Abstract
A variety of bulk polymers for the selective separation of chloramphenicol were synthesised from 2-vinylpyridine, diethylaminoethyl methacrylate or methacrylic acid monomers. Chromatographic evaluation indicated that chloramphenicol was retained under nonpolar elution conditions (k = 58.65) through selective hydrogen bonding and ionic interactions. The retention of chloramphenicol under aqueous elution conditions (k > 100) results from nonselective hydrophobic interactions. Under nonpolar elution conditions, the functional monomer employed imparted a significant influence on the recognition properties of the corresponding polymer. After solid-phase extraction using a molecularly imprinted polymer as sorbent and either an organic or aqueous washing solvent, nearly 100% recovery from the chloramphenicol standard solution was achieved, and nearly 90% recovery could be attained from spiked honey samples. The molecularly imprinted polymer was well suited to suppress matrix effects, and provided optimal preconcentration of the target molecule (chloramphenicol) prior to chromatographic analysis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chloramphenicol (CAP) is a broad-spectrum antibiotic that was first isolated in 1947 from cultures of Streptomyces venezuelae [1, 2]. It exerts its antimicrobial effect by inhibiting bacterial protein synthesis through binding at the 50S ribosomal subunit, thereby interfering with the requisite peptidyl transferase. It is a potent drug for the treatment of several infectious diseases including bacterial meningitis and typhoid fever. However, CAP has been shown to possess several harmful side effects in humans, such as Grey syndrome, bone marrow suppression, and fatal aplastic anaemia. Due to these health concerns, CAP is only prescribed to humans for life-threatening illnesses that are resistant to other antibiotics. Further, in the EU, application of CAP to food production has been prohibited since 1994 [3]. Moreover, the EU has defined a minimum required performance limit (MRPL) of 0.3 µg kg−1 for CAP in foods of animal origin [4], because a safe level of CAP dosage has yet to be identified. Owing to a broad spectrum of activity, a ready availability and a relatively low cost, the use of CAP in veterinary medicine is still attractive in certain countries. Therefore, sensitive and reproducible detection techniques are needed to control and monitor CAP residues in food.
Over the past several decades, gas chromatography with electron capture detection [5, 6], liquid chromatography with ultraviolet detection [7, 8], thin-layer chromatography [9] and immunoassays [10, 11] have been used to determine CAP levels in various matrices. Today, several reported methods are based on gas chromatography [10, 12] or liquid chromatography [10, 13–15] with mass spectrometric and tandem mass spectrometric detection, respectively. Certain mass spectrometric detection methods involve extraction procedures, such as solid-phase extraction (SPE), for sample cleanup and preconcentration of the target analyte. In recent years, SPE has become commonly employed, as it offers many benefits including convenience, speed, lower cost, and minimal consumption of organic solvents relative to alternative extraction methods. Further, SPE can be readily incorporated into automated analytical procedures.
Molecularly imprinted polymers (MIPs) are capable of meeting the demands of the abovedescribed sample preparation techniques. Molecular imprinting technology is a synthetic approach that imitates natural molecular recognition (for reviews see [16–18]). The imprinting process is performed by copolymerising functional and cross-linking monomers in the presence of a template molecule that corresponds to a certain target analyte, or has a similar molecular structure. The subsequent removal of the imprint molecule reveals binding sites in the polymer network that are complementary to the template in size, shape and position of the functional groups. This allows for a highly selective rebinding of the target analyte. Additionally, MIPs are reusable, inexpensive to produce, exhibit high mechanical and chemical stability, and are applicable to a number of different operating conditions. The use of MIP-SPE in combination with aqueous washing solvents for the determination of CAP has been previously described in different sample matrices [19–22].
We have previously described the preparation of a CAP-imprinted polymer synthesized from a methacrylic acid functional monomer [20]. This polymer was applied as a sorbent in the SPE of honey samples prior to high-performance liquid chromatography (HPLC). This polymer was able to remove matrix components from a honey sample, and allowed for the extraction of CAP under aqueous elution conditions. In order to further increase the utility of this extraction technique, we have studied several MIPs that were synthesised from different functional monomers and different washing solvents including aqueous and nonpolar solvents. Our results are discussed in the present report.
Experimental
Chemicals
The functional monomers 2-vinylpyridine (VPy), diethylaminoethyl methacrylate (DAM) and methacrylic acid (MAA), and the cross-linking monomer ethylene glycol dimethacrylate (EDMA) were purchased from Sigma-Aldrich Chemie GmbH (Steinheim, Germany). The free radical initiator α,α′-azoisobutyronitrile (AIBN) was provided by Fluka Chemie GmbH (Buchs SG, Switzerland). CAP (99.5% pure) was obtained from Ehrenstorfer GmbH (Augsburg, Germany). Prior to application, MAA was distilled under reduced pressure, and EDMA was purified by extraction from 10% sodium hydroxide, dried over anhydrous magnesium sulfate and distilled under reduced pressure. AIBN was recrystallised from methanol. Acetonitrile (MeCN), methanol and ethyl acetate were gradient grade for liquid chromatography, and chloroform, trifluoroacetic acid (TFA) and sodium chloride were gradient grade for analysis. All were supplied by Merck KGaA (Darmstadt, Germany). Water was demineralised and purified by Seralpur PRO 90C, Seral (Ransbach-Baumbach, Germany).
Polymer preparation
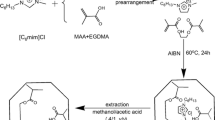
A prepolymerisation solution consisting of 0.42 mmol (0.14 g) CAP, 5 mmol functional monomer (0.54 mL VPy, 1.00 mL DAM, or 0.42 mL MAA), 24.5 mmol (4.62 mL) EDMA, 0.32 mmol (53 mg) AIBN and 7 mL chloroform was prepared in a screw-capped glass vial. The template to monomer molar ratio for the prepared MIPs was 1:12 because of the limited solubility of the template in the porogen chloroform. The molar ratio of the functional monomer and the cross-linking monomer was 1:5. The solution was sonicated for 5 min, and then purged with a stream of nitrogen for 5 min. The polymerisation was thermally initiated at 60 °C, and maintained in a warm-water bath for 24 h. The resulting polymer was a solid block, which was ground and sieved with water. Particles having an average size below 25 µm were collected. In order to remove the template and residues of nonreactive species, the polymer was transferred to a Soxhlet apparatus and heated at reflux over methanol for 6 h. Nonimprinted polymers (NIP) were prepared simultaneously under the same conditions without the addition of the template.
Liquid chromatography
The prepared polymers were slurry packed into stainless steel columns 60 × 4.6-mm ID (CS-Chromatographie Service GmbH, Langerwehe, Germany) using a Hitachi-Pump 655A-12 (Merck KGaA, Darmstadt, Germany). Acetonitrile/water (70:30, v/v) was used as the pushing solvent. A number of mixed mobile phases were investigated as suitable chromatography eluents including methanol/water, acetonitrile/water, and acetonitrile/water containing 0.1% TFA. The mixture ratios were between 100:0 and 10:90 (v/v). Analyses were performed at a flow rate of 1 mL min−1 in an isocratic mode and a detection wavelength of 275 nm. A sample volume of 0.01 mL was injected. The retention factors k = (t r – t 0)/t 0 (t r = retention time of a given analyte; t 0 = retention time of the void volume marker acetone) and the imprinting factors IF = k MIP/k NIP (k MIP = retention factor of the MIP; k NIP = retention factor of the NIP) were calculated.
For CAP quantification, the dried product obtained after SPE was redissolved in 0.2 mL acetonitrile/water (30:70, v/v), and analysis of a 0.01-mL sample was performed on a 125 mm × 4.6-mm ID Superspher®100 RP-18 endcapped column (Merck KGaA, Darmstadt, Germany) at a flow rate of 1 mL min−1 and a detection wavelength of 275 nm. The recoveries of CAP were determined by comparing the analyte peak area ratios with those of an external standard (0.01 µmol mL−1; calibration range 0.0001–0.5 µmol mL−1; correlation coefficient r = 0.9999).
The HPLC system consisted of an HPLC pump P4000 and a UV6000 LP detector from Thermo Separation Products (San Jose, CA, USA). Data were recorded and processed on a Chrom Quest™ Chromatography workstation from Thermo Quest Inc. (San Jose, CA, USA).
SPE
Dry polymers (50 mg for use with aqueous washing solvents and 100 mg for use with nonpolar washing solvents) were packed into empty 10-mL SPE cartridges (Isolute-XL (G), Separtis GmbH, Grenzach-Wyhlen, Germany) between two polyethylene frits (Separtis GmbH, Grenzach-Wyhlen, Germany). Prior to the extraction, the polymer was conditioned by thorough wetting with washing solvent. CAP standard solutions (0.1 mL) or required sample volumes (1 mL) also in washing solvent were applied to the cartridges, which were then washed with the corresponding washing solvent. Finally, elution was carried out with 3 mL methanol at an approximate flow rate of 0.5 mL min−1. The elution fractions were collected and evaporated to dryness for further analysis.
Preparation of honey samples
Details of the sample preparation have been described previously [14]. One gram (± 0.05 g) of a homogenised commercial polyflora honey was weighed into a 10-mL centrifuge tube spiked with 0.1 mL of a standard solution containing 0.01 µmol (3.23 µg) or 0.1 µmol (32.31 µg) CAP, dissolved in 2 mL 4% sodium chloride solution, and combined with 5 mL ethyl acetate. This solution was vortexed for 15 min and then centrifuged at 10,000 g for 5 min. The ethyl acetate was removed from the tube and evaporated to dryness. The residue was redissolved in 1 mL of a mixture of SPE washing solvent (ethyl acetate, acetonitrile or methanol/water 5:95, v/v). This mixture was then loaded onto the SPE cartridge.
Results and discussion
Chromatographic evaluation
Several bulk polymers for the separation of CAP were synthesised using VPy, DAM or MAA as functional monomers. HPLC analyses were performed to evaluate the imprinting effect of the MIPs and to identify the influence of the mobile phase composition on the retention time of CAP.
An increased CAP retention time on the MIP columns relative to the NIP columns was observed with acetonitrile as the mobile phase. As such, the highest imprinting factors were achieved with a nonpolar solvent. Addition of 1% water or 0.1% TFA resulted in a drastic decrease in the retention and imprinting factors (Table 1). Suárez-Rodríguez and Díaz-García have previously demonstrated the disruptive effects of added protic solvents to acetonitrile on the recognition properties of CAP on a CAP-imprinted bulk polymer. Thus, in accordance with the literature [23–25] the analyte was retained on the MIPs under nonpolar elution conditions via CAP-selective hydrogen bonding. The strong decrease in the retention and imprinting factors (Table 1) caused by the addition of 0.1% to acetonitrile implies that ionic interactions are of importance besides hydrogen bondings. The 1,3-diol moiety of CAP is responsible for most of the selective binding under nonpolar elution conditions [23, 24]. Nevertheless, the particular structure of CAP also contributed to the interactions since the substitution of the nitro group (in thiamphenicol) and the dichloroacetyl group (in azidamphenicol) resulted in significant decreases in the affinity for the MIP (results not shown).
The functional monomers used for the synthesis of the corresponding polymers imparted a significant influence on the resulting recognition properties when acetonitrile was used as mobile phase. As shown in Table 1, the MIP synthesized from VPy showed higher retention capabilities and imprinting effects towards CAP than the MIP synthesized from DAM. The retention of CAP in acetonitrile on the MIP synthesized from MAA was relatively low.
In water-rich mobile phases the retention of the target analyte was also significantly affected by the water content (Fig. 1). The retention factor of CAP increased with the water concentration at above 20% water in acetonitrile. On replacing acetonitrile with methanol, the retention of CAP continuously increased with water concentration. The increase of the retention factor obtained with mixtures containing methanol was much greater than with mixtures containing acetonitrile. Thus, CAP was not eluted from the columns within 200 min when the eluent contained more than 80% water in mixtures with methanol, or more than 90% water in mixtures with acetonitrile. The addition of 0.1% TFA to water-rich mobile phases had no influence on the retention behaviour of CAP (results not shown).
The imprinting factors of CAP exhibited values below 2 under aqueous elution conditions, and there were only slight differences between the retention factors on the MIP and NIP columns (Fig. 2). The identities of functional monomers employed in corresponding polymer syntheses showed insignificant effects on the recognition properties. Based on these results, we propose that hydrogen-bonding interactions between CAP and the recognition sites of the polymer are disrupted under these conditions, and that the analyte is retained on the polymer matrix due to additional nonselective hydrophobic interactions.
SPE of CAP from standard solutions
On the basis of the chromatographic evaluations described above, the MIP synthesized from VPy was applied as a sorbent in manual SPE for the selective separation and concentration of CAP. The effects of different washing solvents on these extractions were investigated, and CAP was quantified in each eluted fraction by HPLC. Ethyl acetate and acetonitrile were investigated as SPE washing solvents because they do not interfere with the specific hydrogen bonds formed between CAP and the functional monomer residues within the imprinted cavities. Further, to most effectively harness the nonselective hydrophobic interactions, different mixtures of methanol/water and acetonitrile/water were tested as SPE washing solvents.
After washing with 3 mL ethyl acetate or 1 mL acetonitrile, recoveries of approximately 100% were obtained when the amount of CAP in 0.1 mL was between 0.0025 and 0.01 µmol (Table 2). The recoveries were reproducible with standard deviations below 1.6. We propose that ethyl acetate facilitates the formation of hydrogen bonds more effectively than acetonitrile because it is more nonpolar. As such, the retention of CAP was more pronounced with ethyl acetate (recovery of CAP 98.9% after washing with 5 mL ethyl acetate) than with acetonitrile (recovery of CAP 53.7% after washing with 5 mL acetonitrile) during the washing step. CAP quantities greater than 0.01 µmol resulted in reduced recoveries. These reduced recoveries result from the limited capacity of the MIP to bind selectively to the target analyte since it contains only a limited number of molecular imprints. However, the minimum required performance limit (MRPL) for CAP in food of animal origin is 0.3 µg kg−1 (1 pmol g−1) [4]. Thus, CAP quantities above 0.01 µmol are not of practical importance.
The elution strength of the aqueous washing solvent increased with an increasing content of methanol or acetonitrile due to a disruptive effect on the nonselective hydrophobic interactions (Table 3). With up to 20% methanol or 5% acetonitrile, CAP was strongly retained on the MIP, and recoveries were generally above 98%. Methanol disrupted the hydrophobic interactions to a lesser extent than acetonitrile. Since, the retention of CAP was based on nonselective hydrophobic interactions with the polymer matrix, CAP quantities above 0.01 µmol did not result in reduced recoveries under aqueous conditions, as observed under the nonpolar conditions described previously. Furthermore, the amount of polymer could be reduced from 100 to 50 mg when washing solvents were used that contained water because the hydrophobic interactions under aqueous conditions were much stronger than the nonpolar interactions in acetonitrile or ethyl acetate.
Application of MIP to SPE of CAP from honey samples
The extractions of honey samples were performed under the same conditions as the extractions of the standard solutions. All fractions from the washing step with either ethyl acetate or an aqueous solvent containing up to 20% methanol were found to be free of the analyte (Table 4). Furthermore, under these conditions recoveries of approximately 90% were achieved. After washing with acetonitrile, a 60% recovery was obtained. It is likely that the honey matrix interfered with the recognition of CAP in acetonitrile, which displayed a stronger elution strength relative to both ethyl acetate and mixtures of water/methanol.
The chromatograms obtained with washing and elution fractions obtained by MIP-SPE of a honey sample spiked with CAP are shown in Figs. 3 and 4. A baseline separation of the CAP peak from the honey matrix could be achieved when ethyl acetate was used as washing solvent for preconcentration with MIP-SPE (Fig. 3a). The water-based washing step offered a poor cleanup of the sample because of the unspecific retention of the matrix components. Consequently, less honey matrix was found in the washing fraction 5:95 (v/v) methanol/water (Fig. 4c) than in the washing fraction ethyl acetate (Fig. 3c). Furthermore, a greater concentration of CAP was necessary to identify the CAP peak when 5:95 (v/v) methanol/water was used as washing solvent (Fig. 4a). The chromatograms of honey samples which were not spiked with CAP (Figs. 3b and 4b) indicated that no components of the honey matrix were located at the position of the CAP peak. The improved CAP separation from the honey matrix under the nonpolar conditions further highlights the selective interactions of CAP with the MIP.
Chromatograms (RP-HPLC) obtained by a MIP-SPE, elution fraction of 1 g honey spiked with 0.01 µmol CAP; b MIP-SPE, elution fraction of 1 g honey; c MIP-SPE, washing fraction (3 mL ethyl acetate) of 1 g honey spiked with 0.01 µmol CAP. Conditions for MIP-SPE and HPLC: see footnote of Table 4
Chromatograms (RP-HPLC) obtained by a MIP-SPE, elution fraction of 1 g honey spiked with 0.1 µmol CAP; b MIP-SPE, elution fraction of 1 g honey; c MIP-SPE, washing fraction (5 mL 5:95 (v/v) methanol/water) of 1 g honey spiked with 0.1 µmol CAP. Conditions for MIP-SPE and HPLC: see footnote of Table 4
Conclusion
We have described the first synthesis and subsequent application of a polymer in the separation of CAP in both nonpolar and aqueous solvents systems. The retention of CAP is governed by selective hydrogen bonding in the nonpolar media, and by nonselective hydrophobic interactions in the aqueous media. Further analyses have demonstrated the applicability of the MIP, synthesized with 2-vinylpyridine as functional monomer, as a sorbent in the SPE of CAP from honey samples. This process successfully suppresses matrix effects and preconcentrates the target analyte. During the washing step, a better separation of the honey matrix was achieved with ethyl acetate relative to an aqueous solvent. This further demonstrates the selective interactions of CAP with the MIP under nonpolar conditions. The MIP-SPE/RP-HPLC system employed in this study does not reach the required MRPL of 0.3 µg kg−1 [4]. Therefore, our future studies will be aimed at applying the MIP to an automated online SPE coupled to LC–MS/MS analysis of honey samples and other bee products. The application of nonpolar washing solvents during the MIP-SPE is of special interest.
References
Yunis AA (1988) Ann Rev 28:83–100
Holt D, Harvey D, Hurley R (1993) Adverse Drug React Toxicol Rev 12:83–95
Council Regulation (EEC) No 2377/90, amending regulation No 1430/94 of 22 June 1994, Off J Eur Commun L156 23(6)
Commission Decision 2003/181/EC of 13 March 2003, amending decision 2002/657/, Off J Eur Commun L71/17
Akhtar MH, Danis C, Sauve A, Barry C (1995) J Chromatogr A 696:123–130
Pfenning AP, Roybal JE, Rupp HS, Turnipseed SB, Gonzales SA, Hurlbut JA (2000) J AOAC Int 83:26–30
Roudaut B (1996) J Liq Chromatogr R T 19:1097–1105
Boulaire SL, Bauduret JC, Andre F (1997) J Agric Food Chem 45:2134–2142
Abjean JP (1997) J AOAC Int 80:737–740
Impens S, Reybroeck W, Vercammen J, Courtheyn D, Ooghe S, DeWash K, Smedts W, DeBrabander H (2003) Anal Chim Acta 483:153–163
Scortichini G, Annunziata L, Haouet MN, Benedett F, Krusteva I, Galarini R (2005) Anal Chim Acta 535:43–48
Xie MX, Liu Y, Qiu YM, Han J, Liu YZ (2005) Chin J Anal Chem 33:1–4
Ortelli D, Edder P, Corvi C (2004) Chromatographia 59:61–64
Grotewahl D (2006) Entwicklung von Methoden zur Bestimmung von Chloramphenicol in Bienenprodukten mittels LC-MS. Cuvillier Verlag, Göttingen
Nicolich RS, Werneck-Barroso E, Marques MAS (2006) Anal Chim Acta 565:97–102
Sellergren B (2001) J Chromatogr A 906:227–252
Ye L, Mosbach K (2001) J Incl Phenom Macrocycl Chem 41:107–113
Cormack PAG, Elorza AZ (2004) J Chromatogr B 804:173–182
Mena ML, Agüí L, Martines-Ruiz P, Yáñez-Sedeño P, Reviejo AJ, Pingarrón JM (2003) Anal Bioanal Chem 376:18–25
Schirmer C, Meisel H (2006) J Chromatogr A 1132:325–328
Boyd B, Björk H, Billing J, Shimelis O, Axelsson S, Leonora M, Yilmaz E (2007) J Chromatogr A 1174:63–71
Mohamed R, Richoz-Payot J, Gremaud E, Mottier P, Yilmaz E, Tabet JC, Guy PA (2007) Anal Chem 79:9557–9565
Levi R, McNiven S, Piletsky SA, Cheong SH, Yano K, Karube I (1997) Anal Chem 69:2017–2021
McNiven S, Kato M, Levi R, Yano K, Karube I (1998) Anal Chim Acta 365:69–74
Suárez-Rodríguez JL, Díaz-García ME (2001) Biosens Bioelectron 16:955–961
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schirmer, C., Meisel, H. Molecularly imprinted polymers for the selective solid-phase extraction of chloramphenicol. Anal Bioanal Chem 392, 223–229 (2008). https://doi.org/10.1007/s00216-008-2269-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-008-2269-4