Abstract
Obesity is a key risk factor for many diseases, as cardiovascular disorders, diabetes, infertility, osteoarthritis, sleep apnea, non-alcoholic fatty liver disease (NAFLD) as well as increased risk for many cancers. Telmisartan and Candesartan cilexetil are angiotensin II receptor blockers which had proven to involve in pathogenesis of obesity and NAFLD. Aims: This work is designed to explore the possible mitigated effects of Telmisartan and Candesartan cilexetil on weight gain and fatty liver in high fat diet (HFD) fed rats. Main methods: The HFD rat model was achieved with induction of NAFLD. For Seven weeks either telmisartan or candesartan were orally administered at doses of 5 and 10 mg/kg respectively once daily. The effects of both drugs were evaluated by measurements of rat’s body weight, food intakes, length, body mass index (BMI), liver weight, inguinal and interscapular fat weights. In addition, we assayed lipid profile, liver functions tests, serum inflammatory cytokines, adipokine and leptin. Lastly, liver and adipose tissue histopathological structures were evaluated. Key findings: at end of experiment, telmisartan and candesartan were highly effective in decreasing rat’s body weight from (213.1±2.68 to 191.2±2.54 and 203.5±5.89 gm , respectively), BMI, liver weight, fat weights in addition reduced serum levels of lipid and liver enzymes. Also, inflammatory cytokines were reduced with repaired histopathological insults in liver by significantly damped NAFLD score from (6.5 ±0.17 to 1±0 and 4 ±0, respectively) and decreased areas of adipocytes from (21239.12 to 5355.7 and 11607.1 um2 , respectively).Significance: Telmisartan and candesartan have therapeutic potential against obesity and NAFLD induced by HFD in rats. All the previous indices showed more improvement in telmisartan than candesartan group.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity has reached epidemic proportions in the past few years. Marked changes in food environment, in agricultural policies, physical activity, diet and sleep patterns have all contributed to the increase in the obesity epidemic (M. Ellulu et al. 2014). Obesity is a key risk factor for many diseases, including cardiovascular disorders, diabetes, infertility, osteoarthritis, sleep apnea and non-alcoholic fatty liver disease (NAFLD) as well as increased risk for many cancers of different anatomic sites (Lauby-Secretan et al. 2016). Adipocyte hypertrophy associated with obesity, leads to increased production of several Pro-inflammatory adipokines, chemokines and cytokines that triggers local and systemic inflammation on many organs (Hosogai et al. 2007).
NAFLD is considered one of the most serious obesity complications (M. S. Ellulu et al. 2017). Obesity increases triglycerides (TGs) levels and accumulates in liver leading to steatosis. With more fat accumulation, toxic byproducts of lipid peroxidation and oxidative stress associated with increased fat deposition in the liver, lipotoxins and inflammation leads to steatohepatitis. With prolonged inflammation, death of hepatocytes with collagen deposition and fibrosis lead to cirrhosis which is irreversible (Kořínková et al. 2020).
Adult human fat tissues include the known white adipose tissue (WAT) storing lipids, the transition brite (beige) adipocyte and brown adipose tissue (BAT) responsible for adaptive or non-shivering thermogenesis (NST) and burning of fat (Rosenwald and Wolfrum 2014). BAT is rich in mitochondria, so it is metabolically active; contains uncoupling protein 1 (UCP1) which is a protein present in the inner side of mitochondrial membrane, acting through lowering the production of ATP from ADP with subsequent heat production (Bartelt and Heeren 2014). So, increasing EE by induction of BAT thermogenesis is considered a promising line against obesity (Rosenwald and Wolfrum 2014).
The renin-angiotensin system (RAS) is included in the regulation of many systems as well as regulation of body fat. Functioning RAS has been found in adipose tissue (Rawish et al. 2020). Peroxisome proliferator-activated receptor gamma (PPAR-γ) is essential for mature brown adipocytes to maintain the thermogenic capacity (Lasar et al. 2018).
Telmisartan is an angiotensin II receptor blocker. In addition to blocking the renin angiotensin system, telmisartan acts as a selective modulator of PPAR-γ, a central regulator of insulin and glucose metabolism (Benson et al. 2004). Candesartan cilexetil is another non-peptide angiotensin II receptor antagonist, that blocks the vasoconstrictor effects of angiotensin II by selectively blocking the binding of angiotensin II to the AT1 receptor in many tissues (Meredith 2007).
This study highlighted for the first time the anti-obesity effects of both telmisartan and candesartan and their associated hepato-protective potential against NAFLD. To test our hypothesis, we investigate the possible effects of telmisartan and candesartan on UCP-1, PGC1α expression, WAT Arg-1 and we try to explore the possibly involved signaling cascades.
Material and methods
Drugs
Candesartan cilexetil was purchased (Candalkan® tablets, each tablet contains 8 mg, Future Pharmaceutical Industries, Badr city, Egypt), Telmisartan (Micardis® tablets, each tablet contains 40 mg, Boehringer Ingelheim, Germany) and normal Saline 0.9% solution (E.I.P.I. Com, Egypt).
Animals
Forty male albino rats, at age of six weeks weighing 100-130 g, were purchased from Zagazig farm rats, housed in care of laboratory animal house in the Faculty of Medicine, Zagazig University, Sharkia, Egypt. Animals were housed in individual stainless-steel wire mesh cages and kept in a regulated environment (25±1°C, 50±2% humidity), with 12 h light/dark cycles. The procedures in this experimental study were performed according to the National Guidelines for the Care and Use of Laboratory Animals and approved by the Institutional Animal Care and Use Committee, Zagazig university (ZU-IACUC) with approval no (ZU-IACUC/3/F/159/2021), Duration of approval (27-10-2021) to (27-10-2024).
Experimental design
Animals grouping Fig. 1
After acclimation for 1 week, rats were weighed and randomly assigned to one of four groups, (n=10) in each group.
-
1.
Control group (I): rats were fed on normal basal diet as described in Table 1.
-
2.
HFD group (II): rats were fed on HFD as described in Table 1.
-
3.
Telmisartan group (III): rats were fed on HFD and were received daily oral telmisartan 5 mg/kg (Michael L Mathai et al. 2011).
-
4.
Candesartan group (IV): rats were fed on HFD were received daily oral candesartan 10mg/kg d for 7 weeks (Michael L Mathai et al. 2011).
Normal group was fed on normal diet as in Table 1 and HFD group was fed on HFD as in Table 1 daily for 7 weeks. Both telmisartan group and candesartan group were fed on HFD with co-administration of 5mg/kg/d and 10 mg/kg/d, respectively daily for 7 weeks.
Induction of obesity, NFLD and drugs administrations
HFD group was fed on HFD as in Table 1 daily for 7 weeks. Both telmisartan group and candesartan group were fed on HFD with co-administration of 5mg/kg/d and 10 mg/kg/d, respectively daily for 7 weeks. Oral administrations of telmisartan and Candesartan were given once daily by oral gavage in a volume does not exceed 0.3 ml/100 gm for 7 weeks.
Measurement of animals’ body weights and food intakes
Body weights and food intake of animals were measured at end of 1st, 3rd and 7th weeks. Additionally, at the end of experiment, rats were fastened for 12 hours. Both body weight and length for each individual animal were measured.
Collection and preparation of blood and tissue samples
Rats were anesthetized by pentobarbital 60 mg/kg/i.p then blood samples collected from retro-orbital plexus (Leal Filho et al. 2005). Blood samples were centrifuged at 3000 rpm for 10 minutes to get clear serum to be stored rapidly at ˗ 80°c for biochemical evaluations for hepatic enzymes including (ALT, AST and ALP) lipid profile (total cholesterol, triglycerides, LDL, HDL and VLDL cholesterol) and cytokines including (adiponectin and leptin).
Livers, inguinal and interscapular fat samples were excised and rinsed thoroughly with saline then weighted, each organ was divided into two equal parts, one part was preserved in formalin 10% for histopathological examination and other part was rapidly frozen by at ˗80°c, for later biochemical analyses.
Hepatic and WAT biochemical measurements
We subjected liver and WAT tissues to estimate Tumor necrosis factor alpha (TNFα) and Mitogen-activated protein kinase (AMPK). In addition, we assayed the levels of uncoupling protein-1 (UCP-1), Arginase-1 (Arg-1) and Brain derived neurotropic factor (BDNF) in inguinal WAT.
Biochemical analyses
Estimation of serum levels of Alanine Aminotransferase (ALT), Aspartate Aminotransferase (AST) and alkaline phosphatase (ALP)
Serum ALT, AST and ALP were measured according to (Wang et al. 2012; Almo et al. 1994; Szasz 1969) , respectively and expressed as U/L. Using assay kits supplied by MyBiosource, UK (Rat Aspartate aminotransferase ELISA Kit, catalogue no MBS269614, Rat Aspartate aminotransferase ELISA Kit, catalogue no MBS264975 and Rat Alkaline Phosphatase ELISA Kit, Catalogue No MBS2509314)
Estimation of serum levels of total cholesterol (TC), high density lipoprotein (HDL), low density, very low density lipoproteins (LDL/VLDL) and Triglycerides levels (TGs)
Serum levels of TC, HDL and LDL/VLDL cholesterol in serum were detected by the method of Fossati and Prencipe, 1982. Using kits supplied by CELL BIOLABS, INC., USA (Quantitative Colorimetric Determination method of Cholesterol, catalogue no STA-391). TG levels were detected according to the method described by Bucolo and David. 1973. Using kits supplied by CELL BIOLABS, INC., USA (colorimetric determination of serum triglycerides, Catalogue no STA-396).
Estimation of the serum levels of adipokines [leptin (LEP) and adiponectin (ADP)]
Serum leptin and adiponectin were measured according to the methods of Iwase et al. 2000 and Caselli et al. 2010, respectively. Using kits supplied by CUSABIO, USA (Rat Leptin ELISA Kit, Catalog Number CSB-E07433r and Rat Adiponectin ELISA Kit, Catalog Number CSB-E07271r).
Determination of the levels of inflammatory marker Tumor necrosis factor alpha (TNFα) in liver tissue and inguinal WAT
The levels of TNFα in liver tissue and inguinal WAT were assessed according to Kittigul et al. 2000. Using kits supplied by CUSABIO, USA (Rat TNF-α ELISA Kit, Catalog Number CSB-E11987r).
Determination of the levels of Mitogen-activated protein kinase (AMPK) in liver tissue and inguinal WAT
The levels of AMPK in liver tissue and inguinal WAT were measured following the method of Tan et al. 2017. Using kits supplied by abcam, UK (AMPK alpha pThr172 ELISA Kit, catalogue number ab154468).
Determination of the levels of uncoupling protein-1 (UCP-1), Arginase-1 (Arg-1) and Brain derived neurotropic factor (BDNF) in inguinal WAT
Estimation of UCP-1, Arg1 and BDNF levels in inguinal WAT were measured according to (Arfuso et al. 2021; George et al. 2018; Rabie et al. 2014), respectively. Using kits supplied by CUSABIO, USA [UCP1 (Mouse Mitochondrial Brown Fat Uncoupling Protein ELISA Kit, Catalog Number. CSB-EL025554MO, Rat Arginase 1 (ARG1) ELISA Kit, Catalog Number CSB-E17519r and Rat Brain derived neurotropic factor (BDNF) ELISA Kit, catalogue number CSB-E04504r].
Histological analyses
Liver, inguinal and interscapular adipose tissue specimens were taken from the animals after scarification and fixed in 10% formalin solution, then embedded in paraffin wax. Sections were cut at 4μm thickness to perform the following.
Hematoxylin and eosin (H&E) stain according to method of (Hirsch et al. 1997; Rahn 2001) and then examined under the light microscopy for evaluating the severity of obesity and NAFLD Histopathology scoring of NAFLD was performed using the method described by (Takahashi and Fukusato 2014) Table 2
Histological Scoring System for NAFLD: Grade 1 (mild) (Steatosis: up to 66%, Ballooning: occasional in zone 3, Intralobular inflammation: scattered polymorphs ± lymphocytes, Portal inflammation: no or mild), Grade 2 (moderate) (Steatosis: any degree, Ballooning: obvious, predominantly zone 3, Intralobular inflammation: polymorphs and chronic inflammation noted, Portal inflammation: mild to moderate) and Grade 3 (severe) (Steatosis: panacinar, Ballooning: ballooning and disarray obvious, predominantly in zone 3, Intralobular inflammation: scattered polymorphs ± mild chronic inflammation)
Histomorphometry studies
Morphometric examination of adipose tissue was done on every rat in each group. After H&E perceptive fields across the pictures caught by the light microscope at 400×amplification was chosen to quantify the area of adipocyte from10 rats/group. Furthermore, median values were accounted for Image J analysis software (Fiji ImageJ; 1.51 n, NIH, USA) used at Human Anatomy and Embryology Department, Zagazig University.
Statistical analysis
The obtained results were tabulated as means ± standard error of the mean (SEM). Comparison between different groups was made using one-way analysis of variances (one-way ANOVA) followed by post-Hoc (least significant difference “LSD”) tests. Statistical Package of Social Sciences (SPSS) computer software (SPSS v. 28) was used to carry out the statistical analysis. P value <0.05 is statistically significant. Quantitative data was analyzed using The Kruskal-Wallis’s test followed by dunns test to detect significant between groups. To detect interaction between weight of animals or food intakes in relation to time in weeks, we do repeat measures a nova test to detect significant in same group along with time pass.
Results
Candesartan and telmisartan lower body weight (BW) in gram (gm) and food intakes (FI) gram /day at the end of 1st, 3 rd and 7th week
First for BW, at the end of 1st week, there were no significant differences detected between the mean BW of control, HFD, telmisartan and candesartan groups. At the end of 3rd week, HFD grouped showed a significant increase in the mean BW when compared to the control group " (p-value <0.0001) Table 3". Telmisartan and candesartan administrations caused a significant decrease in the mean BW when compared to the HFD group " (p-value <0.0001, each) Table 3". At the end of experiment, telmisartan and candesartan treatment significant reduced the mean BW when compared to HFD group " (p-value <0.0001) Table 3. The mean BW of telmisartan group was significantly lower when compared to control group " (p-value <0.019) Table 3".
For FI, at the end of 1st week, mean FI of HFD group was significantly increased compared to control group" (p-value <0.009) Table 4". At the end of 3rd week, telmisartan and candesartan administration caused a significant increase of FI as compared to control group " (p-value <0.001, 0.006, respectively) Table 4". The amount of FI in telmisartan and candesartan groups showed no significant difference when compared to each other’s and to HFD group. At the end of experiment, the mean FI of telmisartan and candesartan administered groups showed a significant decrease when compared to HFD group " (p-value <0.0001), each Table 4" with no significant difference when compared to each other’s and to control group Table 4.
Anthropometric parameters (body weight gain at end of the study, length, and body mass index
-
Candesartan and telmisartan decrease body weight gain (gm) associated with HFD fed rats.
the mean BW of rats fed on basal diet for 7 weeks was 213.1±2.68 gm, but with rats that were fed on HFD for 7 weeks, their mean BW significantly increased to 360.9±7.25 gm " (p-value <0.0001) Fig. 2". Interestingly, oral administration of telmisartan and candesartan in for 7weeks in HFD rats significantly decreases BW to (191.2±2.54 and 203.5±5.89 gm), respectively " (p-value <0.0001, each Fig. 2" as compared to HFD group with no significant difference to each other" (p-value <0.32, Fig. 2". Furthermore, the mean BW of candesartan group showed no significant difference in relation to normal control group, but the BW of telmisartan group showed significant difference as compared to normal control group " (p-value <0.019) Fig. 2".
-
Candesartan and telmisartan have no effects on length (cm) in HFD fed rats.
Bar chart representing the effect of administration of single daily oral dose of 5 mg/kg/d telmisartan and 10 mg/kg/d candesartan for 7weeks on rat body weight, length and body mass index in HFD induced obesity in male albino rats. Data were analyzed by one-way ANOVA and represented by (Mean ± SE). Significant differences when (p<0.05) (a significant against control group, b significant against HFD group and c significant against Telmisartan+ HFD). n=10. Abbreviations: (n: number of rats in each group; BW: body weight; gm: gram; L: length; BMI: body mass index and cm: centimeter)
The mean length of rats that were fed on basal diet for 7 weeks was 18.4±0.16 cm. While lengths of rats who were fed on HFD alone or telmisartan and candesartan administration were (18.9±0.31, 18.3±0.15 and 18.3±0.34 cm), respectively with no significant difference between all studied groups "(p-value > 0.05) Fig. 2".
-
Candesartan and telmisartan reversed the elevated body mass index (BMI) (gm/cm2) associated with HFD fed rats.
Mean BMI of rats fed on basal diet for 7 weeks was 0.62±0.01 gm/cm2, but with rats that were fed on HFD for 7 weeks, their BMI significantly increased to 1.02±0.04 gm/cm2 " (p-value <0.0001) Fig. 2". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased BMI to (0.59±0.02 and 0.6±0.01 gm/cm2), respectively " (p-value <0.0001, each) Fig. 2" as compared to HFD group but not significantly different to normal control group as well as to each other " (p-value >0.05) Fig. 2".
Measurements of liver weights, white (inguinal) adipose tissue weights and brown (interscapular) adipose tissue weights
-
Candesartan and telmisartan decrease the high liver weight (LW) (gm) associated with HFD fed rats.
Mean LW of rats fed on basal diet for 7 weeks was 5.7±0.23 gm, but with rats that were fed on HFD for 7 weeks, LW significantly increased to 12.6±0.5 gm " (p-value <0.0001) Fig. 3". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased LW to (6.35±0.22 and 6.45±0.16 gm), respectively " (p-value <0.0001), each Fig. 3" as compared to HFD group, but not significantly different from normal control group nor to each other "(p-value > 0.05) Fig. 2".
-
Candesartan and telmisartan decrease the high inguinal white adipose tissue weight (WAT wt.) (gm) associated with HFD fed rats.
Bar chart representing the effect of administration of single daily oral dose of 5 mg/kg/d telmisartan and 10 mg/kg/d candesartan for 7weeks on liver weight (LW), inguinal white adipose tissue weight (WAT wt) and interscapular adipose tissue weight in HFD induced obesity in male albino rats. Data were analyzed by one-way ANOVA and represented by (Mean ± SE). Significant differences when (p<0.05) (a significant against control group, b significant against HFD group and c significant against Telmisartan+ HFD). n=10. Abbreviations: HFD: high fat diet; n: number of rats in each group; LW: liver weight; gm: gram; wt: weight and WAT: white adipose tissue)
Mean WAT weight of rats that were fed on basal diet for 7 weeks was 1.97±0.08 gm, but with rats that were fed on HFD for 7 weeks WAT wt significantly increased to 3.64±0.3 gm " (p-value <0.0001) Fig. 3". Oral administration of telmisartan for 7weeks in HFD rats significantly decreased WAT wt to (1.43±0.06 and 1.6±0.06 gm), respectively " (p-value <0.0001, each) Fig. 3" ass compared to both HFD and normal control groups but not significantly different to each other " (p-value >0.05) Fig. 2".
-
Candesartan and telmisartan decrease the increased interscapular adipose tissue weight in (gm) associated with HFD fed rats.
Mean interscapular adipose tissue weight of rats fed on basal diet for 7 weeks was 2.3±0.2 gm, but with rats that were fed on HFD for 7 weeks, interscapular adipose tissue weight significantly increased to 2.75±0.1 gm " (p-value <0.007) Fig. 3". Oral administration of telmisartan for 7weeks in HFD rats significantly decreased interscapular adipose tissue weight to 0.89±0.1 and 0.93±0.05 gm, respectively " (p-value <0.0001, each) Fig. 3" as compared to both HFD and normal control groups but not significantly different to each other " (p-value > 0.9) Fig. 3".
Candesartan and telmisartan repelled the elevated serum Levels of total cholesterol TC (mg/dl), TG (mg/dl), LDL (mg/dl) and VLDL (mg/dl) with increase the low serum level of HDL (mg/dl) associated with HFD fed rats
Regarding TC serum levels, the mean serum level of TC of rats fed on basal diet for 7 weeks was 97.32±0.13 (mg/dl), but significantly increased with rats fed on HFD for 7 weeks to 366.78±12.4 (mg/dl) " (p-value <0.0001) Fig. 4". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased serum TC level to 126.62±3.76 and 134.49±2.82 (mg/dl), respectively " (p-value <0.0001, each) Fig. 4" as compared to HFD. There were no significant differences to each other " (p-value > 0.05, each) Fig. 4". .
Bar chart representing the effect of administration of single daily oral dose of 5 mg/kg/d telmisartan and 10 mg/kg/d candesartan for 7weeks on lipid profile: TC, TG, LDL, VLDL and, HDL in HFD induced obesity in male albino rats. Data were analyzed by one-way ANOVA and represented by (Mean ± SE). Significant differences when (p<0.05) (a significant against control group, b significant against HFD group and c significant against Telmisartan+ HFD). n=10. Abbreviations: n: number of rats in each group; TC: total cholesterol; TG: triglyceride; HDL: high density lipoprotein; LDL: low density lipoprotein; mg: milligram; VLDL: very low-density lipoprotein; dl: deciliter)
The mean serum TG of rats fed on basal diet for 7 weeks was 60.07±0.13 (mg/dl). Rats that fed on HFD for 7 weeks, serum level of TG highly significantly increased to 378.94±8.91 (mg/dl) " (p-value <0.0001) Fig. 4". Oral administration of telmisartan and for 7weeks in HFD rats significantly decreased serum TG to (132.09±2.43 and 144.31±2.32 (mg/dl), respectively " (p-value <0.0001, each) Fig. 4" as compared to HFD group but not significantly different to each other. Both telmisartan and candesartan groups were significantly higher than normal control group " (p-value <0.0001, each) Fig. 4".
Mean serum LDL level of rats fed on basal diet for 7 weeks was 40.45±0.3 (mg/dl), but with rats that were fed on HFD for 7 weeks LDL significantly (p-value <0.0001) increased to 216.42±7.38 (mg/dl). Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased LDL to (68.97±3.44 and 98.57±3.17 (mg/dl), respectively as compared to HFD group " (p-value <0.0001, each) Fig. 4", but significantly still higher than normal control group " (p-value <0.0001, each) Fig. 4". Telmisartan group is significantly lower than candesartan group " (p-value <0.0001) Fig. 4".
Mean serum level of VLDL of rats fed on basal diet for 7 weeks was 11.46±0.11 (mg/dl), but with rats that were fed on HFD for 7 weeks serum VLDL significantly increased to 297.16±7.84 (mg/dl) "(p-value <0.0001) Fig. 4". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased VLDL to (32.33±1.4 and 39.99±0.34 (mg/dl), respectively " (p-value <0.0001, each) Fig. 4" in relation to HFD group with no significance to each other (p-value =0.6). Both telmisartan and candesartan groups were significantly higher than normal control group " (p-value <0.0001, 0.002, respectivelly) Fig. 4".
Mean serum level of HDL in control group fed on basal diet for 7 weeks was 49.12±0.67 (mg/dl). In HFD rats, serum level of HDL significantly decreased to 18.27±0.48 (mg/dl) " (p-value <0.0001) Fig. 4". Oral administration of telmisartan and candesartan for 7weeks significantly increased serum HDL level to (48.17±0.72 and 46.64±0.2 (mg/dl), respectively " (p-value <0.0001, each) Fig. 4" as compared to HFD group but was not significantly different to each nor to control group "(p-value > 0.05) Fig. 4".
Candesartan and telmisartan improve liver dysfunctions associated with HFD fed rats
Regarding liver enzymes, Mean serum level of ALT in rats fed on basal diet for 7 weeks was 82.78±0.77 (U/L), but with rats that were fed on HFD for 7 weeks, their serum ALT significantly increased to 303.18±5.33 (U/L) " (p-value <0.0001) Fig. 5". With oral administration of telmisartan and candesartan for 7weeks in HFD rats, serum ALT significantly decreased to (121.58±4.84 and 193.3±3.28 (U/L), respectively " (p-value <0.0001, each) Fig. 5" as compared to HFD, but it was significantly higher than normal control group. Telmisartan group was significantly lower than candesartan group " (p-value <0.0001) Fig. 5".
Bar chart representing the effect of administration of single daily oral dose of 5 mg/kg/d telmisartan and 10 mg/kg/d candesartan for 7weeks on serum liver function parameters: alanine transaminase (ALT), aspartate aminotransferase (AST) and Alkaline phosphatase (ALP) in HFD induced obesity in male albino rats. Data were analyzed by one-way ANOVA and represented by (Mean ± SE). Significant differences when (p<0.05) (a significant against control group, b significant against HFD group and c significant against Telmisartan+ HFD). n=10. Abbreviations: n: number of rats in each group; HFD; high fat diet; ALT: alanine transaminase; AST: aspartate aminotransferase; ALP: Alkaline phosphatase
Mean serum level of AST of rats fed on basal diet for 7 weeks was 137.47±0.85 (U/L). Rats that were fed on HFD for 7 weeks, their serum AST significantly increased to 230.34±13.43 (U/L) " (p-value <0.0001) Fig. 5". Oral administration of telmisartan and candesartan for 7weeks in rats were fed on HFD significantly decreased serum AST to (146.7±3.5 and 157.72±4.07 (U/L), respectively "(p-value <0.0001, each) Fig. 5" as compared to HFD. Serum AST in control, telmisartan and candesartan groups were not significantly different to each other "(p-value > 0.05) Fig. 5".
Mean serum ALP of rats fed on basal diet for 7 weeks was 99.24±0.3 (U/L) but with rats that were fed on HFD for 7 weeks, their serum ALP significantly increased to 293.0±6.73 (U/L) "(p-value <0.0001) Fig. 5". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased serum ALP to (157.26±5.45 and 222.25±3.99 (U/L), respectively "(p-value <0.0001), each Fig. 5" as compared to HFD, but it was significantly higher than control group "(p-value <0.0001), each Fig. 5". Telmisartan group was significantly lower than candesartan group "(p-value <0.0001) Fig. 5".
Candesartan and telmisartan elevate serum levels of leptin and adiponectin in HFD fed rats
Mean serum leptin of rats fed on basal diet for 7 weeks was 15.16±0.11 (ng/mg) but with rats that were fed on HFD for 7 weeks, their serum leptin significantly increased to 23.88±0.3 (ng/mg) "(p-value <0.0001) Fig. 6". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly increased mean serum leptin to (34.89±1.31 and 45.74±1.1 (ng/mg), respectively "(p-value <0.0001, each) Fig. 6" as compared to both normal control and HFD groups. Serum leptin level in candesartan group was significantly higher as compared to its level in telmisartan group "(p-value <0.0001) Fig. 6".
Bar chart representing the effect of administration of single daily oral dose of 5 mg/kg/d telmisartan and 10 mg/kg/d candesartan for 7weeks on serum leptin and adiponectin in HFD induced obesity in male albino rats. Data were analyzed by one-way ANOVA and represented by (Mean ± SE). Significant differences when (p<0.05) (a significant against control group, b significant against HFD group and c significant against Telmisartan+ HFD). n=10. Abbreviations: n: number of rats in each group; mg: milligram; ng: nanogram; HFD: high fat diet)
In addition, mean serum adiponectin of rats fed on basal diet for 7 weeks and rats fed on HFD were 1.62±0.08 and 0.44±0.05 (ng/mg), respectively which were not significantly different to each other "(p-value > 0.005) Fig. 6". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly increased serum adiponectin to (7.94±0.29 and 3.91±0.35 (ng/mg), respectively "(p-value <0.0001, each) Fig. 6" as compared to both normal control and HFD. Serum adiponectin level in telmisartan group was significantly higher than its level in candesartan group "(p-value <0.0001) Fig. 6".
Candesartan and telmisartan had anti-inflammatory effects by modulating hepatic expression of TNF-α and AMPK in HFD induced obesity in male albino rats
TNF-α level in liver tissue of rats fed on basal diet for 7 weeks was 94.7±1.16 (pg/mg). Rats that were fed on HFD for 7 weeks, TNF-α level in liver tissue significantly increased to 1230.6±78.8 (pg/mg) "(p-value <0.0001) Fig. 7". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased TNF-α level in liver tissue to (144.54±2.64 and 246.4±18.8 (pg/mg), respectively as compared to HFD "(p-value <0.0001, each) Fig. 7". Telmisartan group was significantly lower than candesartan group "(p-value <0.05) Fig. 7"
Bar chart representing the effect of administration of single daily oral dose of 5 mg/kg/d telmisartan and 10 mg/kg/d candesartan for 7weeks on TNF-α and AMPK in liver tissue samples in HFD induced obesity in male albino rats. Data were analyzed by one-way ANOVA and represented by (Mean ± SE). Significant differences when (p<0.05) (a significant against control group, b significant against HFD group and c significant against Telmisartan+ HFD). n=10. Abbreviations: tumor necrosis factor-alpha: TNF adenosine monophosphate activated protein kinase: AMPK; n: number of rats in each group; pg: picogram; ng: nanogram; mg: milligram
Also, AMPK level in liver tissue of rats fed on basal diet for 7 weeks was 4.54±0.13 (ng/mg) as rats that were fed on HFD for 7 weeks, their AMPK had no significant change [4.41±0.43 (ng/mg)] "(p-value =1) Fig. 7". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly increased AMPK to (47.38±2.45 and 24.79±1.52 (ng/mg), respectively "(p-value <0.0001, each) Fig. 7" as compared to HFD. Telmisartan group was significantly higher than candesartan group "(p-value <0.0001) Fig. 7".
Candesartan and telmisartan had beneficial roles by modulating inguinal WAT expression of TNF-α, AMPK, Arg-1, UCP-1 and BDNF in inguinal WAT in HFD induced obesity in male albino rats
Mean TNF-α in inguinal WAT of rats fed on basal diet for 7 weeks was 246.2±2.13 (pg/mg), but with rats that were fed on HFD for 7 weeks, inguinal WAT TNF-α significantly increased to 1348.8±21.85 (pg/mg) "(p-value <0.0001) Fig. 8". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased WAT TNF-α to (368.0±13.25 and 684.3±39.8 (pg/mg), respectively in compared to HFD group "(p-value <0.001, each) Fig. 8". Candesartan group was significantly higher than normal control group "(p-value <0.001) Fig. 8". Telmisartan group was significantly lower than candesartan group "(p-value > 0.001) Fig. 8".
Bar chart representing the effect of administration of single daily oral dose of 5 mg/kg/d telmisartan and 10 mg/kg/d candesartan for 7weeks on TNF-α, AMPK, Arg-1, UCP-1 and BDNF in inguinal WAT in HFD induced obesity in male albino rats. Data were analyzed by one-way ANOVA and represented by (Mean ± SE). Significant differences when (p<0.05) (a significant against control group, b significant against HFD group and c significant against Telmisartan+ HFD). n=10. Abbreviations: n: number of rats in each group; WAT: white adipose tissue; TNF-α: tumor necrosis factor-alpha; pg: picogram; mg: milligram; BDNF: brain derived neurotropic factor; ng: nanogram; Arg-1: arginase-1; AMPK: adenosine mono-phosphate activated protein kinase; UCP-1: uncoupling protein-1
Mean AMPK in inguinal WAT of rats that were fed on basal diet for 7 weeks was 12.83±0.25 (ng/mg) but with rats that were fed on HFD for 7 weeks, their inguinal WAT AMPK significantly decreased to 10.91±1.07 (ng/mg) "(p-value <0.07) Fig. 8". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly increased AMPK to (102.73±2.27 and 77.96±1.24 (ng/mg), respectively "(p-value <0.0001, each) Fig. 8" as compared to both normal control and HFD groups. Telmisartan group was significantly (p-value <0.05) higher as compared to candesartan group "(p-value <0.0001) Fig. 8".
Mean Arg-1 in inguinal WAT of rats fed on basal diet for 7 weeks and rats fed on HFD were (1.16±0.05 and 0.48±0.04 (ng/mg), respectively which were not significantly different to each other"(p-value =0.3) Fig. 8". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly increased Arg-1 in inguinal WAT to 14.99±0.4 and 6.63±0.41 (ng/mg), respectively "(p-value <0.0001, each) Fig. 8" as compared to both normal control and HFD groups. Mean Arg-1 in inguinal WAT level in telmisartan group was significantly higher than its level in candesartan group "(p-value <0.0001) Fig. 8".
Mean UCP-1 in inguinal WAT of rats fed on basal diet for 7 weeks and rats fed on HFD were (7.73±0.12 and 2.39±0.29 (ng/mg), respectively, which were not significantly different to each other. Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly increased UCP-1 of inguinal WAT to (55.26±1.63 and 34.0±1.73 (ng/mg), respectively "(p-value <0.0001), each Fig. 8" as compared to both normal control and HFD groups. UCP-1 level in inguinal WAT in telmisartan group was significantly higher than its level in candesartan group "(p-value <0.0001) Fig. 8".
Mean BDNF in inguinal WAT of rats fed on basal diet for 7 weeks and rats fed on HFD were (14.54±0.15 and 8.59±0.37 (ng/mg), respectively which were not significantly different to each other. Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly increased inguinal WAT BDNF to (39.88±1.83 and 33.15±1.35 (ng/mg), respectively "(p-value <0.0001, each) Fig. 8" as compared to both normal control and HFD. Inguinal WAT BDNF level in telmisartan group was significantly higher than its level in candesartan group "(p-value <0.0001) Fig. 8".
Histopathological results
Candesartan and telmisartan reduce size of area of adipocyte (AA) in inguinal adipose tissue in HFD fed male albino rats
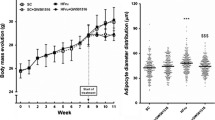
The AA of rats fed on basal diet for 7 weeks was 9893.28 (um2), but with rats that were fed on HFD for7 weeks AA significantly increased to 21239.12 (um2) "(p-value <0.05) Table 5". Oral administration of telmisartan and candesartan significantly decreased AA to (5355.7 and 11607.1 (um2), respectively as compared to rats fed on HFD "(p-value <0.05) Table 5". The AA in telmisartan was not significantly different in compared to normal control, while AA in candesartan group was significantly higher than normal control group "(p-value <0.05) Table 5"
Candesartan and telmisartan repair NAFLD by modulating scoring (steatosis, lobular inflammation, and ballooning degeneration) in HFD fed male albino rat
Effect on steatosis (0-3)
Steatosis was not detected in rats fed on basal diet for 7 weeks. While, HFD significantly induced steatosis which was 2.5±0.53 "(p-value <0.0001) Table 6". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased steatosis scoring "(p-value <0.0001, each) Table 6" as compared to HFD and became similar to control group "(p-value=1, each) Table 6".
Effect on lobular inflammation (0-3)
Lobular inflammation was not detected in rats fed on basal diet for 7 weeks. HFD significantly induce lobular inflammation which was 3±0 "(p-value <0.0001) Table 6". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased lobular inflammation scoring to 0.5±0.2 "(p-value >0.0001) Table 6". While oral co-administration of candesartan for 7weeks in HFD rats didn’t significantly change lobular inflammation 3±0 as compared to HFD group "(p-value =1) Table 6". Telmisartan group was significantly lower than candesartan group "(p-value <0.0001) Table 6".
Effect on ballooning degeneration (0-1)
Ballooning degeneration was not detected in rats fed on basal diet for 7 weeks. HFD significantly induce lobular inflammation which was 1±0 "(p-value <0.0001) Table 6". Oral administration of telmisartan for 7weeks in HFD rats significantly decreased lobular inflammation scoring to 0.5±0.2 in compared to HFD "(p-value <0.0001) Table 6" while oral co-administration of candesartan for 7weeks in HFD rats didn’t significantly change ballooning degeneration scoring in compared to HFD group "(p-value =1) Table 6". The scoring of both telmisartan and candesartan groups were significantly higher than normal control group "(p-value <0.001, 0.0001, respectively) Table 6".
Effect on total NAFLD scoring (0-7)
Total NAFLD was not detected in rats fed on basal diet for 7 weeks. HFD significantly induced NAFLD which was 6.5±0.53"(p-value <0.0001) Table 6". Oral administration of telmisartan and candesartan for 7weeks in HFD rats significantly decreased NAFLD scoring to 1±0 and 4±0, respectively as compared to HFD groups"(p-value <0.0001, respectively) Table 6" but significantly higher than normal control group "(p-value <0.0001) Table 6". Telmisartan group was significantly lower than candesartan group "(p-value <0.0001) Table 6".
Candesartan and telmisartan repair liver histopathological changes induced by an HFD in male rats (H&E staining)
Oral administration of telmisartan and candesartan in rats that were fed on HFD showed, NAFLD improved, the number of inflammatory cells decreased, gradual reduction of degree of steatosis, less prominent hepatocyte ballooning and hepatocyte degeneration Figs. 9 and 10.
A representative image of the four groups showing: control group showing: normal liver tissue with central vein (a) surrounded by rows of hepatocytes with central nuclei and eosinophilic cytoplasm. Two portal tracts could be seen with patent bile duct (circle). No steatosis, hepatocytes degeneration or ballooning could be seen. HFD group showing: diffuse hepatic steatosis with hepatocyte ballooning and degeneration more prominent at zone 3 (circles) with congested central vein (C). Diffuse inflammatory infiltrate could be seen (L). (Steatosis Grade 3). Telmisartan + HFD group showing regression of hepatic steatosis with presence of focal areas confined to zone 3 surrounding central vein (S). Focal areas of periportal inflammation could be seen (circle) (steatosis Grade1). Candesartan + HFD group showing multiple central veins surrounded by rows of regenerated hepatocytes with deep eosinophilic cytoplasm. Focal scattered areas of moderate periportal inflammation could be seen with scattered inflammatory cells (L) among hepatocytes. Areas of steatosis confined to zone 3 around central vein (circle in the right figure) (steatosis grade 2). (H&Ex100)
A representative image of higher magnification of the previous figure showing control group showing: central vein (a) surrounded by rows of normal hepatocytes with deep eosinophilic cytoplasm and central nuclei. No steatosis, ballooning or hepatocyte degeneration could be noticed. HFD group showing congested central vein (a) surrounded by rows of hepatocytes with vacuolated cytoplasm showing ballooning and degeneration (B) with scattered inflammatory infiltrate (L) Foreign body giant cell found at the lower part of the left figure (G) Degenerated apoptotic hepatocyte could be seen (Mallory body) (M) in the right figure. Telmisartan + HFD group showing sinusoidal congestion (C) among rows of regenerating hepatocytes with eosinophilic cytoplasm and central nuclei around central vein (a). Few scattered inflammatory cells could be seen (L). Candesartan + HFD group showing central vein (a) surrounded by rows of regenerating hepatocytes with vacuolated eosinophilic cytoplasm (R) and rows of hepatocytes showing ballooning and degenerative changes (B) (zone 3) (H&Ex400)
Candesartan and telmisartan avert histopathological insults in the adipose tissue (inguinal white adipose tissue (WAT) and interscapular brown adipose tissue (BAT) induced by HFD in male rat (H&E)
Regarding, inguinal white adipose tissue (WAT), Control group: showed normal size adipocytes arranged in lobules separated by microvascular septa, but HFD group: showed hypertrophied adipocytes with mild congestion in fibrovascular septa with inflammatory cell infiltrates (arrows), both treated groups (telmisartan and candesartan): showed regression of adipocytes size with regression of inflammatory cell infiltrates. The improvement was greater in telmisartan group Figs. 11 and 12.
A representative image shows section of white adipose tissue (WAT) formed of multiple lobules of mature adipocytes with large fat vacuole and signet ring nuclei, control group: shows normal size adipocytes arranged in lobules separated by microvascular septa, HFD group: shows hypertrophied adipocytes with mild congestion in fibrovascular septa with inflammatory cell infiltrates (arrows), treated groups (telmisartan and candesartan): shows regression of adipocytes size and regression of inflammatory cell infiltrate with more regression noticed at telmisartan group. (H&E x100)
In addition, Interscapular brown adipose tissue (BAT), Photomicrographs of interscapular brown adipose tissue (BAT) showing significant decrease in BAT number separated by mild congested fibrovascular tissue in HFD indicating low grade inflammation occurring with obesity. Oral administration of telmisartan and candesartan in dose of (5 mg/kg and 10 mg/kg, respectively) for 7weeks in rats were fed on HFD as prophylactic agent showed increased number of multi-locular brown fat cells per unit of area in telmisartan and candesartan groups indicating good response to therapy Figs. 13 and 14.
A representative image of interscapular brown adipose tissue (BAT) showing difference in the number of brown fat cells between control group and HFD group with more brown multi-locular adipocytes present in control group and congested fibrovascular tissue in HFD (arrows). Regression of fibro-vascular septa congestion with restoration of number of brown fat cells in treated groups with more increase noticed in telmisartan group more than candesartan group. (H&Ex100)
A representative image of interscapular brown adipose tissue (BAT) showing multi-locular fat droplets (vacuolated cytoplasm appearance) with sparse of uni-locular fat cells in-between. Control group shows normal distribution in the number of adipocytes with more depletion of brown adipocytes in HFD group. Increase number of multi-locular brown fat cells per unit of area in telmisartan and candesartan groups indicating good response to therapy. Telmisartan and candesartan groups show restoring of brown multi-locular adipocytes number with more increase could be noticed at telmisartan group. (H&E x400)
Discussion
The present study, demonstrated a significant decrease in BW, BMI, LW, WAT, and BAT weights in HFD fed rats administered telmisartan and candesartan. In line with our findings, many previous studies supported our findings, as ARBs have been shown to prevent obesity development in rodent models (Rawish et al. 2020; Huber et al. 2021) and humans (Kintscher et al. 2007).
In our study, serum levels of ALT, AST, and ALP in the HFD group were elevated together with histopathological findings of the liver that indicated NAFLD. Rats were protected by administration of telmisartan, and candesartan. In agreement with our results telmisartan (10 mg/kg/day) can reduce serum levels of ALT, AST, and ALP (Abdelhamid et al. 2021). Murad et al. 2017, also revealed that candesartan (0.1 and 0.3 mg/kg/d) for 14 days improved thioacetamide-induced chronic liver failure in rats.
In our results, there was a significant increase in the serum levels of VLDL, LDL-C, TC and TG with a significant decrease in the serum level of HDL-C in rats consumed HFD. Following telmisartan, and candesartan treatment in HFD-received animals, the lipid profile was significantly decreased while raising HDL levels. In line with our study, numerous studies indicated hepatoprotective effect of telmisartan against lipid buildup and lipotoxicity (Schuster et al. 2018; Rawish et al. 2020). In addition, candesartan has also been shown to have a potential effect on lipid accumulation and dysfunction (Nishida et al. 2011). These findings may be explained by the fact that ARBs have been reported to be a PPARα agonist and a selective PPARγ modulator which have positive effects on lipid and carbohydrate metabolism (Jayapriya et al. 2013), similarly, PPARα plays a very important role in hepatic lipid metabolism where it stimulates the uptake and oxidation of fatty acids (He et al. 2020). Telmisartan is lipophilic, which suggests that compared to other ARBs, it may have a stronger ability to enter intracellular compartments and access to PPAR-γ than other ARBs (Vanitha and Vijayal 2013). Also, telmisartan increases the expression of the phosphoenol-pyruvate carboxykinase (PEPCK) gene in human visceral adipocytes (Yumuk 2006). PEPCK is a 5 key target gene that contributes to the ability of PPAR-γ activators to reduce the levels of fatty acids (Sun et al. 2021) and this could explain the more improvement with telmisartan administration.
In the fact, high FFA levels in the HFD group induce an increase in the transcription factor (NF-kB)”(Feldstein et al. 2004) along with an increase in ROS and TNF-α (Wei et al. 2008), and this activation may be mediated through AngII stimulation of the AT1R present on adipocyte, so blockage of this receptor by ARBs may have an anti-inflammatory role in these cells (Wei et al. 2008). Consistent with these observations, telmisartan and candesartan markedly improved and down regulated TNF-α levels in liver and WAT. Comparable findings were reported by (Wohl et al. 2010), they showed that telmisartan (160 mg/day) treatment for 3 weeks in human decreased plasma TNF-α. Similarly, Abdelhamid et al. 2021, reported that, in an alcohol-induced hepatic damage model, telmisartan (10 mg/kg/day) reduced liver TNF-α expression in adult male BALB/c mice.
BDNF plays an important role in regulating energy balance, promoting fatty acid oxidation (Wohl et al. 2010). In the same context, Hashida et al. 2021, demonstrated that higher serum level of BDNF was associated with a reduction in activity in human patients with NAFLD revealing the suppression action of BDNF on NAFLD activity as well as its favorable action on increasing fatty acid oxidation and lipid metabolism.
We found that rats fed on HFD down-regulated the expression of BDNF in inguinal WAT and AMPK in the liver and inguinal WAT, while administration of telmisartan and candesartan up-regulated the WAT expression of BDNF and AMPK (in the liver tissue and WAT). Similarly, BDNF heterozygous mutant mice and animals with disruption of the BDNF gene exhibit hyperphagia, increased body weight, and adiposity (Kernie et al., 2000; Sha et al. 2007). In accordance with our finding, after 4 weeks of telmisartan treatments (10 mg/kg/day) in streptozotocin-induced diabetic rats, the levels of BDNF were found to be systemically increased in diabetic rats compared to untreated rats (Ola et al. 2013). This may be due to the anti-inflammatory effect of ARBS in the hypothalamus, improving levels of BDNF (Lenart et al. 2019).
Huang et al. 2020 showed that, Telmisartan 3 mg/kg/d improved obesity-induced insulin resistance in C57BL/6J male mice by suppressing endoplasmic reticulum stress via AMPK activation. Similarly, Candesartan (5 mg/kg/day) in drinking water for two weeks significantly increases hypothalamic AMPK expression in both basal and stress-induced models (Marques et al. 2012). Ribeiro-Oliveira et al. 2015) demonstrated that 7 chronic administration of candesartan (5 mg/kg/day) in drinking water for two weeks increased hepatic AMPK activity as measured by a kinase activity assay in male Wistar rats. This may be explained as telmisartan and candesartan increase phosphorylation of AMPK receptor at Thr172 site (the active catalytic site of the receptor) and subsequently its activation (Hwang and Cho 2020; Huang et al. 2020). Because AMPK is a key sensor regulator of energy homeostasis and nutrient metabolism, regulating fatty acid metabolism, protein synthesis, and glucose uptake, its levels are reduced in cases of positive energy balance, such as obesity (Liou et al. 2019). Telmisartan increases AMPK in liver, which in turn increases fatty acid oxidation, decreases fatty acid and TG synthesis, and increases mitochondrial biogenesis. All the previous actions may be implicated in the prevention of NAFLD (Zhou et al. 2019; Pino-de la Fuente et al., 2019).
Adiponectin is an important adipokine which increases fatty acid uptake and oxidation in muscle and other tissues and acts in the brain to increase metabolic rate (Mullen et al. 2009). In liver, adiponectin increases fatty acid oxidation and decreases glucose release and improves insulin resistance (Berg et al. 2001). In adipocytes, it has an autocrine effect on adipose tissue differentiation, browning, increasing lipolysis and fat oxidation (M L Mathai et al. 2008).
In our work, administration of telmisartan or candesartan to HFD-fed rats significantly elevated adiponectin serum levels. In agreement with our finding, (Makita et al. 2008) found that candesartan (8 or 12 mg/d) or telmisartan (40 or 80 mg/d) was found to increase adiponectin in hypertensive patients aged 40-80 years.
In our study, the administration of telmisartan or candesartan to HFD-fed rats increased serum leptin levels. In line with our findings, Schuster et al. 2018 found that HFD induced obesity in male C57BL/6N mice, with marked leptin elevation. Schuster et al. 2018 also reported that telmisartan improved leptin action as it regains leptin transport through the leptin receptor; leptin then can cross the blood brain barrier again and thereby protect against leptin resistance.
Disagreeing with our study, Li et al. 2016 reported that telmisartan treatment in a metabolic syndrome model induced by HFD decreased both serum leptin and adipocyte derived leptin levels.
Macrophages infiltration into adipose tissue is a common feature of obesity (Alexander Rosendah 2015). The importance of transformation of M1 to M2 (M2 polarization) in adipose tissue browning was evidenced by the release of catecholamines from M2 macrophage which subsequently stimulate thermogenesis via β3 adrenergic receptor activation (Nguyen et al. 2011). with its ability to induce UCP-1 with the resultant browning and increasing energy expenditure (Brestoff et al. 2015). Arg-1 is an important marker of M2 macrophage polarization (Ma et al. 2022). PPARγ, which are ligand-activated transcription factors, control macrophage polarization. In fact, PPARγ deficient macrophages have been shown to resist M2 polarization (Mukundan et al. 2009). Consistent with these findings, telmisartan and candesartan administration to HFD fed rats showed significant increase of Arg-1 expression in WAT. Activation of PPARδ, PPARγ, and AMPK in the telmisartan and candesartan groups was shown to favor M2 polarization (Jeon et al. 2019).
The most prominent feature of browning is the induction of UCP-1 expression (Brestoff et al. 2015). With administration of telmisartan or candesartan to HFD-fed rats showed significant increase of UCP-1 expression in WAT. Araki et al. 2006 have reported that telmisartan (5 mg/kg/d) for 14 days induced UCP-1 expression in inguinal and subcutaneous WAT, which was associated with an increase in energy expenditure with decreasing body weight in mature male mice .which could be mediated by maintaining phosphorylation of PPARγ (Jeon et al. 2019).
Telmisartan and candesartan groups increased number of BAT may be attributed to the increase in UCP-1 (Puigserver et al. 1998), AMPK (Hasanvand et al. 2016), BDNF (Kim et al. 2017) and Arg-1(M2 polarization) (Jeon et al. 2019) as shown in our biochemical studies referring to that the anti-obesity effect of telmisartan and candesartan may be due to their browning effect. These results confirmed by Histopathological findings which revealed that both telmisartan and candesartan improved the picture of NAFLD and decrease in WAT diameter while BAT number increased with more improvement and more browning induction occurring with telmisartan group. All the previous indices showed more improvement in telmisartan than candesartan group.
In conclusion
This study highlighted for the first time the potential anti-obesity effect of both telmisartan and candesartan and their associated hepato-protective potential against NAFLD. This suggestion was evidenced by attenuation of body weight, food intake plus correction of lipid profile and liver enzymes. In addition to upregulated AMPK (both in liver tissue and inguinal tissue) and inguinal Arg-1, UCP-1 and BDNF levels with as well as serum adiponectin with M2 macrophage polarization and browning with subsequent increasing energy loss due to increase UCP-1 expression in inguinal WAT adipose tissue. Future research is recommended on telmisartan and candesartan hepato-protective effects especially in humans.
Data availability
Data will be made available on request and requested from corresponding author H.A.H
Email: drhebahassanpharma58@gmail.com, HAHanafi@medicine.zu.edu.eg
References
Abdelhamid AM, Elsheakh AR, Suddek GM, Abdelaziz RR (2021) Telmisartan alleviates alcohol-induced liver injury by activation of PPAR-γ/ Nrf-2 crosstalk in mice. Int Immunopharmacol 99(October):107963. https://doi.org/10.1016/j.intimp.2021.107963
Almo SC, Smith DL, Danishefsky AT, Ringe D (1994) The structural basis for the altered substrate specificity of the R292D Active Site mutant of aspartate aminotransferase from E. coli. Protein Eng 7(3):405–412. https://doi.org/10.1093/protein/7.3.405
Araki K, Masaki T, Katsuragi I, Tanaka K, Kakuma T, Yoshimatsu H (2006) Telmisartan prevents obesity and increases the expression of uncoupling protein 1 in diet-induced obese mice. Hypertension (Dallas, Tex : 1979) 48(1):51–57. https://doi.org/10.1161/01.HYP.0000225402.69580.1d
Arfuso F, Giannetto C, Panzera MF, Fazio F, Piccione G (2021) Uncoupling protein-1 (UCP1) in the adult horse: correlations with body weight, rectal temperature and lipid profile. Animals : Open Access J MDPI 11(6). https://doi.org/10.3390/ani11061836
Bartelt A, Heeren J (2014) Adipose tissue browning and metabolic health. Nat Rev Endocrinol 10(1):24–36. https://doi.org/10.1038/nrendo.2013.204
Benson, Stephen C, Harrihar A Pershadsingh, Christopher I Ho, Amar Chittiboyina, Prashant Desai, Michal Pravenec, Nianning Qi, Jiaming Wang, Mitchell A Avery, and Theodore W Kurtz 2004. ‘Identification of telmisartan as a unique angiotensin II receptor antagonist with selective PPARgamma-modulating activity.’ Hypertension (Dallas, Tex : 1979) 43 (5): 993–1002. 10.1161/01.HYP.0000123072.34629.57
Berg AH, Combs TP, Du X, Brownlee M, Scherer PE (2001) The adipocyte-secreted protein Acrp30 enhances hepatic insulin action. Nat Med 7(8):947–953. https://doi.org/10.1038/90992
Brestoff JR, Kim BS, Saenz SA, Stine RR, Monticelli LA, Sonnenberg GF, Thome JJ et al (2015) Group 2 innate lymphoid cells promote beiging of white adipose tissue and limit obesity. Nature 519(7542):242–246. https://doi.org/10.1038/nature14115
Bucolo G, David H (1973) Quantitative determination of serum triglycerides by the use of enzymes. Clin Chem 19(5):476–482
Caselli C, Melaiu O, Maltinti M, Del Ry S, Cabiati M, Prescimone T, Neglia D, Giannessi D (2010) A methodological reappraisal of total and high molecular weight adiponectin determination in human peripheral circulation: comparison of four immunometric assays. Clin Chem Lab Med 48(4):561–568. https://doi.org/10.1515/CCLM.2010.104
Ellulu M, Abed Y, Rahmat A, Ranneh Y, Ali F (2014) Epidemiology of obesity in developing countries: challenges and prevention. Global Epid Obes 2(1):2. https://doi.org/10.7243/2052-5966-2-2
Ellulu MS, Patimah I, Khaza’ai H, Rahmat A, Abed Y (2017) Obesity and inflammation: the linking mechanism and the complications. Archiv Med Sci : AMS 13(4):851–863. https://doi.org/10.5114/aoms.2016.58928
Feldstein AE, Werneburg NW, Canbay A, Guicciardi ME, Bronk SF, Rydzewski R, Burgart LJ, Gores GJ (2004) Free fatty acids promote hepatic lipotoxicity by stimulating tnf-alpha expression via a lysosomal pathway. Hepatology (Baltimore, Md.) 40(1):185–194. https://doi.org/10.1002/hep.20283
Fossati P, Prencipe L (1982) Serum triglycerides determined colorimetrically with an enzyme that produces hydrogen peroxide. Clin Chem 28(10):2077–2080
Pino-de la Fuente F, Quezada L, Sepúlveda C, Monsalves-Alvarez M, Rodríguez JM, Sacristan C, Chiong M, Llanos M, Espinosa A, Troncoso R (2019) Exercise Regulates Lipid Droplet Dynamics in Normal and Fatty Liver. Biochim biophys acta mol cell biol lipids 1864(12):158519. https://doi.org/10.1016/j.bbalip.2019.158519
George MY, Esmat A, Tadros MG, El-Demerdash E (2018) In vivo cellular and molecular gastroprotective mechanisms of chrysin; emphasis on oxidative stress, inflammation and angiogenesis. Eur J Pharmacol 818(January):486–498. https://doi.org/10.1016/j.ejphar.2017.11.008
Hasanvand A, Amini-Khoei H, Hadian M-R, Abdollahi A, Tavangar SM, Dehpour AR, Semiei E, Mehr SE (2016) Anti-inflammatory effect of ampk signaling pathway in rat model of diabetic neuropathy. Inflammopharmacology 24(5):207–219. https://doi.org/10.1007/s10787-016-0275-2
Hashida R, Nakano D, Yamamura S, Kawaguchi T, Tsutsumi T, Matsuse H, Takahashi H, Gerber L, Younossi ZM, Torimura T (2021) Association between activity and brain-derived neurotrophic factor in patients with non-alcoholic fatty liver disease: a data-mining analysis. Life (Basel, Switzerland) 11(8). https://doi.org/10.3390/life11080799
He J, Zhang P, Shen L, Niu L, Tan Y, Chen L, Zhao Y et al (2020) Short-Chain Fatty Acids and Their Association with Signalling Pathways in Inflammation, Glucose and Lipid Metabolism. Int J Mol Sci 21(17). https://doi.org/10.3390/ijms21176356
Hirsch C, Zouain CS, Alves JB, Goes AM (1997) Induction of protective immunity and modulation of granulomatous hypersensitivity in mice using piii, an anionic fraction of schistosoma mansoni adult worm. Parasitology 115(Pt 1 (July):21–28. https://doi.org/10.1017/s0031182097001078
Hosogai N, Fukuhara A, Oshima K, Miyata Y, Tanaka S, Segawa K, Furukawa S et al (2007) Adipose tissue hypoxia in obesity and its impact on adipocytokine dysregulation. Diabetes 56(4):901–911. https://doi.org/10.2337/db06-0911
Huang Y, Li Y, Liu Q, Zhang J, Zhang Z, Tong W, Tang Q et al (2020) Telmisartan attenuates obesity-induced insulin resistance via suppression of AMPK mediated ER stress. Biochem Biophys Res Commun 523(3):787–794. https://doi.org/10.1016/j.bbrc.2019.12.111
Huber G, Ogrodnik M, Wenzel J, Stölting I, Huber L, Will O, Peschke E et al (2021) Telmisartan prevents high-fat diet-induced neurovascular impairments and reduces anxiety-like behavior. J Cereb Blood Flow Metabolism: Official J Int Soc Cereb Blood Flow Metabolism 41(9):2356–2369. https://doi.org/10.1177/0271678X211003497
Hwang Y-J, Cho D-H (2020) Activation of AMPK/Proteasome/MLCK degradation signaling axis by telmisartan inhibits VSMC contractility and vessel contraction. Biochem Biophys Res Commun 524(4):853–860. https://doi.org/10.1016/j.bbrc.2020.02.007
Iwase M, Kimura K, Komagome R, Sasaki N, Ishioka K, Honjoh T, Saito M (2000) Sandwich enzyme-linked immunosorbent assay of canine leptin. J Vet Med Sci 62(2):207–209. https://doi.org/10.1292/jvms.62.207
Jayapriya B, Thamilarasi S, Shanthi M, and Lourdu Jafrin A. 2013. ‘Effect of Telmisartan on Blood Pressure and Lipid Profile in Hypertensive Patients with Dyslipidemia’. In
Jeon EJ, Kim DY, Lee NH, Choi HE, Cheon HG (2019) Telmisartan induces browning of fully differentiated white adipocytes via M2 macrophage polarization. Sci Rep 9(1):1–15. https://doi.org/10.1038/s41598-018-38399-1
Kernie SG, Liebl DJ, Parada LF (2000) BDNF regulates eating behavior and locomotor activity in mice. EMBO J 19(6):1290–1300. https://doi.org/10.1093/emboj/19.6.1290
Kim E, Lim S-M, Kim M-S, Yoo S-H, Kim Y (2017) Phyllodulcin, a natural sweetener, regulates obesity-related metabolic changes and fat browning-related genes of subcutaneous white adipose tissue in high-fat diet-induced obese mice. Nutrients 9(10). https://doi.org/10.3390/nu9101049
Kintscher U, Peter Bramlage W, Paar D, Thoenes M, Unger T (2007) Irbesartan for the treatment of hypertension in patients with the metabolic syndrome: a sub analysis of the treat to target post authorization survey. prospective observational, two armed study in 14,200 patients. Cardiovasc Diabetol 6(April):12. https://doi.org/10.1186/1475-2840-6-12
Kittigul L, Temprom W, Sujirarat D, Kittigul C (2000) Determination of Tumor necrosis factor-alpha levels in dengue virus infected patients by sensitive biotin-streptavidin enzyme-linked immunosorbent assay. J Virol Methods 90(1):51–57. https://doi.org/10.1016/s0166-0934(00)00215-9
Kořínková L, Pražienková V, Černá L, Karnošová A, Železná B, Kuneš J, Maletínská L (2020) Pathophysiology of NAFLD and NASH in experimental models: the role of food intake regulating peptides. Front Endocrinol 11:597583. https://doi.org/10.3389/fendo.2020.597583
Lasar D, Rosenwald M, Kiehlmann E, Balaz M, Tall B, Opitz L, Lidell ME et al (2018) Peroxisome proliferator activated receptor gamma controls mature brown adipocyte inducibility through glycerol kinase. Cell Rep 22(3):760–773. https://doi.org/10.1016/j.celrep.2017.12.067
Lauby-Secretan B, Scoccianti C, Loomis D, Grosse Y, Bianchini F, Straif K (2016) Body fatness and cancer--viewpoint of the IARC working group. N Engl J Med 375(8):794–798. https://doi.org/10.1056/NEJMsr1606602
Filho L, Baldoino M, Morandin RC, de Almeida AR, Cambiucci EC, Metze K, Borges G, Gontijo JAR (2005) Hemodynamic parameters and neurogenic pulmonary edema following spinal cord injury: an experimental model. Arq Neuropsiquiatr 63(4):990–996. https://doi.org/10.1590/s0004-282x2005000600016
Lenart L, Balogh DB, Lenart N, Barczi A, Hosszu A, Farkas T, Hodrea J et al (2019) Novel therapeutic potential of angiotensin receptor 1 blockade in a rat model of diabetes-associated depression parallels altered BDNF signalling. Diabetologia 62(8):1501–1513. https://doi.org/10.1007/s00125-019-4888-z
Li H, Li M, Liu P, Wang YP, Zhang H, Li HB, Yang SF et al (2016) Telmisartan ameliorates nephropathy in metabolic syndrome by reducing leptin release from perirenal adipose tissue. Hypertension (Dallas, Tex : 1979) 68(2):478–490. https://doi.org/10.1161/HYPERTENSIONAHA.116.07008
Liou C-J, Lee Y-K, Ting N-C, Chen Y-L, Shen S-C, Shu-Ju W, Huang W-C (2019) Protective effects of licochalcone a ameliorates obesity and non-alcoholic fatty liver disease via promotion of the sirt-1/AMPK Pathway in mice fed a high-fat diet. Cells 8(5). https://doi.org/10.3390/cells8050447
Ma W, Ao S, Zhou J, Li J, Liang X, Yang X, Zhang H et al (2022) Methylsulfonylmethane protects against lethal dose MRSA-induced Sepsis through promoting M2 macrophage polarization. Mol Immunol 146(June):69–77. https://doi.org/10.1016/j.molimm.2022.04.001
Makita S, Abiko A, Naganuma Y, Moriai Y, Nakamura M (2008) Effects of telmisartan on adiponectin levels and body weight in hypertensive patients with glucose intolerance. Metab Clin Exp 57(10):1473–1478. https://doi.org/10.1016/j.metabol.2008.05.019
Marques MB, Ribeiro-Oliveira AJ, Guimarães J, Nascimento GF, Anjos AP, Vilas-Boas WW, Santos RAS et al (2012) Modifications in basal and stress-induced hypothalamic AMP-activated protein kinase (AMPK) activity in rats chronically treated with an angiotensin II receptor blocker. Stress (Amsterdam, Netherlands) 15(5):554–561. https://doi.org/10.3109/10253890.2011.648673
Mathai ML, Naik S, Sinclair AJ, Weisinger HS, Weisinger RS (2008) Selective reduction in body fat mass and plasma leptin induced by angiotensin-converting enzyme inhibition in rats. Int J Obes (2005) 32(10):1576–1584. https://doi.org/10.1038/ijo.2008.126
Mathai ML, Chen N, Cornall L, Weisinger RS (2011) the role of angiotensin in obesity and metabolic disease. Endocr Metab Immune Disord Drug Targets 11(3):198–205. https://doi.org/10.2174/187153011796429853
Meredith PA (2007) Candesartan cilexetil--a review of effects on cardiovascular complications in hypertension and chronic heart failure. Curr Med Res Opin 23(7):1693–1705. https://doi.org/10.1185/030079907x210723
Mukundan L, Odegaard JI, Morel CR, Heredia JE, Mwangi JW, Ricardo-Gonzalez RR, Sharon Goh YP et al (2009) PPAR-delta senses and orchestrates clearance of apoptotic cells to promote tolerance. Nat Med 15(11):1266–1272. https://doi.org/10.1038/nm.2048
Mullen KL, Pritchard J, Ritchie I, Snook LA, Chabowski A, Bonen A, Wright D, Dyck DJ (2009) Adiponectin resistance precedes the accumulation of skeletal muscle lipids and insulin resistance in high-fat-fed rats. Am J Physiol Regul Integr Comp Physiol 296(2):R243–R251. https://doi.org/10.1152/ajpregu.90774.2008
Murad HA, Gazzaz ZJ, Ali SS, Ibraheem MS (2017) Candesartan, rather than losartan, improves motor dysfunction in thioacetamide-induced chronic liver failure in rats. Brazilian J Me Research = Revista Brasileira de Pesquisas Medicas e Biolog 50(11):e6665. https://doi.org/10.1590/1414-431X20176665
Nguyen KD, Qiu Y, Xiaojin Cui YP, Goh S, Mwangi J, David T, Mukundan L, Brombacher F, Locksley RM, Chawla A (2011) Alternatively activated macrophages produce catecholamines to sustain adaptive thermogenesis. Nature 480(7375):104–108. https://doi.org/10.1038/nature10653
Nishida Y, Takahashi Y, Nakayama T, Soma M, Asai S (2011) comparative effect of olmesartan and candesartan on lipid metabolism and renal function in patients with hypertension: a retrospective observational study. Cardiovasc Diabetol 10:1–7. https://doi.org/10.1186/1475-2840-10-74
Ola MS, Ahmed MM, Abuohashish HM, Al-Rejaie SS, Alhomida AS (2013) Telmisartan ameliorates neurotrophic support and oxidative stress in the retina of streptozotocin-induced diabetic rats. Neurochem Res 38(8):1572–1579. https://doi.org/10.1007/s11064-013-1058-4
Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM (1998) A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 92(6):829–839. https://doi.org/10.1016/s0092-8674(00)81410-5
Rabie MA, Mohsen M, Ibrahim M, Mahmoud RE-S (2014) Serum level of brain derived neurotrophic factor (bdnf) among patients with bipolar disorder. J Affect Disord 162(June):67–72. https://doi.org/10.1016/j.jad.2014.02.038
Rahn SB (2001) Liver biopsy interpretation in chronic hepatitis. J Insuran Med (New York, N.Y.) 33(1):110–113
Rawish E, Nickel L, Schuster F, Stölting I, Frydrychowicz A, Saar K, Hübner N, Othman A, Kuerschner L, Raasch W (2020) Telmisartan prevents development of obesity and normalizes hypothalamic lipid droplets. J Endocrinol 244(1):95–110. https://doi.org/10.1530/JOE-19-0319
Ribeiro-Oliveira AJ, Marques MB, Vilas-Boas WW, Guimarães J, Coimbra CC, Anjos AP, Fóscolo RB et al (2015) The effects of chronic candesartan treatment on cardiac and hepatic adenosine monophosphate-activated protein kinase in rats submitted to surgical stress. J Renin-Angio-Aldoste Syst : JRAAS 16(3):481–487. https://doi.org/10.1177/1470320313499199
Rosendahl A, Thomsen LH (2015) Polarization of macrophages in metabolic diseases. J Clin Cell Immunol 06(02). https://doi.org/10.4172/2155-9899.1000313
Rosenwald M, Wolfrum C (2014) The origin and definition of brite versus white and classical brown adipocytes. Adipocyte 3(1):4–9. https://doi.org/10.4161/adip.26232
Schuster F, Huber G, Stölting I, Wing EE, Saar K, Hübner N, Banks WA, Raasch W (2018) Telmisartan prevents diet-induced obesity and preserves leptin transport across the blood-brain barrier in high-fat diet-fed mice. Arch Eur J Physiol 470(11):1673–1689. https://doi.org/10.1007/s00424-018-2178-0
Sha H, Jingyue X, Tang J, Ding J, Gong J, Ge X, Kong D, Gao X (2007) Disruption of a novel regulatory locus results in decreased bdnf expression, obesity, and type 2 diabetes in mice. Physiol Genomics 31(2):252–263. https://doi.org/10.1152/physiolgenomics.00093.2007
Sun C, Mao S, Chen S, Zhang W, Liu C (2021) PPARs-Orchestrated Metabolic Homeostasis in the Adipose Tissue. Int J Mol Sci 22(16). https://doi.org/10.3390/ijms22168974
Szasz G (1969) a kinetic photometric method for serum gamma-glutamyl transpeptidase. Clin Chem 15(2):124–136
Takahashi Y, Fukusato T (2014) Histopathology of nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. World J Gastroenterol 20(42):15539–15548. https://doi.org/10.3748/wjg.v20.i42.15539
Tan Y, Kim J, Cheng J, Ong M, Lao W-G, Jin X-L, Lin Y-G, Xiao L, Zhu X-Q, Xian-Qin Q (2017) Green tea polyphenols ameliorate non-alcoholic fatty liver disease through upregulating ampk activation in high fat fed zucker fatty rats. World J Gastroenterol 23(21):3805–3814. https://doi.org/10.3748/wjg.v23.i21.3805
Vanitha M, Vijayal R (2013) Effect of telmisartan on serum lipid profile in patients with hypertension and dyslipidemia. Int J Med Res Health Sci 2(4):745. https://doi.org/10.5958/j.2319-5886.2.4.119
Wang C-S, Chang T-T, Yao W-J, Wang S-T, Chou P (2012) Impact of increasing alanine aminotransferase levels within normal range on incident diabetes. J Formosan Med Assoc = Taiwan Yi Zhi 111(4):201–208. https://doi.org/10.1016/j.jfma.2011.04.004
Wei Y, Sowers JR, Clark SE, Li W, Ferrario CM, Stump CS (2008) Angiotensin II-induced skeletal muscle insulin resistance mediated by NF-KappaB activation via NADPH oxidase. Am J Physiol Endocrinol Metab 294(2):E345–E351. https://doi.org/10.1152/ajpendo.00456.2007
Wohl P, Krusinová E, Hill M, Kratochvílová S, Zídková K, Kopecký J, Neskudla T et al (2010) Effect of telmisartan on selected adipokines, insulin sensitivity, and substrate utilization during insulin-stimulated conditions in patients with metabolic syndrome and impaired fasting glucose. Eur J Endocrinol 163(4):573–583. https://doi.org/10.1530/EJE-10-0436
Yumuk VD (2006) Targeting components of the stress system as potential therapies for the metabolic syndrome: the peroxisome-proliferator-activated receptors. Ann N Y Acad Sci 1083(November):306–318. https://doi.org/10.1196/annals.1367.019
Zhou J, Ho C-T, Long P, Meng Q, Zhang L, Wan X (2019) Preventive efficiency of green tea and its components on nonalcoholic fatty liver disease. J Agric Food Chem 67(19):5306–5317. https://doi.org/10.1021/acs.jafc.8b05032
Funding
Non
Author information
Authors and Affiliations
Contributions
Noha A.T. Abbas: Conceptualization, experimental design, writing original draft, reviewing, and editing. Fawkia A. Fayed: Supervision, Visualization, reviewing, and editing. Rabab Saber El. Sebaey: Investigation, resources, and data curation. Heba A. Hassan: formal analysis, writing original draft, visualization, reviewing, and editing. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethical approval
The procedures in this experimental study were performed according to the National Guidelines for the Care and Use of Laboratory Animals and approved by the Institutional Animal Care and Use Committee, Zagazig university (ZU-IACUC) with approval no (ZU-IACUC/3/F/159/2021), Duration of approval (27-10-2021) to (27-10-2024).
Consent to participate
Non
Consent to publish
Non
Competing interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abbas, N.A., Fayed, F.A., El Sebaey, R.S. et al. Telmisartan and candesartan promote browning of white adipose tissue and reverse fatty liver changes in high fat diet fed male albino rats. Naunyn-Schmiedeberg's Arch Pharmacol 397, 2359–2378 (2024). https://doi.org/10.1007/s00210-023-02771-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02771-4