Abstract.
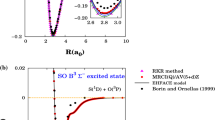
The phenyl cation is known to have two low-energy minima, corresponding to 1 A 1 and 3 B 1 states, the first of which is more stable by ca. 25 kcal/mol. The minimum energy crossing point between these two surfaces, located at various levels including a hybrid method first described here, lies just above the minimum of the triplet, 0.12 kcal/mol at the CCSD(T)/cc-pVDZ//B3LYP/SV level, and there is significant spin-orbit coupling between the surfaces at this point. On the basis of these results, the lifetime of the triplet is expected to be very short.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 24 October 1997 / Accepted: 25 November 1997
Rights and permissions
About this article
Cite this article
Harvey, J., Aschi, M., Schwarz, H. et al. The singlet and triplet states of phenyl cation. A hybrid approach for locating minimum energy crossing points between non-interacting potential energy surfaces. Theor Chem Acc 99, 95–99 (1998). https://doi.org/10.1007/s002140050309
Issue Date:
DOI: https://doi.org/10.1007/s002140050309