Abstract
A fundamental metabolic feature of cancerous tissues is high glucose consumption. The rate of glucose consumption in a cancer cell can be 10–15 times higher than in normal cells. Isolation and cultivation of tumor cells in vitro highlight properties that are associated with intensive glucose utilization, the presence of minimal oxidative metabolism, an increase in lactate concentrations in the culture medium and a reduced rate of oxygen consumption. Although glycolysis is suggested as a general feature of malignant cells and recently identified as a possible contributing factor to tumor progression, several studies highlight distinct metabolic characteristics in some tumors, including a relative decrease in avidity compared to glucose and/or a glutamine dependency of lactate and even proliferative tumor cells. The aim of this review is to determine the particularities in the energy metabolism of cancer cells, focusing on the main nutritional substrates, such as glucose and glutamine, evaluating lactate dehydrogenase as a potential marker of malignancy and estimating activators and inhibitors in cancer treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Every aerobic organism relies primarily on the Krebs cycle, also called TCA (tricarboxylic acid) cycle or citric acid cycle (terms used here indifferently). The Krebs cycle is composed of a series of chemical reactions occurring in the matrix of mitochondria that permit the aerobic organisms to oxidize the biochemical compounds and extract the energy from them. It is the last or concluding metabolic pathway for the lipids, carbohydrates, and amino acids, and it is a well-known and essential biological mechanism for the energy supply, constituting the most vital metabolic pathway that connects almost all the other individual metabolic pathways (Akram 2014). The TCA cycle also provides some macromolecules and redox balance to the aerobic cells. The energy required for cell reproduction and survival usually comes from two sources. The first is glycolysis and the second is the TCA cycle. Both processes are involved in respiration, glycolysis occurring first in the cytoplasmic region, while TCA is the other process that occurs in mitochondria in eukaryotes (in prokaryotes occurs in the cytosol). These processes aim at satisfying the energy requirement of the organism. In glycolysis, a series of reactions metabolize glucose or glycogen into pyruvate to produce two ATP molecules from each glucose molecule. Glycolysis is essential for the brain, whose energy depends on the availability of glucose. It is considered the preferred mode of energy production in different cells, such as anoxic, embryonic or intensively proliferating ones. During the TCA cycle, pyruvate from glycolysis is used to produce 36–38 ATP molecules from each glucose molecule. Two carbon atoms are oxidized to carbon dioxide, whose oxidation energy is coupled with guanosine triphosphate (GTP) or adenosine triphosphate (ATP) synthesis, the dissociation of which will subsequently transfer energy to other metabolic processes that require it. The steps of the TCA cycle are depicted in Fig. 1, whereas its detailed description is performed in the successive Krebs cycle Pathways section.
Krebs cycle synthesizes flavin adenine dinucleotide reduced form (FADH2) and nicotinamide adenine dinucleotide reduced form (NADH) from the acetyl-coenzyme A (acetyl-CoA) compound that is a product that is formed from the decarboxylation of pyruvate (Fernie et al. 2004). NADH generates the huge reductive potential that is essential for the generation of ATP from the mitochondrial respiratory chain complexes (electron transport chain, ETC) during the oxidative phosphorylation (OxPhos) (Fig. 2) (Devin and Rigoulet 2007).
Respiratory chain complexes or electron transport chain (ETC) in mammals. ETC comprises four enzyme complexes (complexes I–IV), two intermediary substrates, coenzyme Q (CoQ) and cytochrome C (CytC), and ATP synthase that, within the electron/proton pathways generate ATP at the matrix side of mitochondria. The NADH, H+, and FADH2 produced by the intermediate metabolism (TCA cycle) are oxidized further by the mitochondrial respiratory chain to establish an electrochemical gradient of protons, which is finally used by the F0F1-ATP synthase (complex V) to produce ATP. The redox systems of glutathione (GSH) are responsible for intrinsic antioxidative defense in mitochondria for preventing cellular damage mediated by ROS
Within mitochondria, during the OxPhos process, the complete degradation of glucose to CO2 and H2O occurs only in the presence of oxygen. Aerobic glycolysis will generate 36–38 molecules of ATP. In the absence of oxygen, as in muscle tissue during intense physical exercise, pyruvate is no longer used in the Krebs cycle and is converted to lactic acid by a process called anaerobic glycolysis (Jiang 2017). The latter is accompanied by the generation of only two ATP molecules.
Throughout ETC, electron leaks can occur and lead to the formation of reactive oxygen species (ROS) and other free radicals. Therefore, this mechanism, although is a universal process among the entire aerobic species, can become dangerous due to the production of an uncontrolled amount of reactive oxidative species. These species further oxidize the cell macromolecules, such as proteins, lipids, and can even cause damage to nucleic acids with harmful consequences within the body (Wei and Lee 2002). A defense mechanism against the ROS exists, and it is the fundamental component of all the aerobic species present on this earth, including humans (Halliwell 1999). Still, it is necessary to evaluate if the Krebs cycle, the major producer of the pro-oxidants, such as NADH, can control intracellularly free radicals production. Attenuation of the TCA cycle can decrease the production of pro-oxidants like NADH, and ultimately this strategy can control the production of reactive oxidative species (Mailloux et al. 2007).
Krebs cycle pathways
Krebs cycle is the main metabolic pathway, in supplying energy to the body, generating about 70% of the ATP. Table 1 shows the main types of reactions involved in the cycle (Krebs and Johnson 1980; Nelson et al. 2017). The oxidation pathways of fatty acids, glucose, amino acids, acetate, ketone bodies generate acetyl-CoA, which constitutes its substrate (Fig. 1).
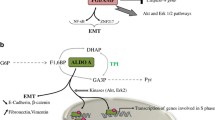
Citric acid, also known as citrate, is formed from oxaloacetate through its aldol condensation. Oxaloacetate is regenerated at the end of each TCA cycle; therefore the cycle continues. The latter comes from the glucose molecule through the glycolysis pathway and then enters into the mitochondria in the form of pyruvate; it can also come from the oxidation of fatty acids during β-oxidation. Within the TCA cycle, citrate is then transformed into isocitrate through the aconitase enzyme (ACO2). Isocitrate dehydrogenase (IDH) will subsequently transform isocitrate into α-ketoglutarate (αKG) during a decarboxylation chemical reaction. In this way, the TCA cycle keeps going on and gives a chief source of cellular ATP and also reducing equivalents that give the electron to ETC (Akram 2014). The mitochondrial citrate carrier (CiC), also referred to as solute carrier family 25 member 1 (Slc25a1), can export citrate from the mitochondria in interchange for malate. In the mitochondrial cytosol, the citrate undergoes breakdown by ATP-Citrate lyase (ACLY) and form acetyl-CoA and oxaloacetate. Oxaloacetate can be transformed into malate by the help of malate dehydrogenase (MDH), which can re-enter the mitochondria with the help of mitochondrial CiC (Palmieri 2004). Acetyl-CoA can be further converted into the malonyl-coenzyme A (malonyl-CoA) with the help of acetyl-CoA carboxylase (ACC). Malonyl-CoA can be incorporated into fatty acids and cholesterol molecules (Saggerson 2008). Fatty acids are used to form phospholipids as in the composition of cellular membranes. Malonyl-CoA can also decrease the β-oxidation of fatty acids since its high amount can prevent carnitine palmitoyltransferase 1 (CPT1) (Paumen et al. 1997). Two forms of ACC exist. One is known as ACC1 that maintains regulation of fatty acid synthesis, and the other one, known as ACC2, which mainly regulates fatty acid oxidation (Brownsey et al. 1997). ACC2 is connected to the outer membrane of mitochondria and can control the levels of malonyl-CoA in the proximity of CPT1 and also regulate its action (Saggerson 2008). Acetyl-CoA is capable of performing a variety of functions in the biochemical processes of cells (Pietrocola et al. 2015). It is not only essential for de novo fatty acid synthesis, but it is also an important cofactor for the acetylation of histone and non-histone proteins, thus playing a role in tuning the expression of several key regulatory enzymes. Citrate, as acetyl supplier, and CiC, as cytosolic citrate transporter, show, therefore, a critical role in epigenetic modifications (Choudhary et al. 2014; Mews et al. 2017).
Citrate inhibits diverse major enzymes used in the glycolytic pathway as a part of a negative feedback loop (Williams and O’Neill 2018). The enzymes utilized in glycolysis, known as phosphofructokinase (PFK) 1 and 2, are directly inhibited by citrate, and another enzyme, known as pyruvate kinase (PK), is indirectly inhibited by it. Citrate is also responsible for decreasing the level of fructose-1,6-bisphosphate that is a known activator of PK (Yalcin et al. 2009). Therefore, citrate is responsible for inhibiting the pathways that synthesize ATP and triggers those that consume it. Citrate also allosterically regulates ACC and the fructose-1, 6-bisphosphate that is a gluconeogenic enzyme (Iacobazzi and Infantino 2014). Citrate is associated with different cellular and metabolic processes comprising those significant for the immune system (Williams and O’Neill 2018). In murine M1 macrophages, increased isocitrate and αKG ratio has been observed along with a reduced level of isocitrate dehydrogenase (IDH1), the enzyme that interconverts these two metabolites, leading to metabolic TCA cycle breakpoint (Jha et al. 2015). The same processes have also been observed in dendritic cells (DCs) (Everts et al. 2014). With the increased glycolytic pathway flux in both macrophages and DCs, pyruvate obtained from glucose goes into the Krebs cycle, but it certainly cannot reach the previous citrate and isocitrate levels. In human macrophages, the export and mitochondrial citrate breakdowns are associated with the synthesis of many essential pro-inflammatory molecules, like nitric oxide (NO), ROS, and a few prostaglandins (Infantino et al. 2013). Prevention of CiC function and its genetic silencing through the small interfering RNA (siRNA), will lead to a substantial decrease in NO, ROS and prostaglandin synthesis in lipopolysaccharides and macrophages that are stimulated to release cytokines. A study has shown that the reduction in prostaglandin synthesis is due to the reduced availability of its precursor molecules, since the addition of exogenous acetate regulates the inhibition activity of CiC on prostaglandin synthesis. Acetate can be transformed into acetyl-CoA synthase (ACSS) (Starai and Escalante-Semerena 2004). In the same way, citrate is also considered vital for the stimulation of DCs.
Energy metabolism of cancer cell
Abnormal functioning of the TCA cycle can result in various pathological conditions in the human body (Anderson et al. 2018). Numerous studies have shown that malignant cells need much smaller amounts of oxygen to grow and that their metabolism follows an anaerobic pattern that leads to lactic acid production. This phenomenon known as the ‘Warburg Effect’, is observed even in the presence of completely functioning mitochondria. It increases cell acidity, lowers oxygen supply, and causes cancer-specific DNA changes (Liberti and Locasale 2016; Vander Heiden et al. 2009). Paradoxically, even in the presence of oxygen, cancer cells prefer to behave as if they were in anaerobic conditions: they avoid the Krebs cycle and therefore have a very reduced efficiency. They quickly consume large amounts of glucose, resulting in only two ATP molecules and lactic acid. In this way, they produce a very acidic environment around them and kill healthy cells. Malignant tumors are characterized by an increased metabolism that conditions a rapid growth which is determined not only by the synthesis of membrane compounds and ribonucleic acids, but also by an intense generation of energy.
Energy metabolism of the tumor is a feature common to all types of cancer cells, from the early stages of transformation to tumor invasion and metastasis, both in vitro and in vivo (Bartkova et al. 2006). Energy metabolism, and in particular glucose metabolism, depends on several factors, both genetic that will dictate the degree and type of expression of the major enzymes involved in glycolysis, as well as external factors, such as glucose concentration in the extracellular environment.
Higher proliferation cells have a lower NADH level, which confirms that anabolic processes are predominant in cancer, and the amount of energy generated is sufficient to maintain transmembrane ion gradients, macromolecule synthesis, counteract oxidative stress, or support to other energy-dependent processes. NADH is an energy-equivalent, mainly produced by the enzymatic activity of aerobic glycolysis dehydrogenases. If the oxygen level in the tissues is low, especially in tumors with high proliferation, the phenomenon of hypoxia and imperfect vascularization determines the transition to predominantly glycolytic energy metabolism. The oxidized glucose in the cells comes either from the degradation of the own glycogen or from the blood that infuses the respective tissue. Cancer cells continue to produce up to 80% of ATP in the process of glycolysis, a phenomenon that occurs in the presence of oxygen, as opposed to proliferating cells that are in hypoxic environments (Diaz-Ruiz et al. 2011). This persistent phenomenon characterized as a hallmark of cancer cell metabolism. Otto Warburg tried to explain the phenomenon of aerobic glycolysis by the appearance of mitochondrial dysfunctions. He considers that two phases characterize the birth of cancer cells from normal cells: the appearance of irreversible damage in the breathing process, and substitution of breath by fermentation (Devic 2016). The factors that lead to the disturbance of tissue respiration are carcinogens, radiation, hypoxia, inflammation, viruses, mutations, oncogenes, and age. The result is a reduction in the number of mitochondria, crystolysis, and disturbance of OxPhos processes. The fermentation process compensates the insufficiency of energy. Only the cell having the fermentation capacity becoming a cancer cell, as this is the only source of energy acquisition required for the activity of the Na+/K+-ATPase pumps. The hypothesis of a causal involvement of mitochondrial dysfunction and glycolysis in the oncogenesis process was, subsequently, abandoned in favor of the genetic theory of cancer occurrence following the involvement of genetic mutations and, in particular, tumor suppressor genes. Mitochondrial biology and aerobic glycolysis or Warburg’s phenomenon are in the focus, when mitochondria are involved in apoptosis or programmed cell death (Vallee and Vallee 2018). The role of the Warburg effect in tumor progression is not fully known (Liberti and Locasale 2016). An adaptive role to an energy substrate deficiency in a rapidly proliferating cell population has been proposed. One of the causes is also the lack of vascular network or angiogenesis, which could meet the rapid growth needs of the tumor. The diffusion capacity of glucose much higher than that of oxygen in tissues has also been postulated, as a possible cause of the transition from oxidative to glycolytic metabolism (Hao et al. 2010). In addition, the role of acidification by the lactate generation of intensive glycolysis could have a toxic effect on normal cells, which also induces invasion and metastasis. Currently, apoptosis inhibition is one of the hallmarks of tumor cells. Apoptosis presents a programmed death mechanism of cancer cells dependent on the cell’s energy reserves. Glycolysis would be one of the important mechanisms, implemented by the tumor tissue, which would allow it to continue to proliferate under conditions of energy stress (evidenced by the high absorption of 2-deoxy-d-glucose) (Zhang et al. 2014). A second evidence, which supports the hypothesis of increased glycolysis, is the increase in lactate concentration. Cancer cell metabolism is associated with marked lactate production and secretion in peritumoral tissues. In tissues, where glucose uptake is not disturbed, the increased level of lactate as the end product of glucose conversion to pyruvate and subsequently lactate under anaerobic conditions reflects, first and foremost, an increased level of glycolysis. Lactate is the rejected product of glycolysis and is normally absent in healthy tissue under normobaric conditions and satisfactory tissue oxygenation conditions (Stepien et al. 2016). In an extracellular environment, there is an intense acidification, mainly due to lactic acid (lactate), which represents a percentage of 80% (the other 20% are the result of CO2 accumulation, generated in the TCA cycle). As stated, some authors attribute this tumor acidity the role of an aggressive factor in tumor invasion (Waniewski and Martin 1998). In recent years, in the specialist literature, the hallmark of the energy metabolism of the tumor, according to the most common hypothesis, is the predominance of glycolytic metabolism over OxPhos (Burns and Manda 2017).
It is obvious that in both normal and cancer cells, the latter being characterized by an undirected proliferation, different mechanisms are evoked to overcome the energy crisis. Some of the mechanisms that may be involved in cancer cell metabolism are angiogenesis and autophagy. Angiogenesis, the process of capillary sprouting from preexisting vessels, is a complex process crucial for sustained primary tumor growth, metastases acceleration and cancer progression. Angiogenesis inhibitors could enable improvements in tumor suppression (Cook and Figg 2010). Autophagy, through its catabolic role, allows recycling of metabolites of dysfunctional organs or useless compounds and contributes, at the same time, to the survival of tumor cells (Isidoro et al. 2005; Kimmelman and White 2017). Thus, autophagy serves as a protective effect for cells, providing an alternative source of nutrients. However, loss of the gene that initiates the autophagy process (Beclin 1), is frequently revealed in tumor tissues, which paradoxically would give it the role as a cell-fate decision machinery (Fei et al. 2016; Kang et al. 2011).
Krebs cycle activators
Glutamine
Cell growth and survival need various metabolic pathways that generate energy, precursor molecules for the production of basic cellular macromolecules, and substrates for other vital activities. Glucose and glutamine are the two most important and abundant nutrients that support the cellular functions mentioned above leading to increase the growth and survival of the cells. Glutamine acts as an interorgan shuttle for nitrogen and carbon; it is also the chief source of nitrogen for the production of nonessential amino acids, hexosamines and nucleotides as well. In culture media, glutamine represents a high percentage of carbon and nitrogen metabolism (Yang et al. 2014). The citric acid cycle assists in ATP synthesis. The pathways start with the condensation reaction of oxaloacetate and acetyl-CoA to synthesize citrate. Further oxidation of citrate produces the reducing equivalents to trigger the ATP synthesis through the OxPhos process. In this pathway, two carbons are released in the form of CO2 per cycle and regenerate oxaloacetate. In cancerous cells, the Krebs cycle works differently. Under such circumstances, oxaloacetate will become a limiting intermediate unless it was provided by another source that certainly did not come from the mitochondrial citrate. Such metabolic pathways that produce oxaloacetate is known as anaplerosis. They are responsible for enabling the Krebs cycle to function as a metabolic pathway that produces ATP (Owen et al. 2002). In the standard culture, several cancer cells utilize a form of Krebs cycle in which acetyl-CoA is generated from glutamine. Glutamine first transforms into glutamate with the help of glutaminase (GLS). This enzyme helps the glutamine to release the amide nitrogen in the form of ammonia. Glutamate is then transformed into a TCA cycle intermediate αKG with the help of two kinds of reactions. In the first reaction, transaminases are responsible for transferring the amino group from glutamate to a keto acid, synthesizing αKG and amino acid. On the other reaction, glutamate undergoes deamination with the help of glutamate dehydrogenases (GDH), releasing ammonia and synthesizing αKG without consuming a keto acid. GLS and transaminases are majorly needed for the growth and survival of glutamine-addicted cells (Yang et al. 2014). Hydrogen peroxide-mediated oxidative stress impairs mitochondrial function. It has been reported that ACO2 is the most sensitive enzyme to the inhibition by H2O2 in the TCA cycle. However, at the concentrations in which ACO2 is inactivated (< 50 µM), glutamate supplies the TCA cycle and NADH generation resulted unaltered. If H2O2 concentration increases (more than twofold), inhibition of α-KT dehydrogenase occurs and limits the amount of NADH available for the ETC. In the early stage of an H2O2-induced oxidative stress, glutamate could be used in a TCA cycle as a metabolite to maintain NADH production (Gibson et al. 1998; Tretter and Adam-Vizi 2000). Studies showed that this glutamate in the body available from glutamine plays also a role in the inhibition of consumption of oxygen caused by H2O2 and NO. This role is only specific for glutamine, not for the other amino acids. The function of glutamine in the prevention of oxygen consumption in sepsis that is caused by the ROS is mediated through the help of glutathione.
Glutathione
When there is a limitation of NADH production to maintain the Krebs cycle, glutathione (GSH) can act as an activator of the Krebs cycle. GSH itself is an antioxidant enzyme that scavenges the ROS and overcomes the oxidative stress to maintain the metabolic pathways in the cell (Fig. 2) (Babu et al. 2001). S-glutathionylation reactions control mitochondrial metabolism and function in response to fluctuations in redox environment (Mailloux et al. 2014). These reactions are performed under oxidative stress conditions or in some cases even under normal conditions (Mailloux and Willmore 2014). Glutathionylation reaction occurs when there is a high concentration of glutathione disulfide (GSSG), in particular in the high oxidative stress condition. Other reactions of GSH also exist that are non-enzymatic and non-specific and totally linked with some pathologies like cardiovascular diseases, neurodegeneration, etc. and linked with oxidative stress (Bjørklund et al. 2020a, b, 2021). In short, GSH can overcome ROS and maintains the cellular microenvironment suitable for producing energy through cellular metabolic pathways (Mailloux et al. 2013). GSH depletion leads to positive regulation of antioxidant genes, many of which are under the Nrf2 (nuclear factor erythroid 2-related factor 2) control. Since this transcription factor binds to the antioxidant responsive element (ARE), a new and indirect approach to cancer therapy is to modulate the Nrf2-ARE metabolic pathway. An effective strategy, for increasing the sensitivity of cancer cells to chemotherapeutic drugs, would be to inhibit Nrf2 and Keap1 (Kelch-Like ECH-Associated Protein 1) interaction. Keap1 is a cysteine-rich protein that tightly interacts with Nrf2 and is encoded by the tumor and metastasis suppressor Keap1gene (Lignitto et al. 2019). Nrf2 shows a dual role in tumorigenesis: a protective activity in the early stages of tumorigenesis, but negative in the later stages. The enhancement of Nrf2 activity remains an important approach in cancer prevention while its inhibition appears to be a valid approach for its treatment. The identification of inhibitors or modulators of the Keap1–Nrf2 pathway is a key topic of current research (Lu et al. 2016). One potential target for redox chemotherapy is heme oxygenase-1 (HO-1). HO-1 inhibitors, including zinc protorporfirine and several soluble derivatives (PEG-ZnPP), have been used successfully to improve cancer cell chemosensitization (Roh et al. 2017).
Iron
Iron has an essential role in the physiology of mammals (Zoroddu et al. 2019). Iron-proteins are crucial components in tissues, for the synthesis of DNA, for oxygen delivery and electron transport. Though iron is an important nutrient, it can also be toxic in high amounts. High levels of iron can induce the generation of toxic oxygen metabolites, which can be dangerous to cells leading their damage. Such conditions can be seen in some diseases, such as hemochromatosis, in which excess amounts of iron can lead to the impairment of tissues due to the oxidative stress damage (Fleming and Ponka 2012; Kang et al. 1998). Organisms have adapted a few regulatory mechanisms to control levels of iron that are actually required for body needs. In advanced eukaryotes, there are iron regulatory proteins (IRPs) that sense iron concentration and post-transcriptionally regulate the expression and translation of iron-related genes to maintain cellular iron homeostasis. However, in case of iron overload, due to the lack of efficient regulatory mechanisms for its excretion in humans, chelation treatment is required. This clinical practice helps to remove iron ions from the body or to mitigate the toxicity by transforming them into less toxic compounds or through their dislocation from the site where they exert a toxic action (Nurchi et al. 2016). Deficiency of iron can also cause many complications in the body (Lachowicz et al. 2014). Several studies showed that deficiency of iron would disturb the synthesis of ACO2 enzyme essential for the regulation of the TCA cycle. The downregulation of ACO2 can limit the capacity of the TCA cycle to synthesize ATP (Ross and Eisenstein 2002). Iron is also essential for the oxidation of acetyl-CoA through the TCA cycle and, ultimately for the synthesis of ATP through OxPhos. For this reason, iron acts as an activator for the Krebs cycle (Oexle et al. 1999). In addition, iron is a cofactor for many enzymes used in the metabolic processes of the cell, such as heme and iron–sulfur clusters (ISCs), and is also required in the form of co-factor for the production of amino acids, sterols, proteins, and even fatty acids. These iron co-factors are vital for the repair mechanism of DNA and the metabolism of xenobiotics as well. It has been observed that during the iron deficiency, many transcripts that encode ISC proteins taking part in glutamate and lipoic acid production are down-regulated (Shakoury-Elizeh et al. 2010). To prevent nutritional iron deficiency, the role of oral iron supplementation has been recommended (Lachowicz et al. 2014).
NADH
NADH is found in the body naturally, and it performs a function in the process of generating energy for cells. NADH has many vital roles in metabolism. Its oxidized form serves as a coenzyme in redox reactions of metabolism. It acts as a secondary messenger molecule. In eukaryotes, the electrons are carried through the NADH. These electrons are produced in the cytoplasm and then transferred into the mitochondria to reduce the mitochondrial NAD+ through the shuttles of mitochondria, e.g. malate-aspartate shuttle. The NADH present in the mitochondria is further oxidized in turn through the electron transport chain which pumps protons across the membrane and synthesizes ATP via OxPhos (Rich 2003). NADH supplements are also available and are used to improve mental clarity, concentration, alertness, and memory (Fricker et al. 2018; Lautrup et al. 2019). Since oral supplementation of NAD+ precursors has been shown to improve synaptic plasticity and neuronal morphology in mouse models of Alzheimer’s disease (AD), it has also been tested for human treatment with promising results (Demarin et al. 2004; Hou et al. 2018). Hydrogen peroxide (H2O2) is responsible for the inhibition of Krebs cycle enzymes and this can limit the production of NADH. The most sensitive enzyme of the Krebs cycle is ACO2; it can be easily affect by oxidative stress or H2O2 in the TCA cycle. Inhibition of α-KGDH by oxidants is responsible for the lower amounts of NADH in the cellular respiration process. When there is a severe exposure of nerve terminals to H2O2, then glutamate acts as an alternative compound. As already mentioned, the synthesis of NADH is therefore maintained in the TCA cycle (Tretter and Adam-Vizi 2000). During the H2O2-induced oxidative stress, the glutamate is possible to be transformed into αKG. When there is a high level of oxidative stress, this phenomenon will rescue a part of the TCA cycle, then ACO2 is completely impaired but α-KGDH is still active and ready to function (the production of αKG from the citrate is restricted). But, α-KGDH will be disturbed by oxidants. In that case, α-KGDH will inhibit the synthesis of NADH and the citric acid cycle activity will be also disturbed (Yudkoff et al. 1994).
Carnosine
Carnosine (β-alanyl-l-histidine) is a natural dipeptide present in many tissues (muscle, brain, liver, eye tissue, kidney, lung tissue), reaching maximum concentrations in skeletal muscle (~ 2.5 mM). The dipeptide is enzymatically synthesized by carnosine synthase enzyme (EC 6.3.2.11) from histidine and β-alanine precursors. The carnosine clearance is mainly due to the hydrolytic action of carnosinase (EC 3.4.13.3). Carnosine manifests a multitude of functions (Artioli et al. 2019; Prokopieva et al. 2016). During intense muscle work, carnosine acts as a proton buffer. It binds the excess protons, derived from lactic acid, and therefore prevents the development of acidosis (Baguet et al. 2010). Moreover, the presence of the histidine residue make it capable of scavenging different radicals, forming chelated compounds with prooxidant metals (such as Cu) (Klebanov et al. 1998; Zhao et al. 2019). Carnosine increases the cell proliferative potential (Hayflick limit) and cell morphological rejuvenation (Vishnyakova et al. 2014). Carnosine increases the longevity of cells grown in vitro 2–3 times and has a rejuvenating effect in senile cells. Together with a direct and indirect antioxidant effect, antiglycating, metal-chelating, chaperone and pH-buffering activity, carnosine is effective in brain neuroprotection and neuromodulation (Berezhnoy et al. 2019). Acute carnosine administration in rats showed an increase of the TCA cycle and OxPhos enzymatic activities, suggesting a preventative or even a therapeutic role in oxidative-driven neurodisorders (Macedo et al. 2016). The anti-neoplastic potential of carnosine has recently been reviewed (Artioli et al. 2019). Several studies claim that carnosine is tangled in cell apoptosis, cell cycle stopping, cell proliferation and glycolytic energy metabolism of some tumor cells (Cheng et al. 2019; Iovine et al. 2012; Lee et al. 2018; Sale et al. 2013). The compound inhibits tumor growth and metabolism processes through its antioxidant activity and its ability to influence glycolysis (Iovine et al. 2012). However, the exact mechanisms that explain this action are not clarified and consequently its antineoplastic properties continue to be investigated (Oppermann et al. 2016). Under the influence of carnosine the activity of isocitrate dehydrogenase, malate dehydrogenase in the TCA cycle together with the activities of mitochondrial electron transport chain complex I, II, III, and IV was markedly decreased in the cultured human cervical gland carcinoma cells (HeLa) but not in cervical squamous carcinoma cells (SiHa). Carnosine could reduce the mRNA and protein expression levels of ATP-dependent Clp protease proteolytic subunit (ClpP), which plays a key role in maintaining the mitochondrial function in HeLa cells (Bao et al. 2018). The influence of carnosine on the activity of chaperonins and a decreased expression of inducible factor of hypoxia alpha (HIF-1α) has been evidenced (Ding et al. 2018; Iovine et al. 2014). Carnosine protects cells from toxic action of aldehydes and ketones. It possesses the ability to form conjugates with toxic oxidized aldehyde products of lipids. The glycated proteins are immunogenic. They take part in the generation in the aging process of autoantigens. It is established that glycosylated carnosine is not mutagenic, unlike amino acids. It inhibits the lysine glycation reaction, modulates the toxicity of the product to cell culture. Protein aging is accompanied by the accumulation of adherent polypeptides, especially those containing carbonyl. It has been found that carnosine not only binds to the carbonyl group in proteins, but modulates their activity, inhibiting the formation of cross-links in proteins, such as result of the generation of the protein–carbonyl-carnosine adduct complex (Castelletto et al. 2019). Carnosine is a selective free-radical nitric oxide NO-dependent activator of guanilate cyclase (Gc) which catalyzes the biosynthesis of cyclic 3′,5′-guanosine monophosphate (cGMP). Therefore, carnosine can be used as an effective remedy for treating sepsis, cancer, asthma, migraine, all being related to the activation of the intracellular signaling system NO-Gc-cGMPc (Severina et al. 2000).
Physical exercise
Exercise significantly changes the average concentrations of metabolites that belong in particular to energy metabolism. An earlier study described that the total concentrations of lactate, pyruvate, malate, citrate, fumarate, fatty acids, acylcarnitines, ketone bodies and TCA cycle intermediates (TCAI) increased gradually during the incremental cycle exercise (Schranner et al. 2020). There is a positive and significant relationship between TCAI and approximately TCA cycle flux in the skeletal muscles of humans during the aerobic exercise. This was the very first study that directly compared the modifications in TCAI pool size with estimates of Krebs cycle flux in human skeletal muscle (Gibala et al. 1998). Comparative modifications of these variables were somewhat complicated, but the reported data demonstrated a substantial increase in TCA cycle flow. This study showed that the association between the pool size of TCAI and the flux of the TCA cycle is not linear (Gibala et al. 1998). The upturn in the citric acid cycle influx can take place by only a little rise in the total concentration of TCAI. For instance, during the shift from rest to submaximal exercise, the ratio of the flux of the Krebs cycle can increase by 70-fold gradually above rest, while the total concentration of intermediates of the citric acid cycle only doubled. Other studies reported that the concentrations of TCAI during the physical activities or strenuous exercise showed that the concentration-induced expansion of the Krebs cycle pool is unrelated to the capability for oxidative energy delivery (Bruce et al. 2001). Howarth et al. reported that after aerobic exercise, the decrease in the muscle TCAI pool would not limit the cycle endurance capacity (Howarth et al. 2004). There is a total of six amino acid molecules that are metabolized in the resting muscle: leucine, valine, isoleucine, aspartate, asparagine, and glutamate. These amino acids give the amino groups and ammonia for the further production of alanine and glutamine and are released in large quantities in post-absorptive conditions and for the period of ingestion of protein food. During exercise, leucine and isoleucine can be oxidized and transformed into acetyl-CoA. The other carbon skeleton compounds are utilized only for the de novo production of TCA-cycle intermediates and for the generation of glutamine (Wagenmakers 1998). Almost half of the glutamine taken up from muscle comes from glutamate from the blood circulation after the ingestion of a large meal. Glutamine that is synthesized in muscle is an essential fuel and the controller of DNA and RNA formation in mucosal cells, and satisfies many other vital functions in human metabolism. The alanine aminotransferase reaction serves to regulate and sustain the high concentrations of Krebs cycle intermediates in muscles during the first ten minutes of exercise. The upturn in the concentration of TCA-cycle intermediates possibly required to increase the flux of the TCA cycle to fulfill the increased energy demand of exercise (Wagenmakers 1998). In the course of intense exercise, the expression of peroxisome proliferator-activated receptor-gamma coactivator 1α (PGC1α) increases in skeletal muscle. PGC1α is a transcriptional coactivator able to activate branched-chain amino acid metabolism, fatty acid oxidation, and the TCA cycle (Hatazawa et al. 2015; Kamei et al. 2020).
Krebs cycle inhibitors
Aconitase inhibition (zinc excess and other inhibitors)
Aconitase is an enzyme involved in the maintenance of cell metabolism. There are two forms of aconitase, one is mitochondrial (ACO2), and the other one is cytosolic (ACO1) (Lushchak et al. 2014). The essential role of ACO2 is to regulate the ATP synthesis in a cell through the regulation of intermediate flux in the TCA cycle. ACO1, in its reduced form works as an enzyme, while, in oxidized form, has a role in iron homeostasis as iron regulatory protein 1 (IRP1).
Research has been done on ACO2 inhibition by zinc. It has been demonstrated that zinc at physiological levels is a substantial inhibitor, in mammalian cells, of mitochondrial ACO2, the enzyme that catalyzes the conversion of citrate to isocitrate, resulting in the accumulation of citrate that is unable to enter the Krebs cycle. Zinc serves as a competitive inhibitor of ACO2, in a mechanism in which only citrate is utilized as a substrate. Inhibition of zinc is specific for the reaction from citrate to cis-aconitase. As a result, the Krebs cycle is affected as the zinc–citrate complex competes with the free citrate to occupy the active site of ACO2 (Costello et al. 1997).
In prostate cancer, the normal citrate-synthesizing glandular secretory epithelial cells undergo a substantial metabolic conversion to malignant citrate-oxidizing cells. The critical step of this metabolic conversion, important to produce malignancy in the prostate, involves ACO2. The reduction of ACO2 activity in the prostate epithelial cells may be due to a reduced level of ACO2 enzyme or its inhibition. Zinc is inhibitor of ACO2 in the prostate epithelial cells and its depletion is the reason for the prevailing of malignancy and metabolic transformation of citrate.
Normally the secretory prostate cells have the specific activity of synthesis and secretion of elevated levels of citrate, with the single limiting ACO2 responsible for the impairment of oxidation of citrate. Conversely, in cancerous cells, the activity of ACO2 is not limiting and consequently, the citrate oxidation is also not impaired (Singh et al. 2006). Metabolic transformation or reprogramming is the key hallmark of cancer. That’s why targeting metabolism can provide an effective way to find promising drug targets to treat cancer (Huang et al. 2020). In prostate cancer, cells endure metabolic reprogramming from the accumulation of zinc. Malignant prostate cancer tissues have low levels of zinc and higher expression of ACO2 which activity is regulated by ROS. It has been postulated that both zinc and a zinc‐dependent metalloprotein P53, which functions in regulating the redox state and metabolism in cancer cells, can impair ACO2 activity and affect cell metabolism trough an increase in ROS levels (Xue et al. 2019). Targeting ACO2 through zinc, P53 or by utilizing another inhibitor compound provide a new strategy for prostate cancer treatment (Xue et al. 2019). In that way, accumulation of ROS causes a decrease in ACO2 activity, consequently Krebs cycle slow down and energy production is reduced. This leads to mitochondrial dysfunction, resulting in apoptosis of malignant cell. Conversely, in breast cancer, expression of ACO2 is reduced and when overexpressed a dysregulation of pyruvate metabolism occurs. This reveals a metabolic vulnerability of cancers associated also with ACO2 loss (Ciccarone et al. 2020).
Toxic metals
The influence of toxic metals on the enzymes of the Krebs cycle and their activity have been evaluated (Oexle et al. 1999; Strydom et al. 2006). These studies demonstrates, for example, that lead and cadmium alone or in combination have a deleterious effect on all TCA enzymes as α-ketoglutarate dehydrogenase, malate dehydrogenase, iso-citrate dehydrogenase and succinate dehydrogenase (Bansal et al. 2002). It has been observed that due to cadmium, muscle glycogen and liver glycogen are reduced (Strydom et al. 2006). In addition, high exposure of iron, and specifically to iron chloride, in human cell lines, can compromise Krebs cycle activity, mitochondrial respiration and oxidative phosphorylation. Elevated amount of iron enhance the expression of ACO2 through a mechanism that involves IRPs. There is a complicated relationship between iron homeostasis, the supply of oxygen, and the energy metabolism of the cell, since, as already mentioned, iron in normal concentration has positive influences on Krebs cycle (Oexle et al. 1999). These relationships, however, are not completely understood yet. The lethal effect of chromium has been also evaluated on the activity of lactate dehydrogenase, succinate dehydrogenase and pyruvate dehydrogenase (Anjum and Shakoori 1997). TCA cycle is also affected by manganese through perturbation of lactate dehydrogenase activity as well as other Krebs cycle enzymes. The change in brain energy metabolism, the increased glycolytic flux and lactate synthesis might contribute to the toxic effects of manganese in the brain (Zwingmann et al. 2004). Also aluminum is able to inhibit several enzymes comprising those related to glycolysis and Krebs cycle, thus interfering with the metabolic pathways of mitochondria (Zatta et al. 2000). Studies have been shown the negative effect of Al on the bioenergy production mechanism in the cells that are consuming oxygen. Aluminum stimulates oxidative stress and triggers the accumulation of succinate (Mailloux et al. 2006). Therefore, the presence of aluminum in a microenvironment of the cell would negatively affect the production of ATP. However the mechanism by which aluminum acts as a pro-oxidant has not been completely clarified (Mailloux et al. 2006). Aluminum is, in fact, a complicated stressor. It affects multiple biological activities. Aluminum affects iron metabolism, and this phenomenon also mediates the formation of ROS intracellularly. As already mentioned, both iron deficiency and overload can affect the synthesis of ATP. Therefore, aluminum toxicity influences Krebs cycle directly and indirectly perturbing the iron dependent enzymes of the TCA cycle (Lemire and Appanna 2011). The lack of ATP synthesis in the brain cells might be a critical cause of neurodegenerative pathologies because the brain is an energy-demanding organ (Kumar and Gill 2009). However, this anomaly in primary metabolic processes may be responsible for the inability of the cytoskeleton to subsidize the appropriate structure of astrocytes. This breakdown in astrocyte morphology will affect the brain because astrocytes provide scaffolding and control the blood–brain barrier through their filopodia (Lemire and Appanna 2011). Ameliorative measures in reducing metal induced oxidative stress and mitochondrial dysfunctions are therefore required. In particular, these strategies include chelation therapy, antioxidants, plant extracts, and drugs, applied alone or in combination (Crisponi et al. 2013; Kumar and Gill 2014; Nurchi et al. 2019). It also seems essential to further evaluate the effect of the metal ions on the central metabolic pathways as potential biomarkers, as they may eventually provide an early indication of ongoing disease (Strydom et al. 2006).
Metformin
Metformin is associated with the biguanide family of drugs utilized for the treatment of diabetes (Inzucchi et al. 2015). Metformin is a well-known drug that inhibits the complex I of the electron transport chain in mitochondria. It results in a decrease in the activity of mitochondrial complex I and ultimately decreased ATP production through oxidative phosphorylation. In diabetic people, metformin initially works in the hepatocytes and inhibits gluconeogenesis. This process is responsible for reducing the hyperglycemia and the related increase in insulin present in the circulation. Metformin has a role in stimulating the LKB1-dependent stress reaction in the hepatocytes, which results in the activation of the AMP-activated protein kinase energy sensor and decreased expression of gluconeogenic enzymes in the liver (Shaw et al. 2005). A pilot case–control study indicated the ability of metformin to reduce the risk of cancer in type 2 diabetic people (Evans et al. 2005). These early observations were subsequently confirmed in several epidemiological studies and meta-analyses (Dinic et al. 2020). Metabolic profiling can be used to evaluate the effect of metformin on cancer cell metabolism. Results indicate that metformin inhibits the flow of carbon source into the Krebs cycle, which affects the metabolic pathways of mitochondria and all the biosynthesis that is occurring within the mitochondria, involving de novo lipogenesis. This indicates that the cancer cells with decreased oxidative mitochondrial activity are responsible for mutations in the electron transport chain under the metformin treatment. Therefore, metformin can suppress tumor cell proliferation by inhibiting the TCA cycle and its metabolic intermediates that are essential for the proliferation of cancer cells (Griss et al. 2015).
At the cellular level, metformin is responsible for the disruption of mitochondrial activity via partially suppressing the NADH dehydrogenase. It also inhibits the glycerol phosphate dehydrogenase in hepatocytes. All these disruptions are responsible for the modifications to the OxPhos. Consequently, electrons present in NADH and Flavin adenine dinucleotide (FADH2) are not efficiently transported via the electron transport chain. Metformin affects several processes in cell, such as AMPK signaling, folate metabolism, anabolic metabolism, and protein kinase A signaling (Liu et al. 2016). The data showed that metformin is responsible for decreasing the mitochondrial respiration. St-Pierre et al. demonstrated that metformin influences mitochondrial bioenergetics in intact cells and in isolated mitochondria, reducing the metabolism of glucose via the Krebs cycle and limiting OxPhos activity. The decrease in mitochondrial function due to the activity of metformin resulted in a compensatory increase in glycolysis. Cells treated with metformin appear energetically inefficient and show increased aerobic glycolysis and reduced glucose metabolism through TCA cycle. Inhibition of all these processes in cancerous cells can substantially reduce their survival and it is very useful in cancer treatment (Andrzejewski et al. 2014). The uses of metformin has been associated with a decreased cancer incidence and mortality. However, its potential use as an immunotherapeutic agent must however be supported by further evidence to confirm its possible benefits in the treatment of malignancies (Chae et al. 2016).
Vitamin B12 deficiency
Vitamins B are a class of vital nutrients that take part in mitochondrial metabolic pathways. They have a role as co-factors or co-enzymes for all the mitochondrial enzymes. Five vitamins B (B1, B2, B3, B5, and B7/B8) are directly taking part in the activity of the Krebs cycle (Janssen et al. 2019). They regulate the activity of all mitochondrial enzymes. Regulating an appropriate pool of vitamins B in mitochondria, is very important to maintain the metabolic and other biochemical processes that are carried out by these mitochondrial enzymes (Janssen et al. 2019). In particular, vitamin B12 (cobalamin) is essential for the production of succinyl-CoA from methylmalonyl-CoA in mitochondria, and is also vital for the biosynthesis of nucleotides and for the metabolism of amino acids. Vitamin B12 deficiency can cause growth retardation and many metabolic disorders in mammals (Green et al. 2017; Solomon 2007). Deoxyadenosylcobalamin, one of the biologically active forms of vitamin B12, is the vital co-enzyme for methylmalonyl-Coenzyme A mutase (MUT) found in mitochondria. This enzyme is responsible for the conversion of methylmalonyl-CoA into succinyl-CoA, and plays a function in the breakdown of amino acids and fatty acids chain. Early investigations on mice indicate that vitamin B12 deficiency was able to disrupt the normal hepatic glucose and glutamic acid metabolism, probably by the TCA inhibition, accompanied by an unusual accumulation of methylmalonic acid (MMA) (Toyoshima et al. 1996). Accumulation of MMA hinders the conversion of methylmalonyl-CoA into succinyl-CoA, and that how succinyl-CoA cannot enter into the TCA cycle. Therefore, TCA gets disrupted due to the deficiency of vitamin B12. This denotes that vitamin B12 is essential for the activity of the MUT enzyme and the proper functioning of the TCA cycle (Green et al. 2016). Another form of vitamin B12, methylcobalamin, serves as a cofactor for the cytosolic enzyme 5-methyltetrahydrofolate-homocysteine methyltransferase (MTR), also referred to as methionine synthase. This enzyme is responsible for the transmethylation of homocysteine through methyltetrahydrofolate (MTHF) to methionine. Moreover, except serving as a coenzyme for methionine synthase, methylcobalamin is also responsible for the production of GSH (Janssen et al. 2019). The mechanisms in the regulation of vitamin B12 metabolism are still under study. Recently, a novel metabolic pathway for vitamin B12 has been described. This linked vitamin B12 with the catabolism of itaconate, an antimicrobial and immunomodulatory metabolite, and with the knockout of citrate lyase subunit beta-like (CLYBL) gene, which encodes a ubiquitously expressed mitochondrial enzyme. During immune activation, itaconate is produced at a high concentration by macrophages, and inhibits vitamin B12 metabolism. This fact underline a novel link between vitamin B12 and immune system (Reid et al. 2017; Shen et al. 2017).
Role of Krebs cycle modulations
Oxidative and nitrosative stress
Aconitase is an enzyme that contains an iron–sulfur cluster sensitive to oxidation. Oxidative stress or ROS play a chief role in aconitase activities maintenance. The catalytic activity of aconitase is regulated by oxidation of [4Fe-4S]2+ cluster and by reversible oxidation of cysteine residues. The redox-dependent posttranslational modifications of aconitase aroused greater interest. These occur due to oxidation, nitrosylation, and thiolation of cysteine residues and tyrosine residues (Lushchak et al. 2014). Oxidative and nitrosative stress is a constant challenge facing various organisms. Nitric oxide can interacts with ROS, like superoxide (O2·−) and hydrogen peroxide (H2O2), generating a group of toxic reactive nitrogen species (RNS). RNS includes nitrous oxide (N2O), peroxynitrite (\({\text{NO}}_{3}^{ - }\)), nitroxyl anion (NO−), and peroxynitrous acid (HNO3). Since also RNS can severely damage cellular components, organisms have developed elaborate detox mechanisms that can control their homeostasis. However, the role of cell metabolism in preventing RNS is not completely understood so far. RNS and ROS can interact with aconitase decreasing its activity and thereby slow down glucose consumption, TCA cycle and OxPhos process (Auger et al. 2011). It has been recognized that TCA cycle intermediates can act effectively in brain cells protection against reactive oxidative species. In particular, it has been demostrated that, between these intermediates, only pyruvate, oxaloacetate, and α-ketoglutarate showed neuroprotective effect against H2O2 mediated toxicity in neuronal cells (Sawa et al. 2017). Other reports suggest their positive effects on neurons by enhancing cell infrastructure, supporting glycolysis and enhanced respiration (Ruban et al. 2015; Zhang et al. 2017). Therefore these intermediates, due for their antioxidant, anti-inflammatory, and anti-aging properties, can be considered perspective tools for many therapeutic interventions to approaches and minimize the deleterious effects of oxidative stress as in preventing or treating chronic neurodegenerative diseases (Bayliak et al. 2016; Wahl et al. 2019).
Inflammation
The innate immune system is the primary line of defense against infection and pathogens. Dendritic cell and macrophages play an essential role for mediating protective immune responses. Citrate has been showed to have a key role in inflammatory pathways. In macrophages, there is a large amount of isocitrate and α-ketoglutarate, and a transcriptional downregulation of isocitrate dehydrogenase 1 (Jha et al. 2015). The same ratio of intermediates is also found in the dendritic cells (Everts et al. 2014). In both macrophages and dendritic cells, it has been observed an increased flux of glycolysis and a TCA cycle fragmentation. Pyruvate that is coming from glycolysis enters into the TCA cycle, but it cannot keep going on past citrate and iso-citrate. The degradation of citrate present in the mitochondria has been associated with the synthesis in human macrophages of various key pro-inflammatory mediators as ROS, NO and prostaglandin E2 (Williams and O’Neill 2018). An increase amount of citrate is observed in both mouse and human necrosis factor α (TNFα) and interferon-γ (IFNγ) macrophages (Tannahill et al. 2013). The gene SLC25A1 codes the mitochondrial citrate carrier (CiC) that triggers the transfer of citrate from the mitochondrial to the cytoplasm compart. In cytosol, citrate is converted to acetyl-CoA and oxalacetate by the ATP-citrate lyase. SCC25A1 is responsible for increasing the synthesis of inflammatory cytokines since acetyl-CoA promotes histones acetylation at the TNFα and interleukin IL-8 promoter regions, increasing the production TNFα, IL-1β, and IFNγ macrophages. These further involve the stimulation of nuclear factor kappa B (NF-κB) and signal transducer and activator of transcription 1 (STAT1), which ultimately produces chronic inflammation in the body (Infantino et al. 2014). Upon lipopolysaccharides (LPS) stimulation, the TCA intermediate, succinate, accumulates in the cytoplasm of monocyte macrophages and DCs. This leads an increase of OxPhos activity, ROS production, HIF-1α stabilization, intracellular sensor NLRP3 inflammasome activation, pro-IL-1β transcription regulation and an increase of IL-1β production (Aguilar-Lopez et al. 2020). Fumarate and its derivatives, together with their bacteriostatic and bactericidal properties, are powerful immunomodulators and antioxidants (Garaude et al. 2016). An anti-inflammatory activity of dimethyl fumarate has been highlighted on murine astrocytes. It exerts in vivo neuroprotective effects in inflammation via activation of the Nrf2 antioxidant pathway and reducing oxidative stress (Linker et al. 2011). Chronic inflammation can be induced due to multiple chronic pathologies involving autoimmune disorders, cancer, and metabolic disorders. There are increasing evidences that innate immune memory involves changes in glycolysis, oxidative phosphorylation, fatty acid and amino acid metabolism (Breda et al. 2019). A future approach in the treatment of diseases, such as inflammatory syndromes and cancer, could be done modulating mitochondrial activity in immune cells, such as the “citrate pathway” (Breda et al. 2019; Swanson et al. 2019; Williams and O’Neill 2018).
Liver detoxification
Mitochondrial failure or dysfunction has been described in many liver diseases, for example, hepatocellular carcinoma, drug toxicity, and it is usually recognized as early modifications in the liver. Body cells developed various mechanisms to maintain the mitochondria integrity and to hinder the effects of mitochondrial lesions. For example, enhanced mitochondria biogenesis and controlled transduction pathways signal make sure the energy metabolism and regulate the apoptosis and various inflammatory reactions (Degli Esposti et al. 2012). TCAI have a role in liver detoxification. One study described that people suffering from fulminant liver failure have disturbed brain metabolism that may be associated with increased ammonia rates. One effect of ammonia is the inhibition of the rate-limiting TCA cycle enzyme α-KGDH and probably pyruvate dehydrogenase. A study of Ott et al. provides a proof about the association between liver detoxification of ammonia through the production of glutamine and lactate (Ott et al. 2005). This showed that the inhibition of both α-KGDH and pyruvate dehydrogenase enhances the detoxification of ammonia through the production of glutamine from α-KG and it also decreases the amount of NADH and oxidative synthesis of ATP in the mitochondria present in the astrocytes (Ott et al. 2005). In the cytosol of astrocytes, this will lead to the synthesis of lactate even in the existence of enough supply of oxygen. There is one other way to synthesize the ATP by utilizing amino acids. In this compensatory mechanism, branched-chain amino acids are used, such as isoleucine and valine, which can supply skeletons of carbon and bypass the α-KGDH inhibition and regulation of activity of TCA cycle (Holecek and Vodenicarovova 2018). A treatment of hyperammonemic patients with glutamine synthetase inhibitors has been also described as potential effective target therapy (Brusilow et al. 2010; Dadsetan et al. 2013; Korf et al. 2019).
Carcinogenesis
Mitochondria dysfunction could cause cancer (Sajnani et al. 2017). Oxidative damage to mitochondrial DNA has been linked with the cancer pathogenesis and many mitochondrial genes are found to be upregulated in the cancer progression. This discovery encouraged scientists to further evaluate the role of mitochondria in the genesis of cancer. Inherited and acquired modification in TCA enzymes, namely citrate synthase, succinate dehydrogenase, fumarate dehydrogenase, and isocitrate dehydrogenase, has been recognized in various types of cancers (Sajnani et al. 2017; Wallace 2005). In addition, several intermediates of the TCA pathway, such as succinate, fumarate, itaconate, 2-hydroxyglutarate isomers, and acetyl-CoA, showed extensive evidence for their “non-metabolic” signalling functions, related to physiological immune or disease contexts, such as the genesis of tumors (Ryan et al. 2019). Abnormalities in the genes that are encoding the TCA cycle’s enzyme lead to aberrant accumulation of oncometabolite and mitochondrial failure since abnormalities in the metabolic enzymes are responsible for the cancer pathogenesis. These alterations mostly occur in aconitase because it is the critical and sensitive enzyme of the TCA cycle. This phenomenon is also responsible for the induction of metabolic reprogramming in cancer cells and increases the use of glucose through glycolysis for the proliferation and survival of cancer cells (Ryan et al. 2019).
Conclusion
The primary energy source of cancer cells is glucose; the metabolism of tumor cells is aerobic glycolysis (Warburg effect), where lactate dehydrogenase is a marker of tumor malignancy. Cancer cells’ dependence on glucose, the possibility of glutamine used in energy processes, inhibition of oxidative metabolism are particularities of tumor metabolism, which can be targeted for selective, basic, and adjuvant treatment. The modulation of energy metabolism is a new strategy in cancer treatment. Krebs cycle is the central metabolic pathway that transfers NADH and FADH to the electron transport chain and produces cellular energy through oxidative phosphorylation. It is linked to several metabolic pathways within cells. All the fuels, such as amino acids, carbohydrates, and fatty acids, molecules end up in the Krebs cycle for the further oxidation process. Many activators can initiate or enhance the functioning of the Krebs cycle, for example, glutamine, glutathione, NADH, iron, and exercise or aerobic training. However, there are some de-activators of the Krebs cycle, also present, that are responsible for decreasing its functionality, for instance, inhibition of aconitase enzyme, heavy metal toxicity, aluminum toxicity, metformin, and vitamin B12 deficiency. These factors reduce the function of the Krebs cycle. It is also associated with some pathologies. Genetic alterations in Krebs cycle enzymes are responsible for the production of many pathologies, including cancer. It is also associated with inflammatory responses and oxidative/nitrosative stress. Krebs cycle also has a role in liver detoxification by eliminating ammonia through glutamine. Considering all the functions and activities of the Krebs cycle, this appears to represent the central and fundamental metabolic pathway in organisms.
Conflict of interest
The authors declare they have no actual or potential competing financial interests.
Abbreviations
- ACC:
-
Acetyl-CoA carboxylase
- ACO2:
-
Aconitase
- αKG:
-
α-Ketoglutarate
- α-KGDH :
-
α-Ketoglutarate dehydrogenase
- ATP:
-
Adenosine triphosphate
- CPT1:
-
Carnitine palmitoyltransferase 1
- CiC:
-
Mitochondrial citrate carrier
- CoA:
-
Acetyl coenzyme A
- COX:
-
Cytochrome c oxidase
- DC:
-
Dendritic cell
- FADH2:
-
Flavin adenine dinucleotide reduced form
- HIF-1α:
-
Inducible factor of hypoxia alpha
- GSH:
-
Glutathione
- GTP:
-
Guanosine triphosphate
- LDH:
-
Lactate dehydrogenase
- NADH:
-
Nicotinamide adenine dinucleotide
- NO:
-
Nitric oxide
- Nrf2:
-
NNuclear factor erythroid 2-related factor 2
- ROS:
-
Reactive oxygen species
- TCA:
-
Tricarboxylic acid
- TCAI:
-
Tricarboxylic acid intermediate
References
Aguilar-Lopez BA, Moreno-Altamirano MMB, Dockrell HM, Duchen MR, Sanchez-Garcia FJ (2020) Mitochondria: an integrative hub coordinating circadian rhythms, metabolism, the microbiome, and immunity. Front Cell Dev Biol 8:51. https://doi.org/10.3389/fcell.2020.00051
Akram M (2014) Citric acid cycle and role of its intermediates in metabolism. Cell Biochem Biophys 68(3):475–478. https://doi.org/10.1007/s12013-013-9750-1
Anderson NM, Mucka P, Kern JG, Feng H (2018) The emerging role and targetability of the TCA cycle in cancer metabolism. Protein Cell 9(2):216–237. https://doi.org/10.1007/s13238-017-0451-1
Andrzejewski S, Gravel SP, Pollak M, St-Pierre J (2014) Metformin directly acts on mitochondria to alter cellular bioenergetics. Cancer Metab 2:12. https://doi.org/10.1186/2049-3002-2-12
Anjum F, Shakoori AR (1997) Sublethal effects of hexavalent chromium on the body growth rate and liver function enzymes of phenobarbitone-pretreated and promethazine-pretreated rabbits. J Environ Pathol Toxicol Oncol 16(1):51–59
Artioli GG, Sale C, Jones RL (2019) Carnosine in health and disease. Eur J Sport Sci 19(1):30–39. https://doi.org/10.1080/17461391.2018.1444096
Auger C, Lemire J, Cecchini D, Bignucolo A, Appanna VD (2011) The metabolic reprogramming evoked by nitrosative stress triggers the anaerobic utilization of citrate in Pseudomonas fluorescens. PLoS ONE 6(12):e28469. https://doi.org/10.1371/journal.pone.0028469
Babu R, Eaton S, Drake DP, Spitz L, Pierro A (2001) Glutamine and glutathione counteract the inhibitory effects of mediators of sepsis in neonatal hepatocytes. J Pediatr Surg 36(2):282–286. https://doi.org/10.1053/jpsu.2001.20690
Baguet A, Bourgois J, Vanhee L, Achten E, Derave W (2010) Important role of muscle carnosine in rowing performance. J Appl Physiol (1985) 109(4):1096–1101. https://doi.org/10.1152/japplphysiol.00141.2010
Bansal P, Sharma P, Goyal V (2002) Impact of lead and cadmium on enzyme of citric acid cycle in germinating pea seeds. Biol Plant 45(1):125–127. https://doi.org/10.1023/a:1015173112842
Bao Y, Ding S, Cheng J, Liu Y, Wang B, Xu H, Shen Y, Lyu J (2018) Carnosine inhibits the proliferation of human cervical gland carcinoma cells through inhibiting both mitochondrial bioenergetics and glycolysis pathways and retarding cell cycle progression. Integr Cancer Ther 17(1):80–91. https://doi.org/10.1177/1534735416684551
Bartkova J, Rezaei N, Liontos M, Karakaidos P, Kletsas D, Issaeva N, Vassiliou L-VF, Kolettas E et al (2006) Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature 444(7119):633–637. https://doi.org/10.1038/nature05268
Bayliak MM, Lylyk MP, Vytvytska OM, Lushchak VI (2016) Assessment of antioxidant properties of alpha-keto acids in vitro and in vivo. Eur Food Res Technol 242(2):179–188. https://doi.org/10.1007/s00217-015-2529-4
Berezhnoy DS, Stvolinsky SL, Lopachev AV, Devyatov AA, Lopacheva OM, Kulikova OI, Abaimov DA, Fedorova TN (2019) Carnosine as an effective neuroprotector in brain pathology and potential neuromodulator in normal conditions. Amino Acids 51(1):139–150. https://doi.org/10.1007/s00726-018-2667-7
Bjørklund G, Peana M, Maes M, Dadar M, Severin B (2020a) The glutathione system in Parkinson’s disease and its progression. Neurosci Biobehav Rev. https://doi.org/10.1016/j.neubiorev.2020.10.004
Bjørklund G, Tinkov AA, Hosnedlova B, Kizek R, Ajsuvakova OP, Chirumbolo S, Skalnaya MG, Peana M et al (2020b) The role of glutathione redox imbalance in autism spectrum disorder: a review. Free Radic Biol Med 160:149–162. https://doi.org/10.1016/j.freeradbiomed.2020.07.017
Bjørklund G, Doşa MD, Maes M, Dadar M, Frye RE, Peana M, Chirumbolo S (2021) The impact of glutathione metabolism in autism spectrum disorder. Pharmacol Res. https://doi.org/10.1016/j.phrs.2021.105437
Breda CNdS, Davanzo GG, Basso PJ, Saraiva Câmara NO, Moraes-Vieira PMM (2019) Mitochondria as central hub of the immune system. Redox Biol 26:101255. https://doi.org/10.1016/j.redox.2019.101255
Brownsey RW, Zhande R, Boone AN (1997) Isoforms of acetyl-CoA carboxylase: structures, regulatory properties and metabolic functions. Biochem Soc Trans 25(4):1232–1238. https://doi.org/10.1042/bst0251232
Bruce M, Constantin-Teodosiu D, Greenhaff PL, Boobis LH, Williams C, Bowtell JL (2001) Glutamine supplementation promotes anaplerosis but not oxidative energy delivery in human skeletal muscle. Am J Physiol Endocrinol Metab 280(4):E669–E675. https://doi.org/10.1152/ajpendo.2001.280.4.E669
Brusilow SW, Koehler RC, Traystman RJ, Cooper AJ (2010) Astrocyte glutamine synthetase: importance in hyperammonemic syndromes and potential target for therapy. Neurotherapeutics 7(4):452–470. https://doi.org/10.1016/j.nurt.2010.05.015
Burns JS, Manda G (2017) Metabolic pathways of the Warburg effect in health and disease: perspectives of choice, chain or chance. Int J Mol Sci 18(12):2755. https://doi.org/10.3390/ijms18122755
Castelletto V, Edwards-Gayle CJC, Greco F, Hamley IW, Seitsonen J, Ruokolainen J (2019) Self-assembly, tunable hydrogel properties, and selective anti-cancer activity of a carnosine-derived lipidated peptide. ACS Appl Mater Interfaces 11(37):33573–33580. https://doi.org/10.1021/acsami.9b09065
Chae YK, Arya A, Malecek MK, Shin DS, Carneiro B, Chandra S, Kaplan J, Kalyan A et al (2016) Repurposing metformin for cancer treatment: current clinical studies. Oncotarget 7(26):40767–40780. https://doi.org/10.18632/oncotarget.8194
Cheng JY, Yang JB, Liu Y, Xu M, Huang YY, Zhang JJ, Cao P, Lyu JX et al (2019) Profiling and targeting of cellular mitochondrial bioenergetics: inhibition of human gastric cancer cell growth by carnosine. Acta Pharmacol Sin 40(7):938–948. https://doi.org/10.1038/s41401-018-0182-8
Choudhary C, Weinert BT, Nishida Y, Verdin E, Mann M (2014) The growing landscape of lysine acetylation links metabolism and cell signalling. Nat Rev Mol Cell Biol 15(8):536–550. https://doi.org/10.1038/nrm3841
Ciccarone F, Di Leo L, Lazzarino G, Maulucci G, Di Giacinto F, Tavazzi B, Ciriolo MR (2020) Aconitase 2 inhibits the proliferation of MCF-7 cells promoting mitochondrial oxidative metabolism and ROS/FoxO1-mediated autophagic response. Br J Cancer 122(2):182–193. https://doi.org/10.1038/s41416-019-0641-0
Cook KM, Figg WD (2010) Angiogenesis inhibitors: current strategies and future prospects. CA Cancer J Clin 60(4):222–243. https://doi.org/10.3322/caac.20075
Costello LC, Liu Y, Franklin RB, Kennedy MC (1997) Zinc inhibition of mitochondrial aconitase and its importance in citrate metabolism of prostate epithelial cells. J Biol Chem 272(46):28875–28881. https://doi.org/10.1074/jbc.272.46.28875
Crisponi G, Dean A, Di Marco V, Lachowicz JI, Nurchi VM, Remelli M, Tapparo A (2013) Different approaches to the study of chelating agents for iron and aluminium overload pathologies. Anal Bioanal Chem 405(2–3):585–601. https://doi.org/10.1007/s00216-012-6468-7
Dadsetan S, Kukolj E, Bak LK, Sørensen M, Ott P, Vilstrup H, Schousboe A, Keiding S et al (2013) Brain alanine formation as an ammonia-scavenging pathway during hyperammonemia: effects of glutamine synthetase inhibition in rats and astrocyte-neuron co-cultures. J Cereb Blood Flow Metab 33(8):1235–1241. https://doi.org/10.1038/jcbfm.2013.73
Degli Esposti D, Hamelin J, Bosselut N, Saffroy R, Sebagh M, Pommier A, Martel C, Lemoine A (2012) Mitochondrial roles and cytoprotection in chronic liver injury. Biochem Res Int 2012:387626. https://doi.org/10.1155/2012/387626
Demarin V, Podobnik SS, Storga-Tomic D, Kay G (2004) Treatment of Alzheimer’s disease with stabilized oral nicotinamide adenine dinucleotide: a randomized, double-blind study. Drugs Exp Clin Res 30(1):27–33
Devic S (2016) Warburg effect—a consequence or the cause of carcinogenesis? J Cancer 7(7):817–822. https://doi.org/10.7150/jca.14274
Devin A, Rigoulet M (2007) Mechanisms of mitochondrial response to variations in energy demand in eukaryotic cells. Am J Physiol Cell Physiol 292(1):C52–C58. https://doi.org/10.1152/ajpcell.00208.2006
Diaz-Ruiz R, Rigoulet M, Devin A (2011) The Warburg and Crabtree effects: on the origin of cancer cell energy metabolism and of yeast glucose repression. Biochim Biophys Acta 1807(6):568–576. https://doi.org/10.1016/j.bbabio.2010.08.010
Ding M, Jiao G, Shi H, Chen Y (2018) Investigations on in vitro anti-carcinogenic potential of l-carnosine in liver cancer cells. Cytotechnology 70(1):163–167. https://doi.org/10.1007/s10616-017-0123-2
Dinic J, Efferth T, Garcia-Sosa AT, Grahovac J, Padron JM, Pajeva I, Rizzolio F, Saponara S et al (2020) Repurposing old drugs to fight multidrug resistant cancers. Drug Resist Updat 52:100713. https://doi.org/10.1016/j.drup.2020.100713
Evans JM, Donnelly LA, Emslie-Smith AM, Alessi DR, Morris AD (2005) Metformin and reduced risk of cancer in diabetic patients. BMJ 330(7503):1304–1305. https://doi.org/10.1136/bmj.38415.708634.F7
Everts B, Amiel E, Huang SC, Smith AM, Chang CH, Lam WY, Redmann V, Freitas TC et al (2014) TLR-driven early glycolytic reprogramming via the kinases TBK1-IKKvarepsilon supports the anabolic demands of dendritic cell activation. Nat Immunol 15(4):323–332. https://doi.org/10.1038/ni.2833
Fei B, Ji F, Chen X, Liu Z, Li S, Mo Z, Fang X (2016) Expression and clinical significance of Beclin-1 in gastric cancer tissues of various clinical stages. Oncol Lett 11(3):2271–2277. https://doi.org/10.3892/ol.2016.4183
Fernie AR, Carrari F, Sweetlove LJ (2004) Respiratory metabolism: glycolysis, the TCA cycle and mitochondrial electron transport. Curr Opin Plant Biol 7(3):254–261. https://doi.org/10.1016/j.pbi.2004.03.007
Fleming RE, Ponka P (2012) Iron overload in human disease. N Engl J Med 366(4):348–359. https://doi.org/10.1056/NEJMra1004967
Fricker RA, Green EL, Jenkins SI, Griffin SM (2018) The influence of nicotinamide on health and disease in the central nervous system. Int J Tryptophan Res 11:1178646918776658. https://doi.org/10.1177/1178646918776658
Garaude J, Acin-Perez R, Martinez-Cano S, Enamorado M, Ugolini M, Nistal-Villan E, Hervas-Stubbs S, Pelegrin P et al (2016) Mitochondrial respiratory-chain adaptations in macrophages contribute to antibacterial host defense. Nat Immunol 17(9):1037–1045. https://doi.org/10.1038/ni.3509
Gibala MJ, MacLean DA, Graham TE, Saltin B (1998) Tricarboxylic acid cycle intermediate pool size and estimated cycle flux in human muscle during exercise. Am J Physiol 275(2):E235–E242. https://doi.org/10.1152/ajpendo.1998.275.2.E235
Gibson GE, Sheu KF, Blass JP (1998) Abnormalities of mitochondrial enzymes in Alzheimer disease. J Neural Transm (Vienna) 105(8–9):855–870. https://doi.org/10.1007/s007020050099
Green CR, Wallace M, Divakaruni AS, Phillips SA, Murphy AN, Ciaraldi TP, Metallo CM (2016) Branched-chain amino acid catabolism fuels adipocyte differentiation and lipogenesis. Nat Chem Biol 12(1):15–21. https://doi.org/10.1038/nchembio.1961
Green R, Allen LH, Bjorke-Monsen AL, Brito A, Gueant JL, Miller JW, Molloy AM, Nexo E et al (2017) Vitamin B12 deficiency. Nat Rev Dis Primers 3:17040. https://doi.org/10.1038/nrdp.2017.40
Griss T, Vincent EE, Egnatchik R, Chen J, Ma EH, Faubert B, Viollet B, DeBerardinis RJ et al (2015) Metformin antagonizes cancer cell proliferation by suppressing mitochondrial-dependent biosynthesis. PLoS Biol 13(12):e1002309. https://doi.org/10.1371/journal.pbio.1002309
Halliwell B (1999) Antioxidant defence mechanisms: from the beginning to the end (of the beginning). Free Radic Res 31(4):261–272. https://doi.org/10.1080/10715769900300841
Hao W, Chang CP, Tsao CC, Xu J (2010) Oligomycin-induced bioenergetic adaptation in cancer cells with heterogeneous bioenergetic organization. J Biol Chem 285(17):12647–12654. https://doi.org/10.1074/jbc.M109.084194
Hatazawa Y, Senoo N, Tadaishi M, Ogawa Y, Ezaki O, Kamei Y, Miura S (2015) Metabolomic analysis of the skeletal muscle of mice overexpressing PGC-1alpha. PLoS ONE 10(6):e0129084. https://doi.org/10.1371/journal.pone.0129084
Holecek M, Vodenicarovova M (2018) Effects of branched-chain amino acids on muscles under hyperammonemic conditions. J Physiol Biochem 74(4):523–530. https://doi.org/10.1007/s13105-018-0646-9
Hou Y, Lautrup S, Cordonnier S, Wang Y, Croteau DL, Zavala E, Zhang Y, Moritoh K et al (2018) NAD+ supplementation normalizes key Alzheimer’s features and DNA damage responses in a new AD mouse model with introduced DNA repair deficiency. Proc Natl Acad Sci 115(8):E1876. https://doi.org/10.1073/pnas.1718819115
Howarth KR, LeBlanc PJ, Heigenhauser GJ (1985) Gibala MJ (2004) Effect of endurance training on muscle TCA cycle metabolism during exercise in humans. J Appl Physiol 97(2):579–584. https://doi.org/10.1152/japplphysiol.01344.2003
Huang L, Wang C, Xu H, Peng G (2020) Targeting citrate as a novel therapeutic strategy in cancer treatment. Biochim Biophys Acta Rev Cancer 1873(1):188332. https://doi.org/10.1016/j.bbcan.2019.188332
Iacobazzi V, Infantino V (2014) Citrate—new functions for an old metabolite. Biol Chem 395(4):387–399. https://doi.org/10.1515/hsz-2013-0271
Infantino V, Iacobazzi V, Palmieri F, Menga A (2013) ATP-citrate lyase is essential for macrophage inflammatory response. Biochem Biophys Res Commun 440(1):105–111. https://doi.org/10.1016/j.bbrc.2013.09.037
Infantino V, Iacobazzi V, Menga A, Avantaggiati ML, Palmieri F (2014) A key role of the mitochondrial citrate carrier (SLC25A1) in TNFalpha- and IFNgamma-triggered inflammation. Biochim Biophys Acta 1839(11):1217–1225. https://doi.org/10.1016/j.bbagrm.2014.07.013
Inzucchi SE, Bergenstal RM, Buse JB, Diamant M, Ferrannini E, Nauck M, Peters AL, Tsapas A et al (2015) Management of hyperglycemia in type 2 diabetes, 2015: a patient-centered approach: update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 38(1):140–149. https://doi.org/10.2337/dc14-2441
Iovine B, Iannella ML, Nocella F, Pricolo MR, Bevilacqua MA (2012) Carnosine inhibits KRAS-mediated HCT116 proliferation by affecting ATP and ROS production. Cancer Lett 315(2):122–128. https://doi.org/10.1016/j.canlet.2011.07.021
Iovine B, Oliviero G, Garofalo M, Orefice M, Nocella F, Borbone N, Piccialli V, Centore R et al (2014) The anti-proliferative effect of l-carnosine correlates with a decreased expression of hypoxia inducible factor 1 alpha in human colon cancer cells. PLoS ONE 9(5):e96755. https://doi.org/10.1371/journal.pone.0096755
Isidoro A, Casado E, Redondo A, Acebo P, Espinosa E, Alonso AM, Cejas P, Hardisson D et al (2005) Breast carcinomas fulfill the Warburg hypothesis and provide metabolic markers of cancer prognosis. Carcinogenesis 26(12):2095–2104. https://doi.org/10.1093/carcin/bgi188
Janssen JJE, Grefte S, Keijer J, de Boer VCJ (2019) Mito-nuclear communication by mitochondrial metabolites and its regulation by B-vitamins. Front Physiol 10:78. https://doi.org/10.3389/fphys.2019.00078
Jha AK, Huang SC, Sergushichev A, Lampropoulou V, Ivanova Y, Loginicheva E, Chmielewski K, Stewart KM et al (2015) Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization. Immunity 42(3):419–430. https://doi.org/10.1016/j.immuni.2015.02.005
Jiang B (2017) Aerobic glycolysis and high level of lactate in cancer metabolism and microenvironment. Genes Dis 4(1):25–27. https://doi.org/10.1016/j.gendis.2017.02.003
Kamei Y, Hatazawa Y, Uchitomi R, Yoshimura R, Miura S (2020) Regulation of skeletal muscle function by amino acids. Nutrients 12(1):261. https://doi.org/10.3390/nu12010261
Kang JO, Jones C, Brothwell B (1998) Toxicity associated with iron overload found in hemochromatosis: possible mechanism in a rat model. Clin Lab Sci 11(6):350
Kang R, Zeh HJ, Lotze MT, Tang D (2011) The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 18(4):571–580. https://doi.org/10.1038/cdd.2010.191
Kimmelman AC, White E (2017) Autophagy and tumor metabolism. Cell Metab 25(5):1037–1043. https://doi.org/10.1016/j.cmet.2017.04.004
Klebanov GI, Teselkin YuO, Babenkova IV, Lyubitsky OB, Rebrova O, Boldyrev AA, Vladimirov YuA (1998) Effect of carnosine and its components on free-radical reactions. Membr Cell Biol 12(1):89–99
Korf H, du Plessis J, van Pelt J, De Groote S, Cassiman D, Verbeke L, Ghesquiere B, Fendt SM et al (2019) Inhibition of glutamine synthetase in monocytes from patients with acute-on-chronic liver failure resuscitates their antibacterial and inflammatory capacity. Gut 68(10):1872–1883. https://doi.org/10.1136/gutjnl-2018-316888
Krebs HA, Johnson WA (1980) The role of citric acid in intermediate metabolism in animal tissues. FEBS Lett 117(Suppl):K1-10. https://doi.org/10.4159/harvard.9780674366701.c143
Kumar V, Gill KD (2009) Aluminium neurotoxicity: neurobehavioural and oxidative aspects. Arch Toxicol 83(11):965–978. https://doi.org/10.1007/s00204-009-0455-6
Kumar V, Gill KD (2014) Oxidative stress and mitochondrial dysfunction in aluminium neurotoxicity and its amelioration: a review. Neurotoxicology 41:154–166. https://doi.org/10.1016/j.neuro.2014.02.004
Lachowicz JI, Nurchi VM, Fanni D, Gerosa C, Peana M, Zoroddu MA (2014) Nutritional iron deficiency: the role of oral iron supplementation. Curr Med Chem 21(33):3775–3784. https://doi.org/10.2174/0929867321666140706143925
Lautrup S, Sinclair DA, Mattson MP, Fang EF (2019) NAD(+) in brain aging and neurodegenerative disorders. Cell Metab 30(4):630–655. https://doi.org/10.1016/j.cmet.2019.09.001
Lee J, Park JR, Lee H, Jang S, Ryu SM, Kim H, Kim D, Jang A et al (2018) L-carnosine induces apoptosis/cell cycle arrest via suppression of NF-kappaB/STAT1 pathway in HCT116 colorectal cancer cells. Vitro Cell Dev Biol Anim 54(7):505–512. https://doi.org/10.1007/s11626-018-0264-4
Lemire J, Appanna VD (2011) Aluminum toxicity and astrocyte dysfunction: a metabolic link to neurological disorders. J Inorg Biochem 105(11):1513–1517. https://doi.org/10.1016/j.jinorgbio.2011.07.001
Liberti MV, Locasale JW (2016) The Warburg effect: how does it benefit cancer cells? Trends Biochem Sci 41(3):211–218. https://doi.org/10.1016/j.tibs.2015.12.001
Lignitto L, LeBoeuf SE, Homer H, Jiang S, Askenazi M, Karakousi TR, Pass HI, Bhutkar AJ et al (2019) Nrf2 activation promotes lung cancer metastasis by inhibiting the degradation of Bach1. Cell 178(2):316–32918. https://doi.org/10.1016/j.cell.2019.06.003
Linker RA, Lee DH, Ryan S, van Dam AM, Conrad R, Bista P, Zeng W, Hronowsky X et al (2011) Fumaric acid esters exert neuroprotective effects in neuroinflammation via activation of the Nrf2 antioxidant pathway. Brain 134(Pt 3):678–692. https://doi.org/10.1093/brain/awq386
Liu X, Romero IL, Litchfield LM, Lengyel E, Locasale JW (2016) Metformin targets central carbon metabolism and reveals mitochondrial requirements in human cancers. Cell Metab 24(5):728–739. https://doi.org/10.1016/j.cmet.2016.09.005
Lu M-C, Ji J-A, Jiang Z-Y, You Q-D (2016) The Keap1–Nrf2–ARE pathway as a potential preventive and therapeutic target: an update. Med Res Rev 36(5):924–963. https://doi.org/10.1002/med.21396
Lushchak OV, Piroddi M, Galli F, Lushchak VI (2014) Aconitase post-translational modification as a key in linkage between Krebs cycle, iron homeostasis, redox signaling, and metabolism of reactive oxygen species. Redox Rep 19(1):8–15. https://doi.org/10.1179/1351000213Y.0000000073
Macedo LW, Cararo JH, Maravai SG, Goncalves CL, Oliveira GM, Kist LW, Guerra Martinez C, Kurtenbach E et al (2016) Acute carnosine administration increases respiratory chain complexes and citric acid cycle enzyme activities in cerebral cortex of young rats. Mol Neurobiol 53(8):5582–5590. https://doi.org/10.1007/s12035-015-9475-9
Mailloux RJ, Willmore WG (2014) S-glutathionylation reactions in mitochondrial function and disease. Front Cell Dev Biol 2:68. https://doi.org/10.3389/fcell.2014.00068
Mailloux RJ, Hamel R, Appanna VD (2006) Aluminum toxicity elicits a dysfunctional TCA cycle and succinate accumulation in hepatocytes. J Biochem Mol Toxicol 20(4):198–208. https://doi.org/10.1002/jbt.20137
Mailloux RJ, Beriault R, Lemire J, Singh R, Chenier DR, Hamel RD, Appanna VD (2007) The tricarboxylic acid cycle, an ancient metabolic network with a novel twist. PLoS ONE 2(8):e690. https://doi.org/10.1371/journal.pone.0000690
Mailloux RJ, McBride SL, Harper ME (2013) Unearthing the secrets of mitochondrial ROS and glutathione in bioenergetics. Trends Biochem Sci 38(12):592–602. https://doi.org/10.1016/j.tibs.2013.09.001
Mailloux RJ, Jin X, Willmore WG (2014) Redox regulation of mitochondrial function with emphasis on cysteine oxidation reactions. Redox Biol 2:123–139. https://doi.org/10.1016/j.redox.2013.12.011
Mews P, Donahue G, Drake AM, Luczak V, Abel T, Berger SL (2017) Acetyl-CoA synthetase regulates histone acetylation and hippocampal memory. Nature 546(7658):381–386. https://doi.org/10.1038/nature22405
Nelson DL, Lehninger AL, Cox MM (2017) The citric acid cycle. Lehninger principles of biochemistry, 7th edn. Macmillan, New York, pp 619–624
Nurchi VM, Crisponi G, Lachowicz JI, Medici S, Peana M, Zoroddu MA (2016) Chemical features of in use and in progress chelators for iron overload. J Trace Elem Med Biol 38:10–18. https://doi.org/10.1016/j.jtemb.2016.05.010
Nurchi VM, Cappai R, Chand K, Chaves S, Gano L, Crisponi G, Peana M, Zoroddu MA et al (2019) New strong extrafunctionalizable tris(3,4-HP) and bis(3,4-HP) metal sequestering agents: synthesis, solution and in vivo metal chelation. Dalton Trans 48(43):16167–16183. https://doi.org/10.1039/c9dt02905b
Oexle H, Gnaiger E, Weiss G (1999) Iron-dependent changes in cellular energy metabolism: influence on citric acid cycle and oxidative phosphorylation. Biochim Biophys Acta 1413(3):99–107. https://doi.org/10.1016/s0005-2728(99)00088-2
Oppermann H, Schnabel L, Meixensberger J, Gaunitz F (2016) Pyruvate attenuates the anti-neoplastic effect of carnosine independently from oxidative phosphorylation. Oncotarget 7(52):85848–85860. https://doi.org/10.18632/oncotarget.13039
Ott P, Clemmesen O, Larsen FS (2005) Cerebral metabolic disturbances in the brain during acute liver failure: from hyperammonemia to energy failure and proteolysis. Neurochem Int 47(1–2):13–18. https://doi.org/10.1016/j.neuint.2005.04.002
Owen OE, Kalhan SC, Hanson RW (2002) The key role of anaplerosis and cataplerosis for citric acid cycle function. J Biol Chem 277(34):30409–30412. https://doi.org/10.1074/jbc.R200006200
Palmieri F (2004) The mitochondrial transporter family (SLC25): physiological and pathological implications. Pflugers Arch 447(5):689–709. https://doi.org/10.1007/s00424-003-1099-7
Paumen MB, Ishida Y, Muramatsu M, Yamamoto M, Honjo T (1997) Inhibition of carnitine palmitoyltransferase I augments sphingolipid synthesis and palmitate-induced apoptosis. J Biol Chem 272(6):3324–3329. https://doi.org/10.1074/jbc.272.6.3324
Pietrocola F, Galluzzi L, Bravo-San Pedro JM, Madeo F, Kroemer G (2015) Acetyl coenzyme A: a central metabolite and second messenger. Cell Metab 21(6):805–821. https://doi.org/10.1016/j.cmet.2015.05.014
Prokopieva VD, Yarygina EG, Bokhan NA, Ivanova SA (2016) Use of carnosine for oxidative stress reduction in different pathologies. Oxid Med Cell Longev 2016:2939087–2939087. https://doi.org/10.1155/2016/2939087
Reid MA, Paik J, Locasale JW (2017) A missing link to vitamin B12 metabolism. Cell 171(4):736–737. https://doi.org/10.1016/j.cell.2017.10.030
Rich PR (2003) The molecular machinery of Keilin’s respiratory chain. Biochem Soc Trans 31(Pt 6):1095–1105. https://doi.org/10.1042/bst0311095
Roh JL, Jang H, Kim EH, Shin D (2017) Targeting of the glutathione, thioredoxin, and Nrf2 antioxidant systems in head and neck cancer. Antioxid Redox Signal 27(2):106–114. https://doi.org/10.1089/ars.2016.6841
Ross KL, Eisenstein RS (2002) Iron deficiency decreases mitochondrial aconitase abundance and citrate concentration without affecting tricarboxylic acid cycle capacity in rat liver. J Nutr 132(4):643–651. https://doi.org/10.1093/jn/132.4.643
Ruban A, Malina KC, Cooper I, Graubardt N, Babakin L, Jona G, Teichberg VI (2015) Combined treatment of an amyotrophic lateral sclerosis rat model with recombinant GOT1 and oxaloacetic acid: a novel neuroprotective treatment. Neurodegener Dis 15(4):233–242. https://doi.org/10.1159/000382034
Ryan DG, Murphy MP, Frezza C, Prag HA, Chouchani ET, O’Neill LA, Mills EL (2019) Coupling Krebs cycle metabolites to signalling in immunity and cancer. Nat Metab 1:16–33. https://doi.org/10.1038/s42255-018-0014-7
Saggerson D (2008) Malonyl-CoA, a key signaling molecule in mammalian cells. Annu Rev Nutr 28:253–272. https://doi.org/10.1146/annurev.nutr.28.061807.155434
Sajnani K, Islam F, Smith RA, Gopalan V, Lam AK (2017) Genetic alterations in Krebs cycle and its impact on cancer pathogenesis. Biochimie 135:164–172. https://doi.org/10.1016/j.biochi.2017.02.008
Sale C, Artioli GG, Gualano B, Saunders B, Hobson RM, Harris RC (2013) Carnosine: from exercise performance to health. Amino Acids 44(6):1477–1491. https://doi.org/10.1007/s00726-013-1476-2
Sawa K, Uematsu T, Korenaga Y, Hirasawa R, Kikuchi M, Murata K, Zhang J, Gai X et al (2017) Krebs cycle intermediates protective against oxidative stress by modulating the level of reactive oxygen species in neuronal HT22 cells. Antioxidants (Basel) 6(1):21. https://doi.org/10.3390/antiox6010021
Schranner D, Kastenmuller G, Schonfelder M, Romisch-Margl W, Wackerhage H (2020) Metabolite concentration changes in humans after a bout of exercise: a systematic review of exercise metabolomics studies. Sports Med Open 6(1):11. https://doi.org/10.1186/s40798-020-0238-4
Severina I, Bussygina O, Pyatakova N (2000) Carnosine as a regulator of soluble guanylate cyclase. Biochemistry (Moscow) 65(7):783–788
Shakoury-Elizeh M, Protchenko O, Berger A, Cox J, Gable K, Dunn TM, Prinz WA, Bard M et al (2010) Metabolic response to iron deficiency in Saccharomyces cerevisiae. J Biol Chem 285(19):14823–14833. https://doi.org/10.1074/jbc.M109.091710
Shaw RJ, Lamia KA, Vasquez D, Koo SH, Bardeesy N, Depinho RA, Montminy M, Cantley LC (2005) The kinase LKB1 mediates glucose homeostasis in liver and therapeutic effects of metformin. Science 310(5754):1642–1646. https://doi.org/10.1126/science.1120781
Shen H, Campanello GC, Flicker D, Grabarek Z, Hu J, Luo C, Banerjee R, Mootha VK (2017) The human knockout gene CLYBL connects itaconate to vitamin B12. Cell 171(4):771-782e11. https://doi.org/10.1016/j.cell.2017.09.051
Singh KK, Desouki MM, Franklin RB, Costello LC (2006) Mitochondrial aconitase and citrate metabolism in malignant and nonmalignant human prostate tissues. Mol Cancer 5:14. https://doi.org/10.1186/1476-4598-5-14
Solomon LR (2007) Disorders of cobalamin (vitamin B12) metabolism: emerging concepts in pathophysiology, diagnosis and treatment. Blood Rev 21(3):113–130. https://doi.org/10.1016/j.blre.2006.05.001
Starai VJ, Escalante-Semerena JC (2004) Acetyl-coenzyme A synthetase (AMP forming). Cell Mol Life Sci 61(16):2020–2030. https://doi.org/10.1007/s00018-004-3448-x
Stepien K, Ostrowski RP, Matyja E (2016) Hyperbaric oxygen as an adjunctive therapy in treatment of malignancies, including brain tumours. Med Oncol 33(9):101. https://doi.org/10.1007/s12032-016-0814-0
Strydom C, Robinson C, Pretorius E, Whitcutt J, Marx J, Bornman M (2006) The effect of selected metals on the central metabolic pathways in biology: a review. Water Sa 32(4):543–554
Swanson KV, Deng M, Ting JP (2019) The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat Rev Immunol 19(8):477–489. https://doi.org/10.1038/s41577-019-0165-0
Tannahill GM, Curtis AM, Adamik J, Palsson-McDermott EM, McGettrick AF, Goel G, Frezza C, Bernard NJ et al (2013) Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature 496(7444):238–242. https://doi.org/10.1038/nature11986
Toyoshima S, Watanabe F, Saido H, Pezacka EH, Jacobsens DW, Miyatake K, Nakano Y (1996) Accumulation of methylmalonic acid caused by vitamin B12-deficiency disrupts normal cellular metabolism in rat liver. Br J Nutr 75(6):929–938. https://doi.org/10.1079/bjn19960198
Tretter L, Adam-Vizi V (2000) Inhibition of Krebs cycle enzymes by hydrogen peroxide: a key role of [alpha]-ketoglutarate dehydrogenase in limiting NADH production under oxidative stress. J Neurosci 20(24):8972–8979
Vallee A, Vallee JN (2018) Warburg effect hypothesis in autism Spectrum disorders. Mol Brain 11(1):1. https://doi.org/10.1186/s13041-017-0343-6
Vander Heiden MG, Cantley LC, Thompson CB (2009) Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324(5930):1029–1033. https://doi.org/10.1126/science.1160809
Vishnyakova K, Babizhayev M, Aliper A, Buzdin A, Kudryavzeva A, Yegorov Y (2014) Stimulation of cell proliferation by carnosine: Cell and transcriptome approaches. Mol Biol (Mosk) 48(5):718–726
Wagenmakers A (1998) Muscle amino acid metabolism at rest and during exercise: role in human physiology and metabolism. Exerc Sport Sci Rev 26:287–314
Wahl D, Anderson RM, Le Couteur DG (2019) Anti-aging therapies, cognitive impairment and dementia. J Gerontol A Biol Sci Med Sci. https://doi.org/10.1093/gerona/glz135
Wallace DC (2005) The mitochondrial genome in human adaptive radiation and disease: on the road to therapeutics and performance enhancement. Gene 354:169–180. https://doi.org/10.1016/j.gene.2005.05.001
Waniewski RA, Martin DL (1998) Preferential utilization of acetate by astrocytes is attributable to transport. J Neurosci 18(14):5225–5233. https://doi.org/10.1523/jneurosci.18-14-05225.1998
Wei YH, Lee HC (2002) Oxidative stress, mitochondrial DNA mutation, and impairment of antioxidant enzymes in aging. Exp Biol Med (Maywood) 227(9):671–682. https://doi.org/10.1177/153537020222700901
Williams NC, O’Neill LAJ (2018) A role for the Krebs cycle intermediate citrate in metabolic reprogramming in innate immunity and inflammation. Front Immunol 9:141. https://doi.org/10.3389/fimmu.2018.00141
Xue Y-N, Liu Y-N, Su J, Li J-L, Wu Y, Guo R, Yu B-B, Yan X-Y et al (2019) Zinc cooperates with p53 to inhibit the activity of mitochondrial aconitase through reactive oxygen species accumulation. Cancer Med 8(5):2462–2473. https://doi.org/10.1002/cam4.2130
Yalcin A, Telang S, Clem B, Chesney J (2009) Regulation of glucose metabolism by 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatases in cancer. Exp Mol Pathol 86(3):174–179. https://doi.org/10.1016/j.yexmp.2009.01.003
Yang C, Ko B, Hensley CT, Jiang L, Wasti AT, Kim J, Sudderth J, Calvaruso MA et al (2014) Glutamine oxidation maintains the TCA cycle and cell survival during impaired mitochondrial pyruvate transport. Mol Cell 56(3):414–424. https://doi.org/10.1016/j.molcel.2014.09.025
Yudkoff M, Nelson D, Daikhin Y, Erecinska M (1994) Tricarboxylic acid cycle in rat brain synaptosomes. Fluxes and interactions with aspartate aminotransferase and malate/aspartate shuttle. J Biol Chem 269(44):27414–27420
Zatta P, Lain E, Cagnolini C (2000) Effects of aluminum on activity of Krebs cycle enzymes and glutamate dehydrogenase in rat brain homogenate. Eur J Biochem 267(10):3049–3055. https://doi.org/10.1046/j.1432-1033.2000.01328.x
Zhang D, Li J, Wang F, Hu J, Wang S, Sun Y (2014) 2-Deoxy-d-glucose targeting of glucose metabolism in cancer cells as a potential therapy. Cancer Lett 355(2):176–183. https://doi.org/10.1016/j.canlet.2014.09.003
Zhang D, Mably AJ, Walsh DM, Rowan MJ (2017) Peripheral interventions enhancing brain glutamate homeostasis relieve amyloid beta- and TNFalpha-mediated synaptic plasticity disruption in the rat hippocampus. Cereb Cortex 27(7):3724–3735. https://doi.org/10.1093/cercor/bhw193
Zhao J, Posa DK, Kumar V, Hoetker D, Kumar A, Ganesan S, Riggs DW, Bhatnagar A et al (2019) Carnosine protects cardiac myocytes against lipid peroxidation products. Amino Acids 51(1):123–138. https://doi.org/10.1007/s00726-018-2676-6
Zoroddu MA, Aaseth J, Crisponi G, Medici S, Peana M, Nurchi VM (2019) The essential metals for humans: a brief overview. J Inorg Biochem 195:120–129. https://doi.org/10.1016/j.jinorgbio.2019.03.013
Zwingmann C, Leibfritz D, Hazell AS (2004) Brain energy metabolism in a sub-acute rat model of manganese neurotoxicity: an ex vivo nuclear magnetic resonance study using [1-13C]glucose. Neurotoxicology 25(4):573–587. https://doi.org/10.1016/j.neuro.2003.08.002
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gasmi, A., Peana, M., Arshad, M. et al. Krebs cycle: activators, inhibitors and their roles in the modulation of carcinogenesis. Arch Toxicol 95, 1161–1178 (2021). https://doi.org/10.1007/s00204-021-02974-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-021-02974-9