Abstract
Inflammation is one of the characteristic features during the development of human tumors. A pro-inflammatory cytokine that is known to promote inflammation during cancer development is the transforming growth factor-β (TGF-β). On the other hand, demethylzeylasteral (T-96) is a natural compound isolated from Tripterygium wilfordii Hook F, which has been reported to have various pharmacological properties including anti-inflammatory and immunosuppressive activities. We investigated the effects of T-96 on the highly metastatic breast cancer cell line, MDA-MB-231. Cell migration was assessed by scratch-wound migration assay, and the molecular mechanisms underlying the effects of T-96 were examined by qPCR and Western blot analyses. We also investigated the suppression effects of T-96 on the pulmonary metastasis in the 4T1 mouse model. T-96 inhibited TGF-β-induced migration and epithelial–mesenchymal transition both in vitro and in vivo. These results demonstrate that T-96 inhibited invasion of MDA-MB-231 and 4T1 cells via suppressing the canonical and non-canonical TGF-β signaling pathways.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is by far the most frequently diagnosed malignant cancers and the second leading cause of cancer deaths in women in the Western world (Weigelt et al. 2005). Metastasis of cancer cells from the primary sites to distant locations is the primary reason for cancer-associated morbidity and mortality in patients with breast cancer. However, the mechanisms that trigger and regulate this process remain poorly understood (Piccolo et al. 2013; Rhodes et al. 2014). Advancements in the early detection and application of adjuvant therapy have reduced disease-related mortality with a 5-year survival rate of 98% in women with localized breast cancer. Unfortunately, women diagnosed with metastasis have a reduced survival rate of only 27% (Yang et al. 2011). Triple-negative breast cancer (TNBC), which is pathologically defined as estrogen receptor–, progesterone receptor–, and HER2–negative disease, accounts for 12–17% of all breast cancers (Foulkes et al. 2010). Despite general improvements in the management of breast cancer, TNBC represents a continuing challenge and is difficult to treat. Compared with other subtypes, it is associated with a higher frequency of recurrence, shorter disease-free survival, the highest rates of metastatic disease, and the poorest overall survival (Al-Mahmood et al. 2018; Lee and Djamgoz 2018). Therefore, targeting the different stages of metastasis, including local tumor cell invasion, entry into the blood circulatory or lymphatic systems followed by the exit of carcinoma cells from the circulation as well as breach basement membranes in distant organs could serve as specific biological targets for anti-metastatic therapeutics in breast cancer treatment (van Zijl et al. 2011; Kim et al. 2014).
Inflammation, mediated by immune cells, is a cellular response to infection and injury by noxious stimuli and has been implicated in the pathogenesis of arthritis, stroke, neurodegenerative, and cardiovascular diseases (Ricciotti and FitzGerald 2011; Thompson et al. 2015). Recent studies have demonstrated the role of inflammation in triggering breast cancer cells to spread to other organs, or metastasize (Gallego-Ortega et al. 2015; Hugo et al. 2015; Qian et al. 2015). Various inflammation-associated cytokines, such as transforming growth factor-β (TGF-β), play a central role in cancer metastasis. TGF-β is known to work as both a tumor suppressor and a tumor promoter. In early-stage tumors, levels of TGF-β, which promote cell cycle arrest and apoptosis, are positively associated with a favorable prognosis (Tian et al. 2011; Drabsch and ten Dijke 2012). While at advanced stages, they are positively associated with tumor aggressiveness and poor prognosis through promoting cancer cell motility, invasion, epithelial-to-mesenchymal transition (EMT), and cell stemness (Tian et al. 2011; Drabsch and ten Dijke 2012). TGF-β is constitutively expressed in metastasizing breast carcinoma and has been long considered as a key mediator for metastasis into other tissues during breast cancer progression (Bai et al. 2014; Xue et al. 2014). TGF-β mediates its effects on cells through two transmembrane proteins known as the type I (TGFBR1) and type II (TGFBR2) receptors, which are serine–threonine kinases that phosphorylate a variety of downstream signaling proteins after activation (Grady 2005).
The best characteristic of these downstream signaling proteins is the Smad family of proteins, which consists of the members of Smad1, Smad2, Smad3, and Smad5 (Grady 2005). Some studies have provided evidence for the Smad3 pathway–mediated pro-metastatic activities of breast cancer cells in bone metastasis (Petersen et al. 2010). Besides the canonical pathway, TGF-β signaling also activates SMAD-independent pathways such as PI3K/AKT as well as NF-κB, Rho/Rac1, Cdc42, FAK, Src, Abl (Neuzillet et al. 2014). In addition, protein kinases, including mitogen-activated protein kinases (MAPKs) and ERK1/2, are known to be downstream effectors of TGF-β and also play important roles in breast cancer metastasis (Gomes et al. 2012; Pang et al. 2016). TGF-β is also known to trigger epithelial-to-mesenchymal transition (EMT) (Xu et al. 2009; Yu et al. 2014; Pang et al. 2016), which is considered as a key event in tumor metastasis whereby epithelial cell layers lose polarity and cell–cell contact that disrupt cell adherence and subsequently undergo a dramatic rearrangement of the cytoskeleton or matrix (Larue and Bellacosa 2005; Martin et al. 2010; Sun et al. 2010). Therefore, inhibiting the TGF-β signaling pathway may be an effective strategy to block breast cancer metastasis.
Demethylzeylasteral (T-96), extracted from the Tripterygium wilfordii Hook F whole plant or peeled wood parts, is reported to possess a broad range of pharmacological activities such as antifertility, estrogen metabolism regulation, immunosuppressive effects, and immune system modulation (Bai et al. 2003; Zhao et al. 2012; Hu et al. 2015). In the present study, we used MDA-MB-231, a highly metastatic breast cancer cell line derived from mammary glands, a well-validated model to explore the effects of T-96 in inhibiting the TGF-β-induced invasion and other changes associated with EMT. Also, we investigated whether T-96 inhibits TGF-β-induced changes in EMT markers via downregulation of the Smad signaling cascade. In addition, we also analyzed whether T-96 inhibits matrix degradation, migration, and invasion of breast cancer cells. This is the first report on the in vitro anti-metastatic potentials of T-96 and provides a scientific basis for its application in therapeutic intervention against tumor procession.
Materials and methods
Cell culture
MDA-MB-231 and 4T1 cell lines were acquired from the China Infrastructure of Cell Line Resources and grown in DMEM medium supplemented with 10% fetal bovine serum (FBS), 100 U/ml penicillin, and 100 mg/ml streptomycin at 37 °C in a humidified incubator with 5% CO2.
Scratch-wound migration assays
For scratch-wound assays, MDA-MB-231 cells were seeded in 6-well plates and cultured to reach 80% confluence. Then, the cells were divided into five groups and treated as follows: control (no treatment), TGF-β (5 ng/ml), TGF-β (5 ng/ml) + T-96 (5 μM), TGF-β (5 ng/ml) + T-96 (10 μM), TGF-β (5 ng/ml) + SB431542 (10 μM). SB431542 is a specific and selective inhibitor of TGF-β as a positive control. Each well then received one scratch to simulate wounding using a 200-μl pipette tip and four images of each well were taken by observing through a microscope (Nikon, Japan).
qPCR assay
Total RNA was extracted using Trizol (Thermo, USA) according to the manufacturer’s instructions, and the first strand cDNA was synthesized by using M-MLV (Promega, USA) after DNase treatment (Promega, USA). Real-time PCR was performed using the SYBR Green PCR Mix Kit (Thermo, USA). Specific primers used in the reactions were the following: α-SMA: sense, 5′-CCGACCGAATGCAGAAG GA-3′, and antisense, 5′-ACAGAGTATTTGCGCTCCGAA-3′ (Knerr et al. 2001); vimentin: sense, 5′-GACAATGCGTCTCTGGCACGTCTT-3′, and antisense, 5′-TCCTCCGCCTCCTGCAGGTTCTT-3′; fibronectin: sense, 5′-TCGCCATCAGTAGAAGGTAGCA-3′, and antisense, 5′-TGTTATACTGAACACCAGGTTGCA-3′ (Kawata et al. 2012); and GAPDH: sense, 5′-GAAGGTGAAGGTCGGAGT-3′, and antisense, 5′-GAAGATGGTGATGGGATTTC-3′ (Goldberg et al. 2007). The PCR reaction conditions consisted of heating at 95 °C for 5 min and then subjecting to 40 cycles of denaturation at 95 °C for 15 s, annealing at 60 °C for 40 s, and extension at 60 °C for 60 s. Relative gene expression was then calculated using the formula 2(−ΔΔCt).
Isolation of nuclear protein
The nuclear protein was isolated using a Nuclear and Cytoplasmic Protein Extraction Kit (Beyotime, China) and following the detailed instructions provided.
Western blot analysis
Treated cells were lysed with RIPA Buffer (Keygentec, China) containing the Halt protease inhibitor Cocktail (Roche, China). Proteins in lysates were separated by electrophoresis and transferred onto PVDF membranes, blocked previously with 5% skim milk, followed by incubations with different primary antibodies (Abcam, USA). Then, the membranes were washed with phosphate-buffered saline Tween (PBST; 0.05% Tween) and incubated with the second antibody. After washing with PBST again, membranes were incubated with the ECL Western Blotting Detection Reagent (Thermo, USA). Finally, signals were detected and analyzed using a Tanon Western Blot 6100 (Tanon, China).
Immunofluorescence assay
Cells were washed three times with ice-cold phosphate-buffered saline (PBS), fixed in 100% methanol, permeabilized with 0.1% Triton-X-100, blocked with 5% BSA-PBS, incubated with primary antibodies at room temperature, and washed three times with PBS before incubating with secondary antibodies for 40 min. Lastly, cells were mounted with Vectashield (Vector Laboratories, USA) and visualized through a fluorescence microscope. Image J was used for densitometric analysis of the target proteins.
Animal study design
Fourteen female BALB/c mice (8–10 weeks) were purchased from Joint Ventures Sipper BK Experimental Animal (Shanghai, China). All animal experiments were performed in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals with the approval of the Nanjing University of Chinese Medicine, Nanjing. All animals were maintained under the standard light–dark cycle (12 h light and 12 h dark) with ad libitum access to food and water. 4T1 cells (1 × 106) in 100 μl PBS were injected into the mammary fat pads of female BALB/c mice. The tumor size was measured using calipers. When the tumor volume reached 200 mm3, mice were randomly divided into two groups. T-96 (4 mg/kg/day) or vehicle (1% DMSO) in PBS was intraperitoneally injected into the two mice groups once a day, and tumor diameters are measured by digital calipers, and the tumor volume is calculated by the formula: volume = (width)2 × length / 2. The animals were sacrificed when they were moribund (35 days after 4T1 cell injection). Tumors and lungs were removed, measured, and analyzed.
Statistical analysis
Data are presented as means ± SD. Student’s t test was used to analyze the statistical significance of the differences between multiple samples. P < 0.05 was considered statistically significant.
Results
T-96-induced changes in MDA-MB-231 morphology
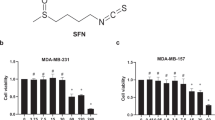
We observed morphological changes on the third day after treatment of the human breast cancer cell line, MDA-MB-231 with TGF-β and T-96, and cell morphology became much more spindle when we only treat the cell with TGF-β, but we treated MDA-MB-231 cells with T-96, and the morphology changed from the spindle shape to a rounded or cobblestone shape in Fig. 1b.
T-96 induces morphology change and decreased the cellular migration. a Chemical structure of T-96. b T-96 induces MDA-MB-231 morphology change. c, d The migration was evaluated by scratch-wound migration assays. This is a representative experiment of three performed in an independent way. *P < 0.05 compared with the control
T-96 inhibits MDA-MB-231 migration in vitro
To screen for effectors to prevent cancer metastasis, we examined the inhibitory effects of T-96 on the migration of MDA-MB-231 cells by scratch-wound migration assays. MDA-MB-231 cells were cultured with 5 or 10 μM of T-96 and TGF-β. The results demonstrated that T-96 significantly reduced the migration of MDA-MB-231 cells. Quantification of cell migration indicated that treatment of MDA-MB-231 cells with 5 or 10 μM of T-96 reduced migration by 31.2% and 50.9%, respectively (Fig. 1b, c).
T-96 inhibits the expression of EMT-related genes and proteins
EMT is a process involved in cancer invasion and metastasis. During EMT, epithelial cells lose their cell polarity and cell–cell adhesion and become mesenchymal cells (Lamouille et al. 2014). We investigated whether T-96 regulates EMT-related vimentin, fibronectin, and α-SMA expression. For this, we extracted both total RNA and proteins after treating MDA-MB-231 cells with T-96 and TGF-β for 24 h. We observed that the mRNA and protein expression of vimentin, fibronectin, and α-SMA increased after treatment with TGF-β (Fig. 2a, b). In contrast, TGF-β-induced mRNA and protein expression of vimentin, fibronectin, and α-SMA were reversed by T-96 (Fig. 2a, b, c). Furthermore, we used immunofluorescence to detect the protein expression of vimentin. Consistent with the Western blot results, immunofluorescence also showed that TGF-β-induced vimentin protein expression was abolished by T-96 treatment (Fig. 2d). Thus, these results revealed that T-96 downregulates the levels of vimentin, fibronectin, and α-SMA expression through the inhibition of TGF-β signaling pathway in MDA-MB-231 cells.
T-96 inhibits EMT-related gene and protein expression in different groups as indicated. a T-96 inhibits EMT-related gene expression by qPCR. b, c T-96 inhibits EMT-related protein expression by Western blotting. d T-96 inhibits Vimentin expression by immunofluorescence. This is a representative experiment of three performed in an independent way. *P < 0.05 compared with the control
T-96 inhibits canonical-TGF-β signaling pathway
SMADs, which are intracellular proteins, include the receptor-regulated Smads (R-SMAD), the common-mediator Smad (co-SMAD), and the antagonistic or inhibitory Smads (I-SMAD). They form a trimer of two receptor-regulated SMADs and one co-SMAD. It has been reported that SMADs transduce extracellular signals from TGF-β to the nucleus and act as transcription factors that regulate the expression of certain genes. Our results revealed that TGF-β significantly induced the phosphorylation of SMAD2/3 (Fig. 3a, b). In contrast, the phosphorylation levels of SMAD2/3 were decreased after treatment with 5 μM or 10 μM T-96. SMAD4, which is the only co-SMAD, interacts with SMAD2/3 to participate in signaling (Shi et al. 1997). Here, we observed that T-96 did not influence the protein expression of SMAD4, but decreased its nuclear translocation (Fig. 3c, d).
T-96 inhibits canonical-TGF-β signaling pathway as indicated. a, b T-96 inhibits the phosphorylation of the Smad2/3 by Western blotting. c, d T-96 inhibits the nucleus translocation of SMAD4. This is a representative experiment of three performed in an independent way. *P < 0.05 compared with the control
T-96 inhibits non-canonical TGF-β signaling pathway
Several studies have suggested that TGF-β can rapidly activate PI3K, which is indicated by the phosphorylation of its downstream effectors, Akt and mTOR (Zhang 2009). Thus, we studied the effect of T-96 on TGF-β-induced phosphorylation of PI3K/AKT/mTOR cell signal in MDA-MB-231 breast cancer cells. We found that T-96 inhibited the activation of this signal pathway (Fig. 4a, b). In breast cancer cells, TGF-β also led to a rapid increase in the activation of the ERK signal (Frey and Mulder 1997). In this study, we found that TGF-β-induced phosphorylation of ERK1/2 was affected by T-96 treatment (Fig. 4c, d).
T-96 inhibits pulmonary metastasis in the 4T1 mouse model
To examine if T-96 can suppress pulmonary metastasis of breast cancer cells, we established a breast cancer mouse model by injecting 4T1 cells. Control mice were injected intraperitoneally with vehicle (1% DMSO) and the experimental mice were injected T-96 (4 mg/kg) in 1 ml PBS once a day. The tumor growth was followed for 35 days when the mice were sacrificed to remove the tumors and lungs for analysis. In the 4T1 model, T-96 treatment inhibited the growth of primary tumor (Fig. 5a, b, c) and significantly reduced cancer lung metastasis (Fig. 5d). Also, we found that T-96 significantly suppressed vimentin and α-SMA protein expression in 4T1 mice (Fig. 5e, f).
T-96 inhibits lung metastasis in BALB/c mice injected with 4T1 cells. a Tumor growth curve of 4T1. b, c Tumor weight was measured at the 35th day. d The number of tumor nodules in the lungs was measured at the 35th day. e, f T-96 inhibits EMT-related protein expression by Western blotting in mice. The treatment group is compared with the model groups. Values are statistically significant at *P < 0.05
Discussion
Triple-negative breast cancers (TNBC), which typically lack the estrogen receptor (ER) and progesterone receptor (PR) and exhibit downregulated expression of human epidermal growth factor receptor 2 (HER2), are associated with poor prognosis, due to aggressive proliferation and metastasis, lack of effective clinical therapeutic options, and only partial response to chemotherapy.
MDA-MB-231 used in this study is a TNBC cell line (Tate et al. 2012). We found that T-96 suppresses the invasion of MDA-MB-231 that displays mesenchymal landscapes by reversing EMT to mesenchymal-to-epithelial transition (MET). This entire event is likely mediated via regulation of the TGF-β signaling pathway, which could trigger the molecular reprogramming switch in breast cancer cells.
EMT is a de-differentiation program, and epithelial cells lose their apical–basal polarity and intercellular adhesive property and gain mesenchymal properties that include fibroblast-like morphology and increased motility (Singh and Settleman 2010; Wang et al. 2016). EMT also bestows tumor cells with an associated therapeutic resistance (Singh and Settleman 2010; Davis et al. 2014; Wang et al. 2016). In this study, we found that T-96 could decrease the mRNA and protein expression of vimentin, fibronectin, and α-SMA. These proteins are important markers of EMT and are the primary regulators of cancer cell migration.
TGF-β is one of the key pro-inflammatory cytokines that regulates the EMT process (Moustakas and Heldin 2014). The activities of TGF-β are dysregulated during malignant tumor progression that eventually triggers a cascade of signaling events and downstream effector proteins through canonical and non-canonical signaling pathways. Moreover, the abnormal activation status of canonical and non-canonical signaling pathway may well underlie the ability of TGF-β to induce EMT, and promote pathophysiological EMT in malignant cells (Wendt et al. 2009).
The Smads protein–dependent signal pathway is the canonical TGF-β signaling pathway, which involves intracellular effectors and translocates to the nucleus to regulate transcription. For example, TGF-β-induced EMT is reported to occur through phosphorylation of Smad2/3 (Valcourt et al. 2005; Xue et al. 2014). We found that T-96 inhibits the phosphorylation of Smad2/3 and increases SMAD2/3 protein accumulation in the cell nucleus as evidenced by Western blotting results. SMAD4, a member of SMAD protein family, interacts with other members of the SMAD protein family such as SMAD2 or SMAD3. Aberrant expression of SMAD4 is more common with a prevalence of 61 to 92% in breast cancer specimens (Xie et al. 2002; Vincent et al. 2009; Tram et al. 2011). In comparison with the surrounding unaffected tissues, SMAD4 mRNA expression level was significantly elevated in human breast tumor tissue (Vincent et al. 2009; Tram et al. 2011). We found that T-96 did not influence the protein expression of SMAD4, but decreased SMAD4 protein translocation into the cell nucleus. Therefore, T-96 could directly inhibit the phosphorylation of Smad2/3, but could not increase the negative feedback loops of the TGF-β signaling pathway.
Apart from the SMAD2/3-dependent cell signal pathway, TGF-β also activates non-Smad signaling pathways such as MAPK, Rho-like GTPase, and PI3K/AKT cell signal pathways (Zhang 2009). Some studies have shown that the activation of PI3K/Akt and ERK signaling pathways can promote EMT in different types of malignancies (Ha et al. 2013; Buonato and Lazzara 2014; Baquero et al. 2015). In this study, we found that TGF-β promotes the phosphorylation of PI3K/AKT/mTOR and ERK signal pathways. These results are consistent with previous reports (Han et al. 2015; Li et al. 2015; Baek et al. 2016). However, the phosphorylation of PI3K/AKT and ERK was suppressed by T-96.
n addition, we used a common 4T1 murine model to study breast cancer lung metastasis to further investigate whether T-96 has any anti-metastasis effect. As expected, T-96 decreased metastatic nodules in the lungs and suppressed vimentin and α-SMA expression in primary 4T1 breast cancer tissues.
Conclusion
In conclusion, our results indicated that T-96 can significantly inhibit EMT-related protein synthesis and metastasis and/or EMT effects in the breast cancer cell line, MDA-MB-231. Molecular analysis clearly revealed that T-96 suppresses TGF-β-induced phosphorylation of SMAD2/3, PI3K/AKT/mTOR, and ERK (Fig. 6). These results suggest for the first time that T-96 may negatively regulate TGF-β signaling through canonical and non-canonical pathways.
References
Al-Mahmood S, Sapiezynski J, Garbuzenko OB, Minko T (2018) Metastatic and triple-negative breast cancer: challenges and treatment options. Drug Deliv Transl Res
Baek SH, Ko JH, Lee JH, Kim C, Lee H, Nam D, Lee J, Lee SG, Yang WM, Um JY, Sethi G, Ahn KS (2016) Ginkgolic acid inhibits invasion and migration and TGF-beta-induced EMT of lung cancer cells through PI3K/Akt/mTOR inactivation. J Cell Physiol 232(2):346–354
Bai JP, Shi YL, Fang X, Shi QX (2003) Effects of demethylzeylasteral and celastrol on spermatogenic cell Ca2+ channels and progesterone-induced sperm acrosome reaction. Eur J Pharmacol 464:9–15
Bai WD, Ye XM, Zhang MY, Zhu HY, Xi WJ, Huang X, Zhao J, Gu B, Zheng GX, Yang AG, Jia LT (2014) MiR-200c suppresses TGF-beta signaling and counteracts trastuzumab resistance and metastasis by targeting ZNF217 and ZEB1 in breast cancer. Int J Cancer 135:1356–1368
Baquero P, Jimenez-Mora E, Santos A, Lasa M, Chiloeches A (2015) TGFbeta induces epithelial-mesenchymal transition of thyroid cancer cells by both the BRAF/MEK/ERK and Src/FAK pathways. Mol Carcinog 55(11):1639–1654
Buonato JM, Lazzara MJ (2014) ERK1/2 blockade prevents epithelial-mesenchymal transition in lung cancer cells and promotes their sensitivity to EGFR inhibition. Cancer Res 74:309–319
Davis FM, Stewart TA, Thompson EW, Monteith GR (2014) Targeting EMT in cancer: opportunities for pharmacological intervention. Trends Pharmacol Sci 35:479–488
Drabsch Y, ten Dijke P (2012) TGF-beta signalling and its role in cancer progression and metastasis. Cancer Metastasis Rev 31:553–568
Foulkes WD, Smith IE, Reis-Filho JS (2010) Triple-negative breast cancer. N Engl J Med 363:1938–1948
Frey RS, Mulder KM (1997) TGFbeta regulation of mitogen-activated protein kinases in human breast cancer cells. Cancer Lett 117:41–50
Gallego-Ortega D, Ledger A, Roden DL, Law AM, Magenau A, Kikhtyak Z, Cho C, Allerdice SL, Lee HJ, Valdes-Mora F, Herrmann D, Salomon R, Young AI, Lee BY, Sergio CM, Kaplan W, Piggin C, Conway JR, Rabinovich B, Millar EK, Oakes SR, Chtanova T, Swarbrick A, Naylor MJ, O'Toole S, Green AR, Timpson P, Gee JM, Ellis IO, Clark SJ, Ormandy CJ (2015) ELF5 drives lung metastasis in luminal breast cancer through recruitment of Gr1+ CD11b+ myeloid-derived suppressor cells. PLoS Biol 13:e1002330
Goldberg MT, Han YP, Yan C, Shaw MC, Garner WL (2007) TNF-alpha suppresses alpha-smooth muscle actin expression in human dermal fibroblasts: an implication for abnormal wound healing. J Invest Dermatol 127:2645–2655
Gomes LR, Terra LF, Wailemann RA, Labriola L, Sogayar MC (2012) TGF-beta1 modulates the homeostasis between MMPs and MMP inhibitors through p38 MAPK and ERK1/2 in highly invasive breast cancer cells. BMC Cancer 12:26
Grady WM (2005) Transforming growth factor-beta, Smads, and cancer. Clin Cancer Res 11:3151–3154
Ha GH, Park JS, Breuer EK (2013) TACC3 promotes epithelial-mesenchymal transition (EMT) through the activation of PI3K/Akt and ERK signaling pathways. Cancer Lett 332:63–73
Han D, Wu G, Chang C, Zhu F, Xiao Y, Li Q, Zhang T, Zhang L (2015) Disulfiram inhibits TGF-beta-induced epithelial-mesenchymal transition and stem-like features in breast cancer via ERK/NF-kappaB/Snail pathway. Oncotarget 6:40907–40919
Hu Q, Yang C, Wang Q, Zeng H, Qin W (2015) Demethylzeylasteral (T-96) treatment ameliorates mice lupus nephritis accompanied by inhibiting activation of NF-kappaB pathway. PLoS One 10:e0133724
Hugo HJ, Saunders C, Ramsay RG, Thompson EW (2015) New insights on COX-2 in chronic inflammation driving breast cancer growth and metastasis. J Mammary Gland Biol Neoplasia 20:109–119
Kawata M, Koinuma D, Ogami T, Umezawa K, Iwata C, Watabe T, Miyazono K (2012) TGF-beta-induced epithelial-mesenchymal transition of A549 lung adenocarcinoma cells is enhanced by pro-inflammatory cytokines derived from RAW 264.7 macrophage cells. J Biochem 151:205–216
Kim S, Yao J, Suyama K, Qian X, Qian BZ, Bandyopadhyay S, Loudig O, De Leon-Rodriguez C, Zhou ZN, Segall J, Macian F, Norton L, Hazan RB (2014) Slug promotes survival during metastasis through suppression of Puma-mediated apoptosis. Cancer Res 74:3695–3706
Knerr I, Dittrich K, Miller J, Kummer W, Rosch W, Weidner W, Rascher W (2001) Alteration of neuronal and endothelial nitric oxide synthase and neuropeptide Y in congenital ureteropelvic junction obstruction. Urol Res 29:134–140
Lamouille S, Xu J, Derynck R (2014) Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol 15:178–196
Larue L, Bellacosa A (2005) Epithelial-mesenchymal transition in development and cancer: role of phosphatidylinositol 3′ kinase/AKT pathways. Oncogene 24:7443–7454
Lee A, Djamgoz MBA (2018) Triple negative breast cancer: emerging therapeutic modalities and novel combination therapies. Cancer Treat Rev 62:110–122
Li Y, Jia L, Liu C, Gong Y, Ren D, Wang N, Zhang X, Zhao Y (2015) Axl as a downstream effector of TGF-beta1 via PI3K/Akt-PAK1 signaling pathway promotes tumor invasion and chemoresistance in breast carcinoma. Tumour Biol 36:1115–1127
Martin FT, Dwyer RM, Kelly J, Khan S, Murphy JM, Curran C, Miller N, Hennessy E, Dockery P, Barry FP, O'Brien T, Kerin MJ (2010) Potential role of mesenchymal stem cells (MSCs) in the breast tumour microenvironment: stimulation of epithelial to mesenchymal transition (EMT). Breast Cancer Res Treat 124:317–326
Moustakas A, Heldin P (2014) TGFbeta and matrix-regulated epithelial to mesenchymal transition. Biochim Biophys Acta 8:18
Neuzillet C, de Gramont A, Tijeras-Raballand A, de Mestier L, Cros J, Faivre S, Raymond E (2014) Perspectives of TGF-beta inhibition in pancreatic and hepatocellular carcinomas. Oncotarget 5:78–94
Pang MF, Georgoudaki AM, Lambut L, Johansson J, Tabor V, Hagikura K, Jin Y, Jansson M, Alexander JS, Nelson CM, Jakobsson L, Betsholtz C, Sund M, Karlsson MC, Fuxe J (2016) TGF-beta1-induced EMT promotes targeted migration of breast cancer cells through the lymphatic system by the activation of CCR7/CCL21-mediated chemotaxis. Oncogene 35:748–760
Petersen M, Pardali E, van der Horst G, Cheung H, van den Hoogen C, van der Pluijm G, Ten Dijke P (2010) Smad2 and Smad3 have opposing roles in breast cancer bone metastasis by differentially affecting tumor angiogenesis. Oncogene 29:1351–1361
Piccolo S, Enzo E, Montagner M (2013) p63, Sharp1, and HIFs: master regulators of metastasis in triple-negative breast cancer. Cancer Res 73:4978–4981
Qian BZ, Zhang H, Li J, He T, Yeo EJ, Soong DY, Carragher NO, Munro A, Chang A, Bresnick AR, Lang RA, Pollard JW (2015) FLT1 signaling in metastasis-associated macrophages activates an inflammatory signature that promotes breast cancer metastasis. J Exp Med 212:1433–1448
Rhodes LV, Tate CR, Segar HC, Burks HE, Phamduy TB, Hoang V, Elliott S, Gilliam D, Pounder FN, Anbalagan M, Chrisey DB, Rowan BG, Burow ME, Collins-Burow BM (2014) Suppression of triple-negative breast cancer metastasis by pan-DAC inhibitor panobinostat via inhibition of ZEB family of EMT master regulators. Breast Cancer Res Treat 145:593–604
Ricciotti E, FitzGerald GA (2011) Prostaglandins and inflammation. Arterioscler Thromb Vasc Biol 31:986–1000
Shi Y, Hata A, Lo RS, Massague J, Pavletich NP (1997) A structural basis for mutational inactivation of the tumour suppressor Smad4. Nature 388:87–93
Singh A, Settleman J (2010) EMT, cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene 29:4741–4751
Sun T, Zhao N, Zhao XL, Gu Q, Zhang SW, Che N, Wang XH, Du J, Liu YX, Sun BC (2010) Expression and functional significance of Twist1 in hepatocellular carcinoma: its role in vasculogenic mimicry. Hepatology 51:545–556
Tate CR, Rhodes LV, Segar HC, Driver JL, Pounder FN, Burow ME, Collins-Burow BM (2012) Targeting triple-negative breast cancer cells with the histone deacetylase inhibitor panobinostat. Breast Cancer Res 14:R79
Thompson PA, Khatami M, Baglole CJ, Sun J, Harris SA, Moon EY, Al-Mulla F, Al-Temaimi R, Brown DG, Colacci A, Mondello C, Raju J, Ryan EP, Woodrick J, Scovassi AI, Singh N, Vaccari M, Roy R, Forte S, Memeo L, Salem HK, Amedei A, Hamid RA, Lowe L, Guarnieri T, Bisson WH (2015) Environmental immune disruptors, inflammation and cancer risk. Carcinogenesis 36(Suppl 1):S232–S253
Tian M, Neil JR, Schiemann WP (2011) Transforming growth factor-beta and the hallmarks of cancer. Cell Signal 23:951–962
Tram E, Ibrahim-Zada I, Briollais L, Knight JA, Andrulis IL, Ozcelik H (2011) Identification of germline alterations of the mad homology 2 domain of SMAD3 and SMAD4 from the Ontario site of the breast cancer family registry (CFR). Breast Cancer Res 13:R77
Valcourt U, Kowanetz M, Niimi H, Heldin CH, Moustakas A (2005) TGF-beta and the Smad signaling pathway support transcriptomic reprogramming during epithelial-mesenchymal cell transition. Mol Biol Cell 16:1987–2002
van Zijl F, Krupitza G, Mikulits W (2011) Initial steps of metastasis: cell invasion and endothelial transmigration. Mutat Res 728:23–34
Vincent T, Neve EP, Johnson JR, Kukalev A, Rojo F, Albanell J, Pietras K, Virtanen I, Philipson L, Leopold PL, Crystal RG, de Herreros AG, Moustakas A, Pettersson RF, Fuxe J (2009) A SNAIL1-SMAD3/4 transcriptional repressor complex promotes TGF-beta mediated epithelial-mesenchymal transition. Nat Cell Biol 11:943–950
Wang Y, Liu J, Ying X, Lin PC, Zhou BP (2016) Twist-mediated epithelial-mesenchymal transition promotes breast tumor cell invasion via inhibition of hippo pathway. Sci Rep 6:24606
Weigelt B, Peterse JL, van ‘t Veer LJ (2005) Breast cancer metastasis: markers and models. Nat Rev Cancer 5:591–602
Wendt MK, Allington TM, Schiemann WP (2009) Mechanisms of the epithelial-mesenchymal transition by TGF-beta. Future Oncol 5:1145–1168
Xie W, Mertens JC, Reiss DJ, Rimm DL, Camp RL, Haffty BG, Reiss M (2002) Alterations of Smad signaling in human breast carcinoma are associated with poor outcome: a tissue microarray study. Cancer Res 62:497–505
Xu J, Lamouille S, Derynck R (2009) TGF-beta-induced epithelial to mesenchymal transition. Cell Res 19:156–172
Xue J, Lin X, Chiu WT, Chen YH, Yu G, Liu M, Feng XH, Sawaya R, Medema RH, Hung MC, Huang S (2014) Sustained activation of SMAD3/SMAD4 by FOXM1 promotes TGF-beta-dependent cancer metastasis. J Clin Invest 124:564–579
Yang HL, Kuo YH, Tsai CT, Huang YT, Chen SC, Chang HW, Lin E, Lin WH, Hseu YC (2011) Anti-metastatic activities of Antrodia camphorata against human breast cancer cells mediated through suppression of the MAPK signaling pathway. Food Chem Toxicol 49:290–298
Yu Y, Xiao CH, Tan LD, Wang QS, Li XQ, Feng YM (2014) Cancer-associated fibroblasts induce epithelial-mesenchymal transition of breast cancer cells through paracrine TGF-beta signalling. Br J Cancer 110:724–732
Zhang YE (2009) Non-Smad pathways in TGF-beta signaling. Cell Res 19:128–139
Zhao JW, Wang GH, Chen M, Cheng LH, Ji XQ (2012) Demethylzeylasteral exhibits strong inhibition towards UDP-glucuronosyltransferase (UGT) 1A6 and 2B7. Molecules 17:9469–9475
Author contribution
C Xu, H Cheng, and M Wu conceived and designed the research. L Liu, Y Ji, and J Fan conducted the experiments. L Li and Y Ji analyzed the data. F Li and Y Li contributed to the revision. C Xu wrote the manuscript. All the authors read and approved the manuscript.
Funding
This work was financially supported by the Young Scientists Fund of the National Natural Science Foundation of China (No. 81302829, 81500530, 81500544), Fund of the National Natural Science Foundation of China (No. 81373511, 81573910), and a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All animal experiments were performed in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals with the approval of the Nanjing University of Chinese Medicine, Nanjing.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, L., Ji, Y., Fan, J. et al. Demethylzeylasteral (T-96) inhibits triple-negative breast cancer invasion by blocking the canonical and non-canonical TGF-β signaling pathways. Naunyn-Schmiedeberg's Arch Pharmacol 392, 593–603 (2019). https://doi.org/10.1007/s00210-019-01614-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-019-01614-5