Abstract
Rationale
The cholinergic system is linked extensively to memory, but its exact role remains controversial. In particular, scopolamine-induced impairment in rodents is not task specific, which may be due to difficulty in developing rodent protocols to assess deficits in recent memory, in which the remembered event is brief and distinct, and/or to non-specific behavioral impairment.
Objectives
The present study sought to determine whether scopolamine-induced deficits in recent memory, using a working memory task, could be dose-specifically dissociated from deficits in associative memory in dogs.
Methods
A Latin-square design was used to determine the effect of scopolamine (5, 10 and 15 μg/kg; SC) on a variable delayed-non-matching-to-position (DNMP) task, which assesses visuospatial working memory. Subsequently, the minimal effective dose (15 μg/kg; SC) was administered prior to testing on a landmark discrimination task, which provides a measure of allocentric spatial ability, a black-white discrimination task, an oddity discrimination task and tests of exploratory behavior. We also investigated the effects of a 30 μg/kg dose (SC) on tests of oddity discrimination and behavioral activity.
Results
A 15 μg/kg dose produced significant impairment on the DNMP task, but did not affect performance of any discrimination task and did not alter behavior on tests of open field or curiosity. A 30 μg/kg dose caused disruption on discrimination performance and on open field measures.
Conclusions
Working memory performance is most sensitive to scopolamine-induced impairment and can be dissociated from scopolamine-induced deficits in discrimination performance and non-cognitive behaviors. The present results indicate that scopolamine-induced impairments of working memory in the dog can serve as a model of age-related cholinergic dysfunction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Reduced central cholinergic neurotransmission is linked to impairment of memory processes in humans. Scopolamine, a non-specific muscarinic acetylcholine receptor antagonist, impairs memory of young subjects to a performance level comparable to drug-free, non-demented elderly subjects (Drachman and Leavitt 1974) and consistently impairs memory in the aged to a greater extent than the young (Flicker et al. 1992; Molchan et al. 1992; Ray et al. 1992; Tariot et al. 1996). Post-mortem studies of Alzheimer’s disease (AD) patients demonstrate degeneration of the cholinergic forebrain (Whitehouse et al. 1981, 1982) and reduced levels of choline acetyltransferase (Davies and Maloney 1976; Perry et al. 1977), which is correlated negatively with pre-morbid intellectual impairments (Perry et al. 1978). Cholinergic dysfunction likely is responsible, in part, for the memory decline associated with aging and AD.
In rodents, scopolamine impairs object-recognition memory (Dodart et al. 1997), working memory (Ravel et al. 1992; Andrews et al. 1994; Kirkby et al. 1995; Pilcher et al. 1997), reference memory (Biggan et al. 1996), conditioned learning (Flood and Cherkin 1986) and conditioned memory (Sessions et al. 1998). Studies in non-human primates, however, suggest that recent memory is particularly sensitive to scopolamine impairment (Bartus and Johnson 1976). Two hypotheses for this inconsistency are of importance to the present study. First, behavioral protocols used in rodents for assessing recent memory, which is particularly sensitive to aging (Flicker et al. 1984) and cholinergic disruption (Bartus and Johnson 1976), may be limited in their utility to model recent memory in the primate (Bartus 2000). Second, the extensive impairing effects of scopolamine in rodents may be due to dose-related non-mnemonic effects on performance (Blokland 1995).
The present study addressed the specificity of scopolamine deficits by assessing the effects of scopolamine on various canine neuropsychological tests that are comparable to tests used in humans (e.g. Jha and McCarthy 2000; Lew et al. 2000). Dogs demonstrate age-dependent cognitive impairment and β-amyloid neuropathology that models human aging and dementia (Cummings et al. 1996; Head et al. 1998, 2000; Adams et al. 2000a), but little is known about the role of the cholinergic system in canine cognition. Two cholinergic agonists, citicoline and carbacholine, improve performance on operant conditioning tasks (Bruhwyler et al. 1998; Shapovalova 1999). Performance on these tasks varies as a function of motivation, impulsivity and motor response; improvement by cholinergic agonists, therefore, is not necessarily indicative of cognitive or mnemonic enhancement.
In the present study, recent memory in aged beagles was examined using a delayed-non-matching-to-position (DNMP) task, which provides an age-sensitive measure of visuospatial working memory (Milgram et al. 1994; Head et al. 1995; Adams et al. 2000b). As defined by Bartus (2002), the operational features of recent memory tasks include a non-repetitive and brief remembered event, a rapid decay of retention, and trial-specific information, which are all characteristics of the DNMP task. An incremental dose protocol was used to determine the minimal dose that impaired performance. Subsequently, the effect of this dose was assessed on a variety of non-mnemonic tasks, including tests of allocentric visuospatial function, object discrimination, reference memory, and on tests of spontaneous behavior. Finally, the effects of a higher dose were investigated on a subset of these tests to explore further the relationship between task-specific deficits and dose.
Materials and methods
Subjects
The subjects were random-source beagle dogs, >8 years of age, obtained from the colony at the University of Toronto. All subjects had extensive experience on various canine neuropsychological tests. Because the experiments were carried out over a number of years and a pre-specified level of performance was required, not all subjects were available for all the experiments. The subjects were housed either individually, or in pairs, in pens measuring approximately 1.07×1.22 m, with constant access to water. They were fed in the afternoon following cognitive testing and were exercised once daily for approximately 15 min. The subjects were given regular veterinary exams to ensure that performance was not affected by health problems, or by neurological or sensory deficits. All procedures were conducted in accordance with the “Guide for the care and use of laboratory animals” (NIH publication No. 85-23, revised 1996) and the guidelines of the Canadian Council on Animal Care.
Test apparatus
As previously described (Milgram et al. 1994), the test apparatus consisted of a 0.61×1.15×1.08 m wooden chamber with adjustable stainless steel bars covering the front of the box. Samples were presented on a sliding Plexiglas tray, which contained one medial and two lateral food wells. Displacing an object covering the appropriate food well revealed the food reward, approximately 1 cc of wet dog food (Hill’s P/D diet). The subject was separated from the tester by a wooden screen with a one-way mirror. A hinged door at the bottom of the wooden screen allowed presentation of the sliding tray. All testing occurred in darkness, except for an incandescent light source attached to the front of the chamber.
Procedures
Visuospatial memory performance
The DNMP task was used to assess visuospatial memory performance and was described by Head et al. (1995). Briefly, each trial consisted of two phases, the sample phase and the choice phase. During the sample phase, a red block was presented to the subject over one of the lateral food wells. After an inspection interval (5 s), the subject was allowed to displace the red block and retrieve the food reward beneath the block. The tray was then retracted and the delay interval started. During the choice phase, which followed the delay, the subjects were presented with two identical red blocks covering the lateral food wells. Displacing the block over the non-match position permitted access to the food reward. The inter-trial interval (ITI) was 1 min, and the animals were given 12 trials daily. The correct response occurred to both possible positions equally throughout a session. For this and all subsequent cognitive testing, food reward was smeared under the non-rewarded object to prevent the dogs from using olfactory cues, a correction procedure was used in which subjects were allowed to correct their response once each session after making their first incorrect response, and randomization procedures were controlled using a dedicated computer program, which indicated correct choices, controlled for delays, and allowed the tester to record responses and response-choice latencies.
For the dose-response study, six male and six female subjects were used. The subjects were first tested on a variable-delay DNMP task, using delays of 20, 70, and 110 s, for 15 days to establish performance stability. Testing then was divided into three 6-day test blocks, each of which consisted of 3 control days, 1 treatment day, and 2 drug-washout days. On the treatment day, each group was administered one of three doses of scopolamine (5, 10, or 15 μg/kg). Dogs were paired according to baseline performance into six groups and a Latin-square design was used to randomize the six possible orders of treatment, thus ensuring that no two pairs received the same order of test doses over the three blocks.
Allocentric visuospatial performance
The landmark task provides a measure of allocentric visuospatial function and was described by Milgram et al. (1999). The two lateral wells were used, each covered by identical coasters. The location of an external landmark, a yellow peg, indicated which coaster was associated with reward such that subjects were rewarded for displacing the coaster closest to the landmark. In the present study, three male and one female subjects were selected for inclusion from a larger group, based on their ability to perform at a criterion level when the distance between the landmark and the coaster was 1 cm. A variable distance protocol was used in this study in which the landmark was placed either on top of, at a distance of 1 cm, or at a distance of 4 cm from the rewarded coaster. Each of the three landmark locations occurred on exactly four trials per session, and the left and right sides each were correct on exactly half of these trials. Testing occurred over 15 days and was divided into four blocks, each of which consisted of 1 control day, 1 vehicle day, 1 treatment day (using the 15 μg/kg dose) and a drug-washout day.
Simple discrimination performance
A black-white discrimination task was used to examine the effects of scopolamine on object discrimination ability. The stimuli for this task consisted of two wooden blocks that were identical except for color; one was white and the other black. Subjects were first trained to approach one of the two blocks to obtain food reward to a performance level of greater than 70% accuracy. Each test session consisted of ten trials separated by a 30-s ITI. For the test protocol, one male and four female subjects were tested on the black-white discrimination task. Testing occurred over 7 days consisting of two blocks, using a design that was identical to that described for the landmark task.
Complex discrimination performance
An oddity discrimination task, as described previously by Milgram et al. (2002), was used to examine the effects of scopolamine on a more complex discrimination task. Briefly, the subjects were presented with two identical objects and a third object that differed. Subjects initially were trained to approach the “odd” object from the three until a similar criterion as in the black-white discrimination test was reached. Each test session consisted of 12 trials separated by a 30-s ITI and the location of the rewarded object occurred equally in the three possible positions within a test session. Seven male and two female subjects were tested on the oddity-discrimination task. Testing occurred over 15 days and was divided into four blocks, as described for the landmark task. A 30-μg/kg and 15-μg/kg dose was administered on the first and third, and on the second and fourth blocks, respectively.
Open field activity and exploratory behavior
Open-field arena
All non-cognitive tests (see Siwak et al. 2001) occurred in a room measuring 274.32×252.73 cm. To facilitate tracking a subject during videotape analysis, strips of electrical tape were applied to the floor in a grid pattern. The grid contained 25 rectangles measuring 49.02×53.34 cm. The floor of the test room was cleansed prior to testing and between animals to reduce olfactory cues from modifying behaviors and disrupting any effects.
Curiosity test
Four female and four male subjects were tested twice on the curiosity test, a test of exploratory behavior, with a 1-day washout period. The subjects were divided randomly into two groups of equal male and female subjects, where one group was administered 15 μg/kg scopolamine the first day, and the second group received the drug on the second day. For the curiosity test, seven objects were placed throughout the open-field arena and the subjects were videotaped for a 10-min period. Behaviors were recorded using a computer with dedicated software and included: locomotion, inactivity, sniffing, urinating, jumping, rearing, vocalization, urinating on the objects, picking up the objects, sniffing the objects and interacting with the objects.
Open-field test
Seven male and seven female subjects were tested on the open-field test over 3 days each separated by a drug-washout day. On each day, subjects pseudo-randomly received one of three possible injections; saline, 15 μg/kg, or 30 μg/kg of scopolamine. The open-field test was identical to the curiosity test except that the arena did not contain any objects. Consequently, behaviors associated with object interactions were not included in the analysis.
Drug and preparation
Scopolamine hydrobromide was obtained from Sigma-Aldrich Inc. The drug was prepared for administration in a 100 μg/ml base solution by dissolution in 0.9% normal saline and was administered subcutaneously 1 h prior to testing. Vehicle injections consisted of an equivalent volume of 0.9% normal saline.
Data analysis
Repeated-measures analyses of variance (ANOVAs) were used to evaluate the effect of scopolamine on all cognitive tests. Percent accuracy, based on the number of correct choices, served as the dependent variable in all cognitive analyses. For the dose-response analyses, no differences were found in accuracy between control days at each dose for the DNMP dose-response experiment, or between control and vehicle days on the oddity experiment. Consequently, the control score used for the dose-response analyses was the mean percentage accuracy over all the control days for the DNMP experiment and the mean percentage accuracy on the control and vehicle days for the oddity experiment. Mean daily latencies were used to examine the effects of scopolamine on latency to respond. For the DNMP task, a two-way ANOVA was conducted with dose (4 levels) and delay (3 levels) as within-subject variables. For the landmark task, a two-way ANOVA was conducted with treatment (3 levels) and distance (3 levels) as within-subject variables. For the black-white discrimination, a one-way ANOVA was conducted with treatment (3 levels) serving as a within-subject variable. The oddity-discrimination results were analyzed using a one-way ANOVA with treatment (3 levels) serving as a within-subject variable. To determine differences in control performance among the four cognitive tests, the mean score on control and saline days were compared using a one-way ANOVA. For the curiosity test and open-field analyses, individual dependent t-tests and one-way ANOVAs were used, respectively. An alpha level of 0.05 was used to determine significance for all comparisons. Appropriate post-hoc tests were used to determine specific effects.
Results
Visuospatial memory performance
Performance accuracy was significantly affected by delay [F(2,22)=6.22, P<0.01] and dose [F(3,33)=14.75, P<0.001]. Dunnett’s post-hoc analysis indicated that only performance under the 15 μg/kg dose differed significantly from control (P<0.005) (Fig. 1) and was, therefore, the minimally effective dose in disrupting behavior. Post-hoc Tukey’s revealed that performance under the 20-s delay differed significantly from the 50- (P<0.05) and 70-s (P<0.01) delays. Performance under the 50- and 70-s delays did not differ. Although latency decreased under scopolamine, this effect was not significant.
Effect of scopolamine on performance accuracy across delays. Compared to control (black circles), performance on the variable-delay DNMP was not affected by the 5 μg/kg (white circles) or the 10 μg/kg (black triangles) doses at any delay. The 15 μg/kg (white triangles) dose significantly impaired performance at all delays. The horizontal dashed line is indicative of chance performance. Error bars represent the SEM
Allocentric visuospatial performance
Performance on the landmark task showed a trend towards decreasing accuracy with increased distance [F(2,6)=4.40, P<0.07], but was not affected by scopolamine.
Simple and complex discrimination performance
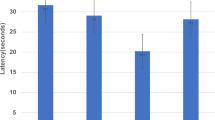
Figure 2 indicates that performance accuracy of the black-white discrimination task under the 15 μg/kg dose of scopolamine decreased slightly compared to control and saline conditions. This effect, however, was not significant. A similar effect was observed with the 15 μg/kg dose on the oddity discrimination task; however, the 30 μg/kg dose severely impaired performance accuracy (Fig. 2). One subject’s data were removed from the analysis due to response absences on all trials following either dose of scopolamine. A significant effect of dose [F(2,14)=22.74; P<0.0001] on performance accuracy was found. Post-hoc Tukey’s indicated that this effect was due to the decrease in performance accuracy following the 30 μg/kg dose of scopolamine compared to both the control (P<0.001) and the 15 μg/kg dose (P<0.001). Performance under the 15 μg/kg dose and control did not differ.
Effect of scopolamine on performance accuracy of the black-white (B-W) and oddity discrimination tasks. Scopolamine (dark gray) did not impair performance on either task at the 15-μg/kg dose compared to control (black) or vehicle (light gray). Performance on the oddity task, however, was significantly impaired by the 30 μg/kg dose. Control and vehicle conditions did not differ from each other or between tasks. Error bars represent SEM
Control performance on cognitive tests
Under the control conditions, response accuracy varied as function of task [F(3,25)=8.18; P<0.001]. Post-hoc Tukey’s indicated that this effect was due to reduced accuracy on the landmark task, when compared to the DNMP (P<0.001), the black-white discrimination (P<0.05) and the oddity discrimination (P<0.01) tasks. Control performance did not differ between the DNMP, black-white discrimination and oddity discrimination tasks.
Open field activity and exploratory behavior
No behavioral changes were observed under the 15 μg/kg dose of scopolamine on the curiosity or open-field tests. By contrast, the 30 μg/kg dose of scopolamine had marked behavioral effects, causing unsteadiness and falling. Consequently, fall frequency was added as a dependent variable to the open-field analysis. Only groom [F(2,26)=5.45; P<0.05] and fall frequency [F(2,26)=6.99; P<0.01] were significantly affected by scopolamine. Post-hoc Tukey’s indicated that groom frequency at the 30 μg/kg dose of scopolamine differed only from control (P<0.001), and fall frequency at the 30 μg/kg dose differed from control (P<0.001) and 15 μg/kg scopolamine (P<0.05). No significant effects were found on the remaining behavioral measures.
Discussion
This study demonstrates that 15 μg/kg scopolamine impairs the performance of aged dogs on a DNMP task. Further, the effect at this dose was selective to working memory performance and was not explicable to obvious disruption in behavioral activity, or to non-specific disruption in cognitive performance. These results suggest an important role of cholinergic function in visuospatial working memory. This study is the first to demonstrate that cholinergic blockade impairs working memory performance in the dog and is consistent with the extensive literature of scopolamine-induced deficits on memory tasks in other species (Bartus and Johnson 1976; Aigner et al. 1991; Ravel et al. 1992; Andrews et al. 1994; Kirkby et al. 1995; Biggan et al. 1996; Pilcher et al. 1997; Taffe et al. 1999). The dose-specific impairment on working memory performance is consistent with the pattern of impairment seen in aging and early AD; that is, impairment in recent memory, but not associative type memory (Flicker et al. 1984; Bartus 2000), which suggests that a 15 μg/kg dose models impairment linked to age-dependent cholinergic dysfunction.
The first experiment demonstrated that a 15 μg/kg dose of scopolamine was the minimal dose required to impair working memory performance in aged dogs; doses of 5 or 10 μg/kg had little effect. The majority of rodent studies use doses of scopolamine between 100 and 1000 μg/kg. By contrast, significant impairments are reported in humans using fixed doses ranging between 300 and 1200 μg (approximately between 4.3 and 17.1 μg/kg; e.g. Rusted 1988; Robbins et al. 1997) and in non-human primates with doses between 3 and 30 μg/kg (e.g. Taffe et al. 1999; Ye et al. 1999). In this respect, canine sensitivity to scopolamine-induced cognitive deficits resembles primate sensitivity.
The dose ranges typically used in rodent studies may not permit the dissociation of working-memory deficits from non-specific performance deficits (Bushnell 1990), although some studies do demonstrate a dose-response relationship in effect specificity (e.g. Biggan et al. 1996). To address the specificity of the working-memory performance impairment, response latencies, behavioral activity, and performance on three discrimination tasks were also examined in the present study. At the effective dose, scopolamine did not affect the response latencies, which provide a measure of reward-based motivation (Kirkby et al. 1995) This suggests that scopolamine, at a dose of 15 μg/kg, does not disrupt motivation or procedural memory, i.e. the ability to perform the task. Nor did the 15 μg/kg dose affect any of the non-cognitive measures examined in this study, which contrasts with observations of increased locomotion and stereotypical behaviors in rodents (Mathur et al. 1997; Laviolette et al. 2000). These studies, however, used dose levels that are at least 1000-fold greater than those used in the present study and that cause non-specific performance impairments in rodent tests of working memory (Godding et al. 1982). In the present study, a dose of 30 μg/kg, which was twice the minimally effective dose for causing impairment on the DNMP task, caused an increase in frequency of falling and a non-significant increase in locomotion.
Scopolamine, at a dose of 15 μg/kg, did not affect performance on any of the discrimination tests used in the present study. There was no effect at any distance on the landmark discrimination task, which requires subjects to go to one of two locations closest to a landmark and becomes increasingly difficult as the distance from the landmark to the correct location increases. The absence of an effect on the landmark task indicates that allocentric visuospatial function is less dependent on cholinergic function than visuospatial working memory performance. These results also suggest that reference, or associative, memory, which refers to rules or information that are invariant between trials, is spared by scopolamine. This interpretation is also supported by the absence of impairments on the black-white and oddity discrimination tasks. Furthermore, the absence of an effect on discrimination performance is probably not due to differences in task difficulty, since accuracy level on two of the discrimination tasks did not differ from the DNMP task under control conditions. Although there was no impairment at 15 μg/kg, a 30 μg/kg dose of scopolamine significantly impaired performance on the oddity discrimination test. As mentioned above, this dose also caused significant non-cognitive behavioral changes, which may be linked to the performance deficit on the oddity task. This finding highlights the importance of testing multiple doses when examining cognitive impairment with various behavioral protocols and the importance of dissociating specific from non-specific effects.
Under control conditions, accuracy on the DNMP task decreased with increasing delay, which validates that the DNMP task was assessing recent memory (Bartus 2002). Scopolamine produced impairment at all delays, which may indicate that the disruptive effects were not due to impairment of memory, but other aspects of task performance, such as attention or stimulus encoding. Both delay-independent (Rosier et al. 1998; Taffe et al. 1999) and delay-dependent (Bartus and Johnson 1976; Biggan et al. 1996) effects of scopolamine are reported. The discrepancies may be due to practice effects (Herremans et al. 1995), tasks used (Pache et al. 1999), or differences in dose levels. In the present study, the absence of a delay-dependent scopolamine effect may also represent a floor effect, since performance at the longest delays following treatment with scopolamine approached chance. Thus, the possibility of detecting further impairment was limited. Alternately, it may be necessary to use a delay shorter than 20 s for performance to be unaffected by scopolamine.
The main goal of the present study was to determine whether scopolamine-induced deficits in recent memory could be dose-specifically dissociated from deficits in associative memory in dogs. The results indicate that a 15 μg/kg dose of scopolamine impairs visuospatial working memory performance, but spares measures of reference, or associative, memory, procedural memory, motivation and non-cognitive behaviors in aged dogs. A 30 μg/kg dose, however, causes less specific behavioral impairment, which demonstrates the importance of testing a range of doses on various behavioral protocols when examining the effects of drugs on cognition. Although the present experiment did not specifically determine if scopolamine disrupts memory, the presence of impairment at all delays on the DNMP suggests that attention, or another non-mnemonic process, was disrupted. We conclude that the canine cholinergic system likely is involved in working memory performance and that scopolamine-induced cognitive disruption using a 15 μg/kg dose can serve as a model of age-related cholinergic dysfunction.
References
Adams B, Chan A, Callahan H, Milgram NW (2000a) The canine as a model of human cognitive aging: recent developments. Prog Neuropsychopharmacol Biol Psychiatry 24:675–692
Adams B, Chan A, Callahan H, Siwak C, Tapp D, Ikeda-Douglas C, Atkinson P, Head E, Cotman CW, Milgram NW (2000b) Use of a delayed non-matching to position task to model age-dependent cognitive decline in the dog. Behav Brain Res 108:47–56
Aigner TG, Walker DL, Mishkin M (1991) Comparison of the effects of scopolamine administered before and after acquisition in a test of visual recognition memory in monkeys. Behav Neural Biol 55:61–67
Andrews JS, Jansen JH, Linders S, Princen A (1994) Effects of disrupting the cholinergic system on short-term spatial memory in rats. Psychopharmacology 115:485–494
Bartus RT (2000) On neurodegenerative diseases, models, and treatment strategies: lessons learned and lessons forgotten a generation following the cholinergic hypothesis. Exp Neurol 163:495–529
Bartus RT, Johnson HR (1976) Short-term memory in the rhesus monkey: disruption from the anti-cholinergic scopolamine. Pharmacol Biochem Behav 5:39–46
Biggan SL, Ingles JL, Beninger RJ (1996) Scopolamine differentially affects memory of 8- and 16-month-old rats in the double Y-maze. Neurobiol Aging 17:25–30
Blokland A (1995) Acetylcholine: a neurotransmitter for learning and memory? Brain Res Brain Res Rev 21:285–300
Bruhwyler J, Liegeois JF, Geczy J (1998) Facilitatory effects of chronically administered citicoline on learning and memory processes in the dog. Prog Neuropsychopharmacol Biol Psychiatry 22:115–128
Bushnell PJ (1990) Modelling working and reference memory in rats: effects of scopolamine on delayed matching-to-position(1,2). Behav Pharmacol 1:419–427
Cummings BJ, Head E, Afagh AJ, Milgram NW, Cotman CW (1996) Beta-amyloid accumulation correlates with cognitive dysfunction in the aged canine. Neurobiol Learn Mem 66:11–23
Davies P, Maloney AJ (1976) Selective loss of central cholinergic neurons in Alzheimer’s disease. Lancet 2:1403
Dodart JC, Mathis C, Ungerer A (1997) Scopolamine-induced deficits in a two-trial object recognition task in mice. Neuroreport 8:1173–1178
Drachman DA, Leavitt J (1974) Human memory and the cholinergic system. A relationship to aging? Arch Neurol 30:113–121
Flicker C, Bartus RT, Crook TH, Ferris SH (1984) Effects of aging and dementia upon recent visuospatial memory. Neurobiol Aging 5:275–283
Flicker C, Ferris SH, Serby M (1992) Hypersensitivity to scopolamine in the elderly. Psychopharmacology 107:437–441
Flood JF, Cherkin A (1986) Scopolamine effects on memory retention in mice: a model of dementia? Behav Neural Biol 45:169–184
Godding PR, Rush JR, Beatty WW (1982) Scopolamine does not disrupt spatial working memory in rats. Pharmacol Biochem Behav 16:919–923
Head E, Mehta R, Hartley J, Kameka M, Cummings BJ, Cotman CW, Ruehl WW, Milgram NW (1995) Spatial learning and memory as a function of age in the dog. Behav Neurosci 109:851–858
Head E, Callahan H, Muggenburg BA, Cotman CW, Milgram NW (1998) Visual-discrimination learning ability and beta-amyloid accumulation in the dog. Neurobiol Aging 19:415–425
Head E, Cotman CW, Milgram NW (2000) Canine cognition, aging and neuropathology. Introduction. Prog Neuropsychopharmacol Biol Psychiatry 24:671–673
Herremans AH, Hijzen TH, Olivier B, Slangen JL (1995) Cholinergic drug effects on a delayed conditional discrimination task in the rat. Behav Neurosci 109:426–435
Jha AP, McCarthy G (2000) The influence of memory load upon delay-interval activity in a working-memory task: an event-related functional MRI study. J Cognit Neurosci 12:S-105
Kirkby DL, Jones DN, Higgins GA (1995) Influence of prefeeding and scopolamine upon performance in a delayed matching-to-position task. Behav Brain Res 67:221–227
Laviolette SR, Priebe RP, Yeomans JS (2000) Role of the laterodorsal tegmental nucleus in scopolamine- and amphetamine-induced locomotion and stereotypy. Pharmacol Biochem Behav 65:163–174
Lew AR, Bremner JG, Lefkovitch LP (2000) The development of relational landmark use in six- to twelve-month-old infants in a spatial orientation task. Child Dev 71:1179–1190
Mathur A, Shandarin A, Laviolette SR, Parker J, Yeomans JS (1997) Locomotion and stereotypy induced by scopolamine: contributions of muscarinic receptors near the pedunculopontine tegmental nucleus. Brain Res 775:144–155
Milgram NW, Head E, Weiner E, Thomas E (1994) Cognitive functions and aging in the dog: acquisition of nonspatial visual tasks. Behav Neurosci 108:57–68
Milgram NW, Adams B, Callahan H, Head E, Mackay B, Thirlwell C, Cotman CW (1999) Landmark discrimination learning in the dog. Learn Mem 6:54–61
Milgram NW, Zicker SC, Head E, Muggenburg BA, Murphey H, Ikeda-Douglas CJ, Cotman CW (2002) Dietary enrichment counteracts age-associated cognitive dysfunction in canines. Neurobiol Aging 23:737–745
Molchan SE, Martinez RA, Hill JL, Weingartner HJ, Thompson K, Vitiello B, Sunderland T (1992) Increased cognitive sensitivity to scopolamine with age and a perspective on the scopolamine model. Brain Res Brain Res Rev 17:215–226
Pache DM, Sewell RD, Spencer PS (1999) Detecting drug effects on short-term memory function using a combined delayed matching and non-matching to position task. J Pharmacol Toxicol Meth 41:135–141
Perry EK, Perry RH, Blessed G, Tomlinson BE (1977) Necropsy evidence of central cholinergic deficits in senile dementia. Lancet 1:189
Perry EK, Tomlinson BE, Blessed G, Bergmann K, Gibson PH, Perry RH (1978) Correlation of cholinergic abnormalities with senile plaques and mental test scores in senile dementia. BMJ 2:1457–1459
Pilcher JJ, Sessions GR, McBride SA (1997) Scopolamine impairs spatial working memory in the radial maze: an analysis by error type and arm choice. Pharmacol Biochem Behav 58:449–459
Ravel N, Vigouroux M, Elaagouby A, Gervais R (1992) Scopolamine impairs delayed matching in an olfactory task in rats. Psychopharmacology 109:439–443
Ray PG, Meador KJ, Loring DW, Zamrini EW, Yang XH, Buccafusco JJ (1992) Central anticholinergic hypersensitivity in aging. J Geriatr Psychiatry Neurol 5:72–77
Robbins TW, Semple J, Kumar R, Truman MI, Shorter J, Ferraro A, Fox B, McKay G, Matthews K (1997) Effects of scopolamine on delayed-matching-to-sample and paired associates tests of visual memory and learning in human subjects: comparison with diazepam and implications for dementia. Psychopharmacology 134:95–106
Rosier A, Cornette L, Orban GA (1998) Scopolamine-induced impairment of delayed recognition of abstract visual shapes. Neuropsychobiology 37:98–103
Rusted JM (1988) Dissociative effects of scopolamine on working memory in healthy young volunteers. Psychopharmacology 96:487–492
Sessions GR, Pilcher JJ, Elsmore TF (1998) Scopolamine-induced impairment in concurrent fixed-interval responding in a radial maze task. Pharmacol Biochem Behav 59:641–647
Shapovalova KB (1999) Activation of the cholinergic system of the striatum improves attention to conditioned reflex stimuli. Neurosci Behav Physiol 29:493–503
Siwak CT, Tapp PD, Milgram NW (2001) Effect of age and level of cognitive function on spontaneous and exploratory behaviors in the beagle dog. Learn Mem 8:317–325
Taffe MA, Weed MR, Gold LH (1999) Scopolamine alters rhesus monkey performance on a novel neuropsychological test battery. Cognit Brain Res 8:203–212
Tariot PN, Patel SV, Cox C, Henderson RE (1996) Age-related decline in central cholinergic function demonstrated with scopolamine. Psychopharmacology 125:50–56
Whitehouse PJ, Price DL, Clark AW, Coyle JT, DeLong MR (1981) Alzheimer disease: evidence for selective loss of cholinergic neurons in the nucleus basalis. Ann Neurol 10:122–126
Whitehouse PJ, Price DL, Struble RG, Clark AW, Coyle JT, Delon MR (1982) Alzheimer’s disease and senile dementia: loss of neurons in the basal forebrain. Science 215:1237–1239
Ye JW, Cai JX, Wang LM, Tang XC (1999) Improving effects of huperzine A on spatial working memory in aged monkeys and young adult monkeys with experimental cognitive impairment. J Pharmacol Exp Ther 288:814–819
Acknowledgements
This work was supported in part by Pfizer Inc. Additional support was provided in the form of postgraduate scholarships to J.A.A. and A.D.F.C. from the Natural Sciences and Engineering Research Council of Canada. A special thanks is extended to Christa Studzinski, Dwight Tapp, Daniel Holowachuk, Christina Siwak, Sara Skoggard, Winnie Lau, Jin Lee, Lisa Lin, Merlin Paes and to the Division of Comparative Medicine’s animal care staff for all their assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Araujo, J.A., Chan, A.D.F., Winka, L.L. et al. Dose-specific effects of scopolamine on canine cognition: Impairment of visuospatial memory, but not visuospatial discrimination. Psychopharmacology 175, 92–98 (2004). https://doi.org/10.1007/s00213-004-1777-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-004-1777-y