Abstract
An analytical protocol including solid-phase extraction and purification is described for the individual quantification of polycyclic aromatic hydrocarbon metabolites (hydroxylated PAHs) in liquid biological matrices such as plasma and bile. The method consists in an enzymatic deconjugation followed by a solid-phase extraction on a C18 cartridge and by a cleanup on an NH2 cartridge. Extracts are then submitted to a derivatization step before gas chromatography/mass spectrometry (GC/MS) analysis. The quantification of PAH metabolites is ensured by adding an internal standard, 1-hydroxypyrene deuterated, at the beginning of the protocol. Recoveries obtained for the entire protocol were for the major part of the compounds between 96 and 70%. However, recoveries were not so satisfying concerning 2-hydroxybiphenyl and especially 3-hydroxybenzo(a)pyrene, with 62 and 36% respectively. Finally, the protocol was applied to different fish bile samples and showed its good applicability to fish bile samples. The NH2 cleanup step has been proved to be a very selective purification step, necessary to remove most of the bile pigments before GC/MS injection. Different PAH metabolites could be detected in those natural samples and their quantification allowed us to distinguish different levels of fish exposure.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Polycyclic aromatic hydrocarbons (PAHs) are ubiquitous contaminants of the environment [1]. The quantification of PAHs in marine organism tissues is usually carried out to evaluate the impact of such a contamination on organisms. However, depending on the ability of organisms to metabolize PAHs, measurement of the bioaccumulated part of the absorbed PAHs can be restrictive and not representative of the contamination [2–6] owing to their transformation into reactive intermediates. The toxicity of PAHs has been evidenced in different studies concerning fish and also mice and human beings [7–13]. In this way, the study of the fate of PAHs in marine organisms appears to be necessary in order to determine the bioavailability and the toxicity of those contaminants.
The concentration of PAH metabolites is usually determined semiquantitatively as global fluorescent aromatic compound equivalents [14–18]. However, some studies also reported quantitative determination of metabolite concentrations [19–21]. This work presents a solid-phase extraction (SPE) based protocol including an essential purification step for the quantitative determination of individual PAH metabolites in the bile of fish. Very few studies have reported the use of a SPE protocol for the analysis of PAH metabolites [22–24]. Moreover, no purification step using SPE cartridges has been used. The metabolites studied, monohydroxylated PAHs, are known to be some of the major metabolites produced by fish, and are present in their free form or more commonly conjugated with glucuronide, sulfate, or glutathione groups [22, 25–28], and are extensively excreted into the gall bladder [22, 26, 29–31]. The following compounds were chosen because they are potential metabolites of priority PAHs listed by the United-States Environmental Protection Agency for their interest in environmental surveys [32]: 1- and 2-hydroxynaphthalene, 9-hydroxyfluorene, 9-hydroxyphenanthrene, 1-hydroxypyrene, 1-hydroxychrysene and 3-hydroxybenzo(a)pyrene (Fig. 1). 2-Hydroxybiphenyl was added to this study owing to the abundance of biphenyl in the environment.
Materials and methods
Chemicals, solvents and reagents
1-and 2-Hydroxynaphthalene were purchased from Lancaster (Bischheim, France), 2-hydroxybiphenyl, 9-hydroxyfluorene and 1-hydroxypyrene from Sigma-Aldrich (Saint Quentin Falavier, France) and 9-hydroxyphenanthrene, 1-hydroxychrysene and 3-hydroxybenzo(a)pyrene from Promochem (Molsheim, France). 1-Hydroxypyrene-d9 and phenanthrene-d10 were obtained from Cambridge Isotope Laboratories (Andover, MA, USA). Ultra Resi-Analysed methylene chloride (Mallinckrodt Baker) and Bakerbond C18 cartridges (500 mg) were provided by Atlantic Labo (Eysines, France), methanol Suprasolv and acetic acid 100% Normapur by VWR (Strasbourg, France), Supelclean LC-NH2 cartridges (500 mg), sodium acetate trihydrate 99% and β-glucuronidaseand aryl sulfatase mixture, 100,000 and 7,500 u/ml, respectively, by Sigma-Aldrich (Saint Quentin Falavier, France) and water (sterilized water for injection) by Aguettant (Lyon, France). 2-Mercaptoethanol and bis(trimethylsylil)trifluoroacetamide (BSTFA) was purchased from Acros Organics (Noisy-Le-Grand, France).
Enzymatic deconjugation and C18 SPE
In order to perform the recovery tests, spiked water was used to mimic a natural liquid sample. To this purpose, a mixture of hydroxylated PAHs was prepared in methanol. Then a dilution was made in buffered water (acetate buffer, pH 5) to obtain spiked water samples of around 1 μg of each compound per gram of solution. The internal standard (1-hydroxypyrened9), 20 μl of 2-mercaptoethanol, used as an antioxidant, and 20 μl of β-glucuronidase and aryl sulfatase mixture were added to the water samples. They were then placed at 37°C in an oven for 20 h for hydrolysis following a modification of the method of Khran et al. [19]. 2-Mercaptoethanol was added at the beginning of the protocol to avoid oxidation phenomena occurring. Glass vials were used for the deconjugation step to avoid adsorption phenomena on inner surfaces.
Spiked water samples were ultrasonicated and centrifuged (5 min, 5,000 rpm, 18°C) after deconjugation and BakerBond C18 cartridges were preconditioned with 2 ml methanol and 4 ml water. The supernatants were loaded on cartridges and washed with 2 ml water and 2ml water/methanol mixture (70:30, v/v). Water traces were then removed by freeze-drying the cartridges. Freeze-drying was performed because of the possible loss of low molecular weight compounds when drying under nitrogen stream at room temperature. Metabolites were then eluted with 6 ml methanol. Extracts were reduced to dryness under nitrogen stream and redissolved in methylene chloride for derivatization and direct injection or in methanol/methylene chloride (20:80, v/v) before NH2 cleanup.
NH2 cleanup
The tests were done with spiked methanol solutions prepared by dilution of the concentrated solution previously prepared. Supelclean LC-NH2 cartridges were used for this purification step. The spiked methanol solutions were first diluted in methylene chloride in order to obtain 1 ml of solution (20:80, v/v). The internal standard and 40 μl of 2-mercaptoethanol were then added. Before loading the samples, the cartridges were first preconditioned with 3 ml methanol/methylene chloride (20:80, v/v). The elution of the compounds was then performed with 6 ml of the same mixture. Extracts were finally reduced to dryness under a nitrogen stream and redissolved in methylene chloride.
Derivatization
A derivatization step using BSTFA was required for reliable detection by gas chromatography/Mass spectrometry (GC/MS) (Mazéas and Budzinski, unpublished data). To this purpose, solutions were kept for 30 min in an oven at 65°C after adding 30 μl of BSTFA.
GC/MS analysis
After adding phenanthrene-d10, prepared in methylene chloride, for recovery control, analyses were performed by GC/MS, using an Agilent Technologies HP GC 6890A gas chromatograph coupled to an Agilent Technologies HP MSD 5973 network mass selective detector both purchased from BIOS Analytique (Union, France), in selected ion monitoring (SIM) mode (automated PTV pulsed splitless injection (25 psi, 1.5 min); purge delay 1.5 min and purge flow 60ml/min; injector temperature 250°C; interface temperature:280°C; oven temperature from 70°C (2 min) to 180°C (1 min) at 5°C/min and from 180° to 290°C (1 min) at 10°C/min; electron impact 70 eV; voltage 2,000 V; source temperature 150°C; 1.53 scan/s; dwell time 50 ms for each ion). The capillary column was an Agilent Technologies HP5/MS (30 m, 0.25 mm; phase 5% diphenylsiloxane, 95% dimethylsiloxane) obtained from BIOS Analytique (l'Union, France) and the carrier gas was helium 6.0 from Linde (Toulouse, France).
The compounds were quantified in SIM mode using the ions 1- hydroxynaphthalene (m/z=201), 2-hydroxynaphthalene (m/z=216), 2-hydroxybiphenyl (m/z=211), 9-hydroxyfluorene (m/z=165), 9-hydroxyphenanthrene (m/z=266), 1-hydroxypyrene (m/z=290), 1-hydroxychrysene (m/z=316) and 3-hydroxybenzo(a)pyrene (m/z=340) (Mazéas and Budzinski, unpublished results).
A mixture constituted of the standard metabolites 1-hydroxypyrene-d9 and phenanthrene-d10 was injected, after derivatization, at the beginning and at the end of each analytical sequence for the determination of response factors. A manipulation blank was performed together with samples for each manipulation series to control potential contamination. The glassware was washed with detergents and heated overnight at 450°C before use.
Natural samples
European flounders (Platichthys flesus) were collected in the Seine Bay (Channel Coast of France) in two locations. One of these locations is the mouth of the Seine Estuary, a place particularly industrialized, and the other one is Antifer, located in the north of the Bay, off a harbor carrying important petroleum activities. Four and ten flounders were collected, respectively . Gall bladders were sampled and stored at -80°C.
Results and discussion
Deconjugation and C18 SPE
The mean recoveries obtained for enzymatic deconjugation followed by C18 SPE are indicated in Table 1. The recoveries range between 82 and 110% for the major part of the compounds; however, for 2-hydroxybiphenyl and the 3-hydroxybenzo(a)pyrene, they are not so satisfying, showing values of 65 and 41%, respectively. The recovery obtained for the internal standard, quantified in comparison with phenanthrene-d9 used as a syringe standard, is 79±5% (n=3). No increase of recoveries is observed by a simple water rinsing (removing methanol) or by using a higher volume of elution. Moreover, recoveries with and without a deconjugation step are similar (Table 1), indicating that the deconjugation step does not induce any significant losses. Those results suggest that 2-hydroxybiphenyl and 3-hydroxybenzo(a)pyrene are partially eluted by water during the rinsing step or too strongly adsorbed on the C18 phase to be eluted with the elution conditions that are used. Increasing the percentage of methanol over 30% during the washing step results in a better purification (removal of more polar compounds) but causes partial elution of the two hydroxynaphthalene isomers and of the 2-hydroxybiphenyl, leading to lower recoveries.
The standard deviations obtained are in the range 3–10% (n=3), except for that of 3-hydroxybenzo(a)pyrene (11%), showing the good reproducibility of the method.
NH2 cleanup
Owing to the nature of the bile extracts obtained after SPE, a purification step was developed. The mean recoveries obtained for the NH2 cleanup step are summarized in Table 2. The recoveries were between 77 and 92%. 2-Hydroxybiphenyl and 3-hydroxybenzo(a)pyrene do not exhibit lower recoveries than other compounds for this step. The recovery obtained for the internal standard is 90±3% (n=3).
Standard deviations are in the range 3–8% (n=3), which is overall slightly better than for the extraction step.
Combined protocol
Spiked waters are then used to test the entire protocol (Fig. 2). The mean recoveries are given in Table 3. They are usually greater than 70% except for 2-hydroxybiphenyl (62%) and 3-hydroxybenzo(a)pyrene (36%) as expected considering the extraction recoveries. The recovery of the internal standard is 74±4% (n=3).
Considering standard deviations, the values are in the range 3–8% (n=3), showing the good reproducibility of the whole protocol. It can be noticed that 1-hydroxypyrene shows the lowest standard deviation of all the compounds studied, underlying the interest in using compounds with similar properties (such as deuterated homologues) for the most accurate quantification. The limits of quantification (S/N>10) were rather good for all the compounds except for 3-hydroxybenzo(a)pyrene, which comprised between 1 and 3 ng/g of bile. In the case of 3-hydroxybenzo(a)pyrene the quantification is higher (30 ng/g) owing to chromatographic discrimination.
Fish bile
The entire protocol was applied to the bile of flounders collected from the Seine Bay. Bile samples were analyzed in triplicates of approximately 100 μl. After the sample loading on the C18 cartridge, the rinsing step with water and more particularly with water/methanol showed the removal of matrix constituents, which resulted in a clearer extract being obtained and a slower clogging of the GC/MS than with water rinsing alone. However, the extract obtained remained strongly concentrated in bile pigments. The NH2 purification step with methanol/methylene chloride elution (20:80, v/v) proved to be efficient, ensuring a strongly selective elution of the metabolites with retention of most of the bile pigments in the first millimeters of the phase.
After GC/MS analysis, different metabolites could be detected and quantified in the bile. 1- and 2-Hydroxynaphthalene, 2-hydroxybiphenyl, 9-hydroxyfluorene and 1-hydroxypyrene were identified, as shown in the chromatogram presented in Fig. 3. 9-Hydroxyphenanthrene was not detected but three other isomers (1-, 2- and 3-hydroxyphenanthrene) could be quantified (Mazéas and Budzinski, unpublished results). 1-Hydroxychrysene and 3-hydroxybenzo(a)pyrene were not detected in any sample either. The average concentrations, standard deviations and coefficients of variation obtained for the two samples are presented in Table 4. The average recovery for 1-hydroxypyrene-d9 (76±5%) is not significantly different from the results obtained with spiked water (74±4%). The results obtained indicate that the variations of concentration are correct, and are generally between 3 and 14%. The results suggest a stronger exposure of flounders from the Seine mouth (La Fosse) to PAHs (global concentration of 3,432±149 ng g) than flounders from Antifer (global concentration of 545±33 ng g−1). For samples from La Fosse the relative abundance of 1-hydroxypyrene is much more important than for Antifer samples (81% and 55%, respectively). This high proportion of 1-hydroxypyrene could be related to a combustion-type input at La Fosse in comparison with a petroleum input at Antifer. But before a definitive conclusion is reached more samples are needed and comparison with other systems is also necessary. Nevertheless the relative distribution of 1-hydroxypyrene could be used as an indication of source pollution.
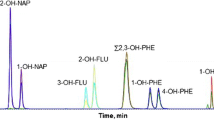
Chromatogram obtained for a fish bile sample by gas chromatography/mass spectrometry in selected ion monitoring SIM mode. 1-OHN 1-hydroxynaphthalene, 2-OHN 2-hydroxynaphthalene, 2-OHBi 2-hydroxybiphenyl, Phe-d10 phenanthrene-d 10, 9-OHFe 9-hydroxyfluorene, 3-OHPhe 3-hydroxyphenanthrene, 1-OHPhe 1-hydroxyphenanthrene, 2-OHPhe 2-hydroxyphenanthrene, 1-OHP-d9 1-hydroxypyrene-d 9, 1-OHP 1-hydroxypyrene
Conclusion
The protocol presented in this study allows the individual quantification of hydroxylated PAHs in liquid matrices such as fish bile by GC/MS analysis after deconjugation, extraction and purification steps. Recoveries for the whole protocol are generally greater than 70%, with standard deviations lower than 10% (n=3). However, owing to the extraction step, 2-hydroxybiphenyl and particularly 3-hydroxybenzo(a)pyrene recoveries are not so satisfying. Conversely, the NH2 cleanup step has been proved to be essential to remove bile pigments before GC/MS injection. The application of the protocol to bile samples from European flounders collected from the Seine Bay has shown its applicability to natural samples, allowing a good quantification of the metabolites with correct reproducibility.
References
McElroy AE, Farrington JW, Teal JM (1989) Varanasi U (ed) Metabolism of polycyclic aromatic hydrocarbons in the aquatic environment. CRC Press, Boca Raton, FL, pp 1–40
Varanasi U, Chan S-L, MacLeod WD, Stein JE, Brown DW, Burrows DG, Tilbury KL, Landahl JT, Wigren CA, Hom T, Pierce SM (1990) NOAA technical memorandum NMFS F/NWC-191. US Department of Commerce, Seattle, WA
McDonald SJ, Kennicutt MC, Liu H, Safe SH (1995) Arch Environ Contam Toxicol 29:232–240
Baumard P, Budzinski H, Garrigues P, Sorbe JC, Burgeot T, Bellocq J (1998) Mar Pollut Bull 36:951–960
Nakata H, Sakai Y, Miyawaki T, Takemura A (2003) Environ Sci Technol 37:3513–3521
Meador JP (2003) In: Douben PET (ed) PAHs: an ecotoxicological perspective. Wiley, Chichester, pp 147–172
International Agency for Research on Cancer (1983) Monographs on the evaluation of the carcinogenic risk of chemicals to humans. International Agency for Research on Cancer, Lyon, France
Pahlman R, Pelkonen O (1987) Carcinogenesis 8:773–778
De Flora S, Bagnasco M, Zanacchi P (1991) Mutat Res 258:285–320
Moore MJ, Myers MS (1994) Malins DC, Ostrander GK (eds) Aquatic toxicology: molecular, biochemical, and cellular perspectives. Lewis, Boca Raton, FL, pp 327–386
IPCS (1998) Environmental health criteria 202. International programme on chemical safety. World Health Organization, Geneva, Switzerland
National Toxicology Program (1999) The 9th report on carcinogens. US Department of Health and Human Services, Public Health Service National Toxicology Program, Washington, DC
Johnson LL, Collier TK, Stein JE (2002) Aquat Conserv Mar Freshw Ecosyst 12:517–538
Krahn MM, Myers M, Burrows DG, Malins DC (1984) Xenobiotica 14:633–646
Krahn MM, Rhodes LD, Myers MS, Moore LK, MacLeod WD, Malins DC (1986) Arch Environ Contam Toxicol 15:61–67
Ariese F, Kok SJ, Verkaik M, Gooijer C, Velthorst NH, Hofstraat JW (1993) Aquat Toxicol 26:273–286
Lin ELC, Cormier SM, Torsella JA (1996) Ecotoxicol Environ Saf 35:16–23
Beyer J, Sandvik M, Hylland K, Fjeld E, Egaas E, Aas E, Skåre JU, Goksøyr A (1996) Aquat Toxicol 36:75–98
Khran MM, Burrows DG, MacLeod WD, Malins DC (1987) Arch Environ Contam Toxicol 16:511–522
Escartín E, Porte C (1999) Mar Pollut Bull 38:1200–1206
Ruddock PJ, Bird DJ, McCalley DV (2002) Ecotoxicol Environ Saf 51:97–105
Hellou J, Payne JF (1987) Environ Toxicol Chem 6:857–862
Law FC, Meng JX, He YT, Chui YC (1994) Xenobiotica 24:221–229
Willett KL, Gardinali PR, Lienesch LA, Di Giulio RT (2000) Toxicol Sci 58:68–76
Varanasi U, Stein JE, Nishimoto M (1989) In: Varanasi U (ed) Metabolism of polycyclic aromatic hydrocarbons in the aquatic environment. CRC, Boca Raton, FL, pp 93–149
Foureman GL (1989) In: Varanasi U (ed) Metabolism of PAH in the aquatic environment. CRC, Boca Raton, FL, pp 185–202
Steward AR, Kandaswami C, Chdambaram S, Ziper C, Rutkowski JP, Sikka HC (1990) Environ Toxicol Chem 9:1503–1512
Leonard JD, Hellou J (2001) Environ Toxicol Chem 20:618–623
Lee RF, Sauerheber R, Dobbs GH (1972) Mar Biol 17:201–208
Statham CN, Melancon MJ, Lech JJ (1976) Science 193:680–681
Varanasi U, Stein JE (1991) Environ Health Perspect 90:93–100
Keith LH, Telliard WA (1979) Environ Sci Technol 13:416–423
Acknowledgements
This research was supported in part by the Erika program (Monitoring of the ecological and ecotoxicological consequences of the Erika wreckage), the Region Aquitaine, the Seine-Aval program and the “Biological Effects of Environmental Pollution in Marine Coastal Ecosystems (BEEP)” European project no. EVK3-CT2000-00543.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mazéas, O., Budzinski, H. Solid-phase extraction and purification for the quantification of polycyclic aromatic hydrocarbon metabolites in fish bile. Anal Bioanal Chem 383, 985–990 (2005). https://doi.org/10.1007/s00216-005-0096-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-005-0096-4