Abstract
Rationale
Methylphenidate, a dopaminergic and noradrenergic reuptake inhibitor, has been shown in young, healthy adult volunteers to produce pronounced effects on working memory and sustained attention. We were interested in assessing whether similar improvements could be conferred upon elderly volunteers in order to gain a more complete understanding of the effects of age on monoaminergic manipulations of working memory and attention, as well as to explore the potential for pharmacological intervention in attention and executive dysfunction disorders in this age group.
Objectives
The main aim of the study was to characterise the dose-related effects of methylphenidate on a range of neuropsychological functions in elderly healthy volunteers.
Methods
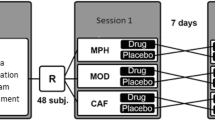
Sixty healthy elderly adult male volunteers received either a single oral dose of placebo, 20 mg or 40 mg methylphenidate prior to performing a variety of tasks designed to assess memory, attention and executive function. A randomised double-blind, between-subjects design was used.
Results
Methylphenidate had significant cardiovascular and subjective effects. However, unlike in younger volunteers, no significant effects of drug on working memory (spatial span and spatial working memory), response inhibition (stop-signal) or sustained attention (rapid visual information processing) were seen. Subtle effects on latency similar to those in younger volunteers were identified: both doses of methylphenidate resulted in a slowing in response time during set-shifting and decision-making.
Conclusions
The results of this study demonstrate that, in elderly subjects, the cognitive effects of methylphenidate are grossly attenuated and distinct from the profile previously described in younger volunteers. It is suggested that methylphenidate may not be appropriate as a pharmacological intervention in elderly patient groups, such as those reporting age-related cognitive decline.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It is well established that healthy subjects undergo age-related decline in cognition (Robbins et al. 1994; Rabbitt and Lowe 2000). The Diagnostic and Statistical Manual of Mental Disorders (DSM)-IV defines age-related cognitive decline as "an objectively identified decline in cognitive functioning consequent to the ageing process that is within normal limits given the person's age. Individuals with this condition may report problems remembering names or appointments or may experience difficulties in solving complex problems" (DSM-IV 1994, 780.9).
Previous work in monkeys (Arnsten et al. 1995), using the dopamine D2 receptor agonist quinpirole to assess the contribution of dopamine (DA) to age-related loss of prefrontal cortical cognitive function, found that DA depletion contributes significantly to age-related cognitive decline. The magnitude of cognitive improvement was less in the aged animals compared with younger monkeys, a finding consistent with a loss of DA from the prefrontal cortex with age. As described by Arnsten and Goldman-Rakic (1985), catecholamine loss in the aged non-human primate is particularly pronounced in the prefrontal and anterior temporal cortices and in the catecholaminergic neurones of the brainstem that project to the cortex.
Methylphenidate, a piperidine-derived central nervous system stimulant, is believed to exert its therapeutic effects via an ability to increase the synaptic concentration of dopamine, through blockade of dopamine transporters (Volkow et al. 1998). Methylphenidate blocks reuptake of DA more effectively than it blocks noradrenaline (NA) and much more than 5-HT (Gatley et al. 1996). In addition, D2 receptor availability in the striatum is correlated with methylphenidate-induced changes in metabolism in frontal and temporal cortices (Volkow et al. 1997). Using therapeutic doses of oral methylphenidate in healthy volunteers, Volkow et al. (2001) have shown that the increase in extracellular dopamine levels in the striatum caused by methylphenidate is negatively correlated with age. Primate studies have shown age-related differences in the cognitive response to methylphenidate (Prendergast et al. 1998), with methylphenidate significantly attenuating distractibility in younger animals, but failing to have this effect in aged monkeys.
Methylphenidate is currently the treatment of choice in attention deficit hyperactivity disorder (ADHD) in children and adults (Solanto 1998), but is being used increasingly in the treatment of elderly patients with disorders such as dementia (Galynker et al. 1997), Alzheimer's disease (Kittur and Hauser 1999), Parkinson's disease (Camicioli et al. 2001) and geriatric depression (Lavretsky and Kumar 2001). There have been no comprehensive assessments of the cognitive effects of methylphenidate in healthy elderly subjects.
Previous research in young volunteers has shown pronounced effects of methylphenidate on working memory and sustained attention. Young volunteers have shown improved accuracy on the Cambridge Neuropsychological Test Automated Battery (CANTAB) spatial working memory task following administration of methylphenidate (Elliott et al. 1997). This finding is consistent with other studies using DA agents in young subjects. Luciana and Collins (1997) showed that spatial delayed working memory functions can be facilitated with the DA agonist bromocriptine and impaired with haloperidol (a DA antagonist). Dietary depletion of DA precursor has been shown to impair spatial recognition memory and spatial working memory (Harmer et al. 2001). Similarly, administration of the dopamine D2 receptor antagonist sulpiride resulted in a dose dependent impairment at a sequence generation spatial working memory task (Mehta et al. 1999).
With regard to sustained attention, Elliott et al. (1997) observed enhanced performance on the rapid visual information processing (RVIP) sustained attention task in terms of the speeding of responses in young male volunteers after they had received 40 mg methylphenidate. d-Amphetamine, which stimulates the release of presynaptic dopamine, has also been shown to improve sustained attention in a number of studies with healthy young adults (Halliday et al. 1994; McKetin et al. 1999). This improvement is attenuated by dietary depletion of dopamine (McTavish et al. 2001). In addition, Koelega (1993) concluded that vigilance tasks appear to be the most sensitive tasks for detecting the effects of stimulants.
It was of interest to assess the effects of age on response inhibition. Mechanisms of inhibitory control may be at the core of age-related cognitive decline (West 1996). Rubia et al. (2000) have demonstrated a significant effect of age on the prefrontal cortex activation during a motor response inhibition task. Methylphenidate has also been shown to improve stop-signal performance in children with ADHD, enhancing their ability to inhibit prepotent responding (Tannock et al. 1989). It is well established that improvement in stop-signal reaction time (SSRT) is correlated with clinical measures of impulsivity in normal children and those children suffering from ADHD (Logan et al. 1997).
Whilst the most robust and replicated effects of methylphenidate have been on measures of working memory and sustained attention, effects of methylphenidate have also been shown on more complex tasks of executive function. In normal young volunteers, Rogers et al. (1999) showed that methylphenidate enhanced learning of the extradimensional shift compared to the intra-dimensional shift in the intradimensional-extradimensional (IDED) set-shifting task. In the same task, response latencies at both of the shift stages were also lengthened by methylphenidate, with the possibility that improvements in task performance may sometimes be accompanied by performance costs on other measures. Methylphenidate has also been shown to improve performance on the Tower of London test of planning (Elliott et al. 1997). Additionally, with respect to decision-making, it has been shown that ageing is associated with poorer decision-making and a decrease in risk-taking in normal volunteers (Deakin et al. 2001).
The present investigation, therefore, sought to extend the current understanding of the relationship between ageing, cognitive function and dopaminergic neurotransmission by characterising the dose-related effects of methylphenidate on neuropsychological functioning in elderly healthy volunteers. The present understanding of the mechanisms of methylphenidate action and the dopaminergic modulations of attentional function does not provide for differential effects across age groups. If loss of catecholamines contributes to age-related cognitive decline, it might be expected that pharmacological treatments that facilitate their transmission may ameliorate these cognitive deficits. Given the current evidence of an inverted "U" function underlying dopaminergic modulation of cognitive function (Zahrt et al. 1997; Mehta et al. 2001), it could be postulated that elderly subjects with reduced levels of baseline dopamine would show an exaggerated cognitive response to methylphenidate. Alternatively, if pre-synaptic loss of dopaminergic terminals or cell bodies underlies the age-related effects (Wenk et al. 1989), elderly subjects may show an attenuated response to DA-stimulating agents.
The neuropsychological battery for assessing effects of methylphenidate in the elderly was designed to assess working memory and sustained attention (shown to be reliably improved in healthy young subjects), response inhibition (a sensitive dopamine-modulated marker of ADHD) and measures of decision-making and problem solving (given the DSM-IV criteria for age-related cognitive decline).
Materials and methods
Subjects and procedures
Sixty healthy male adult volunteers (see Table 1 for demographics) were recruited by advertisement in the local community. Exclusion criteria included any significant psychiatric history, visual or motor impairment, or the concurrent use of any psychotropic medications or any medication contra-indicated with methylphenidate. In addition, subjects with a history of hypertension, cardiac disorders, epilepsy or drug or alcohol abuse were also excluded. All subjects were given the Folstein Mini Mental State Examination (MMSE) (Folstein et al. 1975) to exclude undiagnosed mild dementia: a score of less than 26 resulted in exclusion. All volunteers were advised not to consume alcohol or caffeine-containing drinks for 12 h before the study. The study was approved by the Cambridge Local Research and Ethics Committee and written informed consent was given by all subjects prior to testing.
A double-blind, between subjects design was used, with participants randomised to receive either a single oral dose of a lactose placebo, 20 mg methylphenidate or 40 mg methylphenidate. Groups were well matched for age, NART verbal IQ (as indexed by the National Adult Reading Test, Nelson 1982) and education level (Table 1).
At oral therapeutic doses (0.3–0.6 mg/kg) methylphenidate is estimated to occupy more than half of brain dopamine transporters (Volkow et al. 2001) with accumulation predominantly in the striatum (Challman and Lipsky 2000). This corresponds to a dose range of 20–40 mg methylphenidate for a 70 kg subject. Peak plasma concentration is obtained 1–3 h after oral administration with a mean plasma half-life of 1.5–2.5 h (Challman and Lipsky 2000). Subjects were therefore tested 90 min post drug administration, for approximately 2 h.
Physiological measures
Blood pressure and pulse measurements were taken using a Criticare Systems Inc. Comfort Cuff (Model 507NJ) at four time points: before drug administration, immediately prior to testing (90 min post-drug), 1 h into testing and on completion of testing.
Psychological measures
Subjects were tested using well-validated tests including measures from the CANTAB battery (Cambridge Cognition, http://www.camcog.com) (Sahakian and Owen 1992). The test battery also included novel variants of some of the original CANTAB tasks, to allow for greater sensitivity in normal volunteers. All subjects received the same tests in the same order. All computerised tasks were run on an Advantech personal computer (Model PPC-120T-RT), and responses registered either via the touch-sensitive screen or a response key, depending on the task. The majority of the tasks have been described elsewhere and readers are directed to the cited references, which were selected for their detailed descriptions.
Visual analogue scales
Subjects were asked to complete visual analogue scales (Bond and Lader 1974) before administration of the drug and at two intervals during the testing session: immediately prior to cognitive testing and 1 h into testing. At each time point subjects were asked to rate their feeling in terms of 16 dimensions. The measures used in this study were alert-drowsy, calm-excited, strong-feeble, muzzy-clear headed, well coordinated-clumsy, lethargic-energetic, contented-discontented, troubled-tranquil, mentally slow-quick witted, tense-relaxed, attentive-dreamy, incompetent-proficient, happy-sad, antagonistic-amicable, interested-bored, and withdrawn-gregarious. The dimensions were presented as 100 mm lines, the two extremes of the emotion (e.g. "alert" and "drowsy") written at each end, and subjects marked where they felt they ranked on each line.
Digit span
This task was taken from the Wechsler Adult Intelligence Scale (Wechsler 1981). Subjects were asked to repeat increasingly longer sequences of digits initially forwards and then backwards. Sequences started at 2 digits and went up to a maximum of 9. For each sequence length two different series of digits were presented, with a maximum of two points being awarded if both series were repeated correctly. Failure at the second attempt of any particular stage terminated the test. Scores for both the forwards and backwards tests were summed to yield an overall test score. Actual span, the highest number of digits correctly recalled, was also recorded.
Computerised tests taken from the CANTAB battery
A brief description of the key measures for each of the CANTAB tasks are presented in Table 2. The computerised tests were preceded by a "sensorimotor" task designed to familiarise the subjects with the touch-sensitive computer screen and the procedures. As this was a screening task the response measures are not reported here. Tasks taken from the CANTAB Working Memory and Planning battery were the spatial working memory (SWM) task and the spatial span (SSP) task. Visual memory was tested using the paired associates learning (PAL) task, while tests of attention were the rapid visual information processing (RVIP) task and attentional set-shifting task (IDED). The test battery also included a novel variant of the original CANTAB Stockings of Cambridge task, the "one-touch" Tower of London spatial planning task (NTOL).
Decision making (Gamble) task
This task is described in detail by Rahman et al. (2001). In brief, subjects were shown a display and told that the computer had hidden a yellow token randomly inside one of ten boxes at the top of the screen. The subject was trained to decide whether the computer had hidden the token in a red box or a blue box by touching either the response box marked "red" or "blue". They were then offered bets (in ascending and descending order—the order of presentation of which was randomised across groups) and instructed to try and increase their total points score by placing a bet on their choice being correct. Manipulation of the ratio of red and blue boxes from trial to trial made it possible to examine a subject's decision-making behaviour over a variety of differentially weighted contingencies.
Stop-signal (STOP) task
This classic paradigm (Logan 1994), which measures pre-potent response inhibition, has previously been used to assess ADHD and the remedial effects of methylphenidate in younger subjects (Logan et al. 2000). Subjects were required to make a speeded response on "go" trials (left response for left-pointing arrow, right response for right-pointing arrow), but to withhold their response on "stop" trials (signalled by a 300 Hz tone). Stopping was made difficult by having a preponderance of "go" trials (75%). The timing of the stop-signal was manipulated by means of a tracking algorithm (Osman et al. 1990) in such a way as to allow estimation of SSRT. Subjects performed five blocks of 64 trials each, and were given visual feedback after each block for their average correct "go" reaction time and the number of discrimination errors made (incorrect response on "go" trials). Subjects were not given feedback with regard to successful or failed inhibition, but were urged to do their best to stop, while continuing to respond as fast as possible on "go" trials.
Statistical analysis
To investigate the effect of experimental treatment upon test performance, differences between group mean (or median) performance for single scores were analysed using a one-way ANOVA (three levels) or the equivalent non-parametric Kruskal-Wallis ANOVA (conformity to a normal distribution was assessed with the Kolmogorov-Smirnov test). To clarify the nature of any such differences, planned orthogonal contrasts comparing the effect of the two doses of drug and their relationship to placebo were performed where appropriate. In instances where several readings were taken for the same score, a repeated measures ANOVA was used to test the effects of relevant independent within- and between-subjects variables. Analysis was performed on the raw scores, although for the purposes of Fig. 1 and Fig. 2 these values were adjusted for the baseline score.
Untransformed scores are displayed in tables and figures. However, in order to decrease skew and stabilise variances, data was transformed in preparation for parametric analysis. Logarithmic transformations (x=log10y) were performed for latency and arcsine transformations (\(x = 2\;\arcsin \sqrt y\)) for proportional data.
As the motivation for this study was to determine the overall cognitive profile of methylphenidate in elderly subjects, our interest lies equally in ascertaining the lack of an effect on particular variables, as in identifying the presence of a significant group difference on other variables. The former conclusion of a "lack of effect" is subject to type II errors and the latter, the presence of an effect, to type I errors (Howell 1997). Taking this into consideration, we have taken P>0.1 in reporting "no effect" and P<0.05 in reporting an "effect".
Results
Physiological effects: blood pressure and pulse
Physiological readings were taken at four time points during the experiment and the effects illustrated in Fig. 1. Repeated measures ANOVA revealed a significant main effect of drug on systolic blood pressure [F(2,57)=3.98, P=0.024]. This effect was attributable to subjects in both the drug groups showing higher mean systolic blood pressure than those in the placebo group. There were no significant differences in systolic blood pressure between the two doses of methylphenidate [F(1,38)=2.67, P=0.111]. There was a trend towards increased diastolic blood pressure following drug administration, although this failed to reach significance [F(2,57)=2.57, P=0.085]. There was, however, a highly significant effect of drug on pulse [F(2,57)=9.34, P<0.001], with subjects receiving the high dose of methylphenidate showing a significant elevation in pulse in comparison with the low dose group [F(1,38)=8.40, P=0.006] and the placebo group [F(1,38)=8.91, P=0.005]. Over time, this effect on pulse was highly significant (drug×time interaction, F(6,171)=5.02, P<0.001], with methylphenidate (again, particularly at the high dose) attenuating the natural decrease in heart rate over time (Fig. 1).
Effect of methylphenidate on blood pressure and pulse. A main effect of drug on systolic blood pressure was observed (P=0.024). There was also a trend towards an increase in diastolic blood pressure following drug administration but this failed to reach significance (P=0.085). The 40 mg methylphenidate group showed a significant elevation in heart rate, with the drug attenuating the natural decrease in heart rate over time observed with the placebo and 20 mg groups (P<0.001). *P<0.05; **P≤0.01
Subjective effects: visual analogue scales
As can be seen from Fig. 2, over time, subjects receiving 40 mg methylphenidate reported feeling more alert [drug×time interaction, F(4,114)=3.88, P=0.005], energetic [F(4,114)=5.38, P=0.001] and proficient [F(4,114)=2.74, P=0.040] than subjects in the placebo and low dose groups. Repeated measures ANOVA revealed a between subjects main effect of drug on alertness [F(2,57)=6.20, P=0.004], energy [F(2,57)=8.94, P<0.001] and proficiency [F(2,57)=3.44, P=0.039]. There were no other significant effects of drug or drug×time interactions on any of the other self-reported measures (P>0.05).
Psychological effects
As shown in Table 3, performance on several of the subtests of the CANTAB battery (namely the Digit Span, SSP, RVIP, SWM and STOP tests) did not differ between drug and placebo groups. By contrast, IDED, Gamble and PAL measures did differ significantly between groups. There was a trend towards an effect on latency with the NTOL task. The nature of these group differences is considered in detail below.
IDED
Methylphenidate produced a highly significant increase in the time taken to select a response, with a main effect of drug on total latency [F(2,52)=6.22, P=0.004]. This difference was found to be statistically significant for the compound discrimination reversal (CDR) stage [F(2,59)=4.42, P=0.019], intradimensional shift (IDS) stage [F(2,59)=6.58, P=0.003] and intradimensional reversal (IDR) stage [F(2,59)=3.80, P=0.028]. There was also a tendency towards increased latency at both the extradimensional shift (EDS) [F(2,58)=2.52, P=0.090] and the extradimensional reversal (EDR) [F(2,52)=2.81, P=0.070] stages. There were no significant differences in latency between the low and high dose of methylphenidate [F(1,33)=1.12, P=0.273]. There were no statistically significant effects on total errors [χ2(2)=0.61, P=0.738], pre-extradimensional shift errors [χ2(2)=2.28, P=0.321] or extradimensional shift errors [χ2(2)=0.42, P=0.812] between the groups.
Gamble
In this decision-making task there was a main effect of drug on deliberation time [F(2,53)=5.01, P=0.010], with subjects on drug taking longer to make their decision as to which coloured box the token was hidden in. There was no significant difference between the two doses of methylphenidate [F(1,38)=1.31, P=0.261]. There was also a tendency for subjects on drug to not choose the most likely outcome compared to the placebo group [F(2,53)=2.56, P=0.086]. Although there were no significant differences between the two drug groups [F(1,38)=1.71, P=0.184], subjects in the low dose were the most impaired compared to placebo [F(1,38)=4.87, P=0.043]. There was no effect of drug on the percentage bet placed on the decision [F(2,53)=0.44, P=0.646]. As expected, response time, choice of contingency, and size of bet all differed as a function of the ratio of red to blue boxes: there was a main effect of ratio on the deliberation time, the probability of choosing the most likely outcome and the percentage bet [F(3,57)>5.44, P<0.002]. With regard to the percentage bet, all groups selected significantly higher bets in the descend condition compared to the ascend condition [F(1,53)=34.43, P<0.001], although there were no significant differences between groups for either condition. Thus, subjects receiving methylphenidate performed similarly to those in the placebo group, but with a significant slowing in deliberation time and the low dose group displaying a trend towards choosing the less likely betting option.
PAL
Subjects in the high dose group displayed a trend towards an improved first trial memory score compared to the low-dose group. This effect only tended towards significance between the groups [F(2,59)=3.11, P=0.052], due to a significant difference between the drug groups [F(1,38)=1.36, P=0.020]. However, there were no significant differences between either dose of methylphenidate and the placebo group [F(1,38)<1.50, P>0.141]. There were no statistically significant differences between the groups with regard to total errors [F(2,59)=1.43, P=0.248] or total trials [F(2,59)=1.60, P=0.211] to criterion.
NTOL
In this task of planning, there was no effect of drug on the ability to correctly solve the problems, as measured by the mean attempts to obtain a correct solution [F(2,57)=0.96, P=0.389]. There was also no significant drug×move interaction with respect to accuracy [F(10,285)=0.67, P=0.750]. Subjects in the low dose drug group, however, displayed a trend towards taking longer to make their first attempt compared to the other two groups, with a tendency towards a main effect of drug on total latency [F(2,57)=3.02, P=0.057]. This difference in latency between the groups, however, did not reach significance at any of the individual difficulty levels of the task [F(2,57)<3.02, P>0.056], with only a tendency for subjects in the low dose group to be slower than the other two groups at the two-move [F(2,57)=3.02, P=0.056] and four-move [F(2,57)=2.60, P=0.083] problems. There was no significant drug×move interaction with respect to latency [F(10,285)=1.45, P=0.159].
Discussion
The results of this study demonstrate that, in elderly subjects, the cognitive effects of methylphenidate are grossly attenuated and distinct from the profile previously described in younger volunteers (Elliott et al. 1997). Methylphenidate had significant cardiovascular and subjective effects in elderly subjects. The higher dose of drug was associated with increases in feelings of alertness, energy and proficiency compared to placebo and low dose methylphenidate. Both doses of drug also resulted in a significantly increased systolic blood pressure, with a tendency towards a slightly elevated diastolic blood pressure. In addition, the high dose group showed a significant elevation in heart rate, with the drug attenuating the natural decrease in pulse over the testing session observed in the placebo and low dose groups.
However, unlike in younger volunteers (Elliott et al. 1997), no significant effects of drug were observed on spatial working memory (SWM), spatial memory span (SSP) or sustained attention (RVIP). There was also no effect of methylphenidate on stop-signal reaction time (SSRT) or the number of discrimination errors made in the stop-signal (STOP) task in this older population. Digit span was also unaffected by methylphenidate.
Subtle effects of latency, similar to those shown in younger volunteers (Rogers et al. 1999), were identified in this study. Both doses of methylphenidate resulted in a specific slowing in response time in the IDED and the Gamble tasks. There was also a tendency for subjects in the low dose group to be slower at the NTOL task, although this was not shown in the high dose group and there was no effect on latency at the different difficulty levels of this task between subject groups. Standard reaction time, as measured by the median "go" reaction time on the STOP task and response latency on the RVIP task, remained unaffected by methylphenidate, suggesting that the effect on latency on the IDED, Gamble and NTOL tasks was not a simple psychomotor effect of drug.
Subjects on methylphenidate displayed a trend towards choosing the less likely outcome more often in the Gamble task, particularly at the low dose. However, no differences were observed between the groups in terms of the bets placed on each decision. There was also a trend for methylphenidate to improve performance in the paired-associates learning task at the highest dose, although further analysis revealed no significant differences between the drug groups and the placebo group.
This lack of effect of methylphenidate on spatial working memory and sustained attention is consistent with theories of catecholaminergic loss in aged brains and the inverted "U" pattern of response (Cai and Arnsten 1997). Evidence suggests that either insufficient or excessive stimulation of prefrontal cortex (PFC) dopaminergic receptors is detrimental to cognitive function (Zarht et al. 1997; Mehta et al. 1999, 2001). Arnsten et al. (1994) showed that D1 agonist treatment in aged monkeys produced a biphasic response with low doses improving performance while higher doses resulted in impairment, an effect that mimicked that seen with young, dopamine-depleted monkeys in the same experiment. Full D1-receptor agonists given to elderly rhesus monkeys also impaired (or had no effect on) performance with increasing high doses, despite improving performance following lower doses (Cai and Arnsten 1997). Therefore it is possible that, at the doses used in this study, subjects were displaying similar effects to aged animals on high dose DA agonists and studies with doses lower than 20 mg methylphenidate might reveal improvements in cognitive performance.
Research with animals has indicated that advancing age is associated with marked loss of DA and DA metabolites (Goldman-Rakic and Brown 1981), with a concomitant loss of PFC cognitive function (Rapp and Amaral 1989). Biochemical studies in rats have shown strong correlations between spatial working memory impairments and loss of DA metabolites in the PFC (Luine et al. 1990). In humans, positron emission tomography using 11C-SCH23390 has revealed that the binding potential of dopamine D1 receptors in the frontal cortex decreases by almost 40% with age, despite no evidence of brain atrophy (Suhara et al. 1991). Given the finding in the aged brain of a greater loss of DA from the PFC than from the motor areas (Wenk et al. 1989), it is possible that this explains methylphenidate's lack of effect on performance despite effects of latency similar to those seen in younger subjects being retained.
The significant cardiovascular effects observed after administration of methylphenidate were similar to those observed in younger volunteers (Elliott et al. 1997). These effects of methylphenidate are due to indirect sympathomimetic action as a result of inhibition of the NA transporter (Challman and Lipsky 2000). Indirectly acting sympathomimetics are not selective in their actions at α- and β-adrenoreceptors, causing generalised blood pressure and pulse rate increases. Cardiovascular effects of methylphenidate may therefore be pronounced, in the absence of strong cognitive effects. It is more surprising, however, that the subjective effects of methylphenidate were dissociable from the (lack of) cognitive effects. Rothman et al. (2000) have suggested that the noradrenergic contribution to the subjective effects of stimulant drugs may be underestimated. It is also possible that the increased ratings of alertness and energy may be directly associated with the experience of the cardiovascular effects.
One possible limitation of this study is that the use of a between subjects design might have decreased the statistical power. A between-subjects design was used to avoid practice effects, which would have been anticipated on the more conceptual cognitive tasks such as IDED. However, robust effects of methylphenidate were found on response latency in several of the tests (IDED, Gamble and NTOL), indicating that the lack of performance effects is not due to the study design. The use of higher doses of methylphenidate in this population is not advisable due to the enhanced risk of cardiovascular side-effects. However, it is possible that a repeated dosing schedule of methylphenidate in this population might produce different results. It has been suggested that repeated exposure to stimulants produces a long-lasting change in specific neurochemical systems, particularly the mesolimbic dopamine system (Schenk et al. 1991).
In summary, administration of 20–40 mg orally of methylphenidate to an elderly population of healthy volunteers produces significant cardiovascular effects associated with increased feelings of alertness and energy, but a general lack of effect on most of the cognitive functions monitored. Increases in response latency, similar to those in younger volunteers, were identified. However, these data suggest that methylphenidate may not be beneficial for use in patients reporting age-related memory loss.
References
Arnsten AFT, Goldman-Rakic PS (1985) α2-Adrenergic mechanisms in prefrontal cortex associated with cognitive decline in aged nonhuman primates. Science 230:1273–1276
Arnsten AFT, Cai JX, Murphy BL, Goldman-Rakic PS (1994) Dopamine D1 receptor mechanisms in cognitive performance of young adult and aged monkeys. Psychopharmacology 116:143–151
Arnsten AFT, Cai JX, Steere JC, Goldman-Rakic PS (1995) Dopamine D2 receptor mechanisms contribute to age-related cognitive decline: the effects of quinpirole on memory and motor performance in monkeys. J Neurosci 15:3429–3439
Bond A, Lader M (1974) The use of analogue scales in rating subjective feelings. Br J Med Psychol 47:211–218
Cai JX, Arnsten AFT (1997) Dose-dependent effects of the dopamine D1 receptor agonists A77636 or SKF81297 on spatial working memory in aged monkeys. J Pharmacol Exp Ther 283:183–189
Camicioli R, Lea E, Nutt JG, Sexton G, Oken BS (2001) Methylphenidate increases the motor effects of l-dopa in Parkinson's disease: a pilot study. Clin Neuropharmacol 24:208–213
Challman TD, Lipsky JJ (2000) Methylphenidate: its pharmacology and uses. Mayo Clin Proc 75:711–721
Deakin JB, Robbins TW, Sahakian BJ (2001) Ageing and conservatism in decision making. J Psychopharmacol 15: A38
Elliott R, Sahakian BJ, Matthews K, Bannerjea A, Rimmer J, Robbins TW (1997) Effects of methylphenidate on spatial working memory and planning in healthy young adults. Psychopharmacology 131:196–206
Folstein MF, Folstein SE, McHugh PR (1975) "Mini-Mental State": a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Galynker I, Ieronimo C, Miner C, Rosenblum J, Vilkas N, Rosenthal R (1997) Methylphenidate treatment of negative symptoms in patients with dementia. J Neuropsychiatr Clin Neurosci 9: 231–239
Gatley SJ, Pan D, Chen R, Chaturvedi G, Ding YS (1996) Affinities of methylphenidate derivatives for dopamine, norepinephrine and serotonin transporters. Life Sci 58:231–239
Goldman-Rakic PS, Brown RM (1981) Regional changes of monoamines in cerebral cortex and subcortical structures of aging rhesus monkeys. Neuroscience 6:177–187
Halliday R, Naylor H, Brandeis D, Callaway E, Yano L, Herzig K (1994) The effect of d-amphetamine, clonidine and yohimbine on human information processing. Psychophysiology 31:331–337
Harmer CJ, McTavish SF, Clark L, Goodwin GM, Cowen PJ (2001) Tyrosine depletion attenuates dopamine function in healthy volunteers. Psychopharmacology 154:105–111
Howell DC (1997) Statistical methods for psychology, 4th edn. Wadsworth, London
Kittur S, Hauser P (1999) Improvement of sleep and behaviour by methylphenidate in Alzheimer's disease. Am J Psychiatry 156:1116–1117
Koelega HS (1993) Stimulant drugs and vigilance performance: a review. Psychopharmacology 111:1–6
Lavretsky H, Kumar A (2001) Methylphenidate augmentation of citalopram in elderly depressed patients. Am J Geriatr Psychiatry 9:298–303
Logan GD (1994) On the ability to inhibit thought and action. In: Dagenbach D, Carr TH (eds) Inhibitory processes in attention, memory and language. Academic Press, San Diego, pp 189–239
Logan GD, Schachar RJ, Tannock R (1997) Impulsivity and inhibitory control. Psychol Sci 8: 60–64
Logan GD, Schachar RJ, Tannock R (2000) Executive control problems in childhood psychopathology: stop signal studies of attention deficit hyperactivity disorder. In: Monsell S, Driver J (eds) Control of cognitive processes: attention and performance XVIII. MIT Press, Cambridge, Mass., 653–677
Luciana M, Collins PF (1997) Dopaminergic modulation of working memory for spatial but not object cues in normal humans. J Cognit Neurosci 9:330–347
Luine V, Bowling D, Hearns M (1990) Spatial memory deficits in aged rats: contributions of monoaminergic systems. Brain Res 24:271–278
McKetin R, Ward PB, Catts SV, Mattick RP, Bell JR (1999) Changes in auditory selective attention and event-related potentials following oral administration of d-amphetamine in humans. Neuropsychopharmacology 21:380–390
McTavish SF, McPherson MH, Harmer CJ, Clark L, Sharp T, Goodwin GM, Cowen PJ (2001) Antidopaminergic effects of dietary tyrosine depletion in healthy subjects and patients with manic illness. Br J Psychiatry 179:356–360
Mehta MA, Sahakian BJ, McKenna PJ, Robbins TW (1999) Systemic sulpiride in young adult volunteers simulates the profile of cognitive deficits in Parkinson's disease. Psychopharmacology 146:162–174
Mehta MA, Swainson R, Ogilvie AD, Sahakian BJ, Robbins TW (2001) Improved short-term spatial memory but impaired reversal learning following the dopamine D2 agonist bromocriptine in human volunteers. Psychopharmacology 159:10–20
Nelson H (1982) National adult reading test manual. Windsor NFER-Nelson, UK
Osman A, Kornblum S, Meyer DE (1990) Does motor programming necessitate response execution? J Exp Psychol [Hum Percep Perform] 16:183–198
Owen AM, Downes JJ, Sahakian BJ, Polkey CE, Robbins TW (1990) Planning and spatial working memory following frontal lobe lesions in man. Neuropsychologia 28:1021–1034
Owen AM, Roberts AC, Hodges JR, Summers BA, Polkey CE, Robbins TW (1993) Contrasting mechanisms of impaired attentional set-shifting in patients with frontal lobe damage or Parkinson's disease. Brain 116:1159–1175
Owen AM, Sahakian BJ, Semple JM, Polkey CE, Robbins TW (1995) Visuo-spatial short-term recognition memory and learning after temporal lobe excisions, frontal lobe excisions or amygdalo-hippocampectomy in man. Neuropsychologia 13:1–24
Park SB, Coull JT, McShane RH, Young AH, Sahakian BJ, Robbins TW, Cowen PJ (1994) Tryptophan depletion in normal volunteers produces selective impairments in learning and memory. Neuropharmacology 33:575–588
Prendergast MA, Jackson WJ, Terry AV, Kille NJ, Arneric SP, Decker MW, Buccafusco JJ (1998) Age related differences in distractability and response to methylphenidate in monkeys. Cereb Cortex 8:164–172
Rabbitt P, Lowe C (2000) Patterns of cognitive ageing. Psychol Res 63:308–316
Rahman S, Sahakian BJ, Cardinal RN, Rogers RD, Robbins TW (2001) Decision making and neuropsychiatry. Trends Cognit Sci 5:271–277
Rapp PR, Amaral DG (1989) Evidence for task-dependent memory dysfunction in the aged monkey. J Neurosci 9:3568–3576
Robbins TW, James M, Owen AM, Sahakian BJ, McInnes L, Rabbitt P (1994) Cambridge Neuropsychological Test Automated Battery (CANTAB): a factor analytic study of a large sample of normal elderly volunteers. Dementia 5:266–281
Rogers RD, Blackshaw AJ, Middleton HC, Matthews K, Hawtin K, Crowley C, Hopwood A, Wallace C, Deakin JFW, Sahakian BJ, Robbins TW (1999) Tryptophan depletion impairs stimulus-reward learning while methylphenidate disrupts attentional control in healthy young adults: implication for the monoaminergic basis of impulsive behaviour. Psychopharmacology 146:482–491
Rothman RB, Baumann MH, Dersch CM, Romero DV, Rice KC, Carroll FI, Partilla JS (2001) Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin. Synapse 38:32–41
Rubia K, Overmeyer S, Taylor E, Brammer M, Williams SC, Simmons A, Andrew C, Bullmore ET (2000) Functional frontalisation with age: mapping neurodevelopmental trajectories with fMRI. Neurosci Biobehav Rev 24:13–19
Sahakian BJ, Owen AM (1992) Computerized assessment in neuropsychiatry using CANTAB: discussion paper. J R Soc Med 5:399–402
Sahakian BJ, Morris R, Evenden JL, Heald A, Levy R, Philpot M, Robbins TW (1988) A comparative study of visuospatial memory and learning in Alzheimer type dementia and Parkinson's disease. Brain 111:695–718
Schenk S, Snow S, Horger BA (1991) Pre-exposure to amphetamine but not nicotine sensitizes rats to the motor activating effect of cocaine. Psychopharmacology 103:62–66
Solanto MV (1998) Neuropsychopharmacological mechanisms of stimulant drug action in attention-deficit hyperactivity disorder: a review and integration. Behav Brain Res 94:127–152
Suhara T, Fukuda H, Inoue O, Itoh T, Suzuki K, Yamasaki T, Tateno Y (1991) Age-related changes in human D1 dopamine receptors measured by positron emission tomography. Psychopharmacology 103:41–45
Tannock R, Schachar RJ, Carr RP, Chajczyk D, Logan GD (1989) Effects of methylphenidate on inhibitory control in hyperactive children. J Abnorm Child Psychol. 17:473–491
Volkow ND, Wang G-J, Fowler JS, Logan J, Angrist B, Hitzemann R, Lieberman J, Pappas N (1997) Effects of methylphenidate on regional brain glucose metabolism in humans: relationship to dopamine D2 receptors. Am J Psychiatry 154:50–55
Volkow ND, Wang G-J, Fowler JS, Gatley SJ, Logan J, Ding Y-S, Hitzemann R, Pappas N (1998) Dopamine transporter occupancies in the human brain induced by therapeutic doses of oral methylphenidate. Am J Psychiatry 155:1325–1331
Volkow ND, Wang G-J, Fowler JS, Logan J, Gerasimov M, Maynard L, Ding Yuo Shin, Gatley SJ, Gifford A, Franceschi D (2001) Therapeutic doses of oral methylphenidate significantly increase extracellular dopamine. J Neurosci 21 (RC121):1–5
Wechsler D (1981) Wechsler Adult Intelligence Scale, revised. Psychological Corporation, New York
Wenk GL, Pierce DJ, Struble RG, Price DL, Cork LC (1989) Age-related changes in multiple neurotransmitter systems in the monkey brain. Neurobiol Aging 10:11–19
West RL (1996) An application of prefrontal cortex function theory to cognitive aging. Psychol Bull 120:272–292
Zahrt J, Taylor JR, Matthew RG, Arnsten AFT (1997) Supranormal stimulation of D1 dopamine receptors in the rodent prefrontal cortex impairs spatial working memory performance. J Neurosci 17:8528–8535
Acknowledgements
We thank the volunteers for participating in this study, which was funded by a Wellcome Trust Programme grant awarded to Professors T.W. Robbins, B.J. Everitt and B.J. Sahakian and Dr. A.C. Roberts and completed within the MRC Centre for Behavioural and Clinical Neuroscience. D.C.T. was funded by a MRC Research Studentship. The stop-signal task was developed under a Stroke Association grant to Professors Stephen Monsell and Ian Robertson.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Turner, D.C., Robbins, T.W., Clark, L. et al. Relative lack of cognitive effects of methylphenidate in elderly male volunteers. Psychopharmacology 168, 455–464 (2003). https://doi.org/10.1007/s00213-003-1457-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-003-1457-3