Abstract
Rationale and objective.
It is well established that stress induces reinstatement of drug seeking in an animal model of relapse. Here we studied the role of the medial prefrontal cortex (mPFC) and orbitofrontal cortex (OFC) in foot-shock stress-induced reinstatement of cocaine seeking.
Methods.
Groups of rats were trained to self-administer cocaine (0.5 mg/kg per infusion, i.v., 3 h/day for 9 days) and after ten drug-free days were exposed to extinction and reinstatement test sessions. Each 60 min of extinction was separated by a 30-min time-out period after which the lever and stimulus lights were reintroduced. Rats were given four 1-h extinction sessions on day 1 and then on subsequent days were given two to three 1-h extinction sessions that were followed by a 3-h test for reinstatement. Tests were run every 48 h. In one set of experiments, the effects of inactivation of the prelimbic (PL), infralimbic (IL) or OFC by tetrodotoxin (TTX, 5 ng/0.5 µl per side) on reinstatement induced by foot shock (5 min, intermittent, 1 mA) or priming injections of cocaine (20 mg/kg, i.p.) were determined. In a second set, the effects of infusions of the D1-like and D2-like dopamine receptor antagonists (SCH 23390 and raclopride) were studied using the same methods.
Results.
TTX infusions into the PL cortex blocked both foot shock and cocaine-induced reinstatement. TTX into OFC attenuated foot-shock-induced, but not cocaine-induced reinstatement. Infusions into IL were ineffective. Infusions of SCH 22390 (0.25 µg/0.5 µl per side) into either PL or OFC blocked foot-shock-induced reinstatement, but infusions into PL had no effect on cocaine-induced reinstatement. Raclopride (5 µg/0.5 µl per side) had no effect on foot-shock-induced reinstatement in either PL or OFC or on cocaine-induced reinstatement when infused into PL. Neither TTX nor SCH23390 infusions into PL or OFC had any effect on lever pressing for sucrose.
Conclusions.
These results suggest that the PL and OFC regions form part of the circuitry mediating the effects of foot shock stress on reinstatement of drug seeking and that the PL region may be a common pathway for cue, drug and foot-shock stress-induced reinstatement of drug seeking.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Studies of reinstatement of drug seeking in laboratory animals show that, as in humans, there are three major classes of events that lead to reinstatement following periods of abstinence: presentation of cues previously associated with drug, re-exposure to the drug itself, and exposure to acute stress (Stewart 2000; Shalev et al. 2002). A major finding from these studies in the rat is that different constellations of neurochemical events and anatomical pathways mediate reinstatement induced by these different events. For example, cocaine cue-induced reinstatement depends on the integrity of the basolateral amygdala (BLA) and mesocorticolimbic dopaminergic projections to this region (See 2002), whereas reinstatement induced by cocaine does not; rather the latter is mediated by the mesocorticolimbic dopamine system and dopaminergic/glutamatergic interactions in the nucleus accumbens and ventral pallidum (McFarland and Kalivas 2001; Shaham et al. 2002; Shalev et al. 2002). In our studies, we have identified two brain systems that play critical roles in foot-shock stress-induced reinstatement but have little effect on drug-induced reinstatement. These are the corticotropin-releasing factor (CRF) and the noradrenergic (NA) systems of the central amygdala (CeA) and ventrolateral bed nucleus of the stria terminalis (BNST; Shaham et al. 2000; Erb et al. 2001; Leri et al. 2002).
Despite these clear dissociations, there can be little doubt that the systems preferentially mediating reinstatement of drug seeking by cues, drugs, and stressors are intimately connected (Stewart 2003). It is likely, therefore, that activation within one subsystem can affect other subsystems and may lead finally to the engagement of a common pathway that controls the re-initiation of responding in all these circumstances. In a search for such a pathway, we chose to explore the role of the medial prefrontal (mPFC) and orbitofrontal cortex (OFC) on stress- and cocaine-induced reinstatement in cocaine-trained rats.
Several studies point to the involvement of dopaminergic mechanisms in mPFC and OFC in cocaine self-administration. The self-administration of cocaine into the mPFC is attenuated by 6-hydroxydopamine (6-OHDA) depletion of dopamine in this region, and the substitution of dopamine for cocaine reinstates responding (Goeders and Smith 1983, 1986). Metabolic mapping studies have shown increases in glucose utilization in mPFC during cocaine self-administration (Graham and Porrino 1995). It has been reported, as well, that in early withdrawal from cocaine, metabolism in the OFC is significantly higher in addicts than in non-abusers, and this increase is positively correlated with the intensity of craving (Volkow et al. 1991; Volkow and Fowler 2000). In addition, structural deficiencies in the prefrontal cortex have been found in patients being treated for chronic cocaine addiction (Franklin et al. 2002). More recently, it was found that inactivation of the prelimbic (PL) area by infusions of a mixture of the γ-aminobutyric acid (GABA)A agonist muscimol and the GABAB agonist baclofen blocks cocaine-induced reinstatement in cocaine-trained rats (McFarland and Kalivas 2001). In the present experiments, we used tetrodotoxin (TTX) to reversibly block neural activity and, in other experiments, D1- and D2-like dopamine receptor antagonists to interfere with dopaminergic transmission in the mPFC [PL and infralimbic (IL) regions] and OFC. We compared the effects of these manipulations on foot-shock stress-induced reinstatement with reinstatement induced by priming injections of cocaine in rats trained to self-administer cocaine.
Materials and methods
Subjects
Male Long Evans rats (350–400 g, Charles River, Canada) were used in the studies of cocaine-seeking behavior. Rats were maintained on a reversed light/dark schedule (lights on 1930 hours/off 0730 hours) and were given free access to standard laboratory chow and water except during actual testing sessions. The experimental procedures followed the Canadian Council on Animal Care (CCAC) Guidelines and were approved by the Animal Care Committee, Concordia University. During training, rats were housed in a colony room and were brought daily to the self-administration chambers for the 3-h sessions. During extinction and reinstatement, they were housed 24 h per day in the self-administration chambers. Sessions always occurred during the dark period of the cycle but did not begin until 2 h after dark onset to ensure that animals had time to feed.
Surgery
Intravenous catheterization
Before surgery, rats were anesthetized with sodium pentobarbital (65 mg/kg, i.p.; MTC Pharmaceutical, Cambridge, Ontario, Canada) and were injected with atropine sulfate (0.6 mg/ml; 0.1 ml/rat, s.c.; MTC Pharmaceutical) and antibiotic (Pentlog, Rogar/STB Inc.; 0.1 ml/rat, i.m.; Wyeth-Ayerst, Montreal, Quebec, Canada). An intravenous catheter (Dow Corning, Midland, Mich.) was implanted in the right jugular vein. The catheter was secured to the vein with silk sutures and was passed subcutaneously to the top of the skull where it exited into a connector (a modified 22-gauge cannula; Plastics One, Roanoke, Va.) mounted to the skull with jeweler's screws and dental cement. A plastic blocker was placed over the opening of the connector during the recovery period.
Intracranial cannulation
During surgery, double cannulae (for PL and IL) or two single cannulae (for OFC; 20 gauge; Plastics One) were implanted bilaterally. The stereotaxic coordinates used for the cannulae (relative to bregma and the skull surface) were (Paxinos and Watson 1997):
-
PL and IL: AP +3.2 mm, L ±0.75 mm, DV –3.0 mm
-
OFC: AP +3.2 mm, L ±2.4 mm, DV –4.0 mm
The incisor bar was positioned –3.3 mm below the interaural line. Rats were allowed at least 5 days to recover from surgery. For infusions into PL and IL, injectors of different lengths were used, 4 mm and 5.5 mm, extending 1 mm and 2.5 mm beyond the tip of the cannula, respectively. Note that PL is coincident with the anterior cingulate cortex [Area Cing 3 of Zilles (1985)] in the rat and that, therefore, infusions into PL cannot be differentiated from infusions into anterior cingulate (Cing 3). For the infusions into OFC, a 5-mm injector was used.
Apparatus
The self-administration chambers were equipped with two levers, one retractable lever – "active" (Med Associates, St. Albans, Vt.) – and another non-retractable – "inactive" – lever. At the beginning of a session, the active lever was inserted, and a white stimulus light just above it was illuminated for 30 s. An infusion pump (Razel Scientific Instruments, Stamford, Conn.) was activated by responses on the active lever. Responses on the inactive lever were recorded, but did not result in activation of the pump. Drug solution was delivered over a 10-s period in a volume of 65 µl. The white stimulus light above the lever was illuminated during each 10-s infusion. Responses made during this time were recorded but did not result in reactivation of the pump. Each self-administration chamber was fitted to deliver constant-current, intermittent, inescapable, electric foot shock (5 min, 1 mA; 0.5 s on; mean interval between shocks 40 s; 10–70 s range) through a scrambler to the grid floor (Med Associates, Shaham and Stewart 1995).
Drugs
The drugs used were cocaine HCl (BDH Chemicals, Dumex Medical, Pickering, Ontario), SCH-23390 (R(+)-7-chloro-8-hydroxy-3-methyl-1-phenyl-2,3,4,5-tetrahydro-1H-3-benzazepine hydrochloride), and its inactive enantiomer SCH 23388 (R(–)-7-chloro-8-hydroxy-3-methyl-1-phenyl-2,3,4,5-tetrahydro-1H-3-benzazepine hydrochloride), Research Biochemical International, Natick, Mass., USA), and raclopride (S-3,5-dichloro-N-[(1-ethyl-2-pyrrolidinyl)methyl]-2-hydroxy-6-methoxy-benzamide L-tartrate, Sigma, St. Louis, Mo.); all were dissolved in physiological saline. Tetradotoxin (TTX Sigma, St. Louis, Mo.) was dissolved in distilled water. All intracranial infusions were 0.5 µl in volume.
Procedures
Training
Rats were trained to self-administer cocaine (0.5 mg/kg per infusion, i.v.) on a fixed-ratio (FR) 1 schedule of reinforcement during daily 3-h self-administration sessions over 9 days. A maximum of 50 infusions per session was allowed. The time of the daily training session alternated between the morning and afternoon. Rats were weighed daily, just before entering the self-administration chamber, and their catheters were flushed with a saline–heparin solution (30 U/ml; ICN Biochemicals, Montreal, Quebec, Canada) at the end of each self-administration session. At the end of training, rats were left undisturbed in the colony room for 9–11 days.
Extinction and reinstatement tests
During this period rats were housed in the self-administration chambers and given free access to food and water, except during the actual extinction and test sessions. During extinction and reinstatement testing, all of the conditions present during training were maintained, except that lever presses did not result in drug infusions. On day 1 of extinction, rats were given four 60-min extinction sessions with 30-min intervening periods during which the active lever was withdrawn. As described above, each session started by the entry of the lever and onset of the cue light above the lever, and presses on the lever activated the cue light. Thus, in addition to the lever pressing response, appetitive responses to the cue light as a discriminative stimulus and as a conditioned reinforcer underwent extinction. On subsequent days, rats were given 60-min extinction sessions with 30-min intervening periods until they made 15 or fewer responses on the active lever in 60 min; this criterion was reached after two to three 60-min sessions. When all rats reached the baseline criterion for that day, a test for reinstatement was given. These extinction/reinstatement sessions were given at 48-h intervals. On intervening days, rats were left undisturbed in the self-administration chambers.
Experiment 1: effects of TTX-induced inactivation of the PL, IL, or OFC on foot-shock-induced reinstatement
Here we used TTX to reversibly block neural activity in the different parts of the PFC to determine whether any or all of these regions plays a role in the re-initiation of lever pressing following foot-shock stress. In this first study, we were unsure of what to predict from these manipulations. As reviewed in the introduction, one set of studies might lead one to predict that reduced activity in mPFC would facilitate drug seeking, whereas other studies might lead to the prediction that blockade of activity would reduce drug seeking induced by cocaine or cocaine-related stimuli. For this reason, we used a short period of intermittent foot shock (5 min) sufficient to induce reinstatement, but which would allow for the possibility of enhancement of responding during reinstatement. In this experiment, there were three groups of rats with cannulae directed at one of the following regions: PL (n=18), IL (n=11), or OFC (n=14). Each rat was given four tests for reinstatement, two with foot shock and two without. TTX (5 ng/0.5 µl per side) or SAL (0.5 µl per side) was infused into the appropriate brain site, 25–40 min before insertion of the active lever into the chamber (Highfield et al. 2000). In the foot-shock tests, rats were exposed to 5 min of intermittent foot-shock immediately before lever insertion. The order of the tests with and without foot shock, as well as the infusions of TTX or SAL, was counterbalanced for the four reinstatement tests.
Experiment 2: effects of the TTX-induced inactivation of the PL, IL, or OFC on cocaine priming-induced reinstatement
To compare the effect of TTX-induced inactivation of PL, IL, or OFC on reinstatement induced by foot shock to those of priming injections of cocaine, three additional groups of rats (n=7–8 per group) were studied in this experiment. As in experiment 1, different groups were used for each of the cortical regions studied. The procedures used were similar to those described above for experiment 1. Each rat was given four tests for reinstatement, two with cocaine and two with saline priming injections. TTX or SAL was infused, 25–40 min before insertion of the active lever into the chamber. In the priming tests, rats were injected with cocaine (20 mg/kg, i.p.) or saline (physiological saline, 1 ml/kg, i.p.) 5 min before lever insertion. The order of the priming injections with cocaine and saline, as well as the infusions of TTX or SAL, was counterbalanced for the four reinstatement tests.
Experiment 3: effects of infusions of the D1-like dopamine receptor antagonist SCH 23390 into PL and OFC on foot-shock-induced reinstatement
To study the potential role of D1-like dopamine receptor activation in PL and OFC in foot-shock-induced reinstatement, eight groups of rats (n=5–9 per group) were used. The D1-like dopamine receptor antagonist SCH 23390 or its inactive enantiomer SCH 23388 was infused into PL or PFC. Different groups of rats were used in each condition because of the previous finding that SCH 23390 can have long-lasting effects when infused into the PFC (Vezina et al. 1994). Although SCH 23390 is a D1/D5 dopamine receptor antagonist with some affinity for the 5HT2A receptor, we have found in other experiments that its effects are mimicked by the highly selective D1/D5 receptor antagonist SCH 39166 (Emmi et al. 1997). The procedures used in this experiment were similar to those used in experiment 1, except that SCH 23390 (0.25 µg/0.5 µl per side) or its inactive enantiomer SCH 23388 (0.25 µg/0.5µl per side) was infused into PL or OFC 10 min before the period of foot shock. The dose of SCH 23390 was chosen on the basis of a previous study where it was found that SCH 23390 infusions into PFC had effects similar to those seen after dopamine depletion (Vezina et al. 1994). The dose of SCH 23388 was the same.
Experiment 4: effects of SCH 23390 in PL on the reinstatement induced by cocaine
To compare the effect of infusions of SCH 22390 into PL on reinstatement induced by foot shock to those of priming injections of cocaine, four additional groups of rats were used. Thirty rats were implanted with cannulae aimed at the PL and separated into four groups for testing, as in experiment 3 (n=6–8 per group). The procedures used in this experiment were similar to those used in experiment 2, with the exception that SCH 23390 (0.25 µg/0.5 µl per side) and SCH 23388 (0.25 µg/0.5 µl per side) were infused 10 min before the beginning of the session and 5 min before the priming injections of cocaine or saline.
Experiment 5: effects of the D2-like dopamine receptor antagonist raclopride infused into PL or OFC on foot-shock-induced reinstatement
Experiments 5 and 6 were done to determine whether a D2 receptor antagonist infused into the same regions would affect foot shock or cocaine-induced reinstatement. Forty-one rats, separated into groups of 5–7, were used in the foot-shock study. The procedures used in this experiment were similar to those used in experiment 1, except that the D2 dopamine receptor antagonist raclopride (5 µg/0.5 µl per side) or saline was infused into PL or OFC 10 min before the start of the session. The dose of raclopride was chosen on the basis of a previous study by See et al. (2001).
Experiment 6: effects of raclopride in PL on reinstatement induced by cocaine
As a follow-up to experiment 5, eight additional rats with cannulae directed at PL were tested for cocaine-induced reinstatement. In this experiment, following training and extinction procedures described above, all animals were given four tests for reinstatement (saline–saline, saline–cocaine, raclopride–saline, and raclopride–cocaine) once every second day in a counterbalanced order. As in experiment 5, raclopride (5 µg/0.5 µl per side) or saline was infused into PL 10 min before the start of the session and 5 min before the priming injections of cocaine or saline.
Experiment 7: effects of infusions of TTX and SCH 23390 infused into the PL or OFC on lever pressing for sucrose pellets
In order to test for any general debilitating effects of the infusions of TTX and SCH 23390 on behavior, two additional groups of rats were surgically prepared with cannulae directed at PL (n=6) or OFC (n=7) and were subsequently trained to lever press for sucrose pellets (45 mg, Bio-Serv, Frenchtown, N.J.). Following training these rats were given tests during which they were allowed to lever press for sucrose pellets following infusions of one of the two compounds. In order to initiate lever presses, the rats were partially food deprived for the first four training days until they reached 80% of their original body weight. The rats were given free access to normal food in the home cage for the remainder of the experiment. During training, rats were placed into the chambers where they were allowed to lever press on a FR-1 schedule for sucrose pellets for 1 h each day for 11 days when responding was stable. Four tests were given over a period of 4 days: SAL (0.5 µl per side), TTX (5 ng/0.5 µl per side), SAL (0.5 µl per side), and SCH 23390 (0.25 µg/0.5 µl per side). The total number of pellets received and lever presses were recorded for each 60-min session.
Statistics
The data from the experiments were analyzed using analyses of variance (ANOVAs) with cortical subregion as the between factor and drug pretreatment and stress or priming injections as either within or between factors depending on the design. Post-hoc comparisons of means were made using Fisher's Protected LSD (P<0.05).
Results
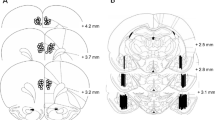
Figure 1 shows the location of the injector tips in PL, IL, and OFC regions. Following histological examination of the injector sites, the data from those rats with the probes placed appropriately were used in the analysis. From a total of 201 rats trained to self-administer cocaine, 10 had misplaced cannulae (their data were eliminated from the analyses) and 2 rats died during the abstinence period. Two rats trained to lever press for sucrose pellets had misplaced cannulae and their data were eliminated.
Representations of brain sections illustrating the placement of the injectors within the prelimbic, infralimbic, and orbitofrontal cortices. Adapted from the Rat Brain Atlas program (Paxinos and Watson 1997)
Experiment 1: effects of TTX-induced inactivation of PL, IL, or OFC on foot-shock-induced reinstatement
Training and extinction
The mean (±SEM) numbers of infusions of 0.5 mg/kg cocaine taken in the 3-h session on the last 3 days of training were 29.70±2.55, 31.88±2.22, and 33.95±2.26, respectively. Mean total responses during the last 3 days of training were 39.81±4.87, 43.72±4.45, and 44.23±3.92 on the active lever and 13.74±9.17, 6.98±5.12, and 8.21±5.25 on the inactive lever. The mean (±SEM) numbers of responses made during the first four 60-min sessions on day 1 of extinction were 53.71±6.18, 19.18±2.47, 15.4±2.15, and 13.33±3.19.
Test for reinstatement
Figure 2 shows the mean number of responses made on the active lever for rats with cannulae placed bilaterally into either PL, IL, or OFC on the tests for reinstatement with and without foot shock and with and without TTX. It can be seen that TTX given into PL blocked foot-shock-induced reinstatement (P<0.05). Infusions of TTX into OFC significantly attenuated foot-shock-induced reinstatement and infusions into IL had no significant effects. The ANOVA revealed significant main effects for drug pretreatment (F 1,40=11.92, P<0.02) and foot shock (F 1,40=16.07, P<0.0005), and there was a significant drug pretreatment by foot shock interaction (F 1,40=9.85, P<0.005).
Mean (±SEM) number of responses on the previously active lever during a 3-h session following exposure to 5 min of intermittent foot-shock stress. Rats were given infusions of saline or tetrodotoxin (TTX) into the prelimbic (PL; n=18), infralimbic (IL; n=11), or orbitofrontal cortex (OFC; n=14) regions before exposure to foot-shock (see Methods). **Significantly different from all remaining test conditions, P<0.02. *Significantly different from their no-foot shock condition, P<0.02
Experiment 2: effects of the TTX-induced inactivation of PL, IL, or OFC on the cocaine priming-induced reinstatement
Training and extinction
The mean (±SEM) numbers of infusions taken by these groups of rats on the last 3 days of training were 31.6±3.06, 34.7±3.18, and 34.5±3.26 in each 3-h session. Mean total responses were 34.9±3.44, 38.5±3.61, and 39.1±4.33 on the active lever and 1.1±0.34, 1.4±0.65, and 1.0±0.35 on the inactive lever. The mean (±SEM) numbers of responses made during the first four 60-min sessions on day 1 of extinction were 36.7±6.9, 17.8±3.0, 15.1±3.6, and 10.4±2.2.
Tests for reinstatement
Figure 3 shows the number of responses made on the active lever by rats with infusions into the PL, IL, or OFC. The ANOVA carried out on the total number of responses in 3 h revealed significant main effects of drug pretreatment (F 1,20=8.58, P<0.01), priming injection (F 1,20=12.99, P<0.02), and a significant drug pretreatment by priming injection interaction (F 1,20=4.70, P<0.05). Post-hoc comparisons between the drug pretreatment conditions indicated that TTX pretreatment in the PL subregion significantly suppressed cocaine-induced reinstatement relative to SAL pretreatment. TTX infusions into the other two cortical regions did not block cocaine-induced reinstatement, nor did they significantly reduce the levels of responding from that seen after SAL.
Mean (±SEM) number of responses on the previously active lever during 3-h session following a priming injection of cocaine (20 mg/kg, i.p.). Rats were given infusions of saline or tetrodotoxin (TTX) into prelimbic (PL; n=7), infralimbic (IL; n=8), or orbitofrontal cortex (OFC; n=8) regions before cocaine (see Methods). **Significantly different from all remaining test conditions, P<0.03. *Significantly different from their response following a saline priming injection, P<0.02
Experiment 3: effects of infusions of the D1 dopamine receptor antagonist SCH 23390 in PL or OFC on foot-shock-induced reinstatement
Training and extinction
The mean (±SEM) numbers of infusions made on the last 3 days of training were 31.19±2.77, 31.69±2.9, and 31.61±3.04 in each 3-h session. Mean total responses were 36.86±3.64, 37.92±3.82, and 36.17±3.69 on the active lever, and 1.75±0.51, 2.67±0.68, and 2.0±0.71 on the inactive lever. The mean (±SEM) numbers of responses made during the first four 60-min sessions on day 1 of extinction were 35.56±4.04, 16.64±2.22, 14.53±2.21, and 11.94±2.13.
Test for reinstatement
Figure 4 (left panel) shows the mean number of responses made on the active lever by rats with cannulae in either PL or OFC. The ANOVA carried out on the total number of responses in 3 h revealed significant main effects of drug pretreatment (SCH 23390 or inactive enantiomer SCH 23388; F 1,43=8.96, P<0.01; foot shock, F 1,43=7.18, P<0.02) and a significant drug pretreatment by foot-shock interaction (F 1,43=6.94, P<0.02). Post-hoc comparisons between the drug pretreatment conditions indicated that SCH 23390 pretreatment in either PL or OFC significantly suppressed cocaine-induced reinstatement relative to SCH 23388 pretreatment.
Mean (±SEM) number of responses on the previously active lever in the 3-h session following exposure to 5 min of intermittent foot-shock stress (left panel) or a priming injection of cocaine (20 mg/kg, i.p.; right panel) in 30 rats with cannulae aimed at the prelimbic (PL) region. Different groups of rats (n=6–8) were given infusions of SCH 23390 or its inactive enantiomer SCH 23388 10 min or 5 min before exposure to foot shock or cocaine, respectively. **Significantly different from all remaining groups, P<0.05 (left panel). *Significantly different from the groups given saline priming injections, P<0.05 (right panel)
Experiment 4: effects of PL infusions SCH 23390 on the reinstatement induced by cocaine
Training and extinction
The mean (±SEM) numbers of infusions made on the last 3 days of training were 20.52±2.85, 19.16±3.22, and 21.40±3.01 in each 3-h session. Mean total responses were 23.60±2.99, 22.96±3.82, and 23.36±3.07 on the active lever and 3.44±1.30, 1.92±0.52, and 2.80±0.86 on the inactive lever. The mean (±SEM) numbers of responses made during the first four 60-min sessions on day 1 of extinction were 27.72±4.98, 13.84±4.34, 8.92±2.03, and 3.72±0.97.
Test for reinstatement
Figure 4 (right panel) shows that pretreatment with SCH 23390 into PL had no effect on cocaine-induced reinstatement. The ANOVA carried out on the total number of responses in 3 h revealed significant main effects of priming injection (F 1,24=6.87, P<0.02) but no pretreatment effect. Both SCH 23390- and SCH 23388-pretreated rats showed cocaine-induced reinstatement, but there was no difference between the active and the inactive enantiomer.
Experiment 5: effects of infusions into PL or OFC of the D2-like dopamine receptor antagonist raclopride on foot-shock-induced reinstatement
Training and extinction
The mean (±SEM) numbers of infusions made on the last 3 days of training were 19.09±2.86, 18.63±2.53, and 18.00±2.07 in each 3-h session. Mean total responses were 23.37±3.69, 22.29±3.04, and 21.83±2.36 on the active lever and 2.34±1.08, 4.06±1.69, and 2.37±0.60 on the inactive lever. The mean (±SEM) numbers of responses made during the first four 60-min sessions on day 1 of extinction were 22.59±2.86, 8.22±1.79, 8.16±1.60, and 5.63±1.38.
Test for reinstatement
Figure 5 (left panel) shows that infusions of the D2 dopamine receptor antagonist raclopride into either PL or OFC had no effect on foot-shock-induced reinstatement. The ANOVA carried out on the total number of responses in 3 h revealed a significant main effect of foot shock (F 1,35=46.69, P<0.001) but no pretreatment effect. Post-hoc comparisons between the drug pretreatment conditions indicated that raclopride pretreatment in PL or OFC did not suppress foot-shock-induced reinstatement when compared with the saline pretreatment.
Mean (±SEM) number of responses on the previously active lever in the 3-h session following exposure to 5 min of intermittent foot-shock stress (left panel) in rats with cannulae aimed at prelimbic (PL; n=22) or orbitofrontal cortex (OFC; n=21) regions. Groups of rats (n=5–7) were given infusions of saline or raclopride 10 min before exposure to foot shock. The right panel shows the effects of raclopride on cocaine-induced reinstatement in a separate group of rats with cannulae aimed at PL (n=8). *Significantly different from the no foot shock group, P<0.05
Experiment 6: effects of raclopride infusion in PL on reinstatement induced by cocaine
Training and extinction
The mean (±SEM) numbers of infusions made on the last 3 days of training were 30.13±2.29, 31.25±5.55, and 41.63±2.90 in each 3-h session. Mean total numbers of responses were 36.63±3.08, 42.38±9.78, and 50.63±5.88 on the active lever and 1.50±0.82, 5.75±4.66, and 2.38±1.27 on the inactive lever. The mean (±SEM) numbers of responses made during the first four 60-min sessions on day 1 of extinction were 67.00±8.00, 35.63±6.10, 33.13±11.71, and 25.63±4.91.
Test for reinstatement
Figure 5 (right panel) shows that infusions of the D2 dopamine receptor antagonist raclopride into PL had no effect on cocaine-induced reinstatement. The ANOVA carried out on the total number of responses in 3 h revealed a significant main effect of cocaine (F 1,7=28.60, P=0.001) but no pretreatment effect. Both raclopride- and saline-pretreated rats showed cocaine-induced reinstatement and there was no difference between the conditions.
Experiment 7: effects of TTX and SCH 23390 into PL or OFC on lever pressing for sucrose pellets
Training
The mean (±SEM) numbers of sucrose pellets obtained on the last 3 days of training were 53.75±11.92, 62.92±15.01, and 61.5±12.14 for each 60-min session. Mean total responses were 93.58±23.09, 104.3±30.2, and 99.75±21.63 on the active lever and 7.5±2.2, 6.7±1.9, and 6.42±1.77 on the inactive lever.
Test
Table 1 shows the means (±SEM) for the number of pellets received and responses made by rats trained to respond for sucrose pellets after infusions of TTX or SCH 23390 into either PL or OFC. There were no effects of TTX infusions into either PL or OFC on the number of pellets received (infusion × group; F 1,11=1.02, P=0.33) or on the number of responses made (infusion × group; F 1,11=1.49, P=0.24). Similarly, there were no effects of infusions of the D1 dopamine receptor antagonist SCH 23390 on either the number of pellets (infusion × group; F 1,11=0.19, P=0.67) or the number of responses (infusion × group, F 1,11=0.13, P=0.72).
Discussion
A major finding from these experiments is that TTX inactivation of the PL/anterior cingulate cortex blocked reinstatement of cocaine seeking induced by both foot-shock stress and cocaine priming injections. These effects occurred at a dose that was without effect when infused into the same regions in rats lever pressing for sucrose pellets, indicating that the blockade of reinstatement was not due simply to motor incapacity. The present data, taken together with those of McFarland and Kalivas (2001) showing attenuation of cocaine-induced reinstatement by infusions of muscimol in same area, and those of See (2002) showing that TTX infusions in the area can also block cue-induced reinstatement, suggest that this region of the PFC may serve as a possible common pathway for reinstatement of drug seeking by stressors, priming injections of drugs, and drug-related cues. Note that the effect of TTX in PL was relatively selective in that infusions into the IL region just ventral to the PL region did not affect foot-shock- or cocaine-induced reinstatement. This finding is particularly important in that the injectors would have passed directly through the PL area opening up the possibility that infusions directed into IL could have seeped up the outer wall of the injector into the area above.
The effects of infusions into the OFC were mixed. These infusions attenuated foot-shock-induced reinstatement, but did not significantly affect cocaine-induced reinstatement. One can speculate that this latter difference might arise from the importance of the OFC in mediating the affective consequences of foot-shock stress (Johansen et al. 2001). The OFC, however, is known to be involved in the appetitive and aversive motivational control of goal-directed behavior (O'Doherty et al. 2001; Small et al. 2001), modulating decisions about expected outcomes (Bechara et al. 2000; Hollerman et al. 2000). Thus, although neither TTX nor SCH 23390 infusions blocked reinstatement induced by cocaine, itself, such infusions might block reinstatement induced by non-extinguished cocaine-associated cues that predict cocaine.
A second major finding from these experiments is that the D1-like dopamine receptor antagonist SCH 23390, infused into either PL or OFC, blocked foot-shock-induced reinstatement. As mentioned above, these effects occurred at a dose that was without effect when infused into the same regions in rats lever pressing for sucrose pellets. A somewhat surprising aspect of this finding is that infusions of SCH 23390 into PL and OFC had a more profound effect on foot-shock-induced reinstatement than did infusions of TTX. One might have predicted, for example, that complete inactivation of cells and fibers of passage by TTX would cause greater disruption than the more selective effect of D1-like dopamine receptor blockade. The fact that it did not suggests that D1-like dopamine receptor blockade interferes with specific processes that may be masked by the more general disruption caused by infusions of TTX. The lack of effects of the D2-like dopamine receptor antagonist raclopride, at least at the dose tested, points also to the importance of activity at D1-like receptors in these regions (Castner et al. 2000).
A somewhat unexpected finding in view of the results mentioned above, that dopamine infusions into the region reinstates responding for cocaine after 6-OHDA lesions of the mPFC (Goeders and Smith 1986), was that the D1-like dopamine receptor antagonist did not block cocaine-induced reinstatement when infused into PL. In fact, it was recently shown in two studies that intra-mPFC infusions of the non-selective dopamine receptor antagonist, flupenthixol, blocked reinstatement induced by systemic cocaine (McFarland and Kalivas 2001; Park et al. 2002). A determination of the effectiveness of the selective antagonists will have to await the testing of more doses of each. Finally, although no effects of D2 dopamine receptor infusion into the PFC were seen in these experiments, one cannot rule out the possibility that higher doses might be effective.
The data from the present experiments and those of McFarland and Kalivas (2001), Park (2002), and See (2002) are remarkably consistent pointing to the importance of the mPFC in reinstatement to drug seeking induced by cues, drugs, and stressors. When we originally began these experiments, however, it was not at all obvious what should have been predicted from inactivation of these frontal cortical regions. As discussed in the introduction, there was evidence from metabolic mapping studies for increased glucose utilization in mPFC regions during cocaine self-administration in monkeys (Graham and Porrino 1995) as well as increased metabolism in the OFC in cocaine abusers during early withdrawal that was positively correlated with the intensity of drug craving (Volkow et al. 1991; Volkow and Fowler 2000). On the other hand, structural deficiencies in the PFC have been found in patients being treated for chronic cocaine addiction (Franklin et al. 2002), and decreases in functional activity in the mPFC and OFC have been found in monkeys and human addicts following long-term exposure to cocaine self-administration (Volkow et al. 1992; Lyons et al. 1996). It has been suggested that dysfunction of activity in these regions is correlated with a decreased ability to withhold responding and impulsive behaviors (Jentsch and Taylor 1999; Rogers et al. 1999). In light of these suggestions, the finding that inactivation of the PL region blocks the re-initiation of drug seeking behaviors in rats that have undergone extinction might be seen as counterintuitive. However, the consistency of the finding across different laboratories suggests that the effects are reliable and should be studied further.
References
Bechara A, Damasio H, Damasio AR (2000) Emotion, decision making and the orbitofrontal cortex. Cereb Cortex 10:295–307
Castner SA, Williams GV, Goldman-Rakic PS (2000) Reversal of antipsychotic-induced working memory deficits by short-term dopamine D1 receptor stimulation. Science 287:2020–2022
Emmi A, Rajabi H, Stewart J (1997) Behavioral and neurochemical recovery from partial 6-hydroxydopamine lesions of the substantia nigra is blocked by daily treatment with D1/D5, but not D2, dopamine receptor antagonists. J Neurosci 17:3840–3846
Erb S, Salmaso N, Rodaros D, Stewart J (2001) A role for the CRF-containing pathway from central nucleus of the amygdala to the bed nucleus of the stria terminalis in stress-induced relapse to cocaine seeking in rats. Psychopharmacology 158:360–365
Franklin TR, Acton P, Maldjian JA, Gray JD, Croft JR, Dackis CA, O'Brien CP, Childress AR (2002) Deacreased gray matter concentration in the insular, orbitofrontal, cingulate, and temporal cortices of cocaine patients. Biol Psychiatry 51:134–142
Goeders NE, Smith JE (1983) Cortical dopaminergic involvement in cocaine reinforcement. Science 221:773–775
Goeders NE, Smith JE (1986) Reinforcing properties of cocaine in the medial prefrontal cortex: primary action on presynaptic dopaminergic terminals. Pharmacol Biochem Behav 25:191–199
Graham JH, Porrino LJ (1995) Neuroanatomical substrates of cocaine self-administration. In: Hammer RP Jr (ed) Neurobiology of cocaine. CRC Press, Boca Raton, pp 3–14
Highfield D, Clements A, Shalev U, McDonald R, Featherstone R, Stewart J, Shaham Y (2000) Involvement of the medial septum in stress-induced relapse to heroin seeking in rats. Eur J Neurosci 12:1705–1713
Hollerman JR, Tremblay L, Schultz W (2000) Involvement of basal ganglia and orbitofrontal cortex in goal-directed behavior. Prog Brain Res 126:193–215
Jentsch JD, Taylor JR (1999) Impulsivity resulting from frontostriatal dysfunction in drug abuse: implications for the control of behavior by reward-related stimuli. Psychopharmacology 146:373–390
Johansen JP, Fields HL, Manning BH (2001) The affective component of pain in rodents: direct evidence for a contribution of the anterior cingulate cortex. Proc Natl Acad Sci USA 98:8077–8082
Leri F, Bruneau J, Stewart J (2002) Understanding poly-drug use: heroin and cocaine. Addiction (in press)
Lyons D, Friedman DP, Nader MA, Porrino LJ (1996) Cocaine alters cerebral metabolism within the ventral striatum and limbic cortex of monkeys. J Neurosci 16:1230–1238
McFarland K, Kalivas PW (2001) The circuitry mediating cocaine-induced reinstatement of drug-seeking behavior. J Neurosci 21:8655–8663
O'Doherty J, Kringelbach ML, Rolls ET, Hornak J, Andrews C (2001) Abstract reward and punishment representations in the human orbitofrontal cortex. Nat Neurosci 4:95–102
Park WK, Bari AA, Jey AR, Anderson SM, Spealman RD, Rowlett JK, Pierce RC (2002) Cocaine administered into the medial prefrontal cortex reinstates cocaine-seeking behavior by increasing AMPA receptor-mediated glutamate transmission in the nucleus accumbens. J Neurosci 22:2916–2925
Paxinos G, Watson C (1997) The rat brain in stereotaxic coordinates, 3rd edn. Academic Press, New York
Rogers RD, Everitt BJ, Baldacchino A, Blackshaw AJ, Swainson R, Wynne K, Baker NB, Hunter J, Carthy T, Booker E, London M, Deakin JF, Sahakian BJ, Robbins TW (1999) Dissociable deficits in the decision-making cognition of chronic amphetamine abusers, opiate abusers, patients with focal damage to prefrontal cortex, and tryptophan-depleted normal volunteers: evidence for monoaminergic mechanisms. Neuropsychopharmacology 20:322–339
See RE (2002) Neural substrates of conditioned-cued relapse to drug-seeking behavior. Pharmacol Biochem Behav 71:517–529
See RE, Kruzich PJ, Grimm JW (2001) Dopamine, but not glutamate, receptor blockade in the basolateral amygdala attenuates conditioned reward in a rat model of relapse to cocaine-seeking behavior. Psychopharmacology 154:301–310
Shaham Y, Stewart J (1995) Stress reinstates heroin-seeking in drug-free animals: an effect mimicking heroin, not withdrawal. Psychopharmacology 119:334–341
Shaham Y, Erb S, Stewart J (2000) Stress-induced relapse to heroin and cocaine seeking in rats: a review. Brain Res Rev 33:13–33
Shaham Y, Shalev U, Lu L, de Wit H, Stewart J (2002) The reinstatement model of drug relapse: history, methodology and major findings. Psychopharmacology DOI 10.1007/s00213-002-1224-x
Shalev U, Grimm JW, Shaham Y (2002) Neurobiology of relapse to heroin and cocaine seeking: a review. Pharmacol Rev 54:1–42
Small DM, Zatorre RJ, Dagher A, Evans AC, Jones-Gotman M (2001) Changes in brain activity related to eating chocolate: from pleasure to aversion. Brain 124:1720–1733
Stewart J (2000) Pathways to relapse: the neurobiology of drug- and stress-induced relapse to drug-taking. J Psychiatr Neurosci 25:125–136
Stewart J (2003) Pathways to relapse: factors controlling the reinitiation of drug seeking after abstinence. In: Bardo MT (ed) The Nebraska symposium on motivation: motivational factors in the etiology of drug abuse. University of Nebraska Press, Lincoln (in press)
Vezina P, Blanc G, Glowinski J, Tassin J-P (1994) Blockade of D-1 dopamine receptors in the medial prefrontal cortex produces delayed effects on pre- and postsynaptic indices of dopamine function in the nucleus accumbens. Synapse 16:104–112
Volkow ND, Fowler JS (2000) Addiction, a disease of compulsion and drive: involvement of the orbitofrontal cortex. Cereb Cortex 10:318–325
Volkow ND, Fowler JS, Wolf AP, Hitzemann R, Dewey S, Bendriem B, Alpert R, Hoff A (1991) Changes in brain glucose metabolism in cocaine dependence and withdrawal. Am J Psychiatry 148:621–626
Volkow ND, Hitzemann R, Wang GJ, Fowler JS, Wolf AP, Dewey SL, Handlesman L (1992) Long-term frontal brain metabolic changes in cocaine abusers. Synapse 11:184–190
Zilles K (1985) The cortex of the rat: a stereotaxic atlas. Springer, Berlin Heidelberg New York
Acknowledgements.
We thank Francesco Leri for his help with the study on sucrose reinforcement. Supported by grants from the National Institute of Drug Abuse (USA) and Fonds pour la Formation de Chercheurs et l'Aide à la Recherche du Québec (FCAR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Capriles, N., Rodaros, D., Sorge, R.E. et al. A role for the prefrontal cortex in stress- and cocaine-induced reinstatement of cocaine seeking in rats. Psychopharmacology 168, 66–74 (2003). https://doi.org/10.1007/s00213-002-1283-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-002-1283-z