Abstract
Both vestibular and neck proprioceptive inputs contribute towards maintaining a walking trajectory. We investigated how aging alters neck proprioceptive and vestibular interaction for preserving equilibrium and spatial orientation during locomotion. Young and healthy elderly were exposed to two sensory manipulations as they walked, eyes closed, to a target located straight ahead: (1) right side dorsal neck muscle vibration (Vib), and (2) Vib + transmastoidal galvanic vestibular stimulation (Vib + GVS). The maximum path deviation, average frontal centre of mass velocity and average trunk roll were evaluated. Trunk yaw rotation was computed at every metre of the path. We observed that directional responses to neck muscle stimulation were very sensitive to the reference frame generated by vestibular information. The attenuation of path deviation in older adults can be attributed to a reduced sensitivity of the neck proprioceptive system rather than the vestibular system.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
When a visual reference is not available, both vestibular and neck muscle receptor inputs increase their contribution to postural control and to planning and maintaining a walking orientation for efficient progression (Bove et al. 2001; Fitzpatrick et al. 1999). Tonic vibration applied unilaterally to the dorsal neck muscles, during straight ahead locomotion in the absence of visual cues, causes human subjects to veer in a direction opposite to that of the vibration. This directional response has been attributed to an altered perception of egocentric space and the direct influence of the neck proprioceptive input on spinal neural circuits, possibly through reticulospinal or vestibulospinal pathways, for postural control to respond to this altered body centred reference frame (Courtine et al. 2003; Bove et al. 2001). Similarly, the application of bipolar galvanic vestibular stimulation (GVS), which creates an imbalance in the vestibular input, also causes direction-specific deviations (towards the anodal electrode) from the intended path. Altered spatial orientation as well as a direction-specific postural instability, generated by vestibular imbalance, has been postulated to contribute to this effect (Fitzpatrick et al. 1999).
It is proposed that the central nervous system (CNS) deciphers and integrates muscle proprioceptive information with respect to a reference frame generated by the vestibular input, for the maintenance of vertical posture (Popov et al. 1999; Hlavacka et al. 1996). In particular, postural responses to unilateral dorsal neck muscle vibration during standing are recalibrated with respect to the status of the vestibular input (Popov et al. 1996). However, task-specific modulation of the gain of the proprioceptive system (Courtine et al. 2003), as well as posture-dependent modification of responses to vestibular perturbation (Britton et al. 1993), indicates that the sensory-motor processing by the CNS in standing may differ from that during a dynamic task such as locomotion.
Age-related deterioration in the proprioceptive and vestibular systems (Liu et al. 2005; Lopez et al. 1997; Bergstrom 1973) and deficits in central integrative mechanisms (Hay et al. 1996) may further modify sensory integration strategies in older adults. Strupp et al. (1999) have proposed that age-related deterioration of the vestibular system may, in fact, increase the gain of the neck proprioceptive system, leading to larger spatial errors in responses to neck muscle vibration in older adults. However, they investigated the perception of spatial orientation in a sitting position and did not record any postural responses. Quoniam et al. (1995) have shown reduced postural responses (position, velocity and acceleration) in older adults, as compared to young adults, to lower limb muscle vibration in standing. How neck proprioceptive–vestibular interaction during locomotion may be altered by the aging process is not known. Therefore, the objective of the present study was to compare postural and spatial orientation responses to unilateral dorsal neck muscle vibration during locomotion, under normal and altered vestibular conditions, in the absence of a visual input, in young and healthy active older adults.
Materials and methods
Participants
Seven young (age 21.57±4.03, three females, four males) and seven elderly participants (age 70.00±6.22, three females, four males) participated after signing an informed consent form approved by the University of Waterloo, Office of Research Ethics, in confirmation with the standards established by the Declaration of Helsinki. None of them reported vestibular, neuromuscular deficits or cervical neck syndrome. The older adults were independent, healthy and active community dwellers.
Procedures
Task
The participants were asked to walk (stepping first with the right foot) to a previously seen target, located 6.5 m straight ahead, with their eyes closed. In the beginning of each trial, the subjects were allowed to see the target to reinforce the straight ahead location of the target. After completion of a trial, the participants were guided back to the starting position, with their eyes closed, along a random trajectory by a spotter, to avoid knowledge of result.
During the initial three walking trials, tonic vibration stimulation was applied unilaterally to participants’ right dorsal neck muscles, using a custom-made mechanical vibrator, at a frequency of 85 Hz with amplitude of 2 mm (Vib condition: control condition). The vibration was initiated 3 s prior to the signal for commencing walking and was maintained throughout the walking trial. On subsequent trials, transmastoidal bipolar GVS (Grass Medical Instruments, MA, USA) was simultaneously applied to provide a concomitant direction-specific perturbation of the vestibular system. The anode was placed on the left mastoid for congruent vestibular information (Vib + L condition) and on the right mastoid for in-conflict information (Vib + R condition). The threshold stimulation intensity for GVS was calculated by slowly increasing the intensity in a stepwise manner (a step of 0.05 mA) to a level where the participants demonstrated a visible postural sway in the anodal direction (Bent et al. 2000). In addition, the onset of cutaneous (pin-prick) sensation was monitored. Two experimental intensities were computed for each participant as the second (2t) and fourth (4t) multiple of the threshold, to provide an insight into the scaling effect of vestibular perturbation. Thus, the task was performed in the following five experimental sensory conditions: Vib, Vib + 2tL, Vib + 4tL, Vib + 2tR and Vib + 4tR. A total of three trials were performed in a block in each condition and, except for the Vib condition, the conditions were presented randomly. The participants also performed three walking trials with eyes closed (no stimulation). Ten infrared light emitting diodes (IREDs) were attached to the participant’s body on the following anatomical landmarks: the acromium processes, the spine of the 12th thoracic vertebra, above the ears, the occiput, the spines of the 7th cervical and 2nd sacral vertebrae, and the left and right heels. The IREDs were tracked by three OPTOTRAK camera banks (Northern Digital, Waterloo, ON, Canada).
Outcome measures
The trunk IRED coordinates were used to compute the trunk centre of mass (CoM) location during walking, the profile of which represented the walking trajectory. Global performance was measured as the maximum deviation towards the left (MaxDev) from the intended straight path and the average CoM velocity (CoMVel) in the medio-lateral (M-L) direction. The average position of the trunk in the frontal plane was evaluated by measuring the average roll angle (TRAvg). Trunk orientation pattern in the horizontal plane was assessed by computing trunk yaw angles (TYaw) at every 1 m distance from the starting position up to 5 m (Ym1, Ym2, Ym3, Ym4 and Ym5). Walking velocity (Vel) was computed for each trial.
Data analysis
Data were analyzed using a two-way mix factor ANOVA (condition × age) for MaxDev, CoMVel, TRAvg and Vel, and a three-way mix factor ANOVA (condition × location × age) for TYaw. Data were further analyzed if a significant interaction was found. Backward deletion multiple regression assessed the comparative weighting of the average CoM velocity and trunk kinematic parameters in predicting the maximum path deviation. A statistical program SPSS version 12 (SPSS Inc., San Rafael, CA, USA) was used for data analysis. Statistical significance was set at α=0.05 and post hoc analysis was performed if a significant main effect or interaction effect was observed. The data are presented as means and standard error.
Results
The threshold GVS intensity did not differ between the groups (F (1,12)=1.09, P=0.317) and ranged from 0.2 to 0.3 mA. The walking velocity was not affected by the sensory conditions (F (4,48)=1.93, P=0.167) and was not different in the young and older adults (F (1,12)=1.68, P=0.219) (young 75.99 cm/s, old 72.22 cm/s).
Initially, to rule out the differential effect of any a priori systematic bias in the two age groups on the walking trajectory, MaxDev was compared during walking with eyes closed (EC). A one-way ANOVA showed that the groups were not different in the EC condition (F (1,12)=0.122, P=0.733) (Y 13.17 cm, O 14.99 cm). Since the groups were not different in the baseline condition (EC), for all subsequent analysis the Vib condition was used as the control condition.
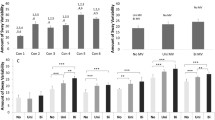
MaxDev was significantly affected by the sensory manipulation conditions in both groups (F (4,48)=23.42, P=0.001) (Fig. 1a, b). However, the main effect of age showed that the deviations of the older group were significantly smaller than the young group (F (1,12)=5.12, P=0.04) (Fig. 2a). All the participants veered to the left when unilateral dorsal neck muscle vibration was applied on the right side (Vib). The subjects deviated farther to left when the GVS was simultaneously applied with the anode on the left mastoid process. Conversely, the leftward deviation was significantly attenuated when the anode was placed on the right mastoid process. Further, multiple comparisons demonstrated that path deviations were significantly different between Vib + 4tL and Vib + 2tL (P=0.001), as well as between Vib + 2tR and Vib + 4tR (P=0.018). When the GVS-induced path deviation was evaluated by subtracting the vibration-induced deviation from the dual stimulation conditions, no differences were found between the groups (F (1,12)=0.45, P=0.51). All the four conditions were statistically different from each other (Fig. 2b).
A bird’s eye view of average locomotor trajectories in the young (a) and in the elderly (b) in five experimental conditions. Negative values indicate leftward deviation. Vib: Vibration only; Vib + 2tL: Vibration + GVS with two times threshold, anode on the left mastoid; Vib + 4tL: Vibration + GVS with four times threshold, anode on the left mastoid; Vib + 2tR: Vibration + GVS with two times threshold, anode on the right mastoid; Vib + 4tR: Vibration + GVS with four times threshold, anode on the right mastoid. Black plot indicates EC condition
a The average (mean ± SEM) values of maximum leftward deviation from the intended path. The differences in the path deviation between Vib and Vib + 4tL, Vib + 2tR and Vib + 4tR were statistically significant. Negative values indicate deviation to the left. Black bars represent the young group and grey bars represent the older group. b The mean values (mean ± SEM) of absolute GVS-induced change in the maximum path deviation. All the conditions were statistically different from each other. Negative values indicate an increase in path deviation towards the left and positive values indicate reduction in path deviation
The sensory manipulations significantly affected CoMVel in the M-L direction (F (4,48)=15.73, P=0.001) (Fig. 3). The negative values indicate that the CoM moved faster to the left side with neck vibration, which was further augmented when the GVS anode was placed on the left side. With the anode on the right side, the leftward CoMVel reduced significantly at a 2t intensity. At a 4t intensity, the direction of the velocity reversed and the CoM moved faster to the right side, as shown by the positive values. The trunk velocity of the young group was considerably, although not statistically, higher (F (1,12)=2.42, P=0.14).
The TRAvg and TYaw did not differ between the groups. Therefore, the data were pooled for further analysis. The trunk roll angle was significantly influenced by sensory manipulation (F (4,52)=23.99, P=0.001) (Fig. 4a). The average trunk roll was towards the left in the Vib condition. The angular displacement to the left was increased with the left anodal GVS and attenuated with the anode on the right. In the Vib + 4tR condition, the trunk tilted slightly to the right. A significant interaction was found between the condition and the spatial location for the TYaw (F (16,208)=3.27, P=0.001) (Fig. 4b). One-way ANOVAs with multiple comparisons, conducted separately for each spatial location, showed that the yaw angles in only the Vib + 4tR condition were significantly more (to the right) at Ym3 (F (4,52)=2.78, P=0.03), Ym4 (F (4,52)=3.55, P=0.01) and Ym5 (F (4,52)=4.01, P=0.007).
a Average (mean ± SEM) trunk position in the frontal plane during walking. The differences in average trunk position between Vib and Vib + 4tL, Vib + 2tR and Vib + 4tR were statistically significant. Positive values indicate trunk tilt to the left. b Mean (mean ± SEM) values of trunk rotation in five conditions at 1 m (Ym1), 2 m (Ym2), 3 m (Ym3), 4 m (Ym4) and 5 m (Ym5). Ym3, Ym4 and Ym5 were statistically different in Vib + 4tR condition. Positive values indicate trunk rotation to the left
A multiple regression analysis (backward deletion) was performed separately for the young and the older groups, with MaxDev as a criterion variable and CoMVel, TRAvg and Ym3 as predictor variables. Only the regression coefficient of CoMVel was found to be significantly greater than zero in both the groups (young group y = −169.64 + 4.17x, older group y = −266.47 + 5.14x) (Fig. 5). The CoM velocity in the M-L direction was a strong predictor of the magnitude of the path deviation; it explained 76 and 87% variance in path deviation in the young and the elderly, respectively.
Discussion
In the absence of a visual reference, neck proprioceptive and vestibular system inputs are integrated for an internal representation of postural and spatial orientation. Psychophysical evidence has shown the interactive effects of these two systems in a sitting position for the perception of a straight ahead direction (Karnath 1994) and for the perception of body movement (Schweigart et al. 2002). In the present study, the path trajectory while walking to a previously seen target located straight ahead, was significantly influenced by the experimental manipulation of the afferent information coming from these sensory channels. Similar to previous findings (Courtine et al. 2003; Bove et al. 2001), all subjects deviated to the left from their intended straight ahead path, with right-sided dorsal neck muscle vibration. Furthermore, congruent and in-conflict vestibular information had systematic additive and negating effects on the path deviation, respectively. For the first time, these results demonstrate that the neck proprioceptive and vestibular signals are linearly integrated during walking for maintaining path trajectory. The anatomical and physiological convergence of these two sensory channels at various levels (e.g. at the level of vestibular nuclei (Gdowski and McCrea 2000) or at the cortical level (Bottini et al. 2001)) may contribute to this effect. Additionally, path deviations during dual stimulation were scaled as a function of the magnitude of the vestibular perturbation in both groups. These results also suggest that the neck proprioceptive and vestibular input integration during walking, is very sensitive to the available vestibular input. It would be interesting to know if similar sensitivity is demonstrated to the neck muscle spindle output by manipulating the frequency of vibration.
The present results suggest that the central integrative mechanism for neck proprioceptive and vestibular inputs, for maintaining path trajectory during locomotion, was preserved in the older participants. However, the overall path deviations were attenuated in the older group, irrespective of the sensory condition. When the absolute GVS-induced modification in path deviations was compared, the effect of age disappeared, implying that the age-related reduction in path deviation primarily originated from a decline in the response to neck muscle vibration. Evidence from animal studies has shown age-related atrophic structural changes of the muscle spindle complex leading to reduced afferent information and dynamic sensitivity (Miwa et al. 1995). A human study that investigated the effects of aging, on vibration induced postural responses in the standing posture, has revealed attenuation of kinetic and kinematic responses to vibration-induced leg proprioceptive information in the elderly (Quoniam et al. 1995). Therefore, it is possible that both peripheral degeneration and central integrative factors contributed to reduced sensitivity to neck muscle vibration in the older adults.
Considering age-related morphological changes in various components of the vestibular system (Lopez et al. 1997; Bergstrom 1973) and current evidence from psychophysical studies (Schweigart et al. 2002; Strupp et al. 1999), we expected reduced responses to the GVS during locomotion in older adults. However, the subjects in these psychophysical studies were in a seated position. Task differences as well as reduced sensory-motor demands of postural control in sitting, may have contributed to these differences. A relative increase in vestibular gain in the absence of visual input (Fitzpatrick et al. 1999) may be higher in older adults for the control of equilibrium, considering the dynamic nature of the locomotor task. On the other hand, it is also speculated that age-related degeneration in cerebellar components, particularly Purkinje cells (Torvik et al. 1986), may reduce the inhibitory influence on vestibulospinal tract and, thus preserve the GVS-induced postural responses (Welgampola and Colebatch 2002) in older adults.
It is proposed that the head acceleration information from the vestibular system is incorporated to maintain lateral stability during locomotion. When vision is removed, frontal plane instability can potentially result in deviations from the planned trajectory in labyrinthine deficient subjects (Glasauer et al. 1994). A direction-specific path deviation, with the GVS-induced lateralized vestibular input, has been partially attributed to directional postural instability (Fitzpatrick et al. 1999). On the other hand, the role of the neck’s proprioceptive system, in postural stability during locomotion, is not clear. It is postulated that the path deviation in response to unilateral neck vibrations is primarily due to an altered perception of straight ahead (Courtine et al. 2003; Bove et al. 2001). None of these studies have evaluated direction-specific postural instability similar to that reported with lateralized vestibular input (Fitzpatrick et al. 1999).
The average CoM velocity in the M-L direction was significantly influenced by the sensory conditions in this study. It is possible that, as a secondary effect, the magnitude and the direction of CoM velocity in the frontal plane influenced the lateral location of foot placement to keep the CoM within the base of support. A cumulative effect of such directional response can further result in path deviation. The M-L CoM velocity was considerably lesser in the older group and replicated the results of path deviations, suggesting that age-related decrease of CoM velocity responses could primarily be attributed to attenuated responses to neck muscle vibration. Compared to young adults, a lesser gain in sway velocity, following lower limb muscle vibration, has been previously reported in the standing posture in older adults (Quoniam et al. 1995). It is possible that the magnitude differences in the M-L CoM velocity contributed to attenuated path deviations.
Segmental kinematics in the frontal plane was significantly modified by the sensory manipulations in both young and older adults. On an average, the trunk maintained a leftward inclination with tonic neck vibration. Although there is no direct evidence, it is possible that the neck proprioceptive input can directly elicit trunk muscle responses in addition to its influence on the lower limb muscles (Anderson and Magnusson 2002). The trunk tilt response in the frontal plane was directionally modified by the simultaneous GVS, possibly through its direct influence on the trunk muscles (Ali et al. 2003).
An appropriate adaptation of body orientation is a key factor for efficient progression during locomotion (Hollands et al. 2001). Input from the neck proprioceptive and the vestibular systems is incorporated by the CNS for egocentric spatial orientation during movement, by interpreting the trunk motion in space (Mergner et al. 1991). The results demonstrate that the vibration-induced leftward path deviation was accompanied by orientation responses to the left. However, concurrently applied GVS did not further alter the orientation in spite of its effect on path deviation, unless when high magnitude in-conflict vestibular stimulation was used. It can be noticed from the trajectories (Fig. 1a, b) that only in the aforementioned condition did the direction of the path trajectory actually reverse to the right. Therefore, the reset in body orientation was probably required to optimize the locomotor performance under this condition. It is not clear from the present study why the trunk kinematic responses did not demonstrate the effects of aging.
Multiple regression analysis demonstrated that the CoM velocity in the M-L direction was a strong predictor of the magnitude of path deviation. In contrast, trunk orientation in the frontal (trunk tilt) or horizontal plane (trunk rotation) did not show significant weighting in prediction of the global response in both age groups, possibly due to a low magnitude of the actual average angular displacement in response to the sensory stimulations (average range, 3° and 8°, respectively). It is also possible that the organization of the global responses by the CNS, during locomotion, were primarily aimed at maintaining balance under the challenging sensory conditions used in this study, rather than simply guided by the body orientation.
In conclusion, neck proprioceptive and vestibular inputs were centrally integrated during locomotion for the purpose of maintaining a walking trajectory. The present data suggest that directional locomotor responses to neck muscle vibrations were very sensitive to the reference frame generated by vestibular information. This relationship was preserved in older adults; however, locomotor responses were significantly attenuated in older adults. Unlike the evidence from studies performed in a sitting position (Schweigart et al. 2002; Strupp et al. 1999), these results may be attributed to a reduced sensitivity of the neck proprioceptive system rather than the vestibular system. A high predictive value of the CoM velocity suggests that organization of the global responses by the CNS were primarily directed towards maintaining equilibrium under the challenging sensory conditions used in this study rather than simply guided by the body orientation.
References
Ali AS, Rowen KA, Iles JF (2003) Vestibular actions on back and lower limb muscles during postural tasks in man. J Physiol 546(2):615–624
Anderson G, Magnusson M (2002) Neck vibration causes short-latency electromyographic activation of lower leg muscles in postural reactions of the standing human. Acta Laryngol 122(3):284–288
Bent LR, McFadyen BJ, Merkley VF, Kennedy PM, Inglis JT (2000) Magnitude effects of galvanic vestibular stimulation on the trajectory of human gait. Neurosci Lett 279:157–160
Bergstrom B (1973) Morphology of the vestibular nerve. Analysis of the calibers of the myelinated vestibular nerve fibers in man at various ages. Acta Otolaryngol 76(5):331–338
Bottini G, Karnath HO, Vallar G, Sterzi R, Frith CD, Frackowiak RS, Paulesu E (2001) Cerebral representations for egocentric space: functional–anatomical evidence from caloric vestibular stimulation and neck vibration. Brain 124:1182–1196
Bove M, Diverio M, Pozzo T, Schieppati M (2001) Neck muscle vibration disrupts steering of locomotion. J Appl Physiol 91(2):581–588
Britton TC, Day BL, Brown P, Rothwell JC, Thompson PD, Marsden CD (1993) Postural electromyographic responses in the arm and leg following galvanic vestibular stimulation in man. Exp Brain Res 94(1):143–151
Courtine G, Papaxanthis C, Laroche D, Pozzo T (2003) Gait-dependent integration of neck muscle afferent input. Neuroreport 14(18):2365–2368
Fitzpatrick RC, Wardman DL, Taylor JL (1999) Effects of galvanic vestibular stimulation during human walking. J Physiol 517:931–939
Gdowski GT, McCrea RA (2000) Neck proprioceptive inputs to primate vestibular nucleus neurons. Exp Brain Res 135(4):511–526
Glasauer S, Amorim MA, Vitte E, Berthoz A (1994) Goal-directed linear locomotion in normal and labyrinthine-defective subjects. Exp Brain Res 98(2):323–335
Hay L, Bard C, Fleury M, Teasdale N (1996) Availability of visual and proprioceptive afferent messages and postural control in elderly adults. Exp Brain Res 108(1):129–139
Hlavacka F, Mergner T, Krizkova M (1996) Control of the body vertical by vestibular and proprioceptive inputs. Brain Res Bull 40(5–6):431–434; discussion 434–435
Hollands MA, Sorensen KL, Patla AE (2001) Effects of head immobilization on the coordination and control of head and body reorientation and translation during steering. Exp Brain Res 140(2):223–233
Karnath HO (1994) Subjective body orientation in neglect and the interactive contribution of neck muscle proprioception and vestibular stimulation. Brain 117:1001–1012
Liu JX, Eriksson PO, Thornell LE, Pedrosa-Domellof F (2005) Fiber content and myosin heavy chain composition of muscle spindles in aged human biceps brachii. J Histochem Cytochem 53(4):445–454
Lopez I, Honrubia V, Baloh RW (1997) Aging and the human vestibular nucleus. J Vestib Res 7(1):77–85
Mergner T, Siebold C, Schweigart G, Becker W (1991) Human perception of horizontal trunk and head rotation in space during vestibular and neck stimulation. Exp Brain Res 85(2):389–404
Miwa T, Miwa Y, Kanda K (1995) Dynamic and static sensitivities of muscle spindle primary endings in aged rats to ramped stretch. Neurosci Lett 201(1):179–182
Popov KE, Lekhel H, Bronstein A, Gresty M (1996) Postural responses to vibration of neck muscles in patients with unilateral vestibular lesions. Neurosci Lett 214:202–204
Popov KE, Kozhina GV, Smetanin BN, Shikov VY (1999) Postural responses to combined vestibular and hip proprioceptive stimulation in man. Eur J Neurosci 11:3307–3311
Quoniam C, Hay L, Roll J-P, Harlay F (1995) Age effects on reflex and postural responses to proprioceptive inputs generated by tendon vibration. J Gerontol 50:B155–B165
Schweigart G, Chien RD, Mergner T (2002) Neck proprioception compensates for age-related deterioration of vestibular self-motion perception. Exp Brain Res 147(1):89–97
Strupp M, Arbusow V, Borges Pereira C, Dieterich M, Brandt T (1999) Subjective straight-ahead during neck muscle vibration: effects of ageing. Neuroreport 10(15):3191–3194
Torvik A, Torp L, Lindboe CF (1986) Atrophy of cerebellar vermis in ageing. A morphometric and histologic study. J Neurol Sci 76:283–294
Welgampola MS, Colebatch JG (2002) Selective effects of ageing on vestibular-dependent lower limb responses following galvanic stimulation. Clin Neurophysiol 113(4):528–534
Acknowledgements
This work was supported by NSERC, Canada and the Ontario Neurotrauma Foundation, Canada. We thank Sebastian Tomescu for his assistance during data collection.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deshpande, N., Patla, A.E. Postural responses and spatial orientation to neck proprioceptive and vestibular inputs during locomotion in young and older adults. Exp Brain Res 167, 468–474 (2005). https://doi.org/10.1007/s00221-005-0182-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-005-0182-z