Abstract
Rationale
The serotonin precursor l-tryptophan (TRP) is available as a nutritional supplement and is licensed as an antidepressant in a number of countries. However, evidence of its efficacy as the primary treatment for depression is limited, and the direct action of TRP on the symptoms of depression and anxiety has not been well-characterised.
Objectives
The present study assessed whether TRP induces cognitive changes opposite to the negative biases found in depression and characteristic of those induced by serotonergic antidepressants in healthy volunteers.
Materials and methods
Thirty eight healthy volunteers were randomised to receive 14 days double-blind intervention with TRP (1 g 3× a day) or placebo. On the final day, emotional processing was assessed using four tasks: facial expression recognition, emotion-potentiated startle, attentional probe and emotional categorisation and memory.
Results
TRP increased the recognition of happy facial expressions and decreased the recognition of disgusted facial expressions in female, but not male, volunteers. TRP also reduced attentional vigilance towards negative words and decreased baseline startle responsivity in the females.
Conclusions
These findings provide evidence that TRP supplementation in women induces a positive bias in the processing of emotional material that is reminiscent of the actions of serotonergic antidepressants. This highlights a key role for serotonin in emotional processing and lends support to the use of TRP as a nutritional supplement in people with mild depression or for prevention in those at risk. Future studies are needed to clarify the effect of tryptophan on these measures in men.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As the precursor to serotonin synthesis, l-tryptophan (TRP), which is contained in dietary protein, has a plausible physiological basis for acting as an antidepressant. Indeed, for many years TRP has been available as a nutritional supplement and is licensed as an antidepressant in a number of countries. However, although there are a number of studies documenting its efficacy as an adjunct to other antidepressant treatments, in particular monoamine oxidase inhibitors (Pare 1963; Coppen et al. 1963; Glassman and Platman 1969), there is limited evidence of its efficacy as the primary treatment for depression (Shaw et al. 2002). As a result, the direct action of TRP on the symptoms of depression and anxiety has not been well-characterised.
Some of the clearest evidence to support a role for tryptophan in mood comes from studies in which TRP levels are acutely depleted by the administration of a mixture of essential amino acids from which TRP has been omitted. Studies have shown a significant, transient reappearance of depressive symptoms after acute tryptophan depletion (ATD) in both medicated and non-medicated depressed patients in remission (Delgado et al. 1990; Smith et al. 1997). In addition, there is some evidence of a mood-lowering effect of ATD in healthy, never depressed volunteers (Young et al. 1985), although these effects are more variable and most robust in those that have a family history of depression (Benkelfat et al. 1994). Interestingly, previous studies have reported larger effects of ATD on mood in female volunteers (Booij et al. 2002), which is consistent with the finding that ATD has a greater biochemical effect in women than men, as demonstrated using radiolabelled alpha-methyl-l-tryptophan with positron emission tomography (Nishizawa et al. 1997).
Manipulations of the serotonin (5-HT) system have also been shown to affect social and cognitive functioning. Primate and human studies demonstrate that increasing 5-HT function, through administration of either TRP or a selective serotonin reuptake inhibitor (SSRI), results in increased affiliative and socially constructive behaviour (Raleigh et al. 1991; Knutson et al. 1998; Moskowitz et al. 2001). Conversely, lowering serotonergic function in monkeys and humans has been shown to increase aggression and decrease affiliative behaviour (Raleigh et al. 1980; Raleigh and McGuire 1991; Pihl et al. 1995).
As well as influencing social behaviour, there is emerging evidence that 5-HT is involved in emotional biases in the perception of socially relevant stimuli. For example, acute TRP depletion has been shown to increase reaction times for happy but not sad targets in an affective go/no-go task, suggesting the induction of a negative perceptual bias in the processing of emotional material (Murphy et al. 2002). Such negative distortions in information processing have been repeatedly reported in depression and are thought to play a fundamental role in the maintenance of mood disorders (Beck et al. 1979). For example, depression has been associated with a decreased perception of happy facial expressions (Surguladze et al. 2004), and increased attentional vigilance towards negative stimuli has been reported in both depression and anxiety disorders (Bradley et al. 1997; MacLeod et al. 1986; Mogg et al. 1995). The emotion-potentiated startle reflex has also been shown to be altered in depression and anxiety disorders (Grillon et al. 1994, 1998; Allen et al. 1999).
We have recently demonstrated that serotonergic antidepressants have the opposite effect on information processing and induce a positive bias on a number of emotion-related tasks. After a 7-day treatment with the SSRI, citalopram, healthy volunteers showed decreased recognition of the negative facial expressions of fear, disgust and anger, increased recall of positive (relative to negative) emotional material and reduced startle responses to threatening pictures compared to placebo (Harmer et al. 2004). These changes in emotional processing occurred in the absence of significant changes in mood.
It is not known whether increasing serotonin synthesis has similar effects on emotional processing to those seen after the inhibition of serotonin reuptake. To investigate this, the present study examined the effect of a 2-week administration of TRP or placebo on similar measures of emotional processing in healthy volunteers. The use of healthy volunteers allows the elucidation of the direct effects of TRP on cognitive function unconfounded by mood changes as a result of symptom remission. It was hypothesised that TRP would induce cognitive changes and emotional biases opposite to those found in depression and characteristic of those induced by serotonergic antidepressants. Given the previous findings from tryptophan depletion studies, a particular question of interest was whether there is a gender difference in the effects of TRP on emotional processing.
Materials and methods
Subjects and design
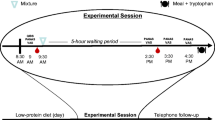
A total of 38 healthy volunteers (18 male, 20 female), between the ages of 18 and 40 years (mean age: 25.6 years) took part in this study. Participants were recruited using adverts in University departments and screened to exclude those with a current or previous history of psychiatric disorder (assessed using the Structured Clinical Interview for DSM-IV) or significant physical illness. Six of the participants were regular but not heavy smokers (defined as less than ten cigarettes per day) and these were evenly distributed between the two experimental groups (placebo: 3; tryptophan: 3). All participants gave their written consent to participate in the study, which was approved by the local ethics committee. Volunteers were randomly allocated to receive tryptophan (1 g 3×a day) or placebo for a period of 14 days. The groups were well-matched in terms of gender, verbal IQ (as assessed with the National Adult Reading Test), weight, age (see Table 1) and family history of mood disorder (placebo: 3; tryptophan: 1). On average, the male volunteers were significantly heavier than the female volunteers (p>0.001), but there were no significant weight differences between the two groups. The premenstrual week was avoided for the study period in women.
The 3-g daily dose of tryptophan was chosen because a previous study suggested that this dose should keep levels of tryptophan hydroxylase close to saturation for most of the day (Young and Gauthier 1981). Fourteen days of treatment with this dose has also previously been shown to alter social interaction (Moskowitz et al. 2001) and a previous study has shown that this dose was more effective than placebo in treating mild to moderate depression (Thomson et al. 1982).
Subjective state was recorded every day throughout treatment using the Befindlichkeits scale of mood and energy (von Zerssen et al. 1974) and the Positive and Negative Affect Scale (Watson et al. 1988). Together, these gave a measure of day-by-day variation in mood. The following questionnaires were given pre- and post-treatment: State–Trait Anxiety Inventory (Spielberger et al. 1983), Buss–Durkee Hostility Inventory (Buss and Durkee 1957), Beck Depression Inventory (Beck et al. 1961), Dysfunctional Attitudes Scale (Weisman and Beck 1978). Participants were also asked to record any possible side effects that they experienced on a daily side effects checklist.
A battery of tests was administered on the 14th day of intervention. All volunteers were given the same cognitive tests in the same order and the test session lasted 2 ½ h in total. During the test session, participants did not smoke or consume caffeine.
Facial expression recognition task
The facial recognition task featured six basic emotions—happiness, sadness, fear, anger, disgust and surprise—taken from the Ekman and Friesen (1976) Pictures of Affect series. These had been morphed between each prototype and neutral using techniques described by Young et al. (1997). This procedure involved taking a variable percentage of the shape and texture differences between the two standard images 0% (neutral) and 100% (full emotion) in 10% steps. Four examples of each emotion at each intensity were given. Each face was also given in a neutral expression, giving a total of 250 stimuli presentations. Each face was presented for 500 ms and was immediately replaced by a blank screen. Subjects made their response by pressing a labelled key on a response keyboard. The task was broken down into three parts, with an untimed rest period between each to minimise fatigue. Subjects were asked to respond as quickly and as accurately as possible.
Recognition threshold was designated as the level of emotional intensity at which participants correctly identified 75% or more of the facial expressions of emotion for two consecutive intensities. Reaction time for correct choices was also recorded.
Emotional categorisation and memory task
Emotional categorisation
Sixty personality characteristics selected to be extremely disagreeable (e.g. domineering, untidy, hostile) or agreeable (cheerful, honest, optimistic) (Anderson 1968) were presented on the computer screen for 500 ms with an intertrial interval of 2 s. These words were matched in terms of word length and ratings of frequency and meaningfulness. Volunteers were asked to categorise these personality traits as likeable or dislikeable as quickly and as accurately as possible. Specifically, they were asked to imagine whether they would be pleased or upset if they overheard someone else referring to them as possessing this characteristic, so that the judgement was in part self-referring. Classifications and reaction times for correct identification were recorded.
Emotional memory
Fifteen minutes after completion of the emotional categorisation task, volunteers were asked to recall as many of the personality traits as possible. This task, therefore, allowed the assessment of incidental memory for positive and negative characteristics. After the free recall task, recognition memory was also assessed by asking volunteers to respond with a ‘Yes’ or ‘No’ to each item on a list comprising the 60 targets plus 60 matched distractors (30 positive, 30 negative).
Emotion-potentiated startle response
Stimuli
Picture stimuli from the International Affective Picture System (Lang et al. 1998) were used, which were designed to elicit positive, negative or neutral emotions (Larson et al. 2000). These stimuli had been rated and selected, such that the negative and positive pictures were similar in terms of arousal but opposite in valence, whereas, the neutral pictures were low on arousal and average on valence. Stimuli were presented for 13 s (intertrial interval: mean=13 s, range=11–15 s) on a 43-cm computer screen approximately 1 m away from the volunteer. Pictures were presented in three blocks each containing seven pictures of each category in a fixed pseudo-random order with the constraint that no two of the same type (neutral, positive or negative) were presented successively.
Procedure and recording
The eye-blink component of the startle response was recorded from the orbicularis oculi using electromyography (EMG startle response system, San Diego Instruments, San Diego, CA, USA). Acoustic probes were 50 ms, 95-dB bursts of white noise with a nearly instantaneous rise time, and were delivered binaurally through headphones (generated through the noise generator and amplifier component of the EMG startle response system, San Diego Instruments). Probes were delivered at 1.5, 4.5 or 7.5 s after picture onset. Within each block of 21 pictures, probes were delivered during five of each trial type (neutral, positive and negative). To limit expectation of the noise, two trials of each type were delivered with no startle probe and three probes per block were delivered in the intertrial interval. To habituate participants to the startle probes and to orient them to the procedure, participants viewed an introductory set of nine neutral pictures and received nine startle probes (two of which occurred during the intertrial interval).
EMG signals were filtered (low cutoff: 0.5 Hz; high cutoff: 100 Hz), rectified and smoothed using a 20-ms filter window. Eye-blink magnitudes in microvolts were calculated by subtracting the amount of integrated EMG at reflex onset from the peak amplitude maximum amount of integrated EMG between 20 and 120 ms after probe onset. Trials with no perceptible eye-blink reflex were assigned a magnitude of zero and included in the analysis. Eye-blink reflexes with excessive noise during a 20-ms prestartle baseline period were excluded.
Of the 38 volunteers, 16 volunteers were not included in the analysis because of equipment failure (N=7), electrode interference (N=3), or because they displayed fewer than 25% satisfactory blink responses in the paradigm (N=6), leaving n=11 and n=11 for the tryptophan and placebo groups, respectively. Eye-blink magnitudes were analysed both as raw data and also z-transformed within participants to allow direct comparison of the acoustic startle response during unpleasant, pleasant and neutral pictorial stimuli presentation. Median amplitude was computed under the three different conditions for each participant.
Attentional probe task
Stimuli
Two types of emotional words were used in this task: 60 social threatening negative words and 60 positive words. Each emotional word was paired with a neutral word that began with the same letter and was matched for length. There were, therefore, 60 socially threatening-neutral pairs and 60 positive-neutral pairs. In addition, there were 60 neutral-neutral pairs.
Procedure
Each trial started with a fixation cross for 500 ms, followed by a word pair. On each trial, one of the word pairs appeared above and the other below the central fixation position. The emotional words appeared in the top and bottom location with equal frequency. In the unmasked condition, the word pair was presented for 500 ms and then a probe appeared in the location of one of the preceding words. The probe was either one or two stars, and participants were asked to press one of two labelled keys on a keyboard to indicate the number of stars present on the screen; their response terminated the probe display. Participants were asked to respond as quickly and as accurately as possible.
The probe appeared at the top and the bottom of the screen with equal frequency. Thus, on half of the emotion-neutral word pair trials the probe appeared in the same position as the emotional word and on the other half the probe appeared in the same position as the neutral word. The sequence of events was the same in the masked condition, except the duration of the word pair was 14 ms and the display of the word pair was immediately followed by a mask, which was displayed for 186 ms. The mask was constructed from digits, letters and other non-letter symbols (for example @B%2#) and was matched for word position and length. There were 360 trials in total (masked: 60 positive-neutral, 60 negative-neutral, 60 neutral-neutral; unmasked: 60 positive-neutral, 60 negative-neutral, 60 neutral-neutral), and masked and unmasked trials were presented in a random order.
One female volunteer did not complete this task due to equipment failure. Reaction time and accuracy scores were recorded. To simplify these results, attentional vigilance scores were calculated for each participant by subtracting the mean accuracy or reaction time from trials when probes appeared in the opposite position to the emotional word (incongruent trials) from trials when probes appeared in the same position as the emotional word (congruent trials).
Statistical analysis
Data were analysed by using between-groups one-way (subjective state, emotional memory) or split-plot two-way (facial expression recognition, emotion potentiated startle response, reaction time) analyses of variance (ANOVAs). Group (tryptophan or placebo) and gender were the between-subjects factors, and facial expression or valence was the within-subject factor. Given that a gender difference was an a priori hypothesis, data were also analysed separately for males and females. In all analyses, a Greenhouse–Geisser correction was used. Where necessary, the interpretation of significant interaction effects was aided by the use of simple main effect analyses.
Results
Subjective state and side effects
There was no significant effect of TRP on measures of subjective state, mood or anxiety (p>0.09 for all comparisons, see Table 2). There was a general trend for participants to report feeling more positive post-treatment compared to pre-treatment, but this was not increased in the TRP group. No significant side effects were reported and no participants withdrew from the study due to adverse effects.
Facial expression recognition
Accuracy
There was a significant interaction between group × gender × emotion [F(5,170)=3.66, p=0.007], suggesting that the tryptophan exerted different effects in males and females. Separate analysis of the male and female data revealed that tryptophan affected facial expression recognition in women (group × facial expression [F(5,90)=3.45, p=0.01]) and marginally in males (group × facial expression [F(5,80)=2.72, p=0.045]). In female volunteers, tryptophan selectively decreased the recognition of disgusted facial expressions [t(18)=−3.386, p=0.003] and selectively increased the recognition of happy facial expressions [t(18)=2.714, p=0.014, see Fig. 1a]. In male volunteers, no specific facial expression reached the p<0.05 significance level (see Fig. 1b).
Facial expression recognition in female (a) and male (b) volunteers following 14 days of treatment with placebo or tryptophan. Values represent the mean recognition threshold inverted to give an accuracy score ±1 SEM (calculated by taking the level of intensity at which each participant correctly identified the expression on 75% of trials for two consecutive trials). Asterisks represent significant level of difference from placebo: **p<0.01
Reaction time
There was a significant main effect of group on median reaction time on the facial expression recognition task [F(1,34)=5.256, p=0.028] but no interaction with group or gender. This was driven by generally slower responses in the TRP group, compared to placebo, irrespective of facial expression.
Emotional categorisation and memory task
Emotional categorisation
Participants were generally very accurate in their categorisation of positive and negative personality characteristics as ‘likeable’ and ‘dislikeable’ [mean correct categorisation of positive characteristics: 96.6% (placebo), 94.38% (TRP); mean correct categorisation of negative characteristics 98.6% (placebo), 96.3% (TRP)], and there was no interaction with gender or group. TRP did not significantly affect the speed to classify negative and positive self-referent personality characteristics (main effect of group F(1,34)=1.47, p=0.2; group × valence × gender F(1,34)=0.23, p=0.6). This absence of effect was also apparent when only the responses in females were considered (main effect of group F(1,18)=0.435, p=0.5; group × valence F(1,18)=0.017, p=0.9).
Emotional memory
Recall and recognition of positive and negative personality characteristics were also not affected by group (recall: main effect of group F(1,34)=0.080, p=0.7; group × valence × gender F(1,34)=656, p=0.4; recognition: main effect of group F(1,34)=0.092, p=0.8; group × valence × gender F(1,34)=1.20, p=0.2). Emotional memory in the female volunteers alone was also not affected by group (all comparisons p>0.3).
Emotion-potentiated startle response
There was no significant group × gender × picture type interaction. However, when the startle amplitude scores for the female participants were analysed separately, there was a significant main effect of group [F(1,12)=5.794, p=0.033]. This reflected a significant decrease in the overall startle reactivity in the female TRP group irrespective of picture type (see Fig. 2a). There was no main effect of group or interaction with picture type in the male participants.
Startle eye-blink responses in the female participants to a 95-dB burst of white noise presented during the presentation of neutral (black), pleasant (grey), and negative (light grey) pictures (± 1 SEM). Upper: Mean raw amplitude of eye-blink. Lower: Z-transformed startle responses. The asterisk represents the statistical significance of the comparisons: *p<0.05
Converting the startle amplitudes to standard (z) scores revealed a potentiated startle response to negatively valenced pictures, compared with positive and neutral pictures (see Fig. 2b), which was not significantly affected by group or gender (main effect of picture type [F(2.34)=5.51, p=0.01] interaction with group or gender ns, group × valence in females ns). Hence, in the female participants, startle responses were generally reduced in the TRP group, compared to placebo, irrespective of the picture stimuli presented.
Attentional probe task
Accuracy
There was no significant group × gender × valence interaction. However, when attentional vigilance scores for the female participants were analysed separately, there was a significant interaction between valence and group [F(1,17)=6.83, p=0.018] in the unmasked condition. As can be seen from Fig. 3, TRP significantly decreased attentional vigilance towards the negative words [t(17)=2.102, p=0.05]. There was no significant difference between groups in the male volunteers (main effect of group F(1,16)=0.531, p=0.7; group × valence F(1,16)=0.676, p=0.4) and no significant effect of group in the masked condition for either gender (female: main effect of group F(1,17)=0.144, p=0.7; group × valence F(1,17)=0.547, p=0.5; males: main effect of group F(1,16)=0.846, p=0.4; group × valence F(1,16)=0.001, p=1).
Attentional vigilance in the female participants towards negative and positive words compared to neutral in the unmasked condition of the attentional probe task. Values represent the response accuracy when the probe appeared in the opposite position to the emotional word (positive or negative) subtracted from the response accuracy when the probe appeared in the same position as the emotional word to give a measure of attentional vigilance. The asterisk depicts a significance level of difference from placebo: *p<0.05
Reaction times
Reaction Times on the attentional probe task were not affected by group in the masked or unmasked conditions (all comparisons p>0.2).
Discussion
This study examined the effect of 14 days of administration of tryptophan (TRP) on a number of measures of emotional processing in healthy volunteers. The findings suggest that repeated administration of TRP induces a positive bias in the processing of emotional material in women but not in men. In women, TRP administration increased the recognition of the positive facial expression of happy and decreased the recognition of the negative facial expression of disgust. TRP also reduced attentional vigilance towards negative words and decreased baseline startle responsivity.
Cognitive studies report that depressed patients show a negative bias in the processing of emotional material (Beck et al. 1979; Bradley et al. 1996; Murphy et al. 1999). Such a bias is believed to play an important role in the maintenance of the key symptoms of the disorder. The tasks used in the present study are good markers of such affective biases in information processing. Depression has been associated with a decreased perception of happy facial expressions and an increased classification of ambiguous faces as negative expressions, and this tendency predicts later relapse (Bouhuys et al. 1999; Gur et al. 1992; Surguladze et al. 2004). The processing of disgusted (Hayward et al. 2005) and fearful (Bhagwagar et al. 2004) facial expressions has also been shown to be enhanced in unmedicated recovered depressed patients, suggesting that some of these biases may persist into periods of euthymia. In addition, abnormal elevation of startle responses has also been reported in a number of anxiety disorders (Grillon et al. 1994; Grillon et al. 1998), and both depression and anxiety disorders have been associated with increased attentional vigilance towards negative stimuli (Bradley et al. 1997; MacLeod et al. 1986; Mogg et al. 1995) on the attentional probe task. This profile of effects is directly opposite to that seen in the present study after 14 days of TRP supplementation.
We previously reported that repeated administration of the SSRI citalopram in healthy volunteers (Harmer et al. 2004) produces biases on emotional processing away from negative and towards positive emotional material. Seven days administration of citalopram reduced the recognition of the negative facial expressions of disgust, fear and anger and increased the misclassification of these expressions as happy. Citalopram also abolished the potentiation of the startle response found in the context of negative affective images. A key question of the present study was whether increasing 5-HT synthesis has similar effects to inhibition of 5-HT reuptake. It is, therefore, of great interest that similar changes in some measures of emotional processing are seen after tryptophan supplementation.
As with the SSRI study, the changes in emotional processing seen in the present study occurred in the absence of any change in subjective mood. This suggests that the observed effects are the direct consequence of increased 5-HT levels in the brain, rather than a non-specific consequence of mood improvement. In support of this, we have previously demonstrated that a single dose of nutritionally sourced tryptophan (1.8 g) is sufficient to induce neuropsychological changes in the detection of facial expressions of emotion (Attenburrow et al. 2003) similar to those seen with acute citalopram administration (Harmer et al. 2003) Taken together, these findings suggest that, like serotonergic antidepressant agents, pharmacological doses of TRP can directly modulate the processing of emotional material. As such, this regime of tryptophan administration appears to affect central serotonergic levels in healthy volunteers consuming a normal diet.
Whilst the effects of TRP administration are reminiscent of those seen after SSRI administration, there are a number of differences in the profile of cognitive effects that may be important in understanding the potential therapeutic effects of TRP. Whilst citalopram reduced the recognition of a number of negative facial expressions (anger, disgust and fear), the effect of TRP was limited to disgust. The recall and recognition of positive and negative personality characteristics was also not affected by TRP, in contrast to the increased positive recall found after 7 days of citalopram. Consistent with previous literature, this suggests that TRP may be a milder manipulation of the serotonergic system than SSRIs.
A further difference is that the effect of repeated administration of citalopram on the emotion-potentiated startle task was restricted to the negative emotional condition, whereas, in the present study there was reduced startle responses across all conditions. Such a reduction in baseline startle reactivity is in contrast to the overall enhancement that is reported in a number of anxiety disorders (Grillon et al. 1994, 1998) and in those at high risk of major depressive disorder (Grillon et al. 2005). Consistent with these clinical effects and the present findings, acute TRP depletion (ATD) has also been shown to enhance baseline startle in unmedicated depressed patients (Hayward et al. 2005). Grillon (2002) has proposed that such elevated baseline startle reflects a greater sensitivity to the negative context of the startle experiment.
A similar distinction between specific fear-potentiated startle effects and more context-dependent effects has been established in the animal literature using the rodent startle paradigm, and it has been suggested that these effects are mediated by different neural substrates (Davis and Shi 1999). Specifically, the central nucleus of the amygdala has been shown to be critically involved in startle potentiation to explicit, phasic threat cues, whereas, the bed nucleus of the stria terminals has been linked to more sustained startle potentiation in response to contextual information, such as exposure to a threatening environment. Consistent with this notion, subchronic citalopram administration has recently been shown to reduce the amygdala response to threat-relevant stimuli in healthy volunteers, suggesting a direct effect of the drug on the amygdala which may underpin the specific reduction of the potentiation of the startle response in the presence of negative pictures (Harmer et al. 2006). Future neuroimaging studies are needed to understand the neural mechanisms through which TRP exerts its effects on emotional processing.
In the present study, the effects of TRP on emotional processing were only seen in the female volunteers. Consistent with this, the mood-lowering effect of ATD in both healthy volunteers and depressed patients has been shown to be more consistent in women, compared to men (Ellenbogen et al. 1996), and it is possible that this may be the consequence of ATD having a greater biochemical effect in women than in men, as demonstrated using radiolabelled alpha-methyl-l-tryptophan with positron emission tomography (Nishizawa et al. 1997). Interestingly, it is also possible that women have a better clinical response to SSRI treatment than men (Kornstein et al. 2001).
Taken together, these findings suggest that women may be generally more susceptible to serotonergic manipulation than men. However, it is important to note that the lack of effect in the male volunteers could have been due to pharmacokinetic factors, given the overall difference in weight between the male and female volunteers in this study. Furthermore, given the small male sample size in the startle task (due to equipment failure), it is not possible to draw conclusions about the effect of TRP in males on this task. Future studies are needed to further characterise the effect of TRP supplementation on emotional processing in male volunteers. The present study suggests, however, that TRP supplementation has greater effects on emotional processing in women than men and that this may be an important therapeutic consideration when using TRP clinically.
TRP supplementation also increased reaction times on the facial recognition task, which raises the possibility that the TRP may have had a mild sedative effect. Consistent with this, previous research has demonstrated that TRP has sedative effects in humans when administered in pure form and in sufficient quantity and may be useful in alleviating mild forms of insomnia (Hartmann and Elion 1977). Importantly, however, in the present study, subjects were not generally worse at recognising facial expressions per se, but rather showed specific and opposite changes in the perception of disgusted and happy faces. The consistency of these effects with those previously reported after citalopram administration strengthens the conclusion that they are the result of manipulations of the 5-HT system, rather than a non-specific effect of sedation.
An alternative explanation for the increased reaction times is that TRP decreased impulsivity. Whilst there were no direct tests of impulsivity in the present study, ATD has been shown to have the opposite effect and increase impulsivity, which is consistent with this notion (Walderhaug et al. 2002). This reaction time effect of TRP does raise the interesting possibility that whilst TRP has a positive bias on emotional processing, cognitive performance overall may not be improved. Given that deficits in general cognitive performance, such as memory and executive function, are a feature of depression, it is important that the effect of TRP on such processes is further characterised in future studies.
In summary, these findings provide evidence that TRP supplementation induces a positive bias in emotional processing in females that is reminiscent of the actions of serotonergic antidepressants. This highlights a key role for serotonin in emotional processing and lends support to the use of TRP as a nutritional supplement in people with mild depression or for prevention in those at risk. Future studies are needed to clarify the effect of tryptophan on these measures in men and to extend these findings into a clinical population.
References
Allen NB, Trinder J, Brennan C (1999) Affective startle modulation in clinical depression: preliminary findings. Biol Psychiatry 46:542–550
Anderson NH (1968) Likableness ratings of 555 personality-trait words. J Pers Soc Psychol 9:272–279
Attenburrow MJ, Williams C, Odontiadis J, Reed A, Powell J, Cowen PJ, Harmer CJ (2003) Acute administration of nutritionally sourced tryptophan increases fear recognition. Psychopharmacology (Berl) 169:104–107
Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J (1961) An inventory for measuring depression. Arch Gen Psychiatry 4:561–571
Beck AT, Rush AJ, Shaw BF, Emery G (1979) Cognitive therapy of depression. Guilford, New York
Benkelfat C, Ellenbogen MA, Dean P, Palmour RM, Young SN (1994) Mood-lowering effect of tryptophan depletion. Enhanced susceptibility in young men at genetic risk for major affective disorders. Arch Gen Psychiatry 51:687–697
Bhagwagar Z, Cowen PJ, Goodwin GM, Harmer CJ (2004) Normalization of enhanced fear recognition by acute SSRI treatment in subjects with a previous history of depression. Am J Psychiatry 161:166–168
Booij L, Van der DW, Benkelfat C, Bremner JD, Cowen PJ, Fava M, Gillin C, Leyton M, Moore P, Smith KA, Van der Kloot WA (2002) Predictors of mood response to acute tryptophan depletion. A reanalysis. Neuropsychopharmacology 27:852–861
Bouhuys AL, Geerts E, Gordijn MC (1999) Depressed patients’ perceptions of facial emotions in depressed and remitted states are associated with relapse: a longitudinal study. J Nerv Ment Dis 187:595–602
Bradley BP, Mogg K, Millar N (1996) Implicit memory bias in clinical and non-clinical depression. Behav Res Ther 34:865–879
Bradley BP, Mogg K, Lee SC (1997) Attentional biases for negative information in induced and naturally occurring dysphoria. Behav Res Ther 35:911–927
Buss AH, Durkee A (1957) An inventory for assessing different kinds of hostility. J Consult Psychol 21:343–349
Coppen A, Shaw DM, Farrell JP (1963) Potentiation of the antidepressive effect of a monoamine-oxidase inhibitor by tryptophan. Lancet 1:79–81
Davis M, Shi C (1999) The extended amygdala: are the central nucleus of the amygdala and the bed nucleus of the stria terminalis differentially involved in fear versus anxiety? Ann N Y Acad Sci 877:281–291
Delgado PL, Charney DS, Price LH, Aghajanian GK, Landis H, Heninger GR (1990) Serotonin function and the mechanism of antidepressant action. Reversal of antidepressant-induced remission by rapid depletion of plasma tryptophan. Arch Gen Psychiatry 47:411–418
Ekman P, Friesen WV (1976) Pictures of facial affect. Palo Alto, California
Ellenbogen MA, Young SN, Dean P, Palmour RM, Benkelfat C (1996) Mood response to acute tryptophan depletion in healthy volunteers: sex differences and temporal stability. Neuropsychopharmacology 15:465–474
Glassman AH, Platman SR (1969) Potentiation of a monoamine oxidase inhibitor by tryptophan. J Psychiatr Res 7:83–88
Grillon C (2002) Startle reactivity and anxiety disorders: aversive conditioning, context, and neurobiology. Biol Psychiatry 52:958–975
Grillon C, Ameli R, Goddard A, Woods SW, Davis M (1994) Baseline and fear-potentiated startle in panic disorder patients. Biol Psychiatry 35:431–439
Grillon C, Morgan CA III, Davis M, Southwick SM (1998) Effects of experimental context and explicit threat cues on acoustic startle in Vietnam veterans with posttraumatic stress disorder. Biol Psychiatry 44:1027–1036
Grillon C, Warner V, Hille J, Merikangas KR, Bruder GE, Tenke CE, Nomura Y, Leite P, Weissman MM (2005) Families at high and low risk for depression: a three-generation startle study. Biol Psychiatry 57:953–960
Gur RC, Erwin RJ, Gur RE, Zwil AS, Heimberg C, Kraemer HC (1992) Facial emotion discrimination: II. Behavioral findings in depression. Psychiatry Res 42:241–251
Harmer CJ, Bhagwagar Z, Perrett DI, Vollm BA, Cowen PJ, Goodwin GM (2003) Acute SSRI administration affects the processing of social cues in healthy volunteers. Neuropsychopharmacology 28:148–152
Harmer CJ, Shelley NC, Cowen PJ, Goodwin GM (2004) Increased positive versus negative affective perception and memory in healthy volunteers following selective serotonin and norepinephrine reuptake inhibition. Am J Psychiatry 161:1256–1263
Harmer CJ, Mackay C, Reid CB, Cowen PJ, Goodwin GM (2006) Antidepressant drug treatment modifies the neural processing of non-conscious threat cues. Biol Psychiatry (epub ahead of publication)
Hartmann E, Elion R (1977) The insomnia of ‘sleeping in a strange place’: effects of l-tryptophane. Psychopharmacology (Berl) 53:131–133
Hayward G, Goodwin GM, Cowen PJ, Harmer CJ (2005) Low-dose tryptophan depletion in recovered depressed patients induces changes in cognitive processing without depressive symptoms. Biol Psychiatry 57:517–524
Knutson B, Wolkowitz OM, Cole SW, Chan T, Moore EA, Johnson RC, Terpstra J, Turner RA, Reus VI (1998) Selective alteration of personality and social behavior by serotonergic intervention. Am J Psychiatry 155:373–379
Kornstein SG, Schatzberg AF, Thase ME, Yonkers KA, McCullough JP, Keitner GI, Gelenberg AJ, Davis SM, Harrison WM, Keller MB (2001) Gender differences in treatment response to sertraline versus imipramine in chronic depression. Am J Psychiatry 157:1445–1452
Lang PJ, Bradley MM, Cuthbert BN (1998) International Affective Picture System (IAPS): technical manual and affective ratings. Gainsville, University of Florida, Center for Research in Psychophysiology
Larson CL, Ruffalo D, Nietert JY, Davidson RJ (2000) Temporal stability of the emotion-modulated startle response. Psychophysiology 37:92–101
MacLeod C, Mathews A, Tata P (1986) Attentional bias in emotional disorders. J Abnorm Psychol 95:15–20
Mogg K, Bradley BP, Williams R (1995) Attentional bias in anxiety and depression: the role of awareness. Br J Clin Psychol 34(Pt 1):17–36
Moskowitz DS, Pinard G, Zuroff DC, Annable L, Young SN (2001) The effect of tryptophan on social interaction in everyday life: a placebo-controlled study. Neuropsychopharmacology 25:277–289
Murphy FC, Sahakian BJ, Rubinsztein JS, Michael A, Rogers RD, Robbins TW, Paykel ES (1999) Emotional bias and inhibitory control processes in mania and depression. Psychol Med 29:1307–1321
Murphy FC, Smith KA, Cowen PJ, Robbins TW, Sahakian BJ (2002) The effects of tryptophan depletion on cognitive and affective processing in healthy volunteers. Psychopharmacology (Berl) 163:42–53
Nishizawa S, Benkelfat C, Young SN, Leyton M, Mzengeza S, de MC, Blier P, Diksic M (1997) Differences between males and females in rates of serotonin synthesis in human brain. Proc Natl Acad Sci U S A 94:5308–5313
Pare CM (1963) Potentiation of monoamine-oxidase inhibitors by tryptophan. Lancet 35:527–528
Pihl RO, Young SN, Harden P, Plotnick S, Chamberlain B, Ervin FR (1995) Acute effect of altered tryptophan levels and alcohol on aggression in normal human males. Psychopharmacology (Berl) 119:353–360
Raleigh MJ, McGuire MT (1991) Bidirectional relationships between tryptophan and social behavior in vervet monkeys. Adv Exp Med Biol 294:289–298
Raleigh MJ, Brammer GL, Yuwiler A, Flannery JW, McGuire MT, Geller E (1980) Serotonergic influences on the social behavior of vervet monkeys (Cercopithecus aethiops sabaeus). Exp Neurol 68:322–334
Raleigh MJ, McGuire MT, Brammer GL, Pollack DB, Yuwiler A (1991) Serotonergic mechanisms promote dominance acquisition in adult male vervet monkeys. Brain Res 559:181–190
Shaw K, Turner J, Del MC (2002) Are tryptophan and 5-hydroxytryptophan effective treatments for depression? A meta-analysis. Aust N Z J Psychiatry 36:488–491
Smith KA, Fairburn CG, Cowen PJ (1997) Relapse of depression after rapid depletion of tryptophan. Lancet 349:915–919
Spielberger CD, Gorsuch RL, Lushene RD (1983) Manual for the State-Trait Anxiety Inventory (STAI). Consulting Psychologists, Palo Alto, California
Surguladze SA, Young AW, Senior C, Brebion G, Travis MJ, Phillips ML (2004) Recognition accuracy and response bias to happy and sad facial expressions in patients with major depression. Neuropsychology 18:212–218
Thomson J, Rankin H, Ashcroft GW, Yates CM, McQueen JK, Cummings SW (1982) The treatment of depression in general practice: a comparison of L-tryptophan, amitriptyline and a combination of L-tryptophan and amitriptyline with placebo. Psychol Med 12:741–751
von Zerssen D, Strian F, Schwarz D (1974) Evaluation of depressive states, especially in longitudinal studies. Mod Probl Pharmacopsychiatry 7:189–202
Walderhaug E, Lunde H, Nordvik JE, Landro NI, Refsum H, Magnusson A (2002) Lowering of serotonin by rapid tryptophan depletion increases impulsiveness in normal individuals. Psychopharmacology (Berl) 164:385–391
Watson D, Clark LA, Tellegen A (1988) Development and validation of brief measures of positive and negative affect: the PANAS scales. J Pers Soc Psychol 54:1063–1070
Weisman AN, Beck AT (1978) Development and validation of the Dysfunctional Attitudes Scale: a preliminary investigation. Paper presented at the American Education Research Association conference. Toronto, Canada
Young SN, Gauthier S (1981) Effect of tryptophan administration on tryptophan, 5-hydroxyindoleacetic acid in human lumbar and cisternal cerebrospinal fluid. J Neurol Neorosurg Psychiatry 44:323–328
Young SN, Smith SE, Pihl RO, Ervin FR (1985) Tryptophan depletion causes a rapid lowering of mood in normal males. Psychopharmacology (Berl) 87:173–177
Young AW, Rowland D, Calder AJ, Etcoff NL, Seth A, Perrett DI (1997) Facial expression megamix: tests of dimensional and category accounts of emotion recognition. Cognition 63:271–313
Acknowledgement
SM was supported by a Wellcome Trust studentship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Murphy, S.E., Longhitano, C., Ayres, R.E. et al. Tryptophan supplementation induces a positive bias in the processing of emotional material in healthy female volunteers. Psychopharmacology 187, 121–130 (2006). https://doi.org/10.1007/s00213-006-0401-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0401-8