Abstract
Rationale
The multidrug resistance transporter, P-glycoprotein (P-gp), is involved in efflux transport of several antipsychotics in the blood–brain barrier (BBB).
Objectives
In the present study, we evaluated the inhibitory effect of the antipsychotics, i.e., risperidone, olanzapine, quetiapine, clozapine, haloperidol, chlorpromazine, a major metabolite of risperidone, 9-OH-risperidone, and a positive control inhibitor, PSC833, on the cellular uptake of a prototypic substrate of P-gp, rhodamine (Rhd) 123, in LLC-PK1 and L-MDR1 cells.
Materials and methods
After incubation of the antipsychotics (1–100 μM) and the positive (10 μM PSC833) or negative (1% dimethyl sulfoxide) controls with 5 μM Rhd 123 for 1 h, the effects of the antipsychotics on the intracellular accumulation of Rhd 123 were examined using a flow cytometric method.
Results
All the antipsychotics showed various degrees of inhibitory effects on P-gp activity. The rank order of the concentration of inhibitor to cause 50% of the maximal increment of intracellular Rhd 123 fluorescence (EC50) was: PSC833 (0.5 μM) < olanzapine (3.9 μM) < chlorpromazine (5.8 μM) < risperidone (6.6 μM) < haloperidol (9.1 μM) < quetiapine (9.8 μM) < 9-OH-risperidone (12.5 μM) < clozapine (30 μM). Considering that the antipsychotics’ plasma concentrations are generally lower than 1 μM, the present results suggest that olanzapine and risperidone are the only agents that may inhibit P-gp activity in the BBB. However, most of the antipsychotics are extensively accumulated in tissues. In addition, when given orally, the drug concentrations in the gastrointestinal tract are likely to be high.
Conclusions
Pharmacokinetic interactions due to inhibition of P-gp activity by the antipsychotics appear possible and warrant further investigation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The ABCB1 transporter encoding for P-glycoprotein (P-gp) confers a multidrug resistance phenotype to some cancer cells that become resistant to chemotherapy (Riordan et al. 1985; Roninson et al. 1986). In addition to expression in tumor cells, P-gp is also localized in a variety of normal human tissues including the apical membranes of the gastrointestinal tract, the biliary canalicular membranes of hepatocytes, the luminal membranes of proximal tubular epithelial cells in the kidney, and the plasma membranes of brain capillary endothelial cells forming blood–brain barrier (BBB). P-gp in these tissues functions as a drug efflux pump greatly affecting substrate absorption, distribution, and excretion (Ayrton and Morgan 2001). In the intestine and brain, the role of P-gp is extruding drugs from entering into the systemic circulation and the central nervous system (CNS), thereby significantly attenuating drug bioavailability and brain penetration. Modulation of P-gp function by P-gp inhibitors or inducers was associated with clinically significant drug–drug interactions (Ayrton and Morgan 2001). Drug–drug interactions resulting from inhibition of P-gp in the gastrointestinal tract have been observed with P-gp inhibitors such as ketoconazole (Kageyama et al. 2005), clarithromycin (Tanaka et al. 2003), verapamil (Pedersen et al. 1983a; Verschraagen et al. 1999), quinidine (Dahlqvist et al. 1980; Pedersen et al. 1983b; Angelin et al. 1987), and itraconazole (Partanen et al. 1996; Jalava et al. 1997). Clinically significant drug–drug interactions at the BBB have been difficult to demonstrate when we rely on pharmacodynamic and toxicodynamic changes. However, one example is a clinically significant interaction between quinidine and loperamide. The resultant respiratory depression, a CNS adverse effect after combined treatment of loperamide and quinidine, was attributed to the significant increase of CNS concentrations of loperamide through inhibition of P-gp function in BBB by quinidine (Sadeque et al. 2000).
The atypical antipsychotic agents, i.e., risperidone, olanzapine, quetiapine, and clozapine, are widely used for patients with severe psychotic disorders. In our previous ATPase studies using recombinant P-gp membranes, we have confirmed that several atypical antipsychotics (AAPs), i.e., quetiapine, risperidone, and olanzapine, showed high binding affinities for P-gp (Boulton et al. 2002). In subsequent P-gp knockout mouse experiments, we further confirmed that the brain penetration of risperidone, a major pharmacologically active metabolite of risperidone, 9-OH-risperidone, and olanzapine are greatly limited by P-gp in the BBB (Wang et al. 2004a,b). However, these studies did not address whether the antipsychotics can inhibit the function of P-gp, but determined their status as substrates.
Despite being well-recognized as having a fairly low propensity for causing cytochrome P450-related drug–drug interactions (Prior and Baker 2003; DeVane and Nemeroff 2001), the AAPs have not been thoroughly assessed for their drug interaction potentials with P-gp. In the present study, we evaluated the inhibitory effects of various concentrations (0.1–100 μM) of four atypical antipsychotics, risperidone, olanzapine, clozapine, quetiapine, and 9-OH-risperidone, as well as two conventional antipsychotics, haloperidol and chlorpromazine, on P-gp activity in LLC-PK1/L-MDR1 cells using a flow cytometric method.
Materials and methods
Materials
Risperidone, 9-OH-risperidone, and methyl-risperidone were obtained from Janssen Pharmaceutica (Titusville, NJ, USA). Olanzapine and quetiapine were obtained from Eli Lilly (Indianapolis, IN, USA) and AstraZeneca Pharmaceuticals (Wilmington, DE, USA), respectively. Haloperidol, chlorpromazine hydrochloride, clozapine, vincristine, and rhodamine 123 (Rhd 123) were purchased from Sigma Chemical (St. Louis, MO, USA). PSC833 was obtained from Novartis Pharmaceutical (Basel, Switzerland). Fetal bovine serum, trypsin and Dulbecco’s modified eagle medium (DMEM) were obtained from Hyclone (Logan, UT, USA). MEM nonessential amino acid solution was obtained from Stem Cell Technologies (Vancouver, BC, Canada). Dulbecco’s phosphate-buffered saline (DPBS), penicillin, and streptomycin were purchased from Mediatech (Herndon, VA). All other reagents were of the purest grade available.
LLC-PK1 and L-MDR1 cells
The L-MDR1 cell line was generated by transfection of the porcine kidney epithelial cell line LLC-PK1 (American Type Culture Collection, Manassas, VA, USA) with human MDR1 gene (Schinkel et al. 1996). The LLC-PK1 and L-MDR1 cell lines were kindly provided by Dr. Kari T. Kivistö (Stuttgart, Germany). The cells were cultured under standard cell culture conditions with Dulbecco’s modified eagle medium (DMEM) containing 4,500 mg/l glucose, 4 mM l-glutamine, and sodium pyruvate (Hyclone) supplemented with 10% fetal bovine serum, 1% MEM nonessential amino acids, 100 U/ml penicillin, and 100 μg/ml streptomycin at 37°C in an atmosphere of 5% CO2 and 95% relative humidity. The LLC-PK1 and L-MDR1 cells were cultured under identical conditions except that L-MDR1 cells were cultured in media containing 0.64 μM vincristine to maintain P-gp expression (Smit et al. 1998).
Sample preparation
LLC-PK1 and L-MDR1 cells were grown to 80 to 90% confluence and harvested with 0.25% trypsin/2.21 mM EDTA in HBSS for drug accumulation experiment. The cells were suspended in DMEM containing 5 μM Rhd 123 and filtrated through a 70-μm nylon mesh screen (Spectrum Laboratory, New Brunswick, NJ, USA). The cell numbers were approximately 106 in each reaction in a final incubation volume of 0.5 ml. After adding various concentrations (0–100 μM) of tested drugs and the positive control inhibitor PSC833 (0–50 μM), cells were incubated at 37°C in an atmosphere of 5% CO2 and 95% relative humidity for 1 h to equilibrate drug accumulation into the cells. The final concentration of the dimethyl sulfoxide (DMSO) for the tested inhibitors and their respective controls was 1% (v/v). No effect of the vehicle on Rhd 123 accumulation was observed at this concentration. All incubations were conducted in triplicates. The intracellular fluorescence was then read within 1 h.
Flow cytometric detection of intracellular Rhd 123 accumulation
Intracellular fluorescence of Rhd 123 was analyzed with FACScan flow cytometer (Becton Dickinson Immunocytometry Systems, Mountain View, CA) equipped with an argon laser using a previously established method (Bachmeier and Miller 2005). Intracellular fluorescence of 15,000 events was logarithmically measured through a 535-nm band pass filter at an excitation wavelength of 488 nm. Cell debris was eliminated by gating on forward vs side scatter. The blank histogram, cells in medium containing vehicle (1% DMSO), yielded negligible cellular autofluorescence. The LLC-PK1 and L-MDR1 cells in medium containing Rhd 123 alone or PSC833 (10 μM), or the tested antipsychotics, generated the negative control, positive control, and inhibitor histograms, respectively. The median fluorescence (ΔF) of the tested compounds was used to calculate the percentage of inhibitory effect (E%) of P-gp mediated Rhd 123 intracellular accumulation using the following equation (Eq. 1):
where ΔF i is the median fluorescence of tested inhibitor at concentration i, ΔF 0 is the median fluorescence of tested inhibitor at concentration 0; ΔF max is the median fluorescence with maximal inhibition of P-gp activity by 10 μM PSC 833. The concentration of inhibitor to cause 50% of maximal increment of intracellular Rhd 123 fluorescence (EC50) values were estimated by fitting the mean of triplicate data to the sigmoidal dose–response equation using GraphPad 4.0 software (Intuitive Software for Science, San Diego, CA, USA).
Statistical analysis
Data are presented as mean±SD of three independent determinations. Effects of PSC833 on Rhd 123 accumulation in LLC-PK1 and L-MDR1 cells were analyzed with one-way ANOVA, using the Turkey test for post hoc comparisons. Effects of putative inhibitors on intracellular Rhd 123 accumulation were analyzed using unpaired t test (two-tailed). P<0.05 was considered statistically significant.
Results
After incubation at 37°C for 1 h, the intracellular fluorescence of Rhd 123 in the L-MDR1 cells was 3.1-fold lower than that in the LLC-PK1 cells (P<0.01), indicating that the overexpression of P-gp in the L-MDR1 cell surface significantly extruded Rhd 123 out of the cells (Fig. 1). After co-treatment of the L-MDR1 cells with the positive control inhibitor, PSC833 (10 μM), at 37°C for 1 h, the reduction in intracellular fluorescence of Rhd 123 in the L-MDR1 cells was abrogated. On the other hand, in the LLC-PK1 cells, the PSC833 did not produce a significant effect on intracellular fluorescence of Rhd 123 (Fig. 1). The intracellular fluorescence of Rhd 123 in the L-MDR1 cells in the presence of PSC833 (10 μM) was significantly higher than that in the control LLC-PK1 cells (P<0.01).
Intracellular fluorescence of Rhd 123 in LLC-PK1 and L-MDR1 cells after incubation of the cells with 5 μM Rhd 123 in the absence and presence of 5 μM PSC833. Data represent mean±SD from three experiments. P values were determined by analysis of variance with Dunnett’s multiple comparison test for post hoc pairwise comparison of the results with the LLC-PK1 cells without inhibitor. Two asterisks indicate statistical significance (P<0.01) compared with LLC-PK1
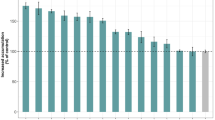
At two clinically relevant concentrations (1 and 5 μM), all tested antipsychotics significantly inhibited intracellular fluorescence of Rhd 123 in the L-MDR1 cells (P<0.01), except for clozapine (at 1 and 5 μM) and 9-OH-risperidone (at 1 μM) (Fig. 2).
The concentration-dependent effects of the putative inhibitors on the intracellular accumulation of Rhd 123 and on P-gp activities are presented in Figs. 3 and 4, respectively. The estimated EC50 values and their ratios to peak plasma concentrations are presented in Table 1. According to the EC50 values, the rank order of the inhibitory potency of the tested antipsychotic agents on P-gp are as follows: PSC833 (0.5 μM) < olanzapine (3.9 μM) < chlorpromazine (5.8 μM) < risperidone (6.6 μM) < haloperidol (9.1 μM) < quetiapine (9.8 μM) < 9-OH-risperidone (12.5 μM) < clozapine (30.0 μM) (Table 1). However, a thorough comparison of the in vivo inhibitory potency of these drugs should take into account both plasma concentrations after normal therapeutic doses, as well as tissue accumulation (see Tables 1 and 2). In this regard, olanzapine, risperidone, 9-OH-risperidone, and chlorpromazine, which have high tissue to plasma partition ratios, may have higher potential to interact with P-gp than other tested drugs (Table 2).
Concentration-dependent effects of the tested inhibitors on intracellular accumulation of Rhd 123 in L-MDR1 cells. Values were calculated by Eq. 1 described in the “Materials and methods.” Data represent mean±SD from three experiments
Concentration-dependent inhibition of the P-gp-mediated Rhd 123 transport by the tested inhibitors in L-MDR1 cells. Values were calculated by the following equation: I%=(100-E) 100%, where the IC50 values (concentration of inhibitor to cause 50% inhibition of original P-gp activity) were determined using SigmaPlot 9.0 (Point Richmont, CA). The IC50 values for PSC833, risperidone, 9-OH-risperidone, olanzapine, clozapine, quetiapine, chlorpromazine, haloperidone were 0.5, 8.5, 25.1, >100, >100, 24.8, >100, and 25.4 μM, respectively. Each data point represents mean±s.d. from three experiments
Discussion
Since being introduced into the market, the AAPs quickly supplanted the conventional antipsychotics, owing to their recognized treatment efficacy in both positive (hallucinations, delusions) and negative (flattening, alogia, and avolition) symptoms of schizophrenia with reduced risks of causing serious neurological side effects such as extrapyramidal symptoms and tardive dyskinesia (Simpson and Lindenmayer 1997). Despite these advantages, the treatment efficacy of the antipsychotics is frequently disappointing. Based on the standard criteria defined for treatment-resistance schizophrenia, the prevalence of treatment resistance rates has varied from 12.9 to 48% (Conley and Buchanan 1997; Meltzer 1992; Juarez-Reyes et al. 1995; Essock et al. 1996). This gives an estimated total of 400,000–1,000,000 patients in the world who are currently experiencing treatment-resistant schizophrenia. Despite the high prevalence and the high economic costs for taking care of these patients, very sparse data are available regarding drug action in this population. The refractory mechanisms are poorly understood.
Similar pharmacoresistance was reported for other major CNS disorders such as epilepsy. In approximately 30% of patients with epilepsy, seizures persist despite the choice of generally effective antiepileptic drugs in conjunction with carefully monitored treatment (Regesta and Tanganelli 1999). It was reported that brain expression of ABCB1, which encodes the multidrug transporter P-gp in humans, is markedly increased in the brain capillary endothelium and astrocytes in the majority of patients with medically intractable partial (mostly temporal lobe) epilepsy (Tishler et al. 1995). This finding was replicated in several subsequent studies (Sisodiya et al. 1999, 2001, 2002). In addition to P-gp, overexpression of multidrug resistance associate protein (MRP) 1 and 2 were also found in surgically resected epileptic foci in epileptogenic tissues (Sisodiya et al. 1999, 2001, 2002; Dombrowski et al. 2001). This indicates that overexpression of P-gp, MRP1, and MRP 2 in the BBB may all contribute to the pharmacoresistance of antiepileptic treatment as the brain transfer of many antiepileptic drugs are affected by these transporters in the BBB (Löscher and Potschka 2002).
In contrast to the rather established relationship of brain overexpression of the multidrug transporters and the pharmacoresistant epilepsy, there are only limited studies that explored the mechanisms of the pharmacoresistant psychiatric disorders.
Quite recently, results from our laboratory revealed that the ABCB1 transporter, P-gp, in BBB greatly limits the brain penetration of several atypical antipsychotics (Boulton et al. 2002; Wang et al. 2004a,b). Thus, the genetic variations of P-gp (Hoffmeyer et al. 2000) in BBB may represent one of the factors contributing to the variable treatment responses in schizophrenia and other psychiatric disorders. Quite recently, a clinical study conducted in our laboratory revealed that the single nucleotide polymorphism of C3435T of P-gp is associated with significant elevation of area under plasma concentration time curve of a standard oral tablet of olanzapine in healthy subjects (Markowitz et al. 2006). In addition, the single nucleotide polymorphisms of the ABCB1 gene, C3435T and G2677T/A, were found to be associated with some therapeutic response to an antipsychotic agent bromperidol (Yasui-Furukori et al. 2006). Yasui-Furukori et al. genotyped 85 schizophrenic patients for the C3435T and G2677T/A variants. However, after 3 mg twice daily of risperidone, the steady-state plasma concentration of risperidone showed no difference between these variants (Yasui-Furukori et al. 2004). In this study, only a single time point (12 h) sample after the last dosing was collected for each individual; therefore, it is very hard to justify the results considering the very large intersubject variations (up to 76-fold). In addition, none of the above-mentioned studies related the pharmacodynamic changes to the brain concentrations of the antipsychotics, which may be more directly related to the functional variation of the BBB transporters.
It needs to be noted that with the advent of positron emission tomography imaging (PET) and single photon emission computerized tomography imaging, it is now well-recognized that all antipsychotics induce a substantial dopamine D2 receptor occupancy (Kapur and Remington 2001). Consistently, findings from different laboratories suggest that significant D2 dopamine receptor occupancy seems to correlate with clinical efficacy and side effects, and also help to understand clinical differences between the available antipsychotics (Kapur et al. 1999; Nyberg and Farde 2000; Bressan et al. 2001). However, contradictory results were also reported by studies in which antipsychotic resistance is not related to D2 receptor occupancy (Sedvall 1992). Although this contradiction can be attributed to the different criteria for selection of the refractory patients and the single time-point characteristic of the reported PET study, the multifactorial pathogenesis of the schizophrenia cannot be excluded.
Reversal of the functional activity of P-gp in BBB may thus increase CNS drug concentrations with improved therapeutic efficacy, and it represents a novel therapeutic intervention. This strategy may be particularly beneficial for those drug combinations in which one of the therapeutic agents possesses a relatively short half-life such as quetiapine. Although speculative, the combination of a short half-life therapeutic agent with potential BBB drug transporter inhibitors such as olanzapine or risperidone may potentially prolong the brain elimination half-life of the short half-life agents and enhance their therapeutic efficacy.
Antipsychotics were routinely combined for the treatment of schizophrenia. Combined treatment of the antipsychotics is currently accounting for approximately 44% of total antipsychotic prescriptions (Keks et al. 1999; Centorrino et al. 2004). By assessing the interaction potential of antipsychotics with P-gp, we herein provide evidence that pharmacokinetic interactions may occur by reversal of P-gp activity in tissues during combined treatment with antipsychotics.
At concentrations ranging from 0.1 to 100 μM, all the tested AAPs (risperidone, 9-OH-risperidone, olanzapine, quetiapine, and clozapine) as well as the two conventional antipsychotics, haloperidol and chlorpromazine, showed concentration-dependent inhibitory effects on P-gp-mediated Rhd 123 intracellular accumulation. However, at a clinically relevant concentration (1 μM), only risperidone and olanzapine showed moderate inhibitory effects on P-gp activity (about 10 to 14%). Other antipsychotics only produced mild (<6%) or negligible effects on P-gp activity. The mean peak plasma concentrations of the tested antipsychotics rarely exceed 1 μM after normal therapeutic doses (except for clozapine, which may reach 2.5 μM in some cases; Aravagiri et al. 1998; Hiemke et al. 2004). Although the exact concentrations of these drugs in the luminal surface of the brain capillary cells where the P-gp localized are unknown, the present results suggest that, among the tested antipsychotics, risperidone and olanzapine are the most likely agents that could inhibit P-gp function in BBB in clinical situations.
Our previous studies have confirmed that many antipsychotics have shown various extent of binding affinities with P-gp (Boulton et al. 2002). In subsequent P-gp knockout mouse experiments, we further provide evidence that olanzapine, risperidone, and 9-OH-risperidone are all transportable substrates of P-gp (Wang et al. 2004a,b). All these data suggest a competitive nature of the inhibitory effect of the tested antipsychotics shown in the present study to compete for the same substrate binding sites in P-gp. However, a more definitive conclusion for the underlying mechanism apparently needs further experiments.
All the antipsychotics have shown various degrees of plasma protein binding and extensive tissue accumulation, with plasma protein binding rates ranging from 88 to 98% (Moffat et al. 2004) and liver or kidney to plasma concentration ratios ranging from 1.5 to 97.7 (Table 2). The extensive tissue accumulation and low free fraction of drugs may counterbalance each other and support the utilization of total plasma drug concentrations in the prediction of drug interaction potentials (Wen et al. 2002). This may suggest that the antipsychotics in certain tissues may reach sufficiently high levels to cause P-gp inhibition. Furthermore, the antipsychotics, when given orally, may produce very high local concentrations in the basolateral surface of the gastrointestinal tract. Inhibition of P-gp function in these tissues may change the pharmacokinetic characteristics of co-administered P-gp substrates.
Only limited studies have evaluated the drug interaction potentials of the antipsychotics with P-gp. In one in vitro study, El Ela et al. (2004) evaluated the inhibitory effects of several antipsychotics on P-gp-mediated talinolol efflux across Caco-2 monolayers. The results indicated that quetiapine, haloperidol, clozapine, and olanzapine showed various degrees of inhibitory effects on talinolol transport with IC50 values of 0.8, 12.4, 92.8, and >250 μM, respectively. In another study, when tested at 100 μM, risperidone, haloperidol, and chlorpromazine inhibited calcein AM cellular uptake in MDCKII-MDR1 cells by 52.6, 37.2, and 46.9%, respectively (Mahar Doan et al. 2002). These previous results are generally in agreement with the present results, except that a preferential inhibitory effect was reported for quetiapine on P-gp-mediated talinolol transport in Caco-2 cells (El Ela et al. 2004). Different probe drugs and different cell lines used in the studies may account for the differences in the results. We searched the literatures for drug interactions of the studied antipsychotics and found very sparse clinical studies reporting drug interactions between the antipsychotics and P-gp. While the present study and also other works (El Ela et al. 2004; Mahar Doan et al. 2002) all support the possible drug–drug interactions between certain antipsychotics and P-gp, especially in certain tissues that the drugs may accumulate in, the lack of clinical reports for such drug interactions may only suggest that clinicians are unaware or unperceptive of such interactions.
In conclusion, the present study indicated that all the antipsychotics showed various degrees of inhibitory effects on P-gp activity at concentrations ranging from 1 to 100 μM. However, risperidone and olanzapine are the most likely agents that may inhibit P-gp activity in BBB. The antipsychotics may accumulate in the liver and kidney, and local concentrations of the antipsychotics may be very high when given orally. Inhibition of P-gp activity by the antipsychotics in the apical membranes of the gastrointestinal tract, the biliary canalicular membranes of the hepatocytes, and the luminal membranes of proximal tubular epithelial cells in the kidney may cause clinically significant drug–drug interactions. The potential for drug–drug interactions involving the antipsychotics and substrates of P-gp warrants further investigation.
Abbreviations
- AAPs:
-
atypical antipsychotics
- BBB:
-
blood–brain barrier
- CNS:
-
central nervous system
- EC50 :
-
the concentration of inhibitor to cause 50% of the maximal increment of intracellular fluorescence
- ΔF:
-
median fluorescence
- I:
-
the percentage of inhibition of P-gp activity
- P-gp:
-
P-glycoprotein
- DMEM:
-
Dulbecco’s modified eagle medium
- Rhd 123:
-
Rhodamine 123
References
Angelin B, Arvidsson A, Dahlqvist R, Hedman A, Schenck-Gustafsson K (1987) Quinidine reduces biliary clearance of digoxin in man. Eur J Clin Invest 17:262–265
Aravagiri M, Marder SR (2002) Brain, plasma and tissue pharmacokinetics of risperidone and 9-hydroxyrisperidone after separate oral administration to rats. Psychopharmacology (Berl) 159:424–431
Aravagiri M, Marder SR, Wirshing D, Wirshing WC (1998) Plasma concentrations of risperidone and its 9-hydroxy metabolite and their relationship to dose in schizophrenic patients: simultaneous determination by a high performance liquid chromatography with electrochemical detection. Pharmacopsychiatry 31:102–109
Aravagiri M, Teper Y, Marder SR (1999) Pharmacokinetics and tissue distribution of olanzapine in rats. Biopharm Drug Dispos 20:369–377
Ayrton A, Morgan P (2001) Role of transport proteins in drug absorption, distribution and excretion. Xenobiotica 31:469–497
Bachmeier CJ, Miller DW (2005) A fluorometric screening assay for drug efflux transporter activity in the blood–brain barrier. Pharm Res 22:113–121
Boulton DW, DeVane CL, Liston HL, Markowitz JS (2002) In vitro P-glycoprotein affinity for atypical and conventional antipsychotics. Life Sci 71:163–169
Bressan RA, Jones HM, Ell PJ, Pilowsky LS (2001) Dopamine D(2) receptor blockade in schizophrenia. Am J Psychiatry 158:971–972
Centorrino F, Goren JL, Hennen J, Salvatore P, Kelleher JP, Baldessarini RJ (2004) Multiple versus single antipsychotic agents for hospitalized psychiatric patients: case-control study of risks versus benefits. Am J Psychiatry 161:700–706
Conley RR, Buchanan RW (1997) Evaluation of treatment-resistant schizophrenia. Schizophr Bull 23:663–674
Dahlqvist R, Ejvinsson G, Schenck-Gustafsson K (1980) Effect of quinidine on plasma concentration and renal clearance of digoxin. A clinically important drug interaction. Br J Clin Pharmacol 9:413–418
DeVane CL, Nemeroff CB (2001) An evaluation of risperidone drug interactions. J Clin Psychopharmacol 21:408–416
Dombrowski SM, Desai SY, Marroni M, Cucullo L, Goodrich K, Bingaman W, Mayberg MR, Bengez L, Janigro D (2001) Overexpression of multiple drug resistance genes in endothelial cells from patients with refractory epilepsy. Epilepsia 42:1501–1506
El Ela AA, Hartter S, Schmitt U, Hiemke C, Spahn-Langguth H, Langguth P (2004) Identification of P-glycoprotein substrates and inhibitors among psychoactive compounds—implications for pharmacokinetics of selected substrates. J Pharm Pharmacol 56:967–975
Essock SM, Hargreaves WA, Dohm FA, Goethe J, Carver L, Hipshman L (1996) Clozapine eligibility among state hospital patients. Schizophr Bull 22:15–25
Fracasso PM, Westervelt P, Fears CL, Rosen DM, Zuhowski EG, Cazenave LA, Litchman M, Egorin MJ (2000) Phase I study of paclitaxel in combination with a multidrug resistance modulator, PSC 833 (Valspodar), in refractory malignancies. J Clin Oncol 18:1124–1134
Hiemke C, Dragicevic A, Grunder G, Hatter S, Sachse J, Vernaleken I, Muller MJ (2004) Therapeutic monitoring of new antipsychotic drugs. Ther Drug Monit 26:156–160
Hoffmeyer S, Burk O, von Richter O, Arnold HP, Brockmoller J, Johne A, Cascorbi I, Gerloff T, Roots I, Eichelbaum M, Brinkmann U (2000) Functional polymorphisms of the human multidrug-resistance gene: multiple sequence variations and correlation of one allele with P-glycoprotein expression and activity in vivo. Proc Natl Acad Sci U S A 97:3473–3478
Hopenwasser J, Mozayani A, Danielson TJ, Harbin J, Narula HS, Posey DH, Shrode PW, Wilson SK, Li R, Sanchez LA (2004) Postmortem distribution of the novel antipsychotic drug quetiapine. J Anal Toxicol 28:264–267
Jalava KM, Partanen J, Neuvonen PJ (1997) Itraconazole decreases renal clearance of digoxin. Ther Drug Monit 19:609–613
Juarez-Reyes MG, Shumway M, Battle C, Bacchetti P, Hansen MS, Hargreaves WA (1995) Effects of stringent criteria on eligibility for clozapine among public mental health clients. Psychiatr Serv 46:801–806
Kageyama M, Namiki H, Fukushima H, Ito Y, Shibata N, Takada K (2005) In vivo effects of cyclosporin A and ketoconazole on the pharmacokinetics of representative substrates for P-glycoprotein and cytochrome P450 (CYP) 3A in rats. Biol Pharm Bull 28:316–322
Kapur S, Remington G (2001) Dopamine D(2) receptors and their role in atypical antipsychotic action: still necessary and may even be sufficient. Biol Psychiatry 50:873–883
Kapur S, Zipursky RB, Remington G (1999) Clinical and theoretical implications of 5-HT2 and D2 receptor occupancy of clozapine, risperidone, and olanzapine in schizophrenia. Am J Psychiatry 156:286–293
Keks NA, Altson K, Hope J, Krapivensky N, Culhane C, Tanaghow A, Doherty P, Bootle A (1999) Use of antipsychosis and adjunctive medications by an inner urban community psychiatric service. Aust N Z J Psychiatry 33:896–901
Loscher W, Potschka H (2002) Role of multidrug transporters in pharmacoresistance to antiepileptic drugs. J Pharmacol Exp Ther 301:7–14
Mahar Doan KM, Humphreys JE, Webster LO, Wring SA, Shampine LJ, Serabjit-Singh CJ, Adkison KK, Polli JW (2002) Passive permeability and P-glycoprotein-mediated efflux differentiate central nervous system (CNS) and non-CNS marketed drugs. J Pharmacol Exp Ther 303:1029–1037
Manjunath K, Venkateswarlu V (2005) Pharmacokinetics, tissue distribution and bioavailability of clozapine solid lipid nanoparticles after intravenous and intraduodenal administration. J Control Release 107:215–228
Markowitz JS, DeVane CL, Malcolm RJ, Gefroh HA, Wang JS, Zhu HJ, Donovan JL (2006) Pharmacokinetics of olanzapine after single-dose oral administration of standard tablet versus normal and sublingual administration of an orally disintegrating tablet in normal volunteers. J Clin Pharmacol 46(2):164–171
Meltzer HY (1992) Treatment of the neuroleptic–nonresponsive schizophrenic patient. Schizophr Bull 18:515–542
Miyazaki H, Matsunaga Y, Nambu K, Oh-e Y, Yoshida K, Hashimoto M (1986) Disposition and metabolism of [14C]-haloperidol in rats. Arzneimittelforschung 36:443–452
Moffat AC, Osselton MD, Widdop B (2004) Clark’s analysis of drugs and poisons, vol. 2. Pharmaceutical Press, London
Nyberg S, Farde L (2000) Non-equipotent doses partly explain differences among antipsychotics—implications of PET studies. Psychopharmacology (Berl) 148:22–23
Partanen J, Jalava KM, Neuvonen PJ (1996) Itraconazole increases serum digoxin concentration. Pharmacol Toxicol 79:274–276
Pedersen KE, Thayssen P, Klitgaard NA, Christiansen BD, Nielsen-Kudsk F (1983a) Influence of verapamil on the inotropism and pharmacokinetics of digoxin. Eur J Clin Pharmacol 25:199–206
Pedersen KE, Christiansen BD, Klitgaard NA, Nielsen-Kudsk F (1983b) Effect of quinidine on digoxin bioavailability. Eur J Clin Pharmacol 24:41–47
Prior TI, Baker GB (2003) Interactions between the cytochrome P450 system and the second-generation antipsychotics. J Psychiatry Neurosci 28:99–112
Regesta G, Tanganelli P (1999) Clinical aspects and biological bases of drug-resistant epilepsies. Epilepsy Res 34:109–122
Riordan JR, Deuchars K, Kartner N, Alon N, Trent J, Ling V (1985) Amplification of P-glycoprotein genes in multidrug-resistant mammalian cell lines. Nature 316:817–819
Roninson IB, Chin JE, Choi KG, Gros P, Housman DE, Fojo A, Shen DW, Gottesman MM, Pastan I (1986) Isolation of human mdr DNA sequences amplified in multidrug-resistant KB carcinoma cells. Proc Natl Acad Sci USA 83:4538–4542
Sadeque AJ, Wandel C, He H, Shah S, Wood AJ (2000) Increased drug delivery to the brain by P-glycoprotein inhibition. Clin Pharmacol Ther 68:231–237
Schinkel AH, Wagenaar E, Mol CA, van Deemter L (1996) P-glycoprotein in the blood-brain barrier of mice influences the brain penetration and pharmocological activity of many drugs. J Clin Invest 97:2517–2524
Sedvall G (1992) The current status of PET scanning with respect to schizophrenia. Neuropsychopharmacology 7(1):41–54 (Aug)
Sgaragli GP, Valoti M, Palmi M, Frosini M, Giovannini MG, Bianchi L, Della Corte L (1995) Rat tissue concentrations of chlorimipramine, chlorpromazine and their N-demethylated metabolites after a single oral dose of the parent compounds. J Pharm Pharmacol 47:782–790
Simpson GM, Lindenmayer JP (1997) Extrapyramidal symptoms in patients treated with RSP. J Clin Psychopharmacol 17:194–201
Sisodiya SM, Heffernan J, Squier MV (1999) Over-expression of P-glycoprotein in malformations of cortical development. Neuroreport 10(16):3437–3441
Sisodiya SM, Lin WR, Squier MV, Thom M (2001) Multidrug-resistance protein 1 in focal cortical dysplasia. Lancet 357:42–43
Sisodiya SM, Lin WR, Harding BN, Squier MV, Thom M (2002) Drug resistance in epilepsy: expression of drug resistance proteins in common causes of refractory epilepsy. Brain 125(Pt 1):22–31
Smit JW, Weert B, Schinkel AH, Meijer DK (1998) Heterologous expression of various P-glycoproteins in polarized epithelial cells induces directional transport of small (type 1) and bulky (type 2) cationic drugs. J Pharmacol Exp Ther 286:321–327
Suzuki Y, Someya T, Shimoda K, Hirokane G, Morita S, Yokono A, Inoue Y, Takahashi S (2001) Importance of the cytochrome P450 2D6 genotype for the drug metabolic interaction between chlorpromazine and haloperidol. Ther Drug Monit 23:363–368
Tanaka H, Matsumoto K, Ueno K, Kodama M, Yoneda K, Katayama Y, Miyatake K (2003) Effect of clarithromycin on steady-state digoxin concentrations. Ann Pharmacother 37:178–181
Tishler DM, Weinberg KI, Hinton DR, Barbaro N, Annett GM, Raffel C (1995) MDR1 gene expression in brain of patients with medically intractable epilepsy. Epilepsia 36:1–6
Verschraagen M, Koks CH, Schellens JH, Beijnen JH (1999) P-glycoprotein system as a determinant of drug interactions: the case of digoxin-verapamil. Pharmacol Res 40:301–306
Yasui-Furukori N, Saito M, Nakagami T, Kaneda A, Tateishi T, Kaneko S (2006) Association between multidrug resistance 1 (MDR1) gene polymorphisms and therapeutic response to bromperidol in schizophrenic patients: a preliminary study. Prog Neuropsychopharmacol Biol Psychiatry 30:286–291
Yasui-Furukori N, Mihara K, Takahata T, Suzuki A, Nakagami T, De Vries R, Tateishi T, Kondo T, Kaneko S (2004) Effects of various factors on steady-state plasma concentrations of risperidone and 9-hydroxyrisperidone: lack of impact of MDR-1 genotypes. Br J Clin Pharmacol 57:569–575
Wang JS, Ruan Y, Taylor RM, Donovan JL, Markowitz JS, DeVane CL (2004a) The brain entry of risperidone and 9-hydroxyrisperidone are greatly limited by P-glycoprotein. Int J Neuropsychopharmacology 7:415–419
Wang JS, Taylor R, Runa Y, Donovan LJ, Markowitz SJ, DeVane CL (2004b) Olanzapine penetration into brain is greater in transgenic mdr1a p-glycoprotein deficient mice than FVB1 (wild-type) aminals. Neuropsychopharmacology 29:551–557
Wen X, Wang JS, Bckman JT, Laitila J, Neuvonen PJ (2002) Trimethoprim and sulfamethoxazole are selective inhibitors of CYP2C8 and CYP2C9, respectively. Drug Metab Dispos 30:631–635
Yeung PK, Hubbard JW, Korchinski ED, Midha KK (1993) Pharmacokinetics of chlorpromazine and key metabolites. Eur J Clin Pharmacol 45:563–569
Acknowledgement
This work was supported by NIH grant MH071811-01A1. None of the authors has conflicting interests that interfere with the integrity of the content of the article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, JS., Zhu, HJ., Markowitz, J.S. et al. Evaluation of antipsychotic drugs as inhibitors of multidrug resistance transporter P-glycoprotein. Psychopharmacology 187, 415–423 (2006). https://doi.org/10.1007/s00213-006-0437-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0437-9