Abstract
Rationale
The number of road fatalities related to the presence of amphetamines in drivers has been relatively constant over the past 10 years. However, there remains uncertainty as to the extent that these drugs induce driving impairment, and whether any such impairments translate to an increase in road fatalities.
Objectives
To examine the acute effects of 0.42 mg/kg dexamphetamine on simulated driving performance, and to establish which, if any, simulated driving abilities become impaired following dexamphetamine administration.
Methods
A repeated-measures, counter-balanced, double-blind, placebo-controlled design was employed. Twenty healthy volunteers completed two treatment conditions—0.42 mg/kg dexamphetamine and placebo. Performance was assessed using a driving simulator task. Blood and saliva samples were obtained prior to the driving tasks and immediately after task completion (120 min and 170 min post-drug administration, respectively).
Results
Mean dexamphetamine blood concentrations were 83 ng/ml and 98 ng/ml at 120 min and 170 min, respectively. Results indicated a decrease in overall simulated driving ability following dexamphetamine administration during the day-time but not the night-time scenario tasks. Contributing to this performance reduction, “incorrect signalling”, “failing to stop at a red traffic light” and “slow reaction times” were the behaviours most strongly affected by dexamphetamine.
Conclusions
The decrease in simulated driving ability observed during the day-time driving tasks are consistent with the perceptual narrowing or tunnel vision that is associated with dexamphetamine consumption.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Previous research has established alcohol as a leading contributor to road fatalities. However, over the last decade there has been an increased awareness of the role of drugs other than alcohol in the causation of road accidents and deaths (Drummer et al. 2003a,b), with the most recent report indicating that 23.5% of Australian road fatalities are drug related (Drummer et al. 2003a,b).
The involvement of psychotropic substances (medicinal or illicit) in road accidents has predominantly been examined by determining which drugs were present in the body tissues, blood or urine of drivers killed or injured in road accidents, or involved in traffic violations. The five drug groups that have been reported to be of most concern are alcohol, cannabis, opiates, stimulants and benzodiazepines, with the most recent report demonstrating that drivers tested positive for cannabis in 13.5% of road fatalities; for opioids, stimulants and benzodiazepines, the respective values were 4.9%, 4.1% and 4.1% (Drummer et al. 2003a,b). These statistics indicate that one of the most common classes of drugs associated with these road-related fatalities is amphetamines.
Amphetamines are non-catecholamine, sympathomimetic amines with central nervous system stimulant activity. While there are numerous subtypes of amphetamines, the most typical action of amphetamine is to facilitate the action of dopamine and noradrenaline by blocking re-uptake from the synapse, inhibiting the action of monoamine oxidase (MAO), and facilitating the release of dopamine and noradrenaline (Feldman et al. 1997). Thus, although amphetamines and related compounds have a strong molecular resemblance to these catecholamines, little of their activity seems to result from a direct agonist action at the receptor level. Amphetamines also enhance post-synaptic serotonin levels; however, their influence on mood and behaviour is thought to result primarily through the enhancement of dopamine and noradrenaline activity. With greater central activity, methamphetamine is a more potent version of dexamphetamine.
There is limited research available on the effects of amphetamines on in situ driving. Landmark culpability studies in Australia have compared drug levels in deceased drivers and have reported stimulants to have an odds ratio of 2.3 (Drummer et al. 2003a,b). Further, Logan (1996) and Logan et al. (1998) noted that typical driving behaviours observed in drivers under the influence of methamphetamine include drifting out of the lane, erratic driving, weaving, speeding, drifting off the road, an increase in risk taking and high-speed collisions. However, although epidemiological studies provide the most accurate representation of driving patterns in situ, they lack experimental control; thus, inferring causation remains problematic.
Experimental studies employing simulated driving have the advantage of greater control over causation but may not easily translate to real-life driving situations. Although there have been two experimental studies examining the effects of methylenedioxymethamphetamine (MDMA) on simulated driving (De Waard et al. 2000; Brookhuis et al. 2004), there has been no such research conducted on dexamphetamine or methamphetamine. It should be noted that although MDMA does share some general central nervous system activation effects with the above amphetamines, there are many affective and entactogenic activities that are quite different and, thus, it is not appropriate to discuss MDMA research here.
In contrast to epidemiological and simulated driving studies, there is a considerable body of knowledge relating the effects of amphetamines on cognition and behaviour. However, this research has also failed to provide consistent results.
In relation to risk-taking behaviour, the literature is contradictory. In terms of clinical research, amphetamines are known to be effective in the treatment of attention deficit hyperactivity disorder, reducing the impulsivity, restlessness and inattention (Zametkin and Rapoport 1987; Kupietz et al. 1985). However, in terms of experimental research, the findings are varied; some studies have reported decreases in impulsive behaviours following acute doses of stimulant drugs (De Wit et al. 2002) and others have shown increases in impulsive behaviours (Hurst 1962; Hurst et al. 1967; Evenden and Ryan 1996). Therefore, although there is a demonstrable relationship between amphetamine and impulsivity, it is clearly complex and dose-, situation- and/or population specific. These latter findings are, however, consistent with the above-mentioned epidemiological research study, where methamphetamine intoxication has been found to be associated with increased risk taking (Logan 1996).
In reference to cognitive performance, the literature indicates that, at lower doses, dexamphetamine appears to improve performance on some cognitive processes. De Wit et al. (2002) found that dexamphetamine improved performance on the digit symbol substitution test (DSST) and digit span—measures of vigilance and memory, respectively—suggesting that dexamphetamine may improve alertness or attention. Similar results have been reported by Wachtel and de Wit (1999) and Cami et al. (2000). Ward et al. (1997) found that 5 mg and 10 mg/70 kg dexamphetamine increased response rate on the DSST without affecting accuracy. In terms of reaction time, dexamphetamine has been shown to increase reaction time (Halliday et al. 1994; Fleming et al. 1995).
By extension, these findings may suggest that at low doses dexamphetamine may improve driving ability. However, contrary to this research, dexamphetamine-induced deficits have been reported on divided attention tasks (Mills et al. 2001). While this appears to be inconsistent with the literature that suggests improvement from dexamphetamine in some cognitive processes (e.g. attention), this may be understood in terms of the phenomena of tunnel vision. For example, Mills et al. (2001) found that 10 mg dexamphetamine induces “tunnel vision”, a phenomenon in which attentional processes become overwhelmed, producing a decrease in an individual’s ability to gather information efficiently. This is thought to occur when an individual experiences sympathetic arousal with a consequent restriction of perception to the focal point (Easterbrook 1959). This might be dangerous when driving as it increases the risk of failing to attend to potential hazards that fall outside of the driver’s attentional focus.
In summary, from the limited research available, it appears that amphetamines might be related to reckless driving, which may in turn contribute to drug-related driving fatalities. However, further research is required into the effects of amphetamine on driving ability. In terms of cognitive ability, amphetamines appear to improve performance at low doses, yet no firm conclusions can be drawn due to the inconsistent results related to differences in amphetamine, amphetamine dose and task type that have been used in the different studies. However, there is indirect evidence suggesting that amphetamine-related impairments in peripheral attention may provide a mechanism for amphetamine-related driving impairment, but this too is in need of further testing.
In order to resolve some of the above ambiguity and help clarify whether amphetamines do impair simulated driving abilities (and if so which abilities), the present study examined the effects of dexamphetamine on a range of simulated driving processes.
Materials and methods
Participants
Twenty healthy participants (10 males; 10 females) aged between 21 years and 32 years (mean±SD=25.4±3.3 years), with an average male weight of 82.1±10.6 kg and an average female weight of 62.2±10.4 kg, were recruited through advertisements. All participants had a minimum of 11 years education. Each was required to have a valid, full driver’s license (no probationary or learner drivers) to ensure that they had at least 3 years of driving experience. The Swinburne University Human Research Ethics Committee approved the research, and all participants provided written informed consent.
All participants were screened by undergoing an interview and a medical examination by a medical practitioner to ensure that they had no history of substance abuse; had no pre-existing physical or neurological conditions; had no history of psychiatric, cardiac, endocrine, gastrointestinal or bleeding disorders; were not pregnant or lactating; were not taking any prescription medication (excluding the contraceptive pill); and were not regular amphetamine users (i.e. they used less than once a month). Only participants who had previously experimented with amphetamines were permitted to participate.
Drug
Dexamphetamine sulphate (5-mg dexamphetamine tablets, Sigma Pharmaceuticals Pty Ltd, Vic., Australia) was prepared by mixing a 0.42-mg/kg dose of dexamphetamine tablets with flour, which was encapsulated in three soft gelatine capsules to render them visually indistinguishable from the placebo capsules, which contained only flour. In order to observe as similar to real-life amphetamine-induced effects as possible, this study administered a dose of 0.42 mg/kg as it is one of the highest doses, to the authors’ knowledge, administered to humans for controlled experimental research purposes.
Experimental design
A repeated-measures, counter-balanced, double-blind, placebo-controlled design was employed. Participants completed two treatment conditions: (i) placebo tablet and (ii) 0.42 mg/kg dexamphetamine tablet. Participants completed the two sessions 1 week apart to reduce traces and any cumulative effects of the drug if it was consumed during the first session. All participants consented to refrain from consuming alcohol for 24 h and no illicit drugs for at least 7 days prior to each session.
Measures
The driving simulator was the CyberCAR LITE driver training and evaluation simulator (Thoroughbred Technologies Pty Ltd). The steering wheel, a “Force Feedback”, with integrated horn, indicators, headlights, ignition, automatic gears and hand brake, was affixed to a bench; brake and accelerator pedals were placed underneath the bench. Participants could adjust the pedal and seat position to suit their height. The simulator task was projected onto a 175×120-cm white screen (distance from steering wheel was 280 cm), Participants observed a two-dimensional computer-generated driving scene, as they would through a vehicle windscreen. The simulated dashboard, which was also projected onto the white screen, included a speedometer, rear-view mirror, and side-mirrors. The tasks administered employed a simulated conventional on-road light motor vehicle with automatic transmission.
The driving simulator program consisted of two modules: the “basic driving module” and the “driving module”. The basic driving module consisted of two tasks that were used to assess basic steering ability and basic speed control. This module was administered for practice, to familiarise participants with the driving simulator and to ensure that they felt confident with the steering, accelerator and brake. The driving module consisted of four tasks—“freeway traffic driving” and “city traffic driving”, under both day and night conditions. Each task took approximately 5 min to complete. The computer program recorded each driver’s performance continuously on a range of variables, in terms of vehicle management and conformance to the pre-programmed set of driver and vehicle standard operating procedures. Following previous research in our laboratory, a subset of 34 relevant variables was analysed, where each reflected an error that can occur during the driving tasks. Each variable score was multiplied by that variable’s “loading factor”, a number which represents the severity of the error, and subsequently all adjusted variable scores were summed to give an overall impairment score. Driving simulator variable scores were summed separately for the day and night conditions. For each, a total score between 0 and 75 was classified as “not impaired” on the driving simulator task, whereas a total score of 76 and above constituted an assessment of “impaired” on the driving task.
Snellen Eye Chart
The Snellen Eye Chart is a standard measure of visual acuity. This was scored such that the number of letters identified correctly was the indication of visual acuity (i.e. higher scores indicated better vision). This was administered to clarify whether any dexamphetamine-related changes in performance were associated with changes in visual acuity.
Blood and saliva samples
Two blood and two saliva samples were taken from each participant during each session. A 10-ml blood sample was obtained with a syringe, by venipuncture from the antecubital vein, and a 1-ml saliva sample was obtained with Cozart Rapiscan (Biomediq DPC Pty Ltd) saliva collection kits, at 120 min and 170 min after drug administration. Blood and saliva samples were immediately stored in a −20°C freezer and subsequently transported to a −70°C freezer after 5–7 days. Blood and saliva samples were analysed for amphetamine levels using the gas chromatography–mass spectroscopy method (Moeller and Kraemer 2002).
Procedure
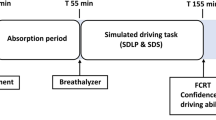
In a preliminary session, on a separate day in which no drug was administered, participants completed the four simulated driving tasks described above as practice. For the two experimental sessions, participants were asked to eat a normal breakfast or lunch before arrival. The experimenter and participant were blind to the treatment condition. A medical practitioner was on-call and a registered nurse was on-site throughout experimental sessions.
Upon arrival on the two experimental days, participants completed the city-traffic simulated driving task (to re-familiarise themselves with the driving simulator). The research nurse then administered the treatment. As dexamphetamine has a peak blood concentration between 120 min and 180 min (Angrist et al. 1987; Kupietz et al. 1985), the first blood and saliva samples were obtained 120 min after drug administration, followed by the Snellen Eye Test and the driving simulator tasks. The second set of blood and saliva samples were then obtained (170 min post-drug administration). The only adverse reaction to amphetamine consumption reported was difficulty in falling asleep and/or disturbed sleep on the night following that session.
Statistical analyses
As the driving simulator task required participants to drive in city and freeway scenarios in two simulated conditions (day and night), data were analysed separately for the day- (freeway and city combined) and night- (freeway and city combined) driving tasks. For each of the day and night conditions, a test of difference in proportions based on paired data (Newcombe 1998) was performed to establish whether there was any relationship between overall simulated driving ability and the presence of dexamphetamine, where the independent variable was drug condition (placebo versus dexamphetamine) and the classification of driving ability (impaired versus not impaired) was the dependent variable. A Bonferroni adjustment was made to correct for type-1 error, resulting in a corrected alpha level of 0.025.
The second set of analyses was a series of Wilcoxon signed-rank tests. These explored the effects of dexamphetamine on each individual driving simulator variable, where drug condition (placebo versus dexamphetamine) was the independent variable and the score for each driving variable was the dependent variable. No correction for multiple comparisons was made, as these analyses were exploratory.
Two paired samples t-tests were performed to determine whether dexamphetamine affected visual acuity. A Pearson’s correlation was performed to determine whether any dexamphetamine-related changes in simulated driving performance were associated with changes in visual acuity.
Results
The mean dexamphetamine concentration levels in blood and saliva at 120 min after drug administration were 83 ng/ml and 236 ng/ml, respectively, and at 170 min after drug administration 98 ng/ml and 242 ng/ml, respectively.
Visual acuity in the left eye significantly decreased under the dexamphetamine condition (t18=2.28, P=0.04); however, there was no difference of acuity in the right eye when comparing the dexamphetamine and placebo conditions (t18=0.62, P=0.55).
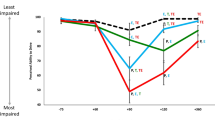
An overall reduction in simulated driving performance was observed under the dexamphetamine condition (19 of 20 participants “impaired”) relative to the placebo condition (13 of 20 “impaired”) for the day-time simulated driving condition (P<0.05, 95% CI = −0.528 to −0.028). However, dexamphetamine did not affect overall simulated driving ability under the night-time driving condition (dexamphetamine 17 of 20 impaired; placebo 17 of 20 impaired; P>0.05, 95% CI = −0.230 to 0.230).
Simulated day driving (city and freeway combined)
As depicted in Table 1, there was an overall trend towards decreased signalling adherence under the dexamphetamine condition, such as at intersections (T=0, P<0.01), when entering a freeway (T=3, P=0.096) and during lane changes, T=46, P=0.08. Additionally, there was a trend found towards drivers failing to stop at a red traffic light more frequently under the dexamphetamine than placebo conditions (T=4, P=0.06). A significant difference was found between the two conditions with regard to the speed the vehicle was travelling on a freeway when an emergency situation occurred, with more drivers under the dexamphetamine condition than placebo condition travelling at a slower speed (T=24, P<0.01).
Poorer visual acuity in the left eye under the dexamphetamine condition was not found to be associated with the observed decrease in simulated day-time driving performance (r19=0.12, P=0.63).
Simulated night driving (city and freeway combined)
As can be seen in Table 1, there was a trend towards a decrease in reaction time under the dexamphetamine condition (T=44, P=0.07).
Discussion
The present study found that 0.42 mg/kg dexamphetamine significantly decreased overall simulated driving performance for the day-time but not night-time driving conditions. Specifically, during the day-time driving tasks, there was a trend towards increased incorrect signalling and failing to stop at a red traffic light when under the dexamphetamine condition. Additionally, drivers travelled at a significantly slower speed on the freeway while under the dexamphetamine condition than the placebo condition. Although no significant decrease in simulated driving ability was found for the night driving condition, there was a trend towards a decreased reaction time under the dexamphetamine condition. As the driving simulator task was completed within the 2- to 3-h post-drug administration period, and as dexamphetamine blood concentrations are relatively constant during this period (Kupietz et al. 1985; Brauer et al. 1996), it is reasonable to conclude that the observed reduction in driving skills corresponded to mean blood and saliva dexamphetamine concentrations of approximately 90 ng/ml and 240 ng/ml, respectively.
Further, poorer left eye visual acuity was found under the dexamphetamine condition. It is not clear what the mechanism for this action is [as the only literature that we are aware of suggests that dexamphetamine may improve visual acuity, and does so bilaterally (Adachi-Usami 1990)]. However, it should be noted that this reduction in visual acuity was not statistically related to the above simulated driving performance decrements.
Since no simulated driving studies examining the effects of dexamphetamine on driving performance have previously been conducted, it is difficult to directly relate the results of the present investigation to previous research. However, the decrease in simulated driving performance for the day-time driving task may be seen as inconsistent with the literature discussed above, which indicates cognitive enhancement following dexamphetamine administration. One possible explanation for this apparent discrepancy is that the present study employed dexamphetamine doses substantially larger (average 30 mg) than were administered in that previous study (where doses ranged from 10 mg to 20 mg). That is, the present performance decrements could be explained by the well-known “inverted-U” phenomena whereby low-level enhancements of dopamine function improve cognitive performance, whereas high-level enhancements of dopamine function result in performance decrements (Williams and Goldman-Rakic 1995). Although only speculative, this explanation is consistent with the epidemiological evidence of a relationship between amphetamine use and road fatalities, in that these higher doses in the present study are representative of the low range of doses used both recreationally and by truck drivers (Logan 1996; Logan et al. 1998).
Another possibility, which is also consistent with the epidemiological findings of a relationship between amphetamine use and road fatalities, is that the performance decrements in the present study were caused by the perceptual narrowing that has been observed following dexamphetamine administration (Mills et al. 2001). Dexamphetamine can cause sympathetic arousal, which results in greater acuity at the focus of attention, with a corresponding loss of acuity peripherally. This phenomenon, first described by Easterbrook (1959), is referred to as “tunnelling” or “tunnel vision” and may diminish a driver’s capacity to gather and organise appropriate information, particularly where this information falls outside of the driver’s focus; consequently this may impair driving ability. This phenomenon has even been observed following administration of 10 mg dexamphetamine, which resulted in blood concentrations (30 ng/ml) substantially lower than those observed in the present study (Mills et al. 2001).
Two findings in particular are consistent with the interpretation that performance decrements were the result of a dexamphetamine-induced tunnel vision. The first is that participants in the dexamphetamine condition travelled significantly slower in the simulated freeway condition (which is inconsistent with previous in situ research that found that drivers under the influence of other derivatives of amphetamine, specifically methamphetamine, tend to speed more; Logan 1996). This speed reduction in the laboratory setting may act as a compensatory mechanism to permit drivers to attend to peripheral information more adequately. The second is that consistent with Logan’s (1996) finding that drivers under the influence of methamphetamine were more likely to fail to stop at a red traffic light, the present trend-level findings of the same performance decrement may also be related to the tunnel vision phenomena as these traffic lights were in the subjects’ periphery.
It is interesting to note that the decrease in simulated driving ability was observed under the day- but not the night driving conditions. Two reasons for this appear plausible. First, the dexamphetamine-induced tunnel vision effect (Mills et al. 2001) may be more relevant during the day-time driving task because visibility is much clearer and more information is available than at night. To avoid an overload of information, drivers may thus need to appropriately select the information that is relevant and subsequently attend to it, a process that requires attention to move between the fovea and periphery. It would thus be useful to obtain data on the times of day that drivers are apprehended for amphetamine-related driving impairment and/or road fatalities (which currently is not available), to determine whether this explanation is consistent with in situ driving and with the culture and driving patterns of both recreational amphetamine users and truck drivers.
A second possibility is that this may be related to the participants’ lack of driving proficiency, as 17 of the 20 participants attained the “impaired” cut-off score under the placebo night condition. This may have produced a floor effect and made it difficult to detect a further performance decrement. The high level of participants reaching the “impaired” cut-off score does not appear to be due to limitations of the driving simulator, as we routinely use it in our laboratory and have not observed a similar level of impairment previously. However, as it was a condition of participation in the study that all participants had used amphetamines in the past (for ethical reasons), it is possible that this unusually high rate of poor driving may be related to prior exposure to the drug.
A possible limitation of the study was that the simulated driving tasks were not randomised, as these computer-based tasks were pre-programmed and not amendable. That is, the night freeway driving task was always completed after the day-time freeway task, and the night city task was always completed following the day-time city driving task, which introduces the possibility of practice and fatigue effects. However, minimising the possibility that practice effects accounted for the day/night discrepancy, participants practiced all driving tasks on a separate day to the experimental sessions, as well as practicing the day city-driving task on each of the testing sessions (prior to drug administration). Fatigue would also be an unlikely explanation of the difference between the day and night results as the order of tasks (i.e. day/night/day/night) meant that any such fatigue effects would be small.
In conclusion, the results of the present study suggest that dexamphetamine does decrease simulated driving performance in recreational users in a day-time driving scenario. It is not clear whether it also occurs under night-time driving conditions due to limitations of the night simulation component of the task. Contributing to this overall reduction in day-time simulated driving performance, there was some evidence to suggest that dexamphetamine affected signalling and traffic light adherence, and drivers were found to travel significantly more slowly under the simulated freeway condition. These results are consistent with perceptual narrowing or tunnel vision effects, where peripheral vision is impaired with dexamphetamine; however, this interpretation remains tentative and further research is needed to clarify this issue.
References
Adachi-Usami E (1990) Senescence of visual function as studied by visually evoked cortical potentials. Jpn J Ophthalmol 34(1):81–94
Angrist B, Corwin J, Bartlik B, Cooper T (1987) Early pharmacokinetics and clinical effects of oral d-amphetamine in normal subjects. Biol Psychiatry 22:1357–1368
Brauer LH, Ambre J, de Wit H (1996) Acute tolerance to subjective but not cardiovascular effects of d-amphetamine in normal healthy men. J Clin Psychopharmacol 16(1):72–76
Brookhuis KA, De Waard D et al (2004) Effects of MDMA (ecstasy), and multiple drugs use on (simulated) driving performance and traffic safety. Psychopharmacology (Berl) 173(3–4):440–445
Cami J, Farre M, Mas M, Roset P, Poudevida S, Mas A, San L, de la Torre R (2000) Human pharmacology of 3, 4-methylenedioxymeth-amphetamine (“Ecstasy”): psychomotor performance and subjective effects. J Clin Psychopharmacol 20(4):455–466
De Waard D, Brookhuis KA, Pernot LMC (2000) A driving simulator study ofthe effects of MDMA (Ecstasy) on driving performance and traffic safety.Proceedings of the International Council on Alcohol Drugs and TrafficSafety (ICADTS), Stockhom Sweden, May 2000
De Wit H, Enggasser JL, Richards JB (2002) Acute administration of d-amphetamine decreases impulsivity in healthy volunteers. Neuropsychopharmacology 27(5):813–825
Drummer OH, Gerostamoulos J, Batziris H, Chu M, Caplehorn J, Robertson MD, Swann P (2003a) The involvement of drugs in drivers of motor vehicles killed in Australian road traffic crashes. Accid Anal Prev 943:1–10
Drummer OH, Gerostamoulos J, Batziris H, Chu M, Caplehorn JRM, Robertson MD, Swann P (2003b). The incidence of drugs in drivers killed in Australian road traffic crashes. Forensic Sci Int 134:154–162
Easterbrook JA (1959) The effect of emotion on cue utilization and the organisation of behaviour. Psychol Rev 66(3):183–201
Evenden Jl, Ryan CN (1996) The pharmacology of impulsive behaviours in rats: the effects of drugs on response choice with varying delays of reinforcement. Psychopharmacology (Berl) 128:161–170
Feldman RS, Meyer JS, Quenzer LF (1997) Principles of neuropsychopharmacology. Sinauer Associates, Incorporated. Sunderland,MA
Fleming K, Bigelow LB, Weinberger DR, Goldberg TE (1995) Neuropsychological effects of amphetamine may correlate with personality characteristics. Psychopharmacol Bull 31(2):357–362
Halliday R, Naylor H, Brandeis D, Callaway E, Yano L, Herzig K (1994) The effect of d-amphetamine, clonidine, and yohimbine on human information processing. Psychophysiology 31:331–337
Hurst PM (1962) The effects of d-amphetamine on risk taking. Psychopharmacologia 3:283–290
Hurst PM, Weidner MF, Radlow R (1967) The effects of amphetamines upon judgement and decisions. Psychopharmacologia 1(5):397–404
Kupietz SS, Bartlik B, Angrist B, Winsberg BG (1985) Psychostimulant plasma concentration and learning performance. J Clin Psychopharmacol 5(5):293–295
Logan BK (1996) Methamphetamine and driving impairment. J Forensic Sci 41(3):457–464
Logan BK, Fligner CL et al (1998) Cause and manner of death in fatalities involving methamphetamine. J Forensic Sci 43(1):28–34
Mills KC, Spruill SE, Kanne RW, Parkman KM, Zhang Y (2001) The influence of stimulants, sedatives, and fatigue on tunnel vision: risk factors for driving and piloting. Hum Factors 43(2):310–327
Moeller MR, Kraemer T (2002) Drugs of abuse monitoring in blood for control of driving under the influence of drugs. Ther Drug Monit 24:210–221
Newcombe RG (1998) Improved confidence intervals for the difference between binomial proportions based on paired data. Stat Med 17:2635–2650
Wachtel SR, de Wit H (1999) Subjective and behavioural effects of repeated d-amphetamine in humans. Behav Pharmacol 10:271–281
Ward AS, Kelly TH, Foltin RW, Fischman MW (1997) Effects of d-amphetamine on task performance and social behaviour of humans in a residential laboratory. Exp Clin Psychopharmacol 5(2):130–136
Williams GV, Goldman-Rakic PS (1995) Modulation of memory fields by dopamine D1 receptors in prefrontal cortex. Nature 376:549–550
Zametkin A, Rapoport J (1987) Neurobiology of attention deficit disorder with hyperactivity: where have we come in 50 years? J Am Acad Child Adolesc Psychiatry 26:676–686
Acknowledgement
Vic Roads, Melbourne, Australia, provided funding for the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Silber, B.Y., Papafotiou, K., Croft, R.J. et al. The effects of dexamphetamine on simulated driving performance. Psychopharmacology 179, 536–543 (2005). https://doi.org/10.1007/s00213-004-2061-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-004-2061-x