant interaction between eye position and adaptation condition [F(1,9) = 6.7, P = 0.029], showing that the slopes were significantly different.
Figure 3a shows the difference between the settings made before and after adaptation (post-pre). Each subject’s data were analysed separately and the results averaged to produce these plots. The two functions (adapt right and adapt left) deviate from true straight-ahead (y = 0) in opposite directions and have opposite slopes. Intriguingly, the largest shift in perceived eye position (post-pre) occurred for test eye positions to the right, irrespective of whether adaptation was to the right or left. This is completely unexpected and is inconsistent with a symmetrical system.
The effect of adaptation on the relationship between eye position and the perceived position of a light compared to a sound is made clear by subtracting the pre-adapt data from the adapted data (a). Filled circles show results after adapting right, open circles after adapting left. The difference between the two functions may be decomposed into two components: a vertical displacement in opposite directions from zero change and a difference in slope, which is positive in one case and negative in the other. The slopes have been subtracted from the data in b, and the vertical displacement has been subtracted in c
To understand the results, it is convenient to decompose the functions in Fig. 3a into two separate effects of adaptation. Firstly, the curves were shifted in the direction of the adaptation: after looking to the left, the curve was shifted in the leftward direction. That is, a light regarded as matching the location of a hidden speaker was displaced to the left after adaptation for all eye positions was tested. This component is isolated in Fig. 3b, in which the slopes of the functions have been subtracted from the post-pre data. The average shift was about 14% of the adapting eccentricity. In Fig. 3c, this shift has been removed by shifting the functions vertically such that both curves go through y = 0 for the straight-ahead test gaze position (x = 0). This clearly illustrates the second effect of adaptation which is that the slopes of the functions have changed in opposite directions.
Discussion
This paper has demonstrated a number of phenomena that need to be taken into account when considering how the brain encodes eye position. Firstly, we have confirmed other studies (McLaughlin and Webster 1967; Park 1969) that have reported that the perceived direction of straight ahead is influenced by the instantaneous direction of gaze. Our control data (Fig. 2a) indicate a shift of 0.23° for every degree of eye eccentricity. This is compatible with the well-known phenomenon that eye position is not accurately taken into account in perceptual tasks (e.g., Bridgeman and Stark 1991). A related phenomenon is that when the speed of a moving target is assessed while it is being tracked (so there is no useful velocity information in the retinal image), the resulting perceived speed is about 70% of the speed estimated when the same motion is observed with the eyes still (the Aubert-Fleischl effect: Fleischl 1882; Aubert 1886, 1887). It seems probable that retinal and extraretinal information are combined in assessing eye position (Freeman and Banks 1998; Blouin et al. 1995). Here we are looking at only the extraretinal contribution.
Maintaining eccentric gaze should cause fatigue in cells coding that particular gaze and might provide a clue concerning the coding mechanism (see Harris 1997 for discussion). We did obtain adaptation effects, indicating that some changes had occurred in the brain as a result of maintaining an eccentric gaze. However, these effects did not seem to be tuned to a particular gaze angle. We found a complex pattern of results that may be thought of as the sum of two simpler effects of adaptation, neither of which was dependent on the test eye position. Firstly, the perceived location of all objects was shifted in the direction of the adapted location. We refer to this symmetric component as a “direction bias” effect. Secondly, there was an asymmetric change in the gain of the function relating perceived straight-ahead (and hence eye position) to true eye position. We refer to this as the “gain change” effect. The fact that effects are seen even when the test eye position was very different from the adapted eye position suggests that the eye position system cannot be modelled as a set of narrowly tuned eye-position channels.
Two components of the eye position code
The finding of two components is reminiscent of the fact that the direction and amplitude of saccadic eye movements seem to be processed separately (Becker and Jurgens 1979). Alteration or recalibration of the direction system (e.g., by prisms) results in a consistent bias added to the direction (McLaughlin and Webster 1967; Templeton et al. 1966; Held 1965). This is comparable to our direction bias effect. Changes to saccadic amplitude (e.g., by jumping the target after a saccade to it has commenced) result in a multiplicative gain change (McLaughlin 1967; McFadden and Wallman 2001) comparable to our gain change effect. It might be convenient if the perceptual coding system matched the motor system. We will therefore describe these effects in terms of direction and amplitude, with the implication that these might reflect two quite independent coding systems. This model is illustrated in Fig. 4. In order to use these parameters for the interpretation of visual space, both direction and amplitude must of course eventually be put together, as they are for the motor control of eye position in the oculomotor nuclei (Fuchs and Luschei 1970).
The direction bias effect
As a result of maintaining an eccentric eye position, the entire function relating the perceived position of light to eye position was shifted by about 14% of the eccentricity of the gaze held. Since we adapted at only one eccentricity on each side, we cannot verify that this is a constant fraction of the gaze eccentricity, but it seems plausible. This displacement is very reminiscent of the change of perceived straight ahead reported after looking through prisms (von Helmholtz 1911; Kohler 1951). When initially donning prisms that displace the visual world to the left, objects to the right of straight ahead are shifted to appear straight ahead. Reaching forwards then moves the hand to objects that appear to be to the left of straight ahead. After a period of wearing such prisms, the perceived straight ahead is shifted to the left, thus aligning the vision with the felt position of objects (von Helmholtz 1911). There is also a displacement of the eyeballs in their sockets after prism adaptation to a new “resting position” which might account for some of that adaptive shift (Kalil and Freedman 1966; McLaughlin and Webster 1967).
The gain change effect
The more unexpected finding was the change in gain (slopes in Figs. 2, 3) induced by adaptation to eccentric gaze. Although the changes appear nicely symmetrical in Fig. 3c, the symmetry is not straightforward. After adapting to a rightward eye position, the post-pre function (Fig. 3a) has a positive slope, that is, the gain of the perceived eye eccentricity was decreased by adapting to a rightward gaze (from 0.77 to 0.73), which means that the error in judging visual direction was increased. In contrast, after adapting to leftward gaze, the post-pre function has a negative slope, that is the gain of perceived eye position was increased (from 0.77 to 0.81) which decreases the error in judging visual direction.
The gain of perceived eye position will determine the perceived amplitude of a change in eye position. When the eye moves through say 10°, a gain of 0.77 suggests that the amplitude will be perceived as less than it really is, around 7.7°. This perceived amplitude seems to be differentially affected by our adaptation procedure, being larger and more accurate after maintaining fixation to the left and smaller and less accurate after maintaining fixation to the right.
Vindras and Viviani (2002) were able to adjust the gain of a visuomotor task such that the modification was transferred to other movements of different sizes. However, in their case the modification was driven by errors introduced by the experimenters. Here there is no obvious reason and no driving error to alter the gain.
The nature of eye position coding
A model of the coding of eye position should be able to explain both the direction bias and the gain change effects that we report. It is not difficult to explain the symmetrical bias change seen following adaptation: this can be seen as a recalibration of the system, such that perceived straight ahead gradually moves towards the current eye position. Since the average eye position corresponds to straight ahead, if this average is shifted by either maintained eccentric viewing, prism exposure or some change in physiological conditions, the straight ahead shifts correspondingly. It is much more difficult to explain the changes in gain. We do not present a full quantitative model of eye position coding but we discuss below some qualitative ideas for explaining the change in gain, based on a population code.
Suppose we have many “fixation neurons”, perhaps in posterior parietal cortex. Each neuron gives a tonic discharge all the time, which is proportional to eye position (Mountcastle et al. 1975). In our proposed scheme, the perceived horizontal eye position is determined by the total summed response of this population. To explain our results, we need to make two assumptions. The first is that there is a right–left asymmetry, either in the number of such neurons or in their responses, as illustrated in Fig. 5a. If, for example, more neurons were active during rightward gaze than during leftward gaze, the total summed activity in the population would be proportional to eye position, as shown in Fig. 5a. The second assumption is that there is mutual inhibition between the left and right pools of neurons, which enhances the gain (sensitivity) of the system.
Neural basis of our population coding of eye position model. a The solid line shows perceived eye eccentricity (right ordinate) as a function of actual eye position and reflects the observed underestimation (perceived eccentricity is 77% of true eccentricity). Also shown (dotted line) is the function (y = x) that would pertain in the case of veridical perception. Positive eccentricities denote rightward gaze, negative indicate leftward. In the model, perceived eye position maps directly onto the total response in a population of eye-position neurons (left ordinate). b Physiologically plausible composition of the model. The neuron population in question is parietal eye-position neurons. These fall into two groups, those with a horizontal component that increases left–right and those that increase right–left. The sum of the horizontal components of the gain planes of all neurons of each type is shown (dotted and dashed line for rightward and leftward populations, respectively). Neurons preferring rightward eye positions are either more numerous or give stronger responses than leftward neurons. The sum of the two profiles gives the population response (solid line)
Figure 6 shows the effect of gaze adaptation on the function shown in Fig. 5a. While gazing to the left, the leftward-gaze cells become less active (adapted) following a period of strong, tonic discharge. The strength of the suppressive signal they apply to the competing rightward-gaze cells is then reduced commensurately. Consequently, the rightward-gaze cells become more active. The slope of the function relating population activity to eye position therefore becomes steeper (dashed line in Fig. 6a) and hence sensitivity to eye position change increases. After adapting to the right, the rightward cells become less active by adaptation and the leftward cells become more active through release from inhibition, with a corresponding reduction of the slope (shown in Fig. 6b). This scheme thus fully explains the change in slope seen in our experiments. However, it addresses only the coding of the horizontal component of eye position; an additional population code would be required to encode the vertical component and it is not known whether this would show an up-down asymmetry. We have attempted to repeat our experiments in the vertical dimension, but vertical sound localization was found not to be sufficiently accurate to yield consistent results and so another measurement method will be required.
Effect of adaptation on the model of perceived eye position. a Effects of adaptation to leftward gaze. Neurons responsive to leftward gaze become adapted, leading to a reduced response during subsequent leftward gaze. Neurons sensitive to rightward gaze are not adapted and experience release from inhibition from adapted leftward neurons, giving an increased response during rightward gaze. Thus, the slope of the function (perceived eye position gain) is increased. b Effects of adaptation to rightward gaze. The same processes result in a decreased slope
A full model would include an alterable set point for overall directional left/right bias control such as has been postulated since Helmholtz to explain the effect of prisms and eccentric gaze. Thus, we suggest the two elements (direction and amplitude) are processed separately (Fig. 4).
Physiological implementation
The gain fields of parietal eye-position neurons can often be approximated by a plane (i.e., the response is maximal along some line in 2D eye-position space and falls off along any orthogonal line). This is the case in area 7a (e.g. Andersen et al. 1990; Sakata et al. 1980; Bremmer et al. 1997a, b) and also in LIP (Andersen et al. 1990; Bremmer et al. 1997a, b) and VIP (Bremmer et al. 1999). Thus, it is not the case that these neurons have a single preferred eye position and a sensitivity profile that falls off in all directions from that position. Instead, the peak response tends to occur for the most eccentric gaze directions (the edge of the plane), whether right, left, up, down or intermediate (depending on the orientation of the plane).
The crucial assumption of our scheme is that rightward gaze yields a stronger population response than leftward gaze among fixation neurons. Compelling physiological evidence for this is lacking, but such evidence as exists points in that direction rather than the opposite. To our knowledge, only Bremmer et al. (1997a, b) have published relevant mean, or population, responses. They employed two measures to study eye position effects in areas 7a and LIP: preferred location of fixation in darkness and preferred direction of pursuit. The two were highly correlated, indicating common eye position influences across tasks. The distribution of response-plane orientations was not significantly different from uniform, lending no support to the hypothesis that rightward-gaze neurons are more numerous than leftward-gaze neurons. However, the mean response plane for the population was not flat, as would be expected for a uniform distribution. For pursuit, the mean response plane was tilted such that the mean response was about 10.7 spikes/s at 20° right (and 0°, or central, vertically) but only 7.0 spikes/s at 20° left (their Fig. 8b; n = 41). LIP showed very little tilt. A tilt in the mean response plane is not necessarily inconsistent with a homogenous distribution of individual planes; it is also possible that neurons coding right and left are balanced in terms of numbers of neurons but that those encoding rightward gaze give stronger responses. For fixation in darkness there was, discouragingly, little apparent tilt for area 7a (their Fig. 13), but this time LIP showed a modest tilt in the predicted direction. In a companion paper (Bremmer et al. 1997a, b), the same group shows corresponding results for MST. The tilt is small, but again it is such that rightward gaze gives a bigger population response than leftward gaze. These findings, based on a single study that involved samples of around 100 neurons per area from two brains, leave us without clear supporting evidence but with enough positive indications to show that our assumption of an asymmetric distribution of activity is at least plausible.
Alternative explanations
We have chosen to interpret our findings in terms of possible underlying physiological mechanisms. However, in estimating perceived eye position, we rely on subjects’ reports of the perceived relative position of a light and a sound. As in many behavioural experiments, it is possible that these reports could be influenced by response biases (see Poulton 1989 for a review). In principle, our results could be entirely explained in this way, if the bias were of the right kind and strong enough. To explain the results obtained with no adaptation (Fig. 2a) in this way, subjects would have to be biased towards reporting that the light was on the right of the sound when looking right, and left of the sound when looking left. This cannot be ruled out, although there is no particular reason to suspect such a gaze-contingent response bias. To explain the results obtained with adaptation (Fig. 2b, c), this initially symmetrical bias would have to change in an asymmetric way, lessening after adapting left and strengthening after adapting right. This seems unlikely.
Finally, it may be that the asymmetry we have demonstrated cannot be mapped directly onto physiological properties of the macaque brain, but instead arises from hemispheric specialization of processing spatial location and attention, for which the evidence is much stronger in humans than in macaques. For example, if left space, including leftward gaze, were encoded preferentially, this might give rise to asymmetries following adaptation.
Conclusions
We have shown, using robust psychophysical measurements, that the perceived position of the eyes over a range of eye positions has systematic errors, and that those errors can be altered as a result of adaptation to eccentric gaze. We posit that eye position is encoded in the brain as a population summed code and provide some quantitative ideas that could form the basis of a model of this process.
References
Andersen RA, Bracewell RM, Barash S, Gnadt JW, Fogassi L (1990) Eye position effects on visual, memory, and saccade-related activity in areas LIP and 7A of Macaque. J Neurosci 10:1176–1196
Aubert H (1886) Die Bewegungsempfindung. Pflügers Archiv Eur J Physiol 39:347–370
Aubert H (1887) Die Bewegungsempfindung. Zweiter Mitteilung. Pflügers Archiv Eur J Physiol 40:459–480
Becker W, Jurgens R (1979) An analysis of the saccadic eye movement system by means of double step stimuli. Vision Res 19:967–983
Blouin J, Gauthier GM, Vercher JL (1995) Internal representation of gaze direction with and without retinal inputs in man. Neurosci Lett 183:187–189
Bremmer F, Distler C, Hoffman KP (1997a) Eye position effects in monkey cortex. 2. Pursuit-related and fixation-related activity in posterior parietal areas LIP and 7A. J Neurophysiol 77:962–977
Bremmer F, Ilg UJ, Distler TC, Hoffmann KP (1997b) Eye position effects in monkey cortex. 1. Visual and pursuit-related activity in extrastriate areas Mt and MST. J Neurophysiol 77:944–961
Bremmer F, Graf W, Ben Hamed S, Duhamel JR (1999) Eye position encoding in the macaque ventral intraparietal area (VIP). Neuroreport 10:873–878
Bridgeman B, Stark L (1991) Ocular proprioception and efference copy in registering visual direction. Vision Res 31:1903–1913
Campbell FW, Maffei L (1970) Electrophysiological evidence for the existence of orientation and size detectors in the human visual system. J Physiol (Lond.) 207:635–652
Fleischl EV (1882) Physiologisch optische Notizen. Sitzungsbez Akad Wissensch 3:7–25
Freeman TC, Banks MS (1998) Perceived head-centric speed is affected by both extra-retinal and retinal errors. Vision Res 38:941–945
Fuchs AF, Luschei ES (1970) Firing patterns of abducens neurons of alert monkeys in relationship to horizontal eye movement. J Neurophysiol 33:382–392
Galletti C, Battaglini PP (1989) Gaze-dependent visual neurons in area-V3A of monkey prestriate cortex. J Neurosci 9:1112–1125
Harris LR (1997) The coding of self motion. In: Harris LR, Jenkin MR (eds) Computational and psychophysical mechanisms of visual coding. Cambridge University Press, Cambridge, pp. 157–183
Held R (1965) Plasticity in sensory-motor systems. Sci Am 213:84–94
Kalil RE, Freedman J (1966) Persistence of ocular rotation following compensation for displaced vision. Percept Mot Skills 22:135–139
Kohler W (1951) Ueber Aufbau und Wandlungen der Wahrnehmungswelt. Sitzungsber Oesterreichishe Akad Wiss 227:1–118
Lewald J (1997) Eye-position effects in directional hearing. Behav Brain Res 87:35–48
Lewald J (1998) The effect of gaze eccentricity on perceived sound direction and its relation to visual localization. Hear Res 115:206–216
Lewald J, Ehrenstein WH (1996) Auditory-visual shift in localization depending on gaze direction. Neuroreport 7:1929–1932
Maffei L, Fiorentini A, Bisti S (1973) Neural correlate of perceptual adaptation to gratings. Science 182:1036–1038
McFadden S, Wallman J (2001) Shifts of attention and saccades are very similar. Are they causally linked? In: Jenkin M, Harris LR (eds) Vision and attention. Springer, New York, pp. 19–39
McLaughlin S (1967) Parametric adjustment in saccadic eye movements. Percept Psychophys 2:359–362
McLaughlin SC, Webster RG (1967) Changes in straight-ahead eye position during adaptation to wedge prisms. Percept Psychophys 2:37–43
Mountcastle VB, Lynch JC, Georgopoulos A, Sakata H, Acuna C (1975) Posterior parietal association cortex of the monkey: command functions for operations within extrapersonal space. J Neurophysiol 38:871–908
Nakamura K, Chung HH, Graziano MSA, Gross CG (1999) Dynamic representation of eye position in the parietooccipital sulcus. J Neurosci 81:2374–2385
Park JN (1969) Displacement of apparent straight ahead as an aftereffect of deviation of the eyes from normal position. Percept Mot Skills 28:591–597
Poulton EC (1989) Bias in quantifying judgements. Erlbaum, Hove
Rosenbluth D, Allman JM (2002) The effect of gaze angle and fixation distance on the responses of neurons in V1, V2, and V4. Neuron 33:143–149
Sakata H, Shibutani H, Kawano K (1980) Spatial properties of visual fixation neurons in posterior parietal association cortex of the monkey. J Neurophysiol 43:1654–1672
Taylor MM, Creelman CD (1967) PEST: efficient estimates on probability functions. J Acoust Soc Am 41:782–787
Templeton WB, Howard IP, Lowman AE (1966) Passively generated adaptation to prismatic distortion. Percept Mot Skills 22:140–142
Trotter Y, Celebrini S (1999) Gaze direction controls response gain in primary visual cortex neurons. Nature 398:239–242
Vindras P, Viviani P (2002) Altering the visuomotor gain. Evidence that motor plans deal with vector quantities. Exp Brain Res 147:280–295
Von Helmholtz H (1911) Handbuch der physiologischen Optik, 3rd edn. Voss, Leipzig
Acknowledgments
LRH was supported by the Natural Sciences and Engineering Research Council of Canada. Many of these experiments were run by Michael Barnett-Cowan and Jeff Sanderson who were graduate students in LRH’s lab.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Harris, L.R., Smith, A.T. The coding of perceived eye position. Exp Brain Res 187, 429–437 (2008). https://doi.org/10.1007/s00221-008-1313-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-008-1313-0