Abstract
Rationale
One of the common neurochemical features of many drugs of abuse is their ability to directly or indirectly enhance dopaminergic activity in the brain, particularly within the ventral tegmental-nucleus accumbens pathway. Dopaminergic pathways in the frontal and limbic cortex also may be targets for these agents, where pharmacological effects could result in heightened attention and/or support self-administration behavior.
Objectives
The purpose of this study was to determine whether drugs from differing pharmacological classes that exhibit abuse potential would share the ability to counter distractability in the delayed matching task.
Methods
Well trained mature macaques performed a computer-assisted delayed matching-to-sample task which included trials associated with three delay intervals and randomly interspersed task-relevant distractors. Drug regimens included four to five doses and subjects were tested no more than twice per week.
Results
All but one of the six compounds (tomoxetine), on average, increased task accuracy for either non-distractor or distractor trials. It was evident that for several compounds, doses required to improve accuracy for non-distractor trials were routinely greater than the doses required to improve accuracy for distractor trials. Data for the individualized Best dose (based upon the subject's optimal level of accuracy during distractor trials) revealed statistically significant distractor-related improvements in task accuracy for the same five compounds. The relative efficacy for reversing distractor-induced decrements in task accuracy was estimated by the level of improvement with respect to baseline: nomifensine (31%)>nicotine (22%)≈morphine (19%)≈caffeine (19%)≈methylphenidate (22%) >tomoxetine (9%). Tomoxetine (noradrenergic preferring) was the only compound that did not produce a significant improvement in accuracy.
Conclusions
These results provide pharmacological support for the concept that attentional mechanisms may play an important role in the "environmental" associative aspects of drug seeking behavior, and as such they may provide the basis for treatment strategies aimed at preventing relapse in detoxified addicts.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The abuse of drugs remains a significant health problem for which current treatment options are often unsuccessful. Abused drugs have been investigated primarily as to their abuse potential and the mechanisms by which they reinforce drug-taking behavior. At least initially, abused drugs have in common the ability to reward their users with a pleasurable experience, an effect that has been linked to elevations of synaptic dopamine concentrations in the nucleus accumbens (for review, see McGinty 1999). The nucleus accumbens also is strategically situated to receive input from higher brain centers in the prefrontal and entorhinal cortex, hippocampus and amygdala. In turn, the nucleus accumbens is the origin of four major efferent pathways to several midbrain, pontine, thalamic and hypothalamic centers (Carelli and Deadwyler 1997; Bardo 1998). The intimate relationship between the nucleus accumbens and cortical and hippocampal centers has engendered the hypothesis that the reinforcing aspects of drugs of abuse require cognitive input which is integrated at the level of the nucleus accumbens, the output of which directs various somatic and visceral efferent and hormonal secretory pathways involved in the consolidation of the reward stimulus and drug-seeking behavior (Bardo 1998; Koob and Le Moal 2001). Return projections from the nucleus accumbens via the thalamus to executive centers in the forebrain also complete a "cognitive" feedback loop in which components of memory, including long-term potentiation (which relates to issues of drug sensitization), are considered to be important for protracted elevated risk to renewed drug-seeking behavior. Indeed, the cognitive aspects of drugs of abuse are of paramount importance in the understanding of reward systems and recidivism (Setlow 1997; Di Chiara et al. 1999; Sarter et al. 1999; Koob and Le Moal 2001). The significance of environmental associative processes in addiction are evidenced by recovered addicts' experience of cravings for the abused drug and even the return to drug use (recidivism) upon encountering drug-associated stimuli.
Implicating cognition as a major component of the mechanism by which drugs of abuse motivate drug-taking behavior invokes reference to the cortical cholinergic system (Bushnell et al. 2000). Such a connection is not surprising. Research into aging and Alzheimer's disease has yielded a strong positive relationship between cholinergic activity and cognitive function (see Bartus 2000). Research into schizophrenia has suggested that abnormal midbrain dopamine projections may result in elevated cortical cholinergic activity. This may be the cause of the hyperattentional problems associated with schizophrenia's positive symptoms (Sarter and Bruno 1999). The relationships between limbic dopamine, with its well-known role in reinforcement, and cortical acetylcholine with its importance in attention and memory are well established. Sarter and Bruno (1999) have summarized the growing literature that implicates abnormal cortical cholinergic excitability as a "final common pathway" that mediates a variety of psychiatric disorders, many of which are believed to result from a pathology that is primarily dopaminergic.
We studied several drugs with varying levels of abuse potential from diverse pharmacological classes in a well-characterized (Prendergast et al. 1998a, 1998b) non-human primate model of attentional aspects of working memory. Caffeine, methylphenidate, morphine, nicotine, nomifensine (primarily a dopamine reuptake inhibitor), and tomoxetine (a norepinephrine reuptake inhibitor) were utilized as drugs with abuse potential that belong to different classes of drugs so as to make possible an examination of cognitive effects that may be common to the group. We tested the possibility that drugs for which abuse liability exists will share a cognitive component that may play an important role in understanding how attention to drug-associated stimuli and environments precipitate recidivism.
Materials and methods
Subjects
Male and female rhesus (Macaca mulatta) and pigtail (Macaca nemestrina) monkeys aged 4–7 years served as experimental subjects. Subject characteristics are presented in Table 1. Monkeys were individually housed at the Animal Behavior Center of the Medical College of Georgia in stainless steel cages composed of multiple 127×71×66 cm units. To promote psychological well-being toys and foraging tubes were provided routinely and monkeys were allowed to observe television programs each afternoon after testing. Delayed matching-to-sample (DMTS) testing was conducted once each weekday. During a test-week, monkeys were maintained on a feeding schedule that allows ~15% of their normal daily food intake to be derived from flavored reinforcement pellets awarded for correct responses during testing. Standard laboratory monkey chow, fresh fruits and vegetables comprised the remainder of their daily food intake. Water was available on an unlimited basis, including during testing. All procedures were reviewed and approved by the Medical College of Georgia Institutional Animal Care and Use Committee and are consistent with AAALAC guidelines. Each subject had previously participated in one or more short-term studies assessing the effects of reversible drugs on DMTS performance and all were well trained in this task. Prior drug experience had produced no observable untoward effects in the animals. A minimal washout period of 4 weeks occurred before the initiation of the current study, and five subjects participated in each dose-response study.
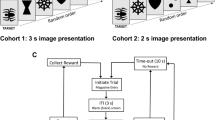
Delayed matching-to-sample (DMTS) procedure
Test panels attached to each animal's home cage presented the task by using a computer-automated system. Stimuli consisted of 2.54 cm diameter colored disks (red, yellow, or green) presented by light-emitting diodes located behind clear push-keys. A trial was initiated by illumination of the sample key with one of three colors. The sample key remained lit until the monkey depressed it to initiate a pre-programmed delay (retention) interval. Following the delay interval, the two choice keys located below the sample key were illuminated. One of the two choice keys was illuminated with the color matching the stimulus, whereas the other (incorrect) choice key was illuminated by one of the two remaining colors. A correct (matching) choice was reinforced. Non-matching choices were neither reinforced nor punished. The inter-trial interval was 5 s and each session consisted of 96 trials. The presentation of stimulus color, choice colors, and choice position were fully counterbalanced so as to relegate non-matching strategies to chance levels of accuracy. Three different presentation sequences were rotated through each daily session to prevent the subjects from memorizing the first several trials. Delay intervals were established during numerous non-drug or vehicle sessions prior to initiating the study. The duration for each delay interval was adjusted for each subject until three levels of performance accuracy were obtained: zero delay (85–100% of trials answered correctly); short delay interval (75–84% correct); medium delay interval (65–74% correct); and long delay interval (55–64% correct). The assignment of retention intervals based upon an individual's baseline task accuracy is necessary to avoid ceiling effects in the most proficient animals during drug studies, while also serving to insure that each animal begins testing at relatively the same level of task difficulty (Paule et al. 1998). Although all study subjects performed within these baseline parameters during standard DMTS sessions, accuracy during baseline non-distractor trials of the DMTS-distractor task were not always maintained at standard DMTS levels for each delay interval. This was probably due to the presence of the distractor trials during the sessions.
Distractor DMTS task (DMTS-D)
Prior to initiating this study, all subjects had adjusted standard DMTS (no distractors) baselines as described in above. Average (±SEM) DMTS accuracies (% trials correct) for the study group were as follows: zero delay=96.5±1.43%; short delay=84.5±2.04%; medium delay=69.4±2.25%; long delay=55.4±2.90%. All of the subsequent DMTS sessions (presented below) were distractor sessions. No further adjustments in baseline or vehicle-associated accuracies were made, even though the presence of distractor trials in a session generally decreased task accuracy of the non-distractor trials relative to standard (no distractor) DMTS sessions. Baseline accuracies for non-distractor trials measured during distractor sessions for the study group were: zero delay=88.49%, short delay=69.29%, medium delay=60.93%, and long delay=53.66%. Therefore, the control/vehicle-associated task accuracies for each of the six drug studies reflected the effect on the specific study group of the presentation of unpredictable distractors during the sessions.
DMTS test sessions with distractors (for review of distractibility in nonhuman primates see Prendergast 2000) were conducted on no more than 3 days every 2 weeks. Allowing 3 days of standard DMTS testing between interference sessions prevents the development of tolerance to distractor-associated impaired performance. To monitor for potential habituation to the distractors, sessions were conducted under vehicle conditions just before and at the completion of each dosing regimen. Distractor stimuli (which occur randomly during 18 of the 96 trials of test sessions) were presented simultaneously on the sample and choice keys 1 s after the stimulus presentation, for a total duration of 3 s. The distraction consisted of a random pattern of the three colored lights flashing in an alternating manner. The flash duration for a given colored light was 0.33 s. Immediately one colored light was extinguished, a different colored light was presented. Each color was presented in random order on each key three separate times during each distractor interval. Distractor stimuli were present an equal number of times on trials with short, medium and long delay intervals. In addition to session accuracy, two response latencies also were measured: the "sample latency", which is the time between presentation of the sample color and the animal pressing in sample key; and the "choice latency", which is the time between presentation of the choice colors and the animal pressing one of the choice keys.
Drug administration
Baseline data for non-distractor and distractor trials were obtained prior to the start of administration of each drug. A minimum washout period of 2 days was maintained between drug administrations. During this time, standard DMTS testing was conducted without vehicle or drug administration. Subsequent drug doses were administered only if a monkey's performance returned to baseline levels during that period. All test compounds were administered in a series of ascending doses. Caffeine (1, 5, 20, 50 mg/kg) was dissolved in water and pipetted onto a sugar cube for oral administration 15 min prior to testing. The total amount of caffeine was considered to be too large for intramuscular delivery. The other test compounds were dissolved in sterile saline (vehicle) within a total injection volume of 0.035 ml/kg. Injections were given in the gastrocnemius muscle 10 min prior to testing. The drugs and doses given were methylphenidate (0.125, 0.25, 0.5, 1.0 mg/kg), nomifensine (0.01, 0.03, 0.1, 0.3, 1.0 mg/kg), tomoxetine (0.01, 0.03, 0.1, 0.3, 1.0 mg/kg), (−)-nicotine (2.5, 5, 10, 20 μg/kg) and morphine (0.5, 1, 2, 4 mg/kg). DuPont Pharmaceuticals supplied the caffeine, nomifensine, and tomoxetine. Nicotine bitartrate and morphine sulfate were purchased from Sigma (St Louis, Mo., USA).
Statistics
Values obtained for each difficulty level (delay interval) were averaged and recorded as the mean percent correct. Drug effects were calculated as the absolute change from vehicle-associated accuracy. Statistical comparisons between vehicle and treated (drug) groups were performed using a repeated measures analysis of variance (JMP Statistical Discovery Software v.4.0, SAS Institute, Inc., Cary, N.C., USA). Post hoc analysis was performed using a multiple comparison procedure with orthogonal contrasts.
Results
General task performance
Task performance was analyzed across all drugs and revealed several important generalities. Overall accuracy in non-distractor and distractor trials was dependent upon the assigned delay interval [F(2,1098)=110.4, P<0.001] such that there was the expected delay-dependent decrement in task accuracy in all experiments. The presence of distractor stimuli decreased performance overall (average of all three delays) from 63.74% to 58.85% [F(1,939)=25.96, P<0.0001]. Distractor-related impairment in task accuracy was obtained for trials with short delays (73.5±0.95 versus 65.0±1.15, t=5.10, P<0.0001) and medium delays (63.0±0.92 versus 58.2±1.15, t=3.61, P=0.0003) but not for trials with long delays (54.1±0.93 versus 52.2±1.18, t=0.87, P=0.39). Sample and choice latencies were recorded to monitor non-cognitive psychomotor effects. There was a marked increase in the latency to depress one of the two choice keys when that choice was incorrect as compared to when that choice was correct. Choice latency for correct responses was shorter (2.75 s) than that for incorrect responses (3.46 s, Student's t=3.24, P=0.001). None of the statistical analyses revealed a statistically significant interaction between treatment and drug dose for any of the compounds tested (i.e. all effects of drug treatment were independent of dosage). Finally, there was no attempt to perform statistical analyses for Best dose-associated non-distractor trials because the Best dose in each case was determined using accuracies associated only with distractor-related trials. Nonetheless, the data for Best dose-associated non-distractor trials are presented in each of Figs. 1, 2, 3, 4, 5 and 6 for comparison.
Caffeine
For non-distractor trials, caffeine treatment (versus vehicle) produced no statistically significant effect on DMTS accuracy either as an independent factor (P>0.05) or as a factor dependent on delay interval (P>0.05). During trials associated with distractors the drug also produced no statistically significant effects [independent of delay: F(4,56)=1.771, P=0.15; dependent on delay: P>0.05]. However, examination of the dose-response data (Fig. 1) shows that, on average, distractor-induced decrements in accuracy were almost completely reversed during sessions following administration of the 5 mg/kg dose. The failure to achieve statistical significance was related at least partly to differences in individual sensitivity to caffeine. Therefore, a Best dose of caffeine was selected for each monkey based upon the dose causing the largest increase in accuracy during distractor trials when averaged over all three delay intervals. The maximal level of improvement in DMTS-D task accuracy was obtained at 5 mg/kg for subjects 18, 23, and 339; 20 mg/kg for 24; and 50 mg/kg for 308, with the average Best dose=17.0±8.7 mg/kg for the group. After administration of each subject's Best dose of caffeine, DMTS-D accuracy significantly increased, independent of delay interval [by 19%; F(1,20)=6.70, P=0.02] not as an interaction with delay (P>0.05). Individual Best dose caffeine treatment significantly decreased Sample latencies [F(1,12)=5.01, P=0.04] and this effect was unrelated to the presence of distractors (P>0.05). Choice latencies were not affected by caffeine treatment (Table 2).
Effect of oral administration of caffeine on the accuracy of five monkeys in their performance of a distractor version of the delayed matching-to-sample (DMTS-D) task. The upper panels show the effect of vehicle and caffeine as a function of delay interval. For non-distractor trials, caffeine treatment produced no significant change from vehicle either as an independent factor (P>0.05) or as a factor dependent on delay interval (P>0.05). During trials associated with distractors the drug produced no statistically significant effects [independent of delay: F(4,56)=1.77, P=0.15; dependent on delay: P>0.05]. The lower panels show the effect the individualized Best dose of caffeine. The Best dose of caffeine significantly increased DMTS-D accuracy, independent of delay interval [F(1,20)=6.70, P=0.02] but not as an interaction with delay (P>0.05). Z, S, M, and L refer to delay intervals (zero, short, medium, and long). "Overall" indicates the accuracy for distractor-related trials for the entire session, i.e., collapsed across delays. Each value indicates the mean±SEM
Methylphenidate
The data presented here for methylphenidate were previously published (Prendergast et al. 1998a). They are reprised here, reanalyzed and reformatted to be consistent with the presentation for the other drugs to facilitate comparisons (Fig. 2). Methylphenidate treatment was associated with a significant improvement in non-distractor accuracy that was dependent upon delay interval [F(12,76)=2.51, P=0.008]. The 0.5 mg/kg dose increased accuracy during the performance of medium delays by 28% (t=2.27, P=0.03), whereas the 1.0 mg/kg dose decreased accuracy during short delay trials by 37% (t=4.76, P<0.0001). For distractor trials, methylphenidate treatment produced no significant change from vehicle levels of DMTS accuracy either as an independent factor, [F(4,56)=1.26, P=0.30] or as a factor dependent on delay interval [F(8,56)=1.48, P=0.18]. However, examination of the dose-response data (Fig. 2) shows that, on average, distractor-induced decrements in short delay accuracy were almost completely reversed during sessions following administration of the 0.25 mg/kg dose. The maximal level of improvement in DMTS-D task accuracy was obtained at 0.125 mg/kg for subjects 146, 218, and 270; 0.25 mg/kg for 284; and 0.5 mg/kg for 13, with the average Best dose=0.23±0.073 mg/kg. After administration of each subject's individual Best dose of methylphenidate, DMTS-D accuracy significantly increased independent of delay interval [by 15%; F(1,20)=5.99, P=0.02] but not as an interaction with delay (P>0.05). Examination of Fig. 2 continues to show that Best dose treatment increased non-distractor trial accuracy particularly during short delay trials (by 35%) and during medium delays (by 22%). Methylphenidate was the only compound of the six to cause a significant decrease in task latencies (Table 2). Drug treatment was associated with a significant decrease in the duration of Choice latencies overall [F(1,35)=11.9, P=0.002] and as an interaction with trial type [i.e. non-distractor versus distractor, F(1,35)=4.60, P=0.039], i.e., methylphenidate decreased the duration of choice latencies specifically for those trials in the session that were associated with distractors. As with the effect of caffeine, methylphenidate treatment decreased durations of Sample latencies [F(1,15)=8.13, P=0.012] and this effect was not specific to trials with or without distractors (P>0.05). Independent of drug treatment, there was a significant increase in Choice latencies associated with incorrect trials relative to correct trials [F(1,35)=9.18, P=0.005]. There also was a significant interaction between Choice latency type (Choice latencies associated with correct versus incorrect trials) and distractor presence [Choice latencies associated with non-distractor trials versus distractor trials, F(1,35)=4.60, P=0.039] such that for non-distractor trials there was a significant increase in the duration of incorrect trial versus correct trial Choice latencies (t=3.66, P=0.0001) ; and for correctly answered trials, there was a significant increase in distractor versus non-distractor Choice latencies (t=2.83, P=0.008).
Effect of IM administration of methylphenidate on the accuracy of five monkeys in their performance of a distractor version of the delayed matching-to-sample (DMTS-D) task. The upper panels show the effect of vehicle and methylphenidate as a function of delay interval. Drug treatment was associated with a significant improvement in non-distractor accuracy that was dependent upon delay interval [F(12,76)=2.51, P=0.008]. The 0.5 mg/kg dose increased accuracy during the performance of medium delays by 28% (t=2.27, P=0.03), whereas the 1.0 mg/kg dose decreased accuracy during short delay trials by 37% (t=4.76, P<0.0001). For distractor trials, drug treatment produced no significant change from vehicle either as an independent factor [F(4,56)=1.26, P=0.30] or as a factor dependent on delay interval [F(8,56)=1.48, P=0.18]. The lower panels show the effect of methylphenidate as the individualized Best dose. The Best dose of methylphenidate significantly increased DMTS-D accuracy independent of delay interval [F(1,20)=5.99, P=0.02] but not as an interaction with delay (P>0.05)
Morphine
For non-distractor trials, morphine treatment produced no significant change in DMTS accuracy either as an independent factor [F(4,76)=1.57, P=0.19] or as a factor dependent on delay interval (P>0.05). During trials associated with distractors the drug also produced no statistically significant effects [independent of delay: P>0.05; dependent on delay: F(8,56)=1.01, P=0.44]. However, examination of the dose-response data (Fig. 3) shows that, on average, non-distractor related accuracy was increased for long delay trials after the highest dose (4 mg/kg) dose, whereas for distractor trials, medium delay trials exhibited improved accuracy after the lowest (0.5 mg/kg) dose. The maximal level of improvement in DMTS-D task accuracy was obtained at 0.5 mg/kg for subjects 146, 218, and 270; 1.0 mg/kg for 284; and 4.0 mg/kg for 13, with the average Best dose=1.3±0.68 mg/kg. After administration of each subject's individual Best dose of morphine, DMTS-D accuracy significantly increased independent of delay interval [by 19%; F(1,20)=6.53, P=0.02] but not as an interaction with delay (P>0.05). Best dose morphine resulted in no significant change in task latencies (Table 2). Independent of drug treatment, there was a significant increase in Choice latencies associated with incorrect trials relative to correct trials [F(1,28)=10.4, P=0.003] and a significant decrease in Choice latencies associated with distractor trials relative to non-distractor trials [F(1,28)=7.84, P=0.009].
Effect of IM administration of morphine on the accuracy of five monkeys in their performance of a distractor version of the delayed matching-to-sample (DMTS-D) task. The upper panels show the effect of vehicle and morphine as a function of delay interval. For non-distractor trials, morphine treatment produced no significant change from vehicle either as an independent factor [F(4,76)=1.57, P=0.19] or as a factor dependent on delay interval (P>0.05). During trials associated with distractors the drug produced no statistically significant effects [independent of delay: P>0.05; dependent on delay: F(8,56)=1.01, P=0.44]. The lower panels show the effect of morphine as the individualized Best dose. The Best dose of morphine significantly increased DMTS-D accuracy independent of delay interval [F(1,20)=6.53, P=0.02] but not as an interaction with delay (P>0.05)
Nicotine
The data presented here for nicotine were previously published (Prendergast et al. 1998b). They are reprised here, reanalyzed and reformatted to be consistent with the presentation for the other drugs to facilitate comparisons (Fig. 4). For non-distractor trials, nicotine treatment produced no significant change from vehicle levels of DMTS accuracy either as an independent factor, [F(4,76)=1.46, P=0.22] or as a factor dependent on delay interval [F(12,76)=1.12, P=0.35]. During trials associated with distractors, the drug also produced no statistically significant effect, but did evoke a trend towards improvement in overall accuracy [independent of delay: F(4,56)=1.75, P=0.15]. Examination of the dose-response data (Fig. 4) shows that this trend was largely reflective of the drug's effect on accuracy at the short delays, which were consistently increased above baseline levels for each of the four doses of nicotine. The maximal level of improvement in DMTS-D task accuracy was obtained at 2.5 μg/kg for subjects 13, and 759; 5.0 μg/kg for 270 and 284; and 10.0 μg/kg for 001, with the average Best dose=5.0±1.37 μg/kg. After administration of each subject's individual Best dose of nicotine, DMTS-D accuracy significantly increased independent of delay interval [by 22%; F(1,8)=10.0, P=0.034] but not as an interaction with delay (P>0.05). Best dose nicotine resulted in no significant change in task latency durations (Table 2). Independent of drug treatment, there was a significant increase in the duration of Choice latencies associated with incorrect trials relative to correct trials [F(1,28)=4.36, P=0.046] and a significant increase in the duration of Choice latencies associated with distractor trials relative to non-distractor trials [F(1,28)=7.01, P=0.013]. There also was a significant interaction between Choice latency type (Choice latencies associated with correct versus incorrect trials) and distractor presence [Choice latencies associated with non-distractor trials versus distractor trials, F(1,28)=10.6, P=0.003] such that for non-distractor trials there was a significant increase in the duration of incorrect trial versus correct trial Choice latencies (t=3.77, P=0.0008); and for correctly answered trials, there was a significant increase in the duration of distractor versus non-distractor Choice latencies (t=4.17, P=0.0003).
Effect of IM administration of nicotine on the accuracy of five monkeys in their performance of a distractor version of the delayed matching-to-sample (DMTS-D) task. The upper panels show the effect of vehicle and nicotine as a function of delay interval. For non-distractor trials, nicotine treatment produced no significant change from vehicle levels of DMTS accuracy either as an independent factor, [F(4,76)=1.46, P=0.22] or as a factor dependent on delay interval [F(12,76)=1.12, P=0.35]. During trials associated with distractors the drug produced no statistically significant effect, but did evoke a trend towards improvement in accuracy overall [independent of delay: F(4,56)=1.75, P=0.15]. The lower panels show the effect of nicotine as the individualized Best dose. The Best dose of nicotine significantly increased DMTS-D accuracy independent of delay interval [by 22%; F(1,8)=10.0, P=0.034] but not as an interaction with delay (P>0.05)
Nomifensine
For non-distractor trials, nomifensine produced no significant change in DMTS accuracy either as an independent factor (P>0.05) or as a factor dependent on delay interval (P>0.05). During trials associated with distractors the drug also produced no statistically significant effects (independent of delay: P>0.05; dependent on delay: P>0.05). However, examination of the dose-response data (Fig. 5) shows that accuracy during long delay trials was consistently increased on average across all five doses. The maximal level of improvement in DMTS-D task accuracy was obtained at 0.03 mg/kg for subjects 18, 24, 308 and 339; 0.1 mg/kg for 23, with the average Best dose=0.044±0.014 mg/kg. After administration of each subject's individual Best dose of nomifensine, DMTS-D accuracy significantly increased independent of delay interval [by 31%; F(1,20)=27.04, P<0.0001] but not as an interaction with delay [F(2,20)=2.49, P=0.11]. Best dose nomifensine resulted in no significant change in task latencies (Table 2). Independent of drug treatment, there was a significant decrease in the duration of Choice latencies associated with incorrect trials relative to correct trials [F(1,28)=7.11, P=0.013]. There also was a significant interaction between Choice latency type (Choice latencies associated with correct versus incorrect trials) and distractor presence [Choice latencies associated with non-distractor trials versus distractor trials, F(1,28)=15.1, P=0.001] such that for non-distractor trials there was a significant increase in the duration of incorrect trial versus correct trial Choice latencies (t=4.62, P<0.0001); and for correctly answered trials, there was a significant increase in the duration of distractor versus non-distractor Choice latencies (t=3.68, P=0.001).
Effect of IM administration of nomifensine on the accuracy of five monkeys in their performance of a distractor version of the delayed matching-to-sample (DMTS-D) task. The upper panels show the effect of vehicle and nomifensine as a function of delay interval. For non-distractor trials, nomifensine treatment produced no significant change from vehicle levels of DMTS accuracy either as an independent factor (P>0.05) or as a factor dependent on delay interval (P>0.05). During trials associated with distractors the drug produced no statistically significant effects (independent of delay: P>0.05; dependent on delay: P>0.05). The lower panels show the effect of nomifensine as the individualized Best dose. The Best dose of nomifensine significantly increased DMTS-D accuracy independent of delay interval [by 31%; F(1,20)=27.04, P<0.0001] but not as an interaction with delay [F(2,20)=2.49, P=0.11]
Tomoxetine
For non-distractor trials, tomoxitine produced no significant change in DMTS accuracy either as an independent factor (P>0.05) or as a factor dependent on delay interval (P>0.05) (Fig. 6). During trials associated with distractors, the drug also produced no statistically significant effects (independent of delay: P>0.05; dependent on delay: P>0.05). The maximal level of improvement in DMTS-D task accuracy was obtained at the 0.01 mg/kg for subject 24; 0.03 mg/kg for 683; 0.3 mg/kg for 308; and 1.0 mg/kg for 23 and 18 with the average Best dose=0.47±0.22 mg/kg. Even after administration of each subject's individual Best dose of tomoxetine, distractor accuracy did not significantly increase above control levels of performance [independent of delay: F(1,20)=1.54, P=0.23; dependent on delay: P>0.05]. This is partly because one animal (308) failed to respond with increased distractor accuracy to any dose of tomoxetine (his average change from baseline for Best dose was –1.6% correct). Even after elimination of 308 from consideration, the overall drug-induced improvement in distractor-associated accuracy for the other four subjects was only 11%. Best dose tomoxetine resulted in no significant change in task latencies (Table 2). Independent of drug treatment, there was a significant decrease in Choice latencies associated with incorrect trials relative to correct trials [F(1,28)=7.09, P=0.013] and a significant decrease in Choice latencies associated with distractor trials relative to non-distractor trials [F(1,28)=6.10, P=0.020].
Effect of IM administration of tomoxetine on the accuracy of five monkeys in their performance of a distractor version of the delayed matching-to-sample (DMTS-D) task. The upper panels show the effect of vehicle and tomoxetine as a function of delay interval. For non-distractor trials, tomoxetine treatment produced no significant change from vehicle levels of DMTS accuracy either as an independent factor (P>0.05) or as a factor dependent on delay interval (P>0.05). During trials associated with distractors, the drug produced no statistically significant effects (independent of delay: P>0.05; dependent on delay: P>0.05). The lower panels show the effect of tomoxetine as the individualized Best dose. The Best dose of tomoxetine did not significantly increase distractor accuracy above control levels of performance [independent of delay: F(1,20)=1.54, P=0.23; dependent on delay: P>0.05]
Discussion
We report that caffeine, methylphenidate, morphine, nicotine, and nomifensine improved DMTS-D accuracy when they were administered at individually optimized doses. The drugs have in common the potential for abuse which is thought to be related to their ability to increase dopamine levels in the nucleus accumbens (Govoni et al. 1984, for review, see Westerink and Kork 1976; Missale et al. 1985; Imperato et al. 1986; Challman and Lipsky 2000). Tomoxetine, the only drug studied that did not significantly improve distractor-related DMTS accuracy, is more selective in its ability to inhibit the uptake of norepinephrine (Zerbe et al. 1985) and does not appear to alter limbic dopamine metabolism.
Previous studies examining the cognitive effects of these drugs have yielded a diversity of findings. Part of this inconsistency may be related to the great variability in individual performance on short-term memory tasks that can mask potential drug effects in a standard dose-response analysis. For example, in the present study, administration of caffeine, methylphenidate, morphine, nicotine and nomifensine consistently improved DMTS-D accuracy, but often at different doses for different subjects. To compare the relative efficacy of each drug it was necessary to determine individualized Best doses (Bartus 2000). Titrating to maximal effect is required for most cognition-enhancing drugs because of the known differences in individual sensitivity, the narrow dose widows, and the inverted U-shaped nature of the dose-response relationship for these kinds of agents (Buccafusco and Terry 2000). In human studies, the ability of methlyphenidate and nicotine to improve attention in both clinical and social settings is well documented (for reviews, see Challman and Lipsky 2000; Rezvani and Levin 2001). Human studies with caffeine have demonstrated enhanced alertness and cognition, with low doses improving visual attention (Warburton 1995). However, squirrel monkeys exhibited no improvement in performance of a DMTS task at 1–30 mg/kg (Hudzik and Wenger 1993). Our findings of caffeine-induced improvement on only the DMTS-D task, along with the decreased Sample response latencies, concur with these previous findings.
Methylphenidate was the only compound to decrease both Sample and Choice latencies. The drug's effect on Choice latencies essentially decreased the overall duration of delay intervals (computer-imposed latency+self-imposed latency). It is possible that the decrease in overall latency duration could have contributed to the drug-induced increase in accuracy during distractor trials (e.g. by providing less difficult problems). However, methylphenidate decreased Choice latencies (correct and incorrect combined) by only 1.2 s (Table 2). Although this difference was highly statistically significant, (Table 1), the difference between recall delays for the assigned short and medium delays and between medium and long delays was 25 and 45 s, respectively. Thus, it is unlikely that such relatively small drug-induced decreases in Choice latencies could effectively decrease task difficulty enough to account for the noted significant changes in accuracy. The observation that for each compound, the duration of Choice latencies was uniformly longer for trials associated with incorrect versus correct responses is consistent with previous studies of sustained attention (see Bushnell 2001). Choice latencies also were longer for distractor versus non-distractor trials, although this may reflect the preponderance of incorrect responses made during distractor trials. The inability of drug treatment (other than methylphenidate) to either decrease Choice latencies or even to normalize the differences between correct and incorrect trial latencies, suggests that these compounds did not improve task accuracies by enhancing psychomotor speed.
Research on the cognitive effects of morphine consistently finds slowed performance. This is perhaps in keeping with the known classification of the drug as a CNS depressant. In a battery of cognitive function tests, humans showed little improvement after being given morphine, demonstrating significant effects only in a choice reaction time test, reported to provide measures of alertness (O'Neill et al. 2000). Accuracy of squirrel monkeys performing a titrating DMTS task did not increase after doses from 1.0 to 1.8 mg/kg (Hudzik and Wenger 1993), nor did morphine increase rhesus monkey accuracy in a DMTS task after doses from 1.0 to 5.6 mg/kg (Schulze and Paule 1991). Although dose was not a statistically significant factor in drug responses, examination of the dose-response data allows the detection of some general trends. For example, in the case of morphine, Fig. 3 shows that, on average, non-distractor related accuracy was increased for long delay trials after the highest dose (4 mg/kg), whereas for distractor trials, the lowest dose (0.5 mg/kg) increased accuracy for medium delay trials. This general trend of greater sensitivity to the enhancing effects drugs for distractor trials versus non-distractor trials seems also to hold for nicotine, methylphenidate, and tomoxetine. If this trend can be confirmed (e.g. by using larger numbers of animals), it may help to explain the diversity of findings for the cognitive effects of these agents in that tests that focus on different aspects of cognitive function may require different optimal therapeutic doses.
The inclusion of nomifensine and tomoxetine in this study permits, perhaps, a glimpse into the possible neurochemical correlates of the cognitive functions assessed by the DMTS and DMTS-D procedures. Nomifensine, an inhibitor of monoamine reuptake preferring the dopamine transporter (Ehsanullah and Turner 1977), while not a drug of abuse per se, has been found to be sufficiently reinforcing to establish lever-pressing behavior in rats (Carlezon et al. 1995). Nomifensine competes with, and can displace cocaine for its binding site on the dopamine transporter (Missale et al. 1985). An early review of the effects of nomifensine in human subjects revealed data supporting some improvement in attention, but (as in this study) without an apparent dose-response relationship (Siegfried and Taeuber 1984). Tomoxetine, a partially selective norepinephrine reuptake inhibitor investigated as a potential therapy for depression and ADHD, was found to produce a highly significant improvement in attention in adults with ADHD (Spencer et al. 1998). Our finding of an improvement in DMTS-D performance by nomifensine but a much weaker effect with tomoxetine suggests a more important role for dopamine than for norepinephrine in the attentional processes required for optimal performance of the DMTS-D task. In previous results with adult ADHD patients, the rated therapeutic effect may depend upon the purported norepinephrine pathology associated with ADHD (for review, see Biederman and Spencer 1999).
The commonality of improved DMTS-D performance across drugs of various classes and mechanisms of action (including psychomotor stimulants and depressants) makes possible an explanation of our results based on the least common denominator among these drugs. If relative efficacy for reversing distractor-induced decrements in task accuracy can be estimated by the level of improvement versus baseline, the six compounds may be ordered as follows: nomifensine (31%)>nicotine (22%)≈morphine (19%)≈caffeine (19%)≈methylphenidate (22%)>tomoxetine (9%). Methylphenidate (Chiueh and Moore 1975), nicotine (Imperato et al. 1986), and nomifensine (Missale et al. 1985) all enhance dopamine release or utilization, whereas morphine does so indirectly through the inhibition of GABAergic neurons. The inclusion of caffeine and the exclusion of tomoxetine in the aforementioned group are supported by drug discrimination studies in which methamphetamine did not substitute for tomoxetine as did cocaine (Tidey and Bergman 1998) whereas methylphenidate substituted for caffeine (Mumford and Holtzman 1991), a result dependent upon dopamine receptors (Powell et al. 1999). Underlying the effects of these drugs is perhaps the significant tonically active GABAergic tract arising from the nucleus accumbens and which innervates the basal forebrain. Agents that enhance the activity of the accumbens dopaminergic pathway thus could disinhibit basal forebrain neurons and increase cortical acetylcholine release, as was found in dialysates collected after systemic administration of either apomorphine or d-amphetamine (Day and Fibiger 1992). That the disinhibition of basal forebrain cholinergic neurons is specific to dopamine is evidenced by lesion studies in which depletion of forebrain dopamine but not norepinephrine attenuated the amphetamine-induced increases in cortical acetylcholine release (Day et al. 1994). While the anatomical substrates of attention have not yet been identified, they have been proposed to exist diffusely in the cortex (Mesulam 1990) and to be dependent upon acetylcholine. Performance of behavioral vigilance tasks by rats has revealed that impairment in the task was correlated with the loss of acetylcholinesterase (AChE)-fiber density (McGaughy et al. 1996).
The finding that caffeine, methylphenidate, morphine, nicotine and nomifensine improved matching accuracy in sessions with interference trials in non-human primates represents pharmacological evidence in support of Robinson and Berridge's (1993) incentive-sensitization theory of drug abuse. This biopsychological theory proposes that the repeated use of addictive drugs confers the memories of, and subsequent attention toward, drugs and drug-related stimuli with extra importance, or salience. Continued use of the drug sensitizes the salient memory possibly increasing the attentional demand of the drug or drug-related stimuli. Our results support the incentive-sensitization theory of drug abuse in that the behavioral improvement in DMTS-D performance may aid our general understanding of the mechanisms of the drugs studied and suggest neurochemical correlates that may underlie the sensitization process by which drugs and drug-related stimuli come to assume more and more importance. It may be that the reinforcement derived from using drugs of abuse may be limited to their ability to elevate dopamine levels in the nucleus accumbens, but that the 'wanting' and craving that are so evident in a drug-dependent person (even when the pleasure of drug use diminishes) may be the result of cortical alterations in drug-associated memories.
In summary, we found that several drugs with abuse potential that belong to various pharmacological classes all seemed to improve aspects of performance during a task of distractability in non-human primates. The relative effectiveness of each drug to reverse distractability did not appear to directly correlate with classical views of the abuse potential for these agents. For example, caffeine was as effective as morphine and nicotine in this regard. But abuse potential does not necessarily speak to aspects of reinforcement as individually perceived by the primate. Even though reinforcement and abuse may not directly correlate with effectiveness in the current study, what is not yet known is how the ability of each of these drugs to enhance attentiveness to drug taking behavior may add to the environmental context of drug taking, and by inference, continued drug abuse. A more comprehensive understanding of these relationships may provide a basis for treatment strategies aimed at preventing relapse in detoxified addicts, and interventions for which there is currently a substantial need.
References
Bardo MT (1998) Neuropharmacological mechanisms of drug reward: beyond dopamine in the nucleus accumbens. Crit Rev Neurobiol 12:37–67
Bartus RT (2000) On neurodegenerative diseases, models, and treatment strategies: lessons learned and lessons forgotten a generation following the cholinergic hypothesis. Exp Neurol 163:495–529
Biederman J, Spencer T (1999) Attention-deficit/hyperactivity disorder (ADHD) as a noradrenergic disorder. Biol Psychiatry 46:1234–1242
Buccafusco JJ, Terry AV Jr (2000) Multiple CNS targets for eliciting beneficial effects on memory and cognition. J Pharmacol Exp Ther 295:438–446
Bushnell PJ (2001) Assessing attention in rats. In: Buccafusco JJ (ed) Methods of behavior analysis in neuroscience. CRC Press, New York, pp 111–121
Bushnell PJ, Levin ED, Marrocco RT, Sarter MF, Strupp BJ, Warburton DM (2000) Attention as a target of intoxication: insights and methods from studies of drug abuse. Neurotoxicol Teratol 22:487–502
Carelli RM, Deadwyler SA (1997) Cellular mechanisms underlying reinforcement-related processing in the nucleus accumbens: electrophysiological studies in behaving animals. Pharmacol Biochem Behav 57:495–504
Carlezon WA Jr, Devine DP, Wise RA (1995) Habit-forming actions of nomifensine in nucleus accumbens. Psychopharmacology 122:194–197
Challman TD, Lipsky JJ (2000) Methylphenidate: its pharmacology and uses. Mayo Clinic Proc 75:711–721
Chiueh CC, Moore KE (1975) Blockade by reserpine of methylphenidate-induced release of brain dopamine. J Pharmacol Exp Ther 193:559–563
Day JC, Fibiger HC (1992) Dopaminergic regulation of cortical acetylcholine release. Synapse 12:281–286
Day JC, Tham CS, Fibiger HC (1994) Dopamine depletion attenuates amphetamine-induced increases of cortical acetylcholine release. Eur J Pharmacol 263:285–292
Di Chiara G, Tanda G, Bassareo V, Pontieri R, Acquas E, Fenu S, Cadoni C, Carboni E (1999) Drug addiction as a disorder of associative learning: role of the nucleus accumbens shell/extended amygdala dopamine. Ann NY Acad Sci 877:461–485
Ehsanullah RS, Turner P (1977) Effects of nomifensine in vitro on uptake of 5-hydroxytryptamine and dopamine into human platelets. Br J Clin Pharmacol 4:159S–163S
Govoni S, Petkov VV, Montefusco O, Missale C, Battaini F, Spano PF, Trabucchi M (1984) Differential effects of caffeine on dihydroxyphenylacetic acid concentrations in various rat brain dopaminergic structures. J Pharm Pharmacol 36:458–460
Hudzik TJ, Wenger GR (1993) Effects of drugs of abuse and cholinergic agents on delayed matching-to-sample responding in the squirrel monkey. J Pharmacol Exp Ther 265:120–127
Imperato A, Mulas A, DiChiara G (1986) Nicotine preferentially stimulates dopamine release in the limbic system of freely moving rats. Eur J Pharmacol 132:337–338
Koob GF, Le Moal M (2001) Drug addiction, dysregulation of reward, and allostasis. Neuropsychopharmacology 24:97–129
McGaughy J, Kaiser T, Sarter M (1996) Behavioral vigilance following infusions of 192IgG-saporin into the basal forebrain: selectivity of the behavioral impairment and relation to cortical AchE-positive fiber density. Behav Neurosci 110:247–265
McGinty JF (1999) Advancing from the ventral striatum to the extended amygdala: implications for neuropsychiatry and drug abuse. Ann NY Acad Sci 877:835
Mesulam MM (1990) Large-scale neurocognitive networks and distributed processing for attention, language, and memory. Ann Neurol 28:597–613
Missale C, Castelletti L, Govoni S, Spano PF, Trabucchi M, Hanbauer I (1985) Dopamine uptake is differentially regulated in rat striatum and nucleus accumbens. J Neurochem 45:51–56
Mumford GK, Holtzman SG (1991) Qualitative differences in the discriminative stimulus effects of low and high doses of caffeine in the rat. J Pharmacol Exp Ther 258:857–865
O'Neill WM, Hanks GW, Simpson P, Fallon MT, Jenkins E, Wesnes K (2000) The cognitive and psychomotor effects of morphine in healthy subjects: a randomized and controlled trial of repeated (four) oral doses of dextropropoxyphene, morphine, lorazepam and placebo. Pain 85:209–215
Paule MG, Bushnell PJ, Maurissen JPJ, Wenger GR, Buccafusco JJ, Chelonis JJ, Elliott R (1998) Symposium overview: the use of delayed matching-to-sample procedures in studies of short-term memory in animals and humans. Neurotoxicol Teratol 20:493–502
Powell KR, Koppelman LF, Holtzman SG (1999) Differential involvement of dopamine in mediating the discriminative stimulu effects of low and high doses of caffeine in rats. Behav Pharmacol 10:707–716
Prendergast MA (2000) Assessment of distractibility in non-human primates performing a delayed matching-to-sample task. In: Buccafusco JJ (ed) Methods of behavior analysis in neuroscience. CRC Press, New York, pp 123–140
Prendergast MA, Jackson WJ, Terry AV Jr, Kille NJ, Arneric SP, Decker MW, Buccafusco JJ (1998a) Age-related differences in distractibility and response to methylphenidate in monkeys. Cereb Cortex 8:164–172
Prendergast MA, Jackson WJ, Terry AV Jr, Decker MW, Arneric SP, Buccafusco JJ (1998b) Central nicotinic receptor agonists ABT-418, ABT-089 and (−)-nicotine reduce distractibility in adult monkeys. Psychopharmacology 136:50–58
Rezvani AH, Levin ED (2001) Cognitive effects of nicotine. Biol Psychiatry 49:258–267
Robinson TE, Berridge KC (1993) The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res Rev 18:247–291
Sarter M, Bruno JP (1999) Abnormal regulation of corticopetal cholinergic neurons and impaired information processing in neuropsychiatric disorders. Trends Neurosci 22:67–74
Sarter M, Bruno JP, Turchi J (1999) Basal forebrain afferent projections modulating cortical acetylcholine, attention, and implications for neuropsychiatric disorders. Ann NY Acad Sci 877:368–382
Schulze GE, Paule MG (1991) Effects of morphine sulfate on operant behavior in rhesus monkeys. Pharmacol Biochem Behav 38:77–83
Setlow B (1997) The nucleus accumbens and learning and memory. J Neurosci Res 49:515–521
Siegfried K, Taeuber K (1984) Pharmacodynamics of nomifensine: a review of studies in healthy subjects. J Clin Psychiatry 45:33–38
Spencer T, Biederman J, Wilens T, Prince J, Hatch M, Jones J, Harding M, Faraone SV, Seidman L (1998) Effectiveness and tolerability of tomoxetine in adults with attention deficit hyperactivity disorder. Am J Psychiatry 155:693–695
Tidey JW, Bergman J (1998) Drug discrimination in methamphetamine-trained monkeys: agonist and antagonist effects of dopaminergic drugs. J Pharmacol Exp Ther 285:1163–1174
Warburton DM (1995) Effects of caffeine on cognition and mood without caffeine abstinence. Psychopharmacology 119:66–70
Westerink BHC, Kork J (1976) Regional rat brain levels of 3,4-dihydroxyphenylacetic acid and homovanillic acid: concurrent fluorometric measurement and influence of drugs. Eur J Pharmacol 38:281–291
Zerbe RL, Rowe H, Enas GG, Wong D, Farid N, Lemberger L (1985) Clinical pharmacology of tomoxetine, a potential antidepressant. J Pharmacol Exp Ther 232:139–143
Acknowledgements
This work was supported in part by the DuPont Pharmaceuticals Company, the Alzheimer's Association, and by the Office of Research and Development, Medical Research Service, Department of Veterans Affairs. The authors also wish to acknowledge the excellent technical assistance provided by Ms. Nancy Kille.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bain, J.N., Prendergast, M.A., Terry, A.V. et al. Enhanced attention in rhesus monkeys as a common factor for the cognitive effects of drugs with abuse potential. Psychopharmacology 169, 150–160 (2003). https://doi.org/10.1007/s00213-003-1483-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-003-1483-1