Abstract
Participants made perceptual judgments about the length of, and manual aiming movements to the opposite end of, formerly visible Müller-Lyer stimuli. The Müller-Lyer illusion affected both perceptual judgments and aiming amplitude. Manipulations of stimulus duration (10 ms or 3,000 ms) and memory delay length (10 ms or 3,000 ms) had no impact on the illusory effect. Aiming movements executed with vision of the hand were less affected by the illusion than movements executed without vision of the hand. The effect of the illusion on aiming amplitude remained the same between peak velocity and the end of the movement even though participants were engaged in on-line control between peak deceleration and the end of the movement. This latter finding was counter to the predictions of a hypothesis (Glover 2002) stating that illusions should only affect the early (planning) stages of movement and not the late (control) stages of movement. We conclude that a single visual representation is used for perception, motor planning, and motor control.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Milner and Goodale (1995) suggested that there are distinct visual processes for action guidance and perception. Potential sources of support for this perception-action hypothesis are studies of actions directed at visual illusions. The logic behind this work is that illusions, which are a consequence of perceptual processing, should not affect action guidance. Some studies have supported this hypothesis while others have not. Thus there is much recent effort devoted to explaining what factors determine whether actions will be affected by illusions (Bruno 2001; Carey 2001; Franz 2001; Smeets and Brenner 2001). One explanation, referred to as the planning-control hypothesis, suggests that illusions only affect the planning of actions, and movements biased by the illusion can be corrected by on-line control (Glover 2002; Glover and Dixon 2001a, 2001b, 2001c, 2002). According to Glover (2002), studies in which illusions show an effect on action typically report measures associated with planning, whereas studies in which illusions show no effect on action typically report post-corrective measures. The primary goal of this study was to test the planning-control hypothesis. To this end, we examined the presence of on-line control, as well as illusion-induced bias at both the early (planning) and late (control) kinematic landmarks of manual aiming movements to Müller-Lyer stimuli. Recent work on the regulation of both blind and visually guided aiming movements indicates that most on-line regulation of the limb occurs between peak deceleration and the end of the movement (Khan et al. 2002, 2003). Given this finding, the planning-control hypothesis would predict that the illusion would have more of an effect at kinematic landmarks associated with planning (e.g., peak velocity) than at portions of the movement associated with control (e.g., the portion of the trajectory between peak deceleration and the end of the movement).
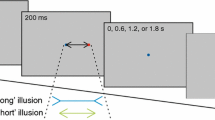
The visual stimuli used in the current study were tails-in, tails-out, and tail-less lines. In the motor task, a hand-held stylus was positioned at one end of the line as the stimulus was presented then extinguished. Following either a short (10 ms) or long (3,000 ms) delay, during which a pattern mask appearedFootnote 1, an auditory signal indicated that the stylus should be moved rapidly to the other end of the line.
In the perceptual task, the delay period was followed by a tail-less comparison line that was short, long, or equal in length, relative to the length of the stimulus. Participants were to verbally indicate whether the comparison was “short”, “long”, or “same”.
The manipulation of delay length was designed to test the planning-control hypothesis, which suggests that movements can be corrected if the delay is shorter than 2,000 ms (Glover 2002). Thus the planning-control hypothesis predicts that the illusion would not impact kinematic measures associated with control for the short (10 ms) delay, but would for the long (3,000 ms) delay. The perception-action hypothesis places a similar emphasis on delays longer than 2,000 ms, suggesting that actions executed under these conditions are guided by perceptual representations, and are thus susceptible to illusory effects.
We also manipulated stimulus duration; the stimulus was presented to participants for either a short (10 ms) or long (3,000 ms) duration. One question that has been raised about studies demonstrating differences between perceptual and motor responses to illusory stimuli has been the differential temporal requirements of the perceptual and motor tasks that have been employed. In the current study, the short duration condition was designed to place perceptual performance under the same time constraints as motor performance. Placing temporal constraints on stimulus exposure also has implications for hypotheses concerning the characteristics of perception and action systems. For example, if visual perceptual processes are slow relative to visual motor processes (Milner and Goodale 1995), then a brief, masked stimulus exposure might reduce the impact of the illusion on perceptual accuracy. Moreover, if visual perceptual processes are distinct from visual motor processes (Milner and Goodale 1995), and thus illusory information must be communicated from perceptual to motor processes, then a brief exposure might also reduce the impact of the illusion on motor accuracy.
In the first experiment of the current study, the aiming movements were executed without visual feedback of the moving limb (no-vision), and in the second experiment they were executed with feedback (vision). This visual feedback manipulation had important implications for understanding memory-guided action. In existing studies of memory-guided manual actions directed at illusions, the reliance on a mnemonic representation of the stimulus was confounded with a lack of visual feedback about hand position. In other words, when the intention of the experimenters was to force participants to use memory by eliminating vision of the stimulus, they have also eliminated vision of the hand. Given that visual feedback about hand position could play an important role in the resistance of movements to illusory effects, we wondered whether the oft-reported susceptibility of memory-guided movements to illusory effects (Bridgeman et al. 1997, 2000; Elliott and Lee 1995; Gentilucci et al. 1996; Post and Welch 1996; Westwood et al. 2000, 2001; Wong and Mack 1981) had been exaggerated because of this confound. In Experiment 2, participants could not see the stimulus, but could see their hand, during movement execution (Carlton 1981). If delayed movements with vision are less susceptible to illusion effects than are no-vision delayed movements, this would suggest that a lack of visual feedback concerning hand position contributes to the susceptibility of delayed movements to illusion effects.
Experiment 1
Method
Participants
Fourteen graduate and undergraduate students (age range 19–26 years) from the McMaster University community participated in this study. The study was conducted in accordance with the ethical guidelines of McMaster University and the Declaration of Helsinki. All participants gave their informed consent prior to their inclusion in the study, and received $15 as compensation for their time. Participants were right-handed, had normal or corrected-to-normal vision, and were naïve to the purpose of the study.
Apparatus
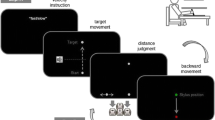
Stimuli were projected onto a piece of black bristol board by an Epson PowerLite 50C projector mounted 100 cm above the surface. E-Prime software (Version 1.0, Psychology Software Tools), running on an AMD 450 MHz computer system, was used to generate stimuli. Stimulus duration was verified with a photo diode and associated hardware (Psychology Software Tools). With their right hand, participants held a stylus (13.5 cm in length) on a switch embedded in a 1.7-cm wooden board beneath the bristol board. An infrared emitting diode (IRED) was attached to the tip of the stylus. In the motor task, an auditory tone, generated by E-Prime and received by a sound-activated relay (Lafayette Instruments Co. Model 63040A), triggered an Optotrak-3020 system (Northern Digital Inc.) to begin recording (200 Hz sampling rate) the position of the IRED. Liquid crystal goggles (see Milgram 1987) were used to occlude vision of the limb for 1.5 s from the time the participants lifted the stylus from the switch. In the perceptual task, the goggles were triggered by the participants’ verbal response into a headset microphone that was connected to a second port in the sound-activated relay. A second microphone (Audio-Technica ATR20), interfaced to the computer with a Serial Response Box Model 200A (Psychology Software Tools), allowed E-Prime to record participants’ vocal reaction time. The experimenter coded the identity of the verbal response manually on a keypad interfaced to the computer. A millisecond timer (Lafayette Instruments Co. Model 50013) was used to interface E-Prime with the Optotrak and the goggles.
Procedure
Each participant performed a perceptual and motor task protocol on separate days. The perceptual task was always performed first, because we were concerned that performance of the motor task would affect the way the perceptual task was performed. Each protocol consisted of 120 trials. Factorial combinations of stimulus duration (10 ms, 3,000 ms) and delay length (10 ms, 3,000 ms) produced four blocks of 30 trials. The order of presentation within and between blocks was randomized. There were three types of Müller-Lyer figures: tail-less, tails-in, and tails-out. The shafts of the Müller-Lyer stimuli were 30 cm in length, and the tails, oriented 45˚ from the horizontal, were 10 cm in length.
Perceptual task
A trial began with the presentation of a white fixation point over the switch. Participants were then presented with a Müller-Lyer stimulus for either 10 ms or 3,000 ms. A random dot mask was then displayed for either 10 ms or 3,000 ms. Following this delay, an auditory tone and one of five tail-less comparison lines (25.5 cm, 27 cm, 30 cm, 33 cm, or 34.5 cm) were presented; the comparison line remained visible for 1.5 s. On hearing the tone, participants were instructed to verbally indicate as quickly as possible if the comparison line was “short”, “long” or “same” compared to the shaft of the initial Müller-Lyer stimulus. The verbal response triggered the goggles to close for 1.5 s, at which time the experimenter coded the response with the keypad. When the participant was ready, the experimenter initiated the subsequent trial. Throughout the protocol participants held the stylus on the switch using their right hand, even though no aiming movement was performed. Within each block of 30 trials, each of the 15 possible stimulus combinations (3 figure types x 5 comparison line lengths) was presented twice in random order.
Motor task
A trial began with the presentation of a white fixation point over the switch, at which time participants depressed the switch with the stylus. Participants were then presented with a Müller-Lyer stimulus for either 10 ms or 3,000 ms. A random dot mask was then displayed for either 10 ms or 3,000 ms. Following this delay, an auditory tone and blank screen were presented. Upon hearing the tone, participants were instructed to make an aiming movement as quickly as possible with the stylus to where they thought the apex of the Müller-Lyer stimulus was located. Lifting the stylus off the switch triggered the goggles to close for 1.5 s, occluding participants’ vision of their moving limb. The auditory tone also triggered Optotrak to record for 1.5 s. Participants were asked to keep the stylus in the final position until the goggles became transparent again (i.e., the Optotrak was finished recording). Within each block of 30 trials, each of the three figure types was presented ten times in random order.
Data analysis
Perceptual task
The effect of the illusion in the perceptual task was assessed by the accuracy of the perceptual comparison. Perceptual accuracy was determined by using Table 1. A negative value was assigned if participants perceived the Müller-Lyer shaft as shorter than the comparison line and a positive value was assigned if participants perceived it as longer. When participants responded “same” to a comparison line that was in fact longer or shorter, the actual difference in centimeters between the Müller-Lyer shaft and the comparison line was used as the amount of error. In order to quantify an inaccurate response of “long” or “short”, 1.5 cm was added to the actual difference between the Müller-Lyer shaft and the comparison line; this value was used as the minimal error because it was the smallest difference between any two comparison lines. Because we converted a perceptual comparison judgment into a quantifiable measure of distance, it is possible that the reported magnitude of the illusion in distance units is an under- or overestimation. Nevertheless, it is reasonable to compare these quantities within the perceptual task (e.g., differences among the three figure types). Vocal reaction time was also measured with E-Prime from the onset of the auditory tone to the registration of participants’ verbal response into the microphone.
Motor task
Displacement data collected from the Optotrak were filtered using a dual-pass Butterworth filter with a cut-off frequency of 10 Hz. Velocity and acceleration profiles were generated by differentiating displacement and velocity, respectively. Movement in the Y-axis (i.e., the primary direction of the movement) was analyzed using custom software (Chua and Elliott 1993). The total displacement in the Y-axis was determined by the difference between location at the finish and start of the movement. Movement start and finish were identified as the frame at which movement velocity rose above or fell below 30 mm/s and remained there for 70 ms (14 frames). Movement time was calculated by multiplying the difference between the start and end frames by 5 ms (i.e., 200 Hz sampling rate). Reaction time was calculated by subtracting the time of the auditory tone (which is the time Optotrak started recording) from the start of the movement. The custom software was used to identify a number of kinematic markers in the Y-axis velocity and acceleration profiles. These included peak velocity and peak deceleration, as well as the spatial location at which these kinematic events occurred. This allowed us to examine both trial-to-trial spatial variability, and amplitude bias not only at the end of the movement, but also at these specific points in the trajectory.
Movement end point location along with the known location of the target was used to calculate constant and variable target-aiming error. Constant error (CE) is the mean algebraic error in the primary direction of the movement (Y-axis) and provides information about a performer’s tendency to undershoot (i.e., negative error) or overshoot (i.e., positive error) a target. Variable error (VE) is the standard deviation of these spatial end points in Y. It provides information about the within-participant consistency of the aiming movements, and when used with spatial variability measures taken at peak velocity and peak deceleration provides us with information about on-line regulation during the “homing phase” of the movement (Woodworth 1899). We also calculated time to peak velocity, and expressed this time period as a proportion of the overall movement time. This proportional time provides an index of the temporal symmetry, with larger proportional times spent after peak velocity being associated with a greater degree of on-line control (Elliott et al. 2001).
Results
Our predictions were specific to a subset of the measured variables. In the interest of brevity, the remaining measures are presented in Tables 2 and 3.
Effect of the illusion on perceptual and motor accuracy
To assess the overall impact of the illusion on the perceptual and motor tasks, we conducted a Figure Type (tails-in, tail-less, tails-out) by Image Duration (10 ms, 3,000 ms) by Delay (10 ms, 3,000 ms) repeated measures analysis of variance (ANOVA) for each of the two protocols (see Tables 2, 3). The perceptual analysis revealed only a main effect for Figure Type, F (1,26)=25.88, p<.001. As is evident in Fig. 1, the tails-in and the tails-out version of the illusion were reliably different from each other. Although the tails-out version of the figure was significantly different from the control figure, the tails-in version failed to be different from the control figure at conventional levels of significance (Tukey HSD, p<.05; critical value = 4.5 mm).
The constant error analysis for the motor protocol yielded main effects for Figure Type, F (2,26)=64.02, p<.001, and Delay, F (1,13)=7.80, p<.05. As in the perceptual situation, participants’ aiming movements were biased by the illusion with all three forms of the figure being reliably different from each other (Tukey HSD, p<.05; see Fig. 2). Moreover, pronounced undershooting at a 10-ms delay (−9.4 mm) became even more dramatic after a 3,000-ms delay (−17.5 mm); such undershooting is typical of memory-guided aiming movements (Elliott and Madalena 1987). Both the planning-control and perception-action hypotheses predicted that the illusion would not impact constant error for the 10-ms delay, but would for the 3,000-ms delay. However, the interaction of Figure Type and Delay was not significant (F<1.0).
Kinematic analyses
Evidence of on-line control late in the movement trajectory was confirmed by an analysis in which we examined the trial-to-trial standard deviations of movement amplitude (i.e., variable error) at three kinematic landmarks: peak velocity, peak deceleration, and movement endpoint (Khan et al. 2003). This analysis revealed a main effect for Kinematic Landmark, F (2,26)=10.51, p<.001, as well as an interaction involving Kinematic Landmark, Figure Type and Delay, F (4,52)=2.62, p<.05. As is evident in Fig. 3, there was a drastic increase in spatial variability between peak velocity and peak deceleration, and a pronounced decrease between peak deceleration and the end of the movement.Footnote 2
Although this pattern of variability over the trajectory was reliable for all figure type and delay combinations (Tukey HSD, p<.05), the presence of the significant interaction begs an explanation. Perhaps the low variability at peak deceleration for tails-in figures in the long delay condition is related to the extreme undershooting for tails-in figures at long delays demonstrated in the constant error analysis (see Fig. 2).
One of the salient features of the planning-control hypothesis is the ability of the control system to correct illusion-induced planning errors as a reaching/aiming movement unfolds. While on-line regulation of the movement certainly seems to be occurring over the final approach to the target, even though participants cannot see their hand, the constant error data indicate that this regulation was not enough to eliminate the impact of the illusion-induced bias. With this in mind, we decided to examine aiming bias at different points in the movement trajectory. To this end, we determined the mean amplitude achieved by the tip of the aiming stylus at peak velocity, peak deceleration and at the termination of the movement. Using the mean for the tail-less figure as a denominator, we then calculated a ratio for aiming movements performed to the tails-in and the tails-out figures. This creates an index of aiming bias that is scaled to the actual distance traveled at a particular kinematic marker. A ratio of greater than 1.0 indicates that the participant moved a greater distance than in the control situation, while a ratio of less than 1.0 reflects a movement of lesser amplitude relative to the tail-less movement. These scores were then analyzed in a Figure Type (tails-in, tails-out) by Image Duration by Delay by Kinematic Landmark repeated measures ANOVA. This analysis revealed only a main effect for Figure Type, F (1,13)=54.07, p<.0001. As is evident in Fig. 4A, tails-in movements covered less distance than tails-out movements at all kinematic landmarks.Footnote 3
Scaled amplitude of manual aiming movements as a function of Müller-Lyer figure type and kinematic landmark. A represents movements performed without visual feedback (Experiment 1) and B represents movements performed with visual feedback (Experiment 2). The amplitudes of movements to tails-in and tails-out stimuli were scaled to the amplitudes of movements to tail-less stimuli. Values greater than one indicate that movements to stimuli with tails overshot movements to tail-less stimuli, and values less than one indicate that movements to stimuli with tails undershot movements to tail-less stimuli
Thus while participants appear to be engaged in on-line control to reduce variability, this regulation during the approach to the target does not reduce the illusion-induced bias.
Movement time and time to peak velocity data also provide relevant information regarding the operation of on-line control in the aiming task (see Table 3). Mean movement times indicate there was plenty of time for the on-line regulation of movement in all conditions (Zelaznik et al. 1983). Proportional time to peak velocity, a subcomponent of movement time, is an index of the symmetry of the velocity profile, and, as such, it provides information about the relative time required to accelerate and to decelerate the limb. The grand mean was .458 indicating that overall participants spent slightly more time decelerating their limb movements than accelerating. Presumably this extra time is used to adjust the movement trajectory during the final stages of the movement (see Elliott et al. 1999).
Experiment 2
In the first experiment, the delayed aiming movements were executed without visual feedback, and the effect of the illusion remained uncorrected even at the end of the movement. These results are problematic for the planning-control hypothesis, which states that on-line corrections can occur under (a) no-vision conditions, and (b) short delay conditions (Glover 2002). Nevertheless, one could argue that the combination of no-vision and delayed conditions might have prevented on-line correction. To address this argument, we conducted a second experiment in which delayed aiming movements were executed with vision. The planning-control hypothesis predicts that the effect of the illusion should be reduced at the end of these movements, at least for short delays.
In the first experiment, memory-guidance was confounded with a lack of visual feedback about hand position. The second experiment eliminated this confound by providing visual feedback. A comparison of delayed movements with vision to delayed movements without vision is critical for understanding why memory-guided movements are so susceptible to illusory effects.
Method
Participants
Fourteen participants ranging from 20 to 27 years of age participated in this study. The study was conducted in accordance with the ethical guidelines of McMaster University and the Declaration of Helsinki. All participants gave their informed consent prior to their inclusion in the study, and received $15 as compensation for their time. Participants were right-handed, had normal or corrected-to-normal vision, and were naïve to the purpose of the study.
Apparatus
All the aspects of the set-up were identical to Experiment 1 except the switch at the home position no longer triggered closure of the goggles at movement initiation.
Procedure
The motor protocol was identical to Experiment 1 except participants were able to see their limb over the course of the aiming movement. A separate perceptual protocol was not conducted.
Analysis
Data collected from the Optotrak were analyzed using the same criteria and techniques as in Experiment 1.
Results
Our predictions were specific to a subset of the measured variables. In the interest of brevity, the remaining measures are presented in Table 4.
Effect of the illusion on motor accuracy
A Figure Type by Image Duration by Delay repeated measures ANOVA of the constant error data revealed a main effect for Figure Type, F (2,26)=24.64, p<.0001, and an Image Duration by Delay interaction, F (1,13)=6.75, p<.05. As can be seen in Fig. 2, in spite of the fact that participants could see their limb over the whole course of the aiming movement, compared to the tail-less condition (.61 mm), they still exhibited reliable undershooting in the tails-in situation (−2.26 mm) and reliable overshooting in the tails-out condition (4.87 mm) (Tukey HSD, p<.05). Unlike Experiment 1, participants did not exhibit an overall pattern of undershooting, and in fact in the 10 ms/10 ms (image/delay) condition participants overshot the target amplitude (3.6 mm) compared to the other three image/delay situations (10 ms /3,000 ms: .4 mm, 3,000 ms/10 ms: 0 mm, 3,000 ms/3,000 ms: .4 mm; Tukey HSD, p<.05).
Kinematic analyses
As in Experiment 1, we examined spatial variability at each of the three kinematic landmarks (i.e., peak velocity, peak deceleration, end of movement). This analysis revealed only a main effect for Kinematic Landmark, F (2, 26)=16.49, p<.0001. As is apparent in Fig. 5, variability increased from peak velocity to peak deceleration, and then was dramatically reduced between peak deceleration and the end of the movement (Tukey HSD, p<.05). As in Experiment 1 (see Fig. 3), this provides strong evidence for on-line control over the final stage of the movement trajectory regardless of figure type, stimulus duration or delay length (Khan et al. 2003).
As in Experiment 1, we calculated ratio scores for each of the two forms of the illusion using the mean amplitude for the tail-less figure as a denominator. A four factor repeated measures ANOVA that included Kinematic Landmark as an independent variable once again revealed a main effect for Figure Type, F (1,13)=45.97 p<.0001, as well as interactions involving Figure Type and Kinematic Landmark, F (2, 26)=3.94, p<.05, and Figure Type, Kinematic Landmark and Image Duration, F (2,26)=4.39, p<.05. As is evident in Fig. 4B, illusion-induced bias was apparent and reliable at all markers in the movement trajectory. Interestingly, post hoc analysis (Tukey HSD, p<.05) revealed that the only significant difference, within a Müller-Lyer configuration, was the increase in bias between peak velocity and peak deceleration for the tails-out form of the illusion (see Fig. 6). There was also a significant Figure Type by Image Duration by Delay interaction, F (1,13)=4.97, p<.05. Post hoc analysis (Tukey HSD, p<.05) indicated that the difference between the two forms of the illusion was reliable in all combinations of duration and delay except the 10 ms duration-3,000 ms delay situation. Although the mediating influence of duration is not obvious, less illusion-induced bias with a long delay is opposite to what both the perception-action and planning-control hypotheses would predict.
Movement time and time to peak velocity data are presented in Table 4. As in Experiment 1, mean movement times indicate there was plenty of time for the on-line regulation of movement in all conditions (Zelaznik et al. 1983). The grand mean of proportional time to peak velocity was .441, indicating that participants spent only slightly more proportional time decelerating the movement than accelerating it when vision of the limb was available.
Between experiment comparisons of movement accuracy and kinematics
In order to determine if the availability of vision of the hand affected the extent of the illusion-induced bias, a Protocol (no-vision, vision) by Figure Type by Image Duration by Delay mixed ANOVA was conducted on the constant error scores. Of interest was the presence of a Protocol by Figure Type interaction, F (2,52)=11.39, p<.001, in the absence of any other interactions involving either of these two variables. Although the illusion-induced bias was significant in both the vision protocol and the no vision protocol (see analyses of individual protocols reported earlier), the effect was only about half the magnitude when vision of the limb was available. Of even greater consequence was the reduction in the overall undershooting in the presence of vision (see Fig. 2). This was reflected in a main effect for Protocol, F (1,26)=8.47, p<.01, in the combined analysis.
Protocol was also added as a factor to the kinematic landmarks analysis, revealing both a main effect for Figure Type, F (1,26)=93.52, p<.0001, and a Protocol by Figure Type interaction, F (1,26)=11.06, p<.01. There was also a significant Protocol by Figure Type by Kinematic Landmark interaction, F (2,52)=4.45, p<.05. As depicted in Fig. 4, the difference between the two forms of the illusion was more pronounced when vision of the hand was not available during aiming. Although post hoc analysis indicated that none of the within protocol-within figure differences was reliable, the three-way interaction appears to be the result of the slight decrease in bias between peak velocity and peak deceleration for the no-vision, tails-out situation, with the opposite tendency when vision was available. We have no explanation for this slightly different pattern.
Discussion
The Müller-Lyer illusion had a notable effect on the perceived length of lines under temporally constrained conditions, in which the durations of stimuli were brief, the stimuli were masked, and the length of lines had to be memorized. Under similar temporal constraints, manual movements aimed at the endpoints of lines were also affected by the illusion. Kinematic analyses of the aiming movements revealed that the effect of the illusion underwent no reduction at late kinematic markers relative to early kinematic markers. This was true for both short (10 ms) and long (3,000 ms) memory delays. This was also true for movements executed without vision (Experiment 1) and for movements executed with vision (Experiment 2), although the effect was more pronounced in the former. Despite their inability to correct the illusion-induced bias, participants were engaged in correction, as evidenced by a reduction of variable error between peak deceleration and the end of the movement. These results are inconsistent with the planning-control hypothesis, which suggests that control processes should reduce the effect of an illusion on a movement executed after a short delay.
The planning-control hypothesis
Glover (2002), in a recent review of the illusion and action literature, cited several studies that were interpreted as consistent with the planning-control hypothesis. How can the differences among the current study and existing studies be reconciled? One distinguishing factor is the illusion that was used in the current study. The Müller-Lyer illusion seems more potent relative to some other visual illusions in terms of its ability to influence action. For example, the Müller-Lyer illusion affects visually guided movements such as manual aiming (e.g., Elliott and Lee 1995; Gentilucci et al. 1996; although see Binsted et al. 2001; Binsted and Elliott 1999a), grasping (e.g., Daprati and Gentilucci 1997; Franz et al. 2001; Westwood et al. 2001; although see Otto-de Haart et al. 1999; Westwood et al. 2000), and saccadic eye movements (e.g., Binsted et al. 2001; Binsted and Elliott 1999a, 1999b).
Other studies examining actions directed at Müller-Lyer stimuli (Westwood et al. 2000, 2001) have been interpreted as supportive of the planning-control hypothesis (Glover 2002). In these studies, participants were required to reach for and grasp the endpoints of a three-dimensional bar that was placed over the shaft of a two-dimensional Müller-Lyer stimulus. In one of the studies (Westwood et al. 2000), when participants had full vision of the stimulus array and their hand, a late kinematic marker (maximum grip aperture) was not significantly affected by the illusion whereas an early kinematic marker (peak aperture velocity) was affected by the illusion. There are reasons to question the degree to which this result is supportive of the planning-control hypothesis. For example, maximum grip aperture was affected by the Müller-Lyer illusion in two other conditions in the same study (Westwood et al. 2000), as it was in several other published studies (Daprati and Gentilucci 1997; Franz et al. 2001; Otto-de Haart et al. 1999; Westwood et al. 2001). In fact, when Westwood et al. (2001) used the identical visual conditions that produced a nonsignificant effect of the illusion on maximum grip aperture in Westwood et al. (2000), they found a significant effect of the illusion on maximum grip aperture. Also, there were other early kinematic markers measured by Westwood et al. (2000) and Westwood et al. (2001) that were not affected by the illusion. Another problem for determining whether Müller-Lyer grasping studies are supportive of the planning-control hypothesis concerns statistical comparisons between early and late kinematic markers. Westwood et al. (2000) did not make a direct comparison between early (peak aperture velocity measured in mm/s) and late (maximum grip aperture measured in mm) kinematic markers. When early and late kinematic markers are analyzed separately, and are both significantly affected by the illusion (as is often the case), it is not possible to say whether the effect of the illusion has increased or decreased over time. We overcame this problem by making direct comparisons of the position of the hand (in mm) at different stages of the movement. It would be more informative to compare our Müller-Lyer aiming results to a Müller-Lyer grasping study in which, for example, grip aperture was monitored throughout the course of the movement, as was done by Glover and Dixon (2002) in their study of the Ebbinghaus illusion. Another issue for grasping studies and the planning-control hypothesis concerns what constitutes an appropriate late kinematic marker, because grip aperture has been criticized as potentially insensitive to the influence of visual illusions (Brenner and Smeets 1996; Jackson and Shaw 2000; Smeets et al. 2002).
Although it is unclear whether the Westwood et al. (2000) results are supportive of the planning-control hypothesis, it is still important to review why our results, in comparison, so clearly demonstrate an effect on late kinematic markers. There are several differences between the current study and Westwood et al. (2000) that could explain why late kinematic markers in the former were more likely to be affected by the illusion. The most obvious difference was that participants in the relevant condition of Westwood et al. (2000) had full vision of the stimulus array and their hand throughout the movement. Under no-vision conditions in the same study, there was a robust effect of the illusion on maximum grip aperture (Westwood et al. 2000). Nevertheless, proponents of the planning-control hypothesis (e.g., Glover 2002) have emphasized that an absence or reduction of illusion effects at late kinematic markers is not dependent upon vision (e.g., Glover and Dixon 2002). A second difference was that participants in the current study received no feedback concerning the accuracy of their movements, whereas participants in Westwood et al. (2000) received tactile feedback from the bar, and thus information about the appropriate proprioceptive posture for future target acquisition. A third difference was that movements in the current study were executed rapidly, with average movement times below 400 msFootnote 4, whereas movements in Westwood et al. (2000) were executed relatively slowly, with average movement times in the range of 666–782 ms.
Another characteristic of the current study that distinguishes it from other studies is the manipulation of stimulus duration, and in particular the use of a short (10 ms) duration. Stimulus duration had no effect on the perceptual task and only minor effects on aiming without vision (movement time, peak velocity) and aiming with vision (reaction time, accuracy). Moreover, these effects were unrelated to the effect of the illusion. This suggests that important differences between our results and those that have been supportive of the planning-control hypothesis cannot be attributed to the temporal constraints placed on our participants.
It seems to us quite reasonable to suggest that, under certain conditions, movements directed at visual illusions can be corrected on-line. Although there are several methodological characteristics of the current study that could have prevented such corrections, the current instantiation of the planning-control hypothesis clearly states that these characteristics should not have prevented correction (Glover 2002).
Memory-guided action
It is often assumed that if illusions affect delayed action, then perceptual memory is the only available means by which action can be guided. For example, Westwood et al. (2000) implied that the decay rate of visuomotor memory can be inferred from the shortest delay at which illusions affect action. Following this logic, one would be forced to make the dubious conclusion that visuomotor memory decays within 10 ms (cf. Elliott et al. 1990), given that the Müller-Lyer illusion affected aiming for the 10 ms delay condition in the current study. We suggest instead that the influence of illusions at short delays says nothing about the decay rate of visuomotor memory; rather, it allows conclusions about the speed with which visual perceptual processes can provide information to visuomotor systems. As for why visually guided movements are more resistant than memory-guided movements to illusion effects, when visual perceptual processes have had ample time to provide illusory information to visuomotor systems, our results suggest that visual feedback concerning the position of the effector plays an important role.
Common versus separate visual representations
Existing models forwarded to explain the effects of illusions on action fall into two classes. Separate representation models suggest that the visual information used for perception is, to some degree, separate from the visual information that is used for action guidance. For example, the planning-control model posits that illusory information is available to motor planning processes, but is not available to on-line control processes. Another example, the perception-action model, has both strong and weak versions (Franz et al. 2001); the strong version suggests that illusory information is not available to motor processes in general, whereas a weak version allows for limited availability of illusory information to motor processes. In contrast to separate representation models, Franz et al. (2001) have forwarded a common representation model, which suggests that a common visual representation is used for perception and action guidance.
Based on the current results, it would be difficult to argue that illusory information is unavailable to motor systems, as is suggested by separate representation models. For example, the illusion affected both the initial and terminal accuracy of pointing movements in both the 10 ms stimulus duration and 10 ms memory delay conditions. The brief stimulus duration and initial accuracy results are problematic for the perception-action model, the weak version of which suggests that perceptual processes are relatively slow to process illusory information and communicate it to motor systems. The brief memory delay results are problematic for the planning-control and the perception-action models, which suggests that illusory information will only inform action after longer delays. The terminal accuracy results are problematic for the planning-control model, which suggests that illusory information is unavailable to control processes.Footnote 5
By comparison, a common representation model has no problems explaining our results, because the illusion had comparable effects on our perceptual and motor tasks. The principle of parsimony also dictates that a single representation model is preferred to a dual representation model. Nevertheless, one might argue that a single representation model cannot explain the results of other published studies in which illusions had larger effects on perceptual measures than motor measures. Franz and colleagues (Franz 2001; Franz et al. 2001) have argued that the perceptual tasks used in these studies have overestimated the impact of illusions on perception. Below, we build upon this argument by pointing out that the motor tasks used in some studies may have underestimated the degree to which illusory information is available to motor systems.
When an illusion has minimal impact on action, there are two possible reasons. One is that the illusory information was prevented from accessing motor processes, due to, for example, a complete or partial structural segregation within the visual system; this is what has been assumed by separate representation models. Alternatively, it is possible that illusory information gained access to motor processes, but was not used by motor processes. Why would available illusory information be ignored by the motor system? Because the illusory information is only one of many sources of information available to motor processes. Other sources of information include visual information about limb position, proprioceptive information about limb, head and eye position, and efference associated with limb, head and eye movements. Under normal circumstances, all of the available information sources are consistent in their analysis of the acting environment. In the laboratory, however, experimenters can create conflict among the different sources of information, by, for example, introducing visual contextual information that produces illusory percepts. We suggest that the degree to which action is informed by the visual processes that are affected by the illusion depends on whether other conflicting sources of information are made available to the actor. This principle has been well supported by the current study and the other published studies of manual aiming movements made to Müller-Lyer stimuli. For example, Gentilucci et al. (1996) found a greater effect of the illusion when neither the stimulus nor the hand was visible during execution, compared to when the stimulus (but not the hand) was visible, and when both stimulus and hand were visible. In the current study, we found a greater effect of the illusion when neither the stimulus nor the hand was visible during execution, compared to when the hand (but not the stimulus) was visible.
To conclude, we suggest that separate representation models have overstated the degree of modularity (Fodor 1983) in visuomotor systems (Goodale 1988). When there is conflict among the processes that inform motor control, the resolution of the conflict requires that one set of processes “wins” and the conflicting set of processes “loses”. We suggest that separate representation models have mistaken this victory for modularity.
Notes
The purpose of the pattern mask was to prevent continued visual processing of the stimulus following the exposure period.
Khan et al. (2003) have demonstrated that decreases in spatial variability do not occur between peak deceleration and the end of the movement if participants have insufficient time or opportunity to use on-line feedback. Thus the changes in variability over the course of the trajectory do not appear to be an artifact of data acquisition or reduction procedures.
Although the analysis of the relative amplitudes revealed no change in the illusion-induced bias from early to late kinematic landmarks, a similar analysis of absolute amplitudes (i.e., not scaled by tail-less amplitudes) revealed an increase in the impact of the illusion at the end of the movement relative to early kinematic landmarks.
Although the aiming movements in the current study were executed rapidly, there was still ample time for feedback utilization (Elliott and Allard 1985; Zelaznik et al. 1983), as is evidenced by the variable and constant error advantages associated with visual feedback availability in Experiment 2.
Other results that are problematic for the planning-control hypothesis were reported by Proteau and Masson (1997) and Brenner and Smeets (1997), who found that a perturbation of the visual background underlying a target had a significant effect on target-directed accuracy, even though the perturbation was not introduced until after a movement was initiated. In other words, visual perceptual information was available to control processes.
References
Binsted G, Elliott D (1999a) Ocular perturbations and retinal/extraretinal information: the coordination of saccadic and manual movements. Exp Brain Res 127:193–206
Binsted G, Elliott D (1999b) The Müller-Lyer illusion as a perturbation to the saccadic system. Hum Mov Sci 18:103–117
Binsted G, Chua R, Helsen W, Elliott D (2001) Eye-hand coordination in goal-directed aiming. Hum Mov Sci 20:563–585
Brenner E, Smeets JBJ (1996) Size illusion influences how we lift but not how we grasp an object. Exp Brain Res 111:473–476
Brenner E, Smeets JBJ (1997) Fast responses of the human hand to changes in target position. J Mot Behav 29:297–310
Bridgeman B, Peery S, Anand S (1997) Interaction of cognitive and sensorimotor maps of visual space. Percept Psychophys 59:456–469
Bridgeman B, Gemmer A, Forsman T, Huemer V (2000) Processing spatial information in the sensorimotor branch of the visual system. Vision Res 40:3539–3552
Bruno N (2001) When does action resist visual illusions? Trends Cogn Sci 5:379–382
Carey DP (2001) Do action systems resist visual illusions? Trends Cogn Sci 5:109–113
Carlton LG (1981) Visual information: the control of aiming movements. Q J Exp Psychol 33A:87–93
Chua R, Elliott D (1993) Visual regulation of manual aiming. Hum Mov Sci 12:365–401
Daprati E, Gentilucci M (1997) Grasping an illusion. Neuropsychologia 35:1577–1582
Elliott D, Allard F (1985) The utilization of visual feedback information during rapid pointing movements. Q J Exp Psychol A 37:407–425
Elliott D, Lee TD (1995) The role of target information on manual-aiming bias. Psychol Res 58:2–9
Elliott D, Madalena J (1987) The influence of premovement visual information on manual aiming. Q J Exp Psychol A 39:541–559
Elliott D, Calvert R, Jaeger M, Jones R (1990) A visual representation and the control of manual aiming movements. J Mot Behav 22:327–346
Elliott D, Carson RG, Goodman D, Chua R (1991) Discrete vs. continuous control of manual aiming. Hum Mov Sci 10:393–418
Elliott D, Binsted G, Heath M (1999) The control of goal-directed limb movements: correcting errors in the trajectory. Hum Mov Sci 18:121–136
Elliott D, Helsen WF, Chua R (2001) A century later: Woodworth’s (1899) two-component model of goal-directed aiming. Psychol Bull 127:342–357
Fodor J (1983) The modularity of mind. MIT Press, Cambridge, MA
Franz VH (2001) Action does not resist visual illusions. Trends Cogn Sci 5:457–459
Franz VH, Fahle M, Bulthoff HH, Gegenfurtner KR (2001) Effects of visual illusions on grasping. J Exp Psychol Hum Percept Perform 27:1124–1144
Gentilucci M, Chieffi S, Deprati E, Saetti MC, Toni I (1996) Visual illusion and action. Neuropsychologia 34:369–376
Glover S (2002) Visual illusions affect planning but not control. Trends Cogn Sci 6:288–292
Glover S, Dixon P (2001a) Dynamic illusion effects in a reaching task: evidence for separate visual representations in the planning and control of reaching. J Exp Psychol Hum Percept Perform 27:560–572
Glover S, Dixon P (2001b) Motor adaptation to an optical illusion. Exp Brain Res 137:254–258
Glover S, Dixon P (2001c) The role of vision in the on-line correction of illusion effects on action. Can J Exp Psychol 55:96–103
Glover S, Dixon P (2002) Dynamic effects of the Ebbinghaus illusion in grasping: support for a planning/control model of action. Percept Psychophys 64:266–278
Goodale MA (1988) Modularity in visuomotor control: from input to output. In: Pylyshyn Z (ed) Computational processes in human vision: an interdisciplinary perspective. Ablex, Norwood, NJ, pp 262–285
Jackson SR, Shaw A (2000) The Ponzo illusion affects grip-force but not grip-aperture scaling during prehension movements. J Exp Psychol Hum Percept Perform 26:418–423
Khan MA, Elliott D, Coull J, Chua R, Lyons J (2002) Optimal control strategies under different feedback schedules: kinematic evidence. J Mot Behav 34:45–57
Khan MA, Lawrence G, Fourkas A, Franks IM, Elliott D, Pembroke S (2003) Online versus offline processing of visual feedback in the control of movement amplitude. Acta Psychol 113:83–97
Milgram P (1987) A spectacle-mounted liquid-crystal tachistoscope. Behav Res Methods Instrum Comput 19:449–456
Milner AD, Goodale MA (1995) The visual brain in action. Oxford University Press, Oxford
Otto-de Haart EG, Carey DP, Milne AB (1999) More thoughts on perceiving and grasping the Müller-Lyer illusion. Neuropsychologia 37:1437–1444
Pepper RL, Herman LM (1970) Decay and interference effects in the short-term retention of a discrete motor act. J Exp Psychol 83:1–18
Post RB, Welch RB (1996) Is there dissociation of perceptual and motor responses to figural illusions? Perception 25:569–581
Proteau L, Masson G (1997) Visual perception modifies goal-directed movement control: supporting evidence from a visual perturbation paradigm. Q J Exp Psychol A 50:726–741
Smeets JBJ, Brenner E (2001) Action beyond our grasp. Trends Cogn Sci 5:287–287
Smeets JBJ, Brenner E, de Grave DDJ, Cuijpers RH (2002) Illusions in action: consequences of inconsistent processing of spatial attributes. Exp Brain Res 147:135–144
Westwood DA, Heath M, Roy EA (2000) The effect of a pictorial illusion on closed-loop and open-loop prehension. Exp Brain Res 134:456–463
Westwood DA, McEachern T, Roy EA (2001) Delayed grasping of a Müller-Lyer figure. Exp Brain Res 141:166–173
Wong E, Mack A (1981) Saccadic programming and perceived location. Acta Psychol 48:123–131
Woodworth RS (1899) The accuracy of voluntary movement. Psychol Rev 3 (Suppl 13):1–119
Zelaznik HN, Hawkins B, Kisselburgh L (1983) Rapid visual feedback processing in single-aiming movements. J Mot Behav 15:217–236
Acknowledgements
This research was supported by the Natural Sciences and Engineering Research Council of Canada and the Canada Research Chair program. We thank John Moroz for technical support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meegan, D.V., Glazebrook, C.M., Dhillon, V.P. et al. The Müller-Lyer illusion affects the planning and control of manual aiming movements. Exp Brain Res 155, 37–47 (2004). https://doi.org/10.1007/s00221-003-1702-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-003-1702-3