Abstract
It has been proposed that somatosensory stimulation in the form of electromyographically triggered neuromuscular electrical stimulation (NMES) to the peripheral nerve can influence functional measures of motor performance in subjects with stroke and can additionally produce changes in cortical excitability. Using a controlled, double-blind design, we studied the effects of intensive (60 h/3 weeks) treatment at home with NMES compared with a sham treatment, applied to the extensor muscles of the hemiplegic forearm to facilitate hand opening in 16 chronic stroke subjects. We investigated improvement in functional use of the hand and change in cortical activation as measured by functional magnetic resonance imaging (fMRI). Following treatment, subjects improved on measures of grasp and release of objects (Box and Block Test and Jebsen Taylor Hand Function Test [JTHFT]: small objects, stacking, heavy cans), isometric finger extension strength, and self-rated Motor Activity Log (MAL): Amount of Use and How Well score. The sham subjects did not improve on any grasp and release measure or self-rated scale, but did improve on isometric finger extension strength. Importantly, however, following crossover, these subjects improved further in the measure of strength, grasp and release (Box and Block [JTHFT]: page turning), and self-rated MAL: Amount of Use score and How Well score. Using fMRI and a finger-tracking task, an index of cortical intensity in the ipsilateral somatosensory cortex increased significantly from pre-test to post-test following treatment. Cortical activation, as measured by voxel count, did not change. These findings suggest that NMES may have an important role in stimulating cortical sensory areas allowing for improved motor function.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Change in somatosensory input has been shown to affect cortical organization in healthy subjects and subjects with brain injury (for review see Nudo et al. 2001), but does it also affect a person’s ability to function after cerebral infarct? The American Heart Association (2000) reports that approximately 600,000 people suffer a first or recurrent stroke each year in the United States. Approximately 69–80% of patients with stroke initially have an impaired upper extremity (Nakayama et al. 1994) and as many as a third of all stroke survivors will have significant residual disability (Brandstater 1998). Often the individual is able to close the fingers into a fist, which is part of the flexion synergy, but is unable to open the fingers. Neuromuscular electrical stimulation (NMES) has been used for many years in clinical settings to help facilitate functional hand opening, but research regarding its benefit is not convincing. Chae and Yu (2000) stated that all randomized controlled studies reported an improvement in motor impairment, with mild to moderately impaired subjects improving the most, but many studies are difficult to interpret because sample sizes were small and designs lacked sham treatment as a control. Other studies have failed to show that NMES has had a positive effect (Hummelsheim et al. 1997; Johansson et al. 2000). In another review, de Kroon and colleagues (2002) found that functional tests were performed in only two studies. They concluded that NMES has a positive effect on motor control but that no conclusions could be drawn with regard to the effect on functional abilities.
Studies to date have used treatment regimes that involve relatively brief periods of use: 30–60 min, three to five times per week (Fields 1987; Kraft et al. 1992; Chae et al. 1998; Francisco et al. 1998; Sonde et al. 1998; Powell et al. 1999; Cauraugh et al. 2000) and are typically used in the clinical setting. We wanted to determine whether a longer duration of use applied in a home use setting would produce more benefit. Thus, we investigated a combination of electromyographically (EMG)-triggered NMES and cyclic NMES in an intensive self-operated home environment. In addition to investigating the functional improvement associated with NMES, we were also interested in investigating whether functional improvement was reflected in cortical activation.
Treatment with NMES to the median nerve performed during functional magnetic resonance imaging (fMRI) in healthy subjects has been shown to activate the primary sensory and primary motor regions of the brain in the hemisphere contralateral to the stimulation (Spiegel et al. 1999; Kampe et al. 2000). Khaslavskaia (2002), using transcranial magnetic stimulation (TMS), found increased amplitude of motor evoked potentials (MEP) in the tibialis anterior following NMES to the peroneal nerve. This increase was maintained for up to 110 min. Recently, Smith and colleagues (2003) demonstrated a dose-response relationship between NMES to the lower extremity and brain-activation in sensory and motor regions contralateral to the stimulation. While these studies have lent evidence supporting the existence of a central effect during NMES in healthy subjects, studies have not been performed on a population suffering from stroke.
The purposes of this study were to investigate whether moderately impaired subjects with chronic stroke would improve on functional tests of ability in the hand after intensive home treatment with NMES and whether evidence of a long-term cortical effect could be seen 48 h after treatment ceased.
Materials and methods
Subjects
Sixteen subjects with stroke were randomly assigned either to a treatment group (N=8) or to a group receiving sham treatment (N=8). Ages were 33–78 years (mean ± standard deviation = 60.1±14.5 years). To control for spontaneous recovery, subjects were at least 6 months post-stroke. The mean time since stroke was 35.5±25.1 months. Subjects were recruited by visits to stroke-support groups, by advertisements in local newspapers, and through contacts with physical and occupational therapists in the region. The neuroanatomical location of the stroke and the subjects’ demographic data are recorded in Table 1.
NMES has previously been shown to be most effective in results for the mild to moderately impaired patient (Chae and Yu 2000), so we required that subjects have at least 10 deg of active flexion/extension movement at the metacarpophalangeal (MP) joint of the index finger. Since independent operation of the stimulator was also a requirement, mental capacity was tested using the Mini-Mental State Examination (Folstein et al. 1975) and we required subjects to have a score of ≥25 out of a possible 30. The University of Minnesota Internal Review Board and the General Clinical Research Center approved this study. All subjects gave informed consent.
Instrumentation and procedure
All subjects were tested a minimum of two times. Those in the treatment-receiving group were tested pre-test and post-test. Those in the group receiving sham treatment were tested pre-test, post-test, and again at post-crossover (after the true treatment had been given).
Functional testing
The following tests were conducted: Box and Block, Motor Activity Log, Jebsen Taylor Hand Function Test, isometric strength of the index finger extension, finger-movement tracking, and fMRI. A trained investigator with 7 years experience as a physical therapist and who was blinded to the designation of the subject’s group performed all testing.
Box and block test
Changes in functional grasp and release of objects were tested using the Box and Block Test (BB) (Mathiowetz et al. 1985). The BB Test has been used previously to examine for functional gains in manual performance following treatment in subjects with stroke (Cauraugh et al. 2000; Carey et al. 2002). Subjects performed three timed (60-s) trials. The dependent variable was the average number of 2.54-cm3 blocks transported from one side of a partitioned box to the other. Good reliability and validity of the BB Test has been reported (Desrosiers et al. 1994; Carey et al. 2002).
Motor Activity Log
The Motor Activity Log (MAL) (Taub et al. 1993) was used because it is an indicator of “real world” function as judged by the subject. The MAL is a structured interview during which patients indicate how often and how well they use their paretic arm in 30 activities of daily living such as using a fork or spoon for eating, picking up the telephone, and using a key to unlock a door. The MAL has been shown to be a reliable instrument (Miltner et al. 1999). For each task the subject rates the ability of the hemiplegic hand on a 0–5 scale for two different categories: amount of use (AS), and how well (HW) the hand performs. A score of zero indicates that the affected hand is not used for that task and a score of 5 indicates that the affected hand performs that task as often as before the stroke or as well as before the stroke. A summary score is used, which is the average of all activities for each category.
Jebsen Taylor Hand Function Test
The Jebsen Taylor Hand Function Test (JTHFT) (Jebsen et al. 1969) is composed of seven timed testing activities, including: 1) writing a sentence, 2) turning over cards, 3) picking up small objects (e.g., pennies, paper clips) and placing them in a container, 4) stacking checkers, 5) simulating eating, 6) moving large empty cans, and 7) moving large weighted cans. Since some of the subjects were using their (pre-stroke) non-dominant hand, we excluded the handwriting portion of the test for all subjects. This test has been shown to have good validity and reliability (Jebsen et al. 1969).
Strength
Strength of finger extensors was measured using a load cell (Model SM-50, Interface, Scottsdale, AZ, USA). With the subject’s wrist in neutral and the metacarpophalangeal (MP) joint in 70 deg of flexion, the proximal phalanx of the index finger was connected to the load cell. The subject performed three 5-s maximal voluntary isometric contractions, separated by 1-min rests. The dependent variable was the average peak force of these three trials, recorded in Newtons (N). The load cell was calibrated before each testing session and was found to have a maximum of 2.5% error in reading compression-forces over the range of 2–200 N.
Finger-movement control
Precision in control of finger movements was measured with a finger-movement tracking task that the subject performed during the fMRI (Carey et al. 1994, 2002). An electrogoniometer attached to the MP joint of the hemiplegic index finger was connected to a computer displaying a sine wave (0.4 Hz) that served as a target for the subject to track using the computer screen cursor. The subject’s uninvolved hand was lightly holding a rubber bulb that he or she could squeeze to gain our attention during the scanning. This served not only as device for communication, but also as a method to deter any mirror movement of the uninvolved hand. Once positioned inside the magnet, the subject tracked for approximately half a minute to adjust to the task. This allowed an investigator to monitor for any mirror movements (Cramer 1999). No subjects displayed mirror movement while under observation.
The complete tracking test lasted 6 min and consisted of the following phases, each of 1 min in duration, making up the model used for activation analysis: control 1, task 1, control 2, task 2, control 3 and task 3. For each “control” phase, the subject merely watched the screen but exerted no effort. For each “task” phase, the subject looked at the screen and attempted to track the sine wave as accurately as possible using careful finger-extension and finger-flexion movements. The amplitude of the sine wave was standardized to each subject’s active range of motion at the MP joint of the index finger, as recorded during set-up. With maximal flexion set as 0% of the subject’s active range and maximal extension set as 100%, the extension (upper) peaks of the sine wave were set at 125% of the range and the flexion (lower) peaks were set at 15% of the range. The range used originally at pre-test was the range used at post-test. The upper peaks were set above the subject’s full range to allow for any increases in extension range that might occur from treatment.
fMRI
Images of the brain were collected on a customized, 4.0 Tesla whole-body research MRI system (Oxford, UK). A head volume coil (Vaughan et al. 2001) was used to allow for the collection of images over the entire volume of the brain. Padding was used around the head to minimize movement.
T1-weighted inversion recovery magnetic resonance (MR) images were obtained in the transverse, coronal, and sagittal planes for anatomical localization (multi-slice turboFLASH sequence, echo time (TE) = 5 ms, repetition time (TR) = 9 ms, inversion time (TI) = 1.2 s, in-plane resolution = 1.56×1.56 mm, slice thickness=5 mm, 2 averages (NEX)). These images were used at the time of the experiment to determine the appropriate volume for the subsequent functional images, and again in post processing to identify landmarks such as the anterior and posterior commissures for standardizing into Talairach space.
Blood oxygen level-dependent (BOLD) T2 *-weighted functional MR images were obtained in the transverse plane using a blipped echo-planar imaging (EPI) sequence with a TE of 25 ms. The total imaged volume extended from the superior pole of the cortex to a depth of 135 mm in 24 slices. Functional images had an axial in-plane resolution of 3.125×3.125 mm and a 5-mm slice thickness. Each set of 24 images was acquired in 3.0 s during the 6-min experiment.
Treatment
Group receiving electrical stimulation
The goal was to investigate intensive home use of electrical stimulation, thus the amount of treatment selected was 60 total hours of use. All subjects were instructed in the operation of the electrical stimulator at the first visit (pre-test). Since the exclusionary criteria required that the subject have good cognitive ability, all subjects were able to apply this machine safely. We maintained contact with the subjects by telephone at least weekly to answer any questions and encourage compliance. Subjects were instructed to use the machine 6 h per day, for 10 days over the course of 3 weeks. Subjects maintained records of use to show compliance. Records were analyzed visually to look for any differing patterns of use. All subjects completed the required total time use and the patterns of use were very similar, typically 3–6 h per day every day or every other day.
The device used for treatment was the Automove Model AM 706 Stimulator (Danmeter, Boulder, CO, USA). In this device the electrodes function as both EMG pickup and stimulation delivery. The EMG activity detected from underlying muscles must reach a set threshold that then triggers electrical stimulation to the muscles, producing stronger contractions than those achieved voluntarily. This then gives sensory bombardment centrally through cutaneous and proprioceptive afferents. The stimulation threshold is pre-programmed by the manufacturer to start at a high level and then is automatically lowered in a step-wise fashion until the subject’s EMG effort does succeed in reaching threshold. Once triggered, the stimulator delivered pulses (pulse width 200 µs, asymmetrical rectangular biphasic, constant current) at 50 Hz and intensity was set to produce finger- and wrist-extension movements. The duration of the resultant evoked contraction following the initial trigger lasted 5 s plus a 1-s ramp-up and a 1-s ramp-down. A 15-s rest period occurred between contractions to minimize fatigue. Half of the treatment time was spent with active effort on the subject’s part required to trigger a stimulated response (trigger mode), while the other half of the time was spent with the machine automatically stimulating the muscle to contract cyclically without any volitional trigger from the patient (cyclic mode). This was done because pilot work suggested that due to the conscious effort required, it was too difficult to schedule the full treatment of trigger mode into each day. No instructions were given to encourage increased hand use or to modify behavior in any other way.
Group receiving sham treatment
Subjects randomly assigned to the group receiving sham treatment received the same instructions as the EMG treatment group. Subjects receiving sham treatment used a very similar device (Automove Model 700S, Danmeter, Boulder, CO, USA) that shows a light when the device is in the “on” phases, and they were told to advance the “current” dial to an indicated level; however, the machine did not deliver any current. They were told that there are different levels of stimulation being studied and that they may not feel the level assigned to them. As with the true treatment, for half the hours of treatment, the subjects were asked to lift the hand actively when the light came on and the “stimulation” started. At the conclusion of the period of sham treatment, all subjects were retested (post-test) and then crossed over to receive the true stimulation, in exactly the same manner as the group receiving true treatment. Subjects in this group also were monitored by phone and maintained records of compliance both during the sham treatment and after the crossover. Seven of eight subjects were then retested following completion of the real treatment (post-crossover). One subject was unable to complete post-treatment testing due to an unrelated hospitalization.
Analysis of data
Tracking
The computer quantified the tracking performance in each of the three task periods of the 6-min tracking test inside the magnet by calculating an accuracy index (AI) (Carey 1990). The maximum possible score is 100%. Negative scores are possible. Previous work showed good reliability in the average accuracy index with ICC values ranging from 0.92 to 0.97 for subjects with stroke (Carey 1990). The validity of the finger-movement tracking test in discriminating between the performance of healthy subjects and subjects with stroke has been demonstrated (Carey et al. 1998).
fMRI
Analysis of the MR images was done on a Sun Ultra 60 (Sun Microsystems, Palo Alto, CA, USA) workstation using the interactive image analysis software Stimulate (Strupp 1996). The raw data for each individual were log-transformed and detrended. The logarithmic transformation was dictated by the lack of normality in the distribution of activation intensities (Lewis et al. 2002). The detrending was necessary due to the presence of time trends; control and task periods were detrended following the log transformation. To eliminate large vessel contributions, a mask was applied to the functional data (Kim et al. 1994). Two types of fMRI analysis were performed: voxel count and intensity.
Voxel count
Our predefined area of investigation for this analysis included bilateral frontal and parietal lobes. Activation was determined using a cross-correlation (“boxcar”) method correlating the hemodynamic response in comparison with the alternating control and task phases in our activation model. The activation model is defined by the sequence of magnetic resonance scans that correspond to each “task” or “control” phase and takes into account the lag in blood flow change that occurs with activation (Ugurbil et al. 1999). A voxel was considered active if it met or exceeded a predetermined threshold of correlation (r=0.40) and formed part of a cluster (>3) of contiguous voxels that also met or exceeded this threshold. In this method of analysis, the question was whether NMES treatment produced a change in the number of active voxels in a given brain area during the finger-tracking task. The anatomical location of each cluster of active voxels was recorded to each hemisphere (either contralateral or ipsilateral to tracking hand) as determined by Talairach coordinates (Talairach and Tournoux 1988). These locations were as follows: gyrus postcentralis, gyrus precentralis, gyrus frontalis superior, gyrus frontalis medius, gyrus frontalis inferior, precuneus, gyrus cinguli, lobulus paracentralis, lobulus parietalis inferior and gyrus frontalis medialis. The investigator analyzing the data was blinded as to the group of each subject. Under these conditions, we estimate the resultant value of significance for active voxels to be at p<0.001 (Xiong et al. 1995).
Intensity
The intersession variability of the MR signal in voxel count has been found to be consistently higher than the variability of signal intensity, suggesting that the latter is a more reliable indicator of cortical activation (Waldvogel et al. 2000). In measurements of intensity, the question was whether subjects who have received NMES show a different intensity of activation during the task period. That is, NMES may not increase the number of voxels that are active but, instead, may increase the BOLD signal intensity for the average of all voxels in a given region of interest. For this measure, analysis was restricted to the gyrus precentralis (GPrC) and gyrus postcentralis (GPoC). The increase in signal suggests that there is increased neural activity (thus increasing BOLD signal) but does not specify whether this is excitatory or inhibitory.
We chose GPoC and GPrC as the most likely areas to demonstrate change in intensity given the results from imaging studies conducted during NMES (Hamdy et al. 1998; Spiegel et al. 1999; Kampe et al. 2000; Stefen et al. 2000; Abo et al. 2001; Backes et al. 2002; Fraser et al. 2002; Kaelin-Lang et al. 2002; McKay et al. 2002; Smith et al. 2003).
For each region separately, we calculated an Intensity Index (II):
where “IntensityTASK” is the average signal intensity during the task periods and “IntensityREST” is the average signal intensity during the rest periods. There is intrinsic variability in signal intensity between and within subjects (Cohen and DuBois 1999), making direct comparisons on raw intensity values difficult. Normalizing the task data to the rest data allows for intersubject and intrasubject comparisons (Georgopoulos et al. 2001). The information was obtained by drawing regions of interest around GPoC and GPrC at each level of the MRI scan containing the specified region. The precentral gyrus was defined as the area bounded anteriorly by the precentral sulcus, posteriorly by the central sulcus, medially by the sagittal fissure, and laterally by the sylvian fissure. The postcentral gyrus was the area bounded anteriorly by the central sulcus, posteriorly by the postcentral sulcus, medially by the sagittal fissure, and laterally by the sylvian fissure. Sulci were identified according to Ono and colleagues (1990). This analysis has an advantage of not having any arbitrary threshold applied. Intensity Index values are in arbitrary units.
Statistical analysis
Two group t-tests were done to determine equality between groups at baseline on all variables. All data from functional tests were analyzed with preplanned, one-tailed, paired t-tests if the data were normally distributed or Wilcoxon Signed-Rank tests if not. The brain activation data (both voxel count and intensity index) were analyzed using two-tailed paired t-tests. Statistical significance was set at p<0.05.
Results
All subjects (N=16) were included in the tests of hand function, but of those subjects the MRI data from four were discarded due to excessive head movement (one subject from the treatment group and one from the sham group). Of the five remaining subjects in the sham group, one subject was unable to complete post-crossover testing due to an unrelated hospitalization. So the brain activation analysis is based on seven subjects in the group receiving EMG treatment, five in the group receiving sham treatment, and four at post-crossover. The analysis to determine equivalence between groups at baseline showed no significant differences between groups for either behavioral or cortical data.
Changes in functional performance
There was significant improvement in performance for the group receiving treatment on the following functional tests: Box and Block, isometric finger-extension strength, MAL (AS and HW) scores, and in the JTHFT for small objects, stacking, and heavy cans. There was a trend toward improvement in the JTHFT for page turning (p=0.053). The group receiving sham treatment improved in isometric finger-extension strength, but no other test. Following crossover for the group receiving sham treatment, subjects improved from post-test on Box and Block, isometric finger-extension strength, MAL (AS and HW) scores, JTHFT for page-turning, and feeding. There was a trend toward improvement in JTHFT for small objects (p=0.060). There was no improvement in the tracking task performed during the fMRI for either group. Table 2 summarizes all data of functional tests.
Cortical activation data
Voxel count
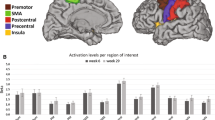
There were no changes in voxel count from pre-test to post-test in any of the designated anatomical areas in either hemisphere for either group. Figures 1 and 2 summarize the data on voxel counts for the ipsilateral and contralateral hemispheres, respectively.
Voxel count analysis data for ipsilateral (nonlesioned hemisphere) anatomical areas while tracking with hemiplegic right hand before (pre) and after (post) NMES or sham. GPoC gyrus postcentralis, GPrC gyrus precentralis, GFs gyrus frontalis superior, GFm gyrus frontalis medius, PCu precuneus, GC gyrus cinguli, LPi lobulus paracentralis, GFd gyrus frontalis medialis
Voxel count data on contralateral (lesioned hemisphere) anatomical areas while tracking with hemiplegic hand before (pre) and after (post) NMES or sham. GPoC gyrus postcentralis, GPrC gyrus precentralis, GFs gyrus frontalis superior, GFm gyrus frontalis medius, PCu precuneus, GC gyrus cinguli, LPi lobulus paracentralis, GFd gyrus frontalis medialis
Intensity
In the group receiving treatment, there was a significant (p=0.046) increase in the Intensity Index from pre-test to post-test in the GPoC area in the hemisphere ipsilateral to the hemiplegic (treated) hand. There was no change in the ipsilateral GPrC or in the contralateral GPoC or GPrC in the group receiving treatment. The Intensity Index did not change in any area in the sham group. The scores for mean Intensity Index are reported in Table 3. Figure 3 is a graph showing individual pre-test and post-test Intensity Index scores for the ipsilateral GPoC.
In the subjects receiving sham treatment, after crossover there were fMRI data from only four remaining people. There was a mean Intensity Index increase in the ipsilateral GPoC, with a pre-test mean of 0.227 and a post-test mean of 0.377. Due to the low N, this was not a significant difference.
Discussion
The important findings of this study were that NMES, when performed in an intensive manner, produced significant improvements in functional activities in the group receiving NMES treatment. The group receiving sham treatment only showed gains in strength without functional improvements from pre-test to post-test; however, after crossing over to receive treatment, they showed significant functional gains that mimicked the group receiving NMES treatment. We did not find a change in the number of active voxels in any neuroanatomical area in the group receiving NMES treatment, but the treatment group had a significant increase in the intensity index for the GPoC ipsilateral to the paretic hand, whereas the group receiving sham treatment did not.
Functional tests
We chose functional tests that would focus on finger movements, particularly finger extension, since controlled release of an object is the function we expected to improve the most with the NMES treatment. The BB test and JTHFT focused on this function. We chose the MAL because we were also interested in the subjects’ own assessment of whether improvements would transfer into hand use in daily activity. We examined isometric finger-extension strength of the index finger as a separate variable.
The BB test and the small objects and stacking components of the JTHFT specifically tested the ability to pick up and release small objects. The group receiving NMES treatment improved performance on all these tests. In addition, this group improved on the component of the JTHFT, which tests grasping and releasing of a heavy can object. This, in combination with the finding that the group receiving sham treatment did not improve on these functions, suggests that the improvement was due to a real treatment effect and not to expectation effects associated with any treatment. The finding that the group receiving sham treatment did improve on the BB test and the page-turning and simulated feeding components of the JTHFT after crossing over to receive the real treatment adds strength to this interpretation. We attribute the absence of significant improvement, although trends were observed, on small objects and stacking components of the JTHFT to the reduced power associated with the loss of one subject following the sham period.
Both the treatment and the sham groups showed significant increases in strength. We believe that the improvement in strength in the group receiving sham treatment stems from repeatedly extending their fingers voluntarily in synchrony with the “stimulation” phases of the sham treatment. We elected to have all subjects perform this voluntary movement so that the only difference between the two groups would be the stimulation itself. Although this repeated movement may have been sufficient to improve strength in the sham subjects, it did not translate into improved functional hand movements. Thus, functional hand movements may depend more on orchestrating synergistic control of multiple muscular forces than on sheer strength alone, and the possibility exists that NMES helps to activate neurons that can improve such control.
The MAL showed significant improvement in the AS and HW scores in both the group receiving treatment and in the group receiving sham treatment after they had crossed over and received the true treatment. The mean changes reported were quite small, however, and it is not clear that the statistically significant changes we observed translate to being clinically significant. The MAL asks a wide variety of questions that could be limited by restricted motion in the shoulder or elbow, which our intervention did not strive to improve, such as “getting out of the car”, “combing your hair”, or “shaving”. Thus, a large improvement in a few activities such as “carrying an object in your hand” and “removing an item of clothing from a drawer” could produce only a modest gain in composite score, but a profound gain in function of particular tasks. Consequently, the MAL may not be the best indicator of improved performance for interventions narrowly focused to such a specific function as finger and wrist extension. It is possible that the increased use of the hand was the source of other effects unique to the treatment group. We feel this is unlikely, however, given that the amount of increased use was quite small (1.5–1.9) and of questionable clinical relevance.
The tracking task failed to show improvement in any group. This task was chosen because it challenges subjects to execute carefully controlled finger movements, which more closely resemble functional activities than the often used “finger opposition task.” Finger-movement tracking requires visual spatial control of flexion and extension movements (Carey et al. 1998). It has previously been shown that tracking performance can be improved in subjects with stroke (Carey et al. 2002) with training in finger tracking, which is intended to challenge and develop such control. In the current study, however, the intervention did not involve visual spatial problem solving or training of precision finger movements. These results, along with earlier results (Carey et al. 2002), invite further research to examine whether NMES combined with tracking training may produce greater gains in functional hand movements than either treatment alone. More research is also required to determine whether a different functional task would have revealed similar cortical results.
Cortical changes
Others have shown in healthy subjects that NMES given during fMRI (Spiegel et al. 1999; Backes et al. 2000; Kampe et al. 2000; Smith et al. 2003) or TMS (Ridding et al. 2000, 2001) increases activation (as measured by voxel count) contralaterally in the sensorimotor cortex. Intensity of activation has not been reported. Testing has not been done on human subjects with stroke, but in rats that have recovered from induced stroke, receiving NMES to the recovered hindlimb during fMRI activates the ipsilateral, non-infarcted hemisphere (Abo et al. 2001).
Importantly, in a related study, patients with acute stroke were studied during pharyngeal stimulation, and it was found that there was a marked increase in the pharyngeal topographic motor representation in the undamaged (ipsilateral) hemisphere as measured by TMS (Fraser et al. 2002). Our results, combined with these findings, provide mounting evidence that there is a cortical component to NMES. However, our finding may simply be demonstrating the consequence of the individual stroke damage interacting with NMES whereby the ipsilateral pathway is the primary mechanism available.
In studies investigating recovery processes after stroke there is an emerging hypothesis that the activation in the stroke hemisphere is predictive of better recovery (for review see Calautti and Baron 2003). Indeed, the uncrossed cortical spinal tract has only marginal physiological function as demonstrated by transcranial magnetic stimulation studies in normal subjects but is activated during complex motor tasks (Carr et al. 1994). This could indicate a role for ipsilateral control in the early recovery stage following stroke (Calautti and Baron 2003) or when an individual with stroke is receiving treatment, as in the current study.
Since there was an increase in the Intensity Index in the ipsilateral GPoC, why was there not also an increase in voxel count? Voxel count and intensity do not mirror each other in what they are measuring. In our voxel count analysis, significance is based upon activation following the time course of the signal relative to the model, whereas the intensity index is based on the amplitude of the signal during the task relative to rest (Carey et al. 2003). The possibility exists that the time course of the changing signal could fail to reach the correlation threshold for “activation,” but have a large difference in signal amplitude creating a significant change in the Intensity Index.
The other question that remains to be answered is why there was evidence of functional improvements, but no change in motor cortex activation (either Intensity Index or voxel count). There are a number of possible reasons. The answer could lie in the type of intervention. The key to achieving voxel count reorganization may be active engagement. In NMES treatment, even with active effort triggering the stimulation, there is repetition, but it does not require any cortical problem solving. It has been shown that active repetitive movements are a key factor in recovery from stroke (Taub et al. 1993; Nudo and Milliken 1996; Nudo et al. 1996b; Feys et al. 1998), but beyond simple repetition, an element of problem solving is also required. This finding was illustrated in the study by Plautz and colleagues (2000) in which monkeys who were trained in a repetitive “easy” task did not produce functional reorganization of cortical maps, whereas monkeys trained in a “difficult” task that required improvements in a motor skill did produce task-related changes in movement representations in the motor cortex (Nudo et al. 1996a). Also using intracortical microstimulation (ICMS) in monkeys recovering from an induced lesion, the use of a restraint jacket to the healthy limb alone resulted in no change in mapped hand representations in the brain beyond that seen with spontaneous recovery, but with the added component of repetitive problem solving, the area of hand representation expanded (Friel et al. 2000).
Relatedly, Kleim and colleagues (1997) showed evidence of significantly greater neuroplastic change in rats that were trained in an acrobatic walkway condition requiring problem solving compared with rats that experienced the simple walkway condition. In our case, the repetitive practice of opening and closing one’s hand (as in the group receiving sham treatment) did increase strength, but did not improve any other measure of functional ability, nor was there any evidence of cortical changes. When the repetitive task of hand opening and closing was paired with electrical stimulation, there was now sufficient intervention to produce improvement in functional tests and produce a change in cortical intensity in the sensory cortex, but not in the motor cortex. Additionally, the result that strength increases alone do not produce cortical reorganization has also been shown in rats in a study that found that skilled forelimb movement, not increased forelimb strength, was associated with a reorganization of forelimb movement representations within the motor cortex (Remple et al. 2001).
With these results we can speculate that simple repetition of movement does not produce changes in cortical activation, but if it is paired with somatosensory stimulation provided by NMES, there is evidence of cortical effects. The fact that the change was in the sensory cortex instead of the motor cortex may also reflect an increased sensitization of those neurons associated with finger movement, which might indicate an LTP-like process (Butefisch et al. 2000).
Another potential reason for the lack of an increased active voxel count may be related to the time interval between the last treatment session and the post-test. McKay and colleagues (2002) suggested that a longer period of stimulation may produce a longer duration of cortical effect. Certainly, our treatment protocol of stimulation over 3 weeks was of long duration, but our post-test was also long after (48 h) the last treatment. This duration was selected because we were most interested in long-term effects, not immediate affects during or after the stimulation.
Somewhat related to simple NMES treatment is a treatment involving low-frequency (0.05 Hz) stimulation to the median nerve paired with TMS to the motor cortex. In one study the investigators treated healthy subjects for 30 min and found an increase in the motor cortex excitability that reverted to pre-stimulation levels within a few hours (Stefen et al. 2000). Another study that followed a similar protocol but repeated it over 3 consecutive days showed that the increased excitability persisted for 2 days (McKay et al. 2002). Further investigation is required to determine whether voxel count changes can be observed immediately after NMES treatment in subjects with stroke.
The third potential reason for lack of increased voxel count may be due to the fact that there was not a behavioral improvement in the tracking test, the task performed during fMRI. Further investigation is needed to determine whether a grasping task would have shown a reorganizational change.
Mechanisms
There are three major processes that have been proposed to be of importance in recovery from stroke: 1) peri-infarct reorganization (Nudo et al. 1996b); 2) neural plasticity, with an increase in dendritic branching in the contralateral hemisphere (Jones and Schallert 1994); and 3) increased activation in the ipsilateral (non-infarcted) hemisphere (Aizawa et al. 1991; Cao et al. 1998). Our finding of an ipsilateral cortical effect from an intervention has been observed often in subjects with stroke (Weiller et al. 1993; Nelles et al. 1999, 2001), but has been shown to be associated with poorer stroke recovery (Calautti and Baron 2003). It is hypothesized that after cerebral damage, normally suppressed ipsilateral motor tracts may become unmasked by a lack of inhibitory control from the affected hemisphere (Caramia et al. 1996; Ziemann et al. 1999) or activated due to excess recruitment (Calautti et al. 2001) and that this may be a pattern that benefits acute or subacute stroke recovery.
Indeed, somatosensory input is required for accurate motor performance and for learning new skills (Panizza et al. 1992) and a reduction of sensory input by local anesthesia impairs motor control in healthy subjects (Aschersleben et al. 2001). Also, in patients with stroke, somatosensory deficits are associated with a slower recovery of motor function (Reding and Potes 1998). Therefore, increased sensory input via ipsilateral pathways may be an important recovery mechanism in motor learning in subjects with stroke.
The majority of work with NMES and the great majority of all rehabilitation procedures has been done on subjects with at least some motor function. More research is needed with single and additive rehabilitation procedures on subjects with severe paralysis to explore effectiveness as well as cortical changes. Current work invites more research to determine whether the augmented proprioceptive feedback from NMES with its possible facilitatory effect on the sensory cortex, as shown here, combined with effects of challenging motor skill training such as tracking training (Carey et al. 2002, 2003) might be more effective than either treatment given alone.
In conclusion, the novel findings of this study were that NMES could be effectively self-administered by stroke patients in a home-use environment in an intensive manner. Repetitive movements, without NMES, did cause an increase in strength, but this increase did not translate into improved hand function nor give evidence of cortical change. Repetitive movements with NMES were effective in producing improvements in hand function and were associated with an increase in the cortical intensity index in the ipsilateral primary sensory cortex. No changes were seen in the motor cortex in either hemisphere, perhaps signifying the need for active engagement in rehabilitation to facilitate motor cortex changes.
References
Abo M, Chen Z, Lai L, Reese T, Bjelke B (2001) Functional recovery after brain lesion-contralateral neuromodulation: an fMRI study. Neuroreport 12:1543–1547
Aizawa H, Inase M, Mushiake H, Shima K, Tanji J (1991) Reorganization of activity in the supplementary motor area associated with motor learning and functional recovery. Exp Brain Res 84:668–671
American Heart Association (2000) Heart and Stroke Statistical Update. In: American Heart Association, Dallas
Aschersleben G, Gehrke J, Prinz W (2001) Tapping with peripheral nerve block, a role for tactile feedback in the timing of movements. Exp Brain Res 136:331–339
Backes WH, Mess WH, van Kranen-Mastenbroek V, Reulen JPH (2000) Somatosensory cortex responses to median nerve stimulation: fMRI effect of current amplitude and selective attention. Clin Neurophysiol 111:1738–1744
Backes WH, Mess WH, van Kranen-Mastenbroek V, Reulen JPH (2002) Somatosensory cortex responses to median nerve stimulation: fMRI effect of current amplitude and selective attention. Clin Neurophysiol 111:1738–1744
Brandstater M (1998) Stroke rehabilitation. In: Gans JDB (ed) Rehabilitation medicine: principles and practice, 3rd edn. Lippincott-Raven, Philadelphia, pp 1165–1190
Butefisch C, Davis B, Wise S, Sawaki L, Kopylev L, Classen J, Cohen LG (2000) Mechanisms of use-dependent plasticity in the human motor cortex. Proc Natl Acad Sci U S A 97:3661–3665
Calautti C, Baron JC (2003) Functional neuroimaging studies of motor recovery after stroke in adults—a review. Stroke 34:1553–1566
Calautti C, Leroy G, Guincestre JY, Baron JC (2001) Dynamics of motor network overactivation after striatocapsular stroke: a longitudinal PET study using a fixed-performance paradigm. Stroke 32:2534–2542
Cao Y, D’Olhaberriague L, Vikingstad EM, Levine SR, Welch KMA (1998) Pilot study of functional MRI to access cerebral activation of motor function after poststroke hemiparesis. Stroke 29:112–122
Caramia MD, Iani C, Bernardi G (1996) Cerebral plasticity after stroke as revealed by ipsilateral responses to magnetic stimulation. Neuroreport 7:1756–1760
Carey J (1990) Manual stretch: effect on finger movement control and force control in subjects with stroke with spastic extrinsic finger flexor muscles. Arch Phys Med Rehabil 71:888–894
Carey J, Bogard C, King B, Suman V (1994) Finger-movement tracking scores in healthy subjects. Percept Mot Skills 79:563–576
Carey J, Baxter T, Di Fabio R (1998) Tracking control in the nonparetic hand of subjects with stroke. Arch Phys Med Rehabil 79:435–441
Carey JR, Kimberley TJ, Lewis SM, Auerbach E, Dorsey L, Rundquist P, Ugurbil K (2002) Analysis of fMRI and finger tracking training in subjects with chronic stroke. Brain 125:773–788
Carey JR, Anderson KM, Kimberley TJ, Lewis SM, Auerbach EJ, Ugurbil K (2003) fMRI analysis of ankle movement tracking training in subject with stroke. Exp Brain Res (in press)
Carr LJ, Harrison LM, Stephens JA (1994) Evidence for bilateral innervation of certain homologous motorneuron pools in man. J Physiol 475:217–227
Cauraugh J, Light K, Kim S, Thigpen M, Behrman A (2000) Chronic motor dysfunction after stroke: recovering wrist and finger extension by electromyography-triggered neuromuscular stimulation. Stroke 31:1360–1364
Chae J, Yu D (2000) A critical review of neuromuscular electrical stimulation for treatment of motor dysfunction in hemiplegia. Assistive Technology 12:33–49
Chae J, Bethoux F, Bohinc T, Dobos L, Davis T, Friedl A (1998) Neuromuscular stimulation for upper extremity motor and functional recovery in acute hemiplegia. Stroke 29:975–979
Cohen MS, DuBois RM (1999) Stability, repeatability, and the expression of signal magnitude in functional magnetic resonance imaging. J Magn Res Imaging 10:33–40
Cramer SC, Finklestein SP, Schaechter JD, Bush G, Rosen, BR (1999) Activation of distant motor cortex regions during ipsilateral and contralateral finger movements. J Neurophysiology 81:383–387
de Kroon JR, van der Lee JH, Ijzerman MJ, Lankhorst GJ (2002) Therapeutic electrical stimulation to improve motor control and functional abilities of the upper extremity after stroke: a systematic review. Clinical Rehabilitation 16:350–360
Desrosiers J, Bravo G, Hebert R, Dutil E, Mercier L (1994) Validation of the Box and Block Test as a measure of dexterity of elderly people: reliability, validity, and norms studies. Arch Phys Med Rehabil 75:751–755
Feys H, De Weerdt W, Selz B, Cox Steck G, Spichger R, Vereeck L, Putman D, Van Hoydonck G (1998) Effect of therapeutic intervention for the hemiplegic upper limb in the acutre phase of stroke. A single-blind, randomized, controlled multicenter trial. Stroke 29:785–792
Fields RW (1987) Electromyographically triggered electric muscle stimulation for chronic hemiplegia. Arch Phys Med Rehabil 68:407–414
Folstein M, Folstein S, McHugh P (1975) “Mini-mental state:” a practical method for grading the cognitive state of patients for the clinician. J Psychiat Res 12:189–198
Francisco G, Chae J, Chawla H, Kirshblum S, Zorowitz R, Lewis G, Pang S (1998) Electromyogram-triggered neuromuscular stimulation for improving the arm function of acute stroke survivors: a randomized pilot study. Arch Phys Med Rehabil 79:570–575
Fraser C, Power M, Hobday D, Hollander I, Tyrell P, Hobson A, Williams S, Thompson D (2002) Driving plasticity in human adult motor cortex is associated with improved motor function after brain injury. Neuron 34:831–840
Friel K, Heddings A, Nudo RJ (2000) Effects of postlesion experience on behavioral recovery and neurophysiologic reorganization after cortical injury in primates. Neurorehabil Neural Repair 14:187–198
Georgopoulos AP, Whang K, Georgopoulos MA, Tagaris GA, Amirikian B, Richter W, Kim SG, Ugurbil K (2001) Functional magnetic resonance imaging of visual object construction and shape discrimination: relations among task, hemispheric lateralization, and gender. J Cogn Neurosci 13:72–89
Hamdy S, Rothwell J, Aziz Q, Singh K, Thompson D (1998) Long-term reorganization of human motor cortex driven by short-term sensory stimulation. Nature Neuroscience 1:64–68
Hummelsheim H, Maier-Loth M, Eickhof C (1997) The functional value of electrical muscle stimulation for the rehabilitation of the hand in stroke patients. Scand J Rehab Med 29:3-10
Jebsen RH, Taylor N, Trieschmann RB, Trotter MJ, Howard LA (1969) An objective and standardized test of hand function. Arch Phys Med Rehabil 50:311–319
Johansson B, Haker E, von Arbin M, Britton M, Langstrom G, Terent A, Ursing D, Asplund K (2000) Acupuncture and transcutaneous nerve stimulation in stroke rehabilitation. Stroke 32:707–713
Jones TA, Schallert T (1994) Use-dependent growth of pyramidal neurons after neocortical damage. J Neurosci 14:2140–2152
Kaelin-Lang A, Luft A, Sawaki L, Burstein A, Sohn Y, Cohen LG (2002) Modulation of human corticomotor excitability by somatosensory input. J Physiol-London 540:623–633
Kampe K, Jones R, Auer D (2000) Frequency dependence of the functional MRI response after electrical median nerve stimulation. Hum Brain Mapp 9:106–114
Khaslavskaia S, Ladouceur M, Sinkjaer T (2002) Increase in tibialis anterior motor cortex excitability following repetitive electrical stimulation of the common peroneal nerve. Exp Brain Res 145:309–315
Kim SG, Hendrick K, Hu X, Merkle H, Ugurbil K (1994) Potential pitfalls of functional MRI using conventional gradient-recalled echo techniques. NMR Biomed 7:69–74
Kleim JA, Vij K, Ballard DH, Greenough WT (1997) Learning-dependent synaptic modifications in the cerebellar cortex of the adult rat persist for at least four weeks. J Neurosci 17:717–721
Kraft GH, Fitts SS, Hammond MC (1992) Techniques to improve function of the arm and hand in chronic hemiplegia. Arch Phys Med Rehabil 73:220–227
Lewis SM, Jerde TA, Tzagarakis C, Tsekos N, Amirikian B, Georgopoulos MA, Kim S-G, Ugurbil K, Georgopoulos AP (2002) Logarithmic transformations for BOLD fMRI data. Society for Neuroscience Abstracts
Mathiowetz V, Volland G, Kashman N, Weber K (1985) Adult norms for the Box and Block Test of manual dexterity. Am J Occup Ther 39:386–391
McKay DR, Ridding MC, Thompson PD, Miles TS (2002) Induction of persistent changes in the organisation of the human motor cortex. Exp Brain Res 143:342–349
Miltner WH, Bauder H, Sommer M, Dettmers C, Taub E (1999) Effects of constraint-induced movement therapy on patients with chronic motor deficits after stroke: a replication. Stroke 30:586–592
Nakayama H, Jorgensen HS, Raaschou HO, Olsen TS (1994) Compensation in recovery of upper extremity function after stroke: the Copenhagen Stroke Study. Arch Phys Med Rehabil 75:852–857
Nelles G, Spiekermann G, Jueptner M, Leonhardt G, Muller S, Gerhard H, Diener HC (1999) Evolution of functional reorganization in hemiplegic stroke: a serial positron emission tomographic activation study. Ann Neurol 46:901–909
Nelles G, Jentzen W, Jueptner M, Muller S, Diener HC (2001) Arm training induced brain plasticity in stroke studied with serial positron Emission Tomography. NeuroImage 13:1146–1154
Nudo R, Milliken G (1996) Reorganization of movement representations in primary motor cortex following focal ischemic infarcts in adult squirrel monkeys. J Neurophys 75:5:2144–2149
Nudo R, Milliken G, Jenkins W, Merzenich M (1996a) Use-dependent alterations of movement representations in primary motor cortex of adult squirrel monkeys. J Neurosci 16(2): 785–807
Nudo R, Wise, Birukte M, SiFuentes, Milliken F, Garrett W (1996b) Neural substrates for the effects of rehabilitative training on motor recovery after ischemic infarct. Science 272:1791–1794
Nudo RJ, Plautz EJ, Frost S (2001) Role of adaptive plasticity in recovery of function after damage to motor cortex. Muscle Nerve 24:1000–1019
Ono M, Kubik S, Abernathey CD (1990) Atlas of the cerebral sulci. Thieme Medical Publishers, New York
Panizza M, Nilsson J, Roth BJ, Basser PJ, Hallett M (1992) Relevance of stimulus duration for activation of motor and sensory fibers; implications for the study of H-reflexes and magnetic stimulation. Electroenceph Clin Neurophysiol 85:22–29
Plautz EJ, Milliken GW, Nudo RJ (2000) Effects of repetitive motor training on movement representations in adult squirrel monkeys: role of use versus learning. Neurobiol Learn Mem 74:27–55
Powell J, Pandyan A, Granat M (1999) Electrical stimulation of wrist extensors in poststroke hemiplegia. Stroke 30:1384–1389
Reding MJ, Potes E (1998) Rehabilitation outcome following initial unilateral hemispheric stroke. Life table analysis approach. Stroke 19:1354–1358
Remple MS, Bruneau RM, VandenBerg PM, Goertzen C, Kleim JA (2001) Sensitivity of cortical movement representations to motor experience: evidence that skill learning but not strength training induces cortical reorganization. Behav Brain Res 123:133–141
Ridding MC, Brouwer B, Miles TS, Pitcher JB, Thompson PD (2000) Changes in muscle responses to stimulation of the motor cortex induced by peripheral nerve stimulation in human subjects. Exp Brain Res 131:135–143
Ridding MC, McKay DR, Thompson PD, Miles TS (2001) Changes in corticomotor representations induced by prolonged peripheral nerve stimulation in humans. Clin Neurophysiol 112:1461–1469
Smith GV, Alon G, Roys SR, Gullapalli RP (2003) Functional MRI determination of a dose-response relationship to lower extremity neuromuscular electrical stimulation in healthy subjects. ExpBrain Res 150: 33–39
Sonde L, Gip C, Fernaeus S, Nilsson C, Viitanen M (1998) Stimulation with low frequency (1/7 Hz) transcutaneous electric nerve stimulation increases motor function of the post-stroke paretic arm. Scand J Rehab Med 30:95–99
Spiegel J, Tintera J, Gawehn J, Stoeter P, Treede R (1999) Functional MRI of human primary somatosensory and motor cortex during median nerve stimulation. Clin Neurophysiol 110:47–52
Stefen K, Kunesch E, Cohen LG, Benecke R, Classen J (2000) Induction of plasticity in the human motor cortex by paired associative stimulation. Brain 123:572–584
Strupp J (1996) Stimulate: a GUI based, fMRI analysis software package. NeuroImage 3:S607
Talairach J, Tournoux P (1988) Co-planar stereotoxic atlas of the human brain. Thieme, New York
Taub E, Miller NE, Novack TA, Cook EWI, Fleming WC, Nepomuceno CS (1993) Technique to improve chronic motor deficit after stroke. Arch Phys Med Rehabil 74:347–354
Ugurbil K, Ogawa S, Kim SG, Chen W, Zhu XH (1999) Imaging brain activity using nuclear spins. In: Maraviglia B (ed) Magnetic resonance and brain function: approaches from physics. IOS Press, Amsterdam, pp 261–310
Vaughan JT, Adriany G, Garwood M, Andersen P, Ugurbil K (2001) The head cradle: an open faced, high performance TEM coil. In: Proceedings of the 9th Annual Meeting of ISMRM, Glasgow, p 15
Waldvogel D, van Gelderen P, Immisch I, Pfeiffer C, Hallett M (2000) The variability of serial fMRI data: correlation between a visual and a motor task. Neuroreport 11:3843–3847
Weiller C, Ramsay SC, Wise RJS, Friston KJ, Frackowiak RSJ (1993) Individual patterns of functional reorganization in the human cerebral cortex after capsular infarction. Ann Neurol 33:181–189
Xiong J, Gao J, Lancaster J, Fox P (1995) Clustered pixels analysis for functional MRI activation studies of the human brain. Hum Brain Mapp 3:287–301
Ziemann U, Ishii K, Borgheresi A, Yaseen Z, Battaglia F, Hallett M, Cincotta M, Wassermann EM (1999) Dissociation of the pathways mediating ipsilateral and contralateral motor-evoked potentials in human hand and arm muscles. J Physiol 518:895–906
Acknowledgements
We gratefully acknowledge the support for this work from the National Institute on Disability and Rehabilitation Research (US Department of Education #H133G010077) and the National Institutes of Health (National Centers for Research Resources P41RR08079 and M01RR00400).
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was completed at the University of Minnesota, Minneapolis, MN 55455, USA
Rights and permissions
About this article
Cite this article
Kimberley, T.J., Lewis, S.M., Auerbach, E.J. et al. Electrical stimulation driving functional improvements and cortical changes in subjects with stroke. Exp Brain Res 154, 450–460 (2004). https://doi.org/10.1007/s00221-003-1695-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-003-1695-y