Abstract
Despite well-known adverse effects associated with cigarette smoking, approximately 20% of the US population continues to smoke and many more are exposed to environmental tobacco smoke. Many of the same individuals are also exposed to environmental neurotoxic chemicals such as the organophosphorus insecticide chlorpyrifos. In the present study, the effects of exposure to low doses of nicotine and chlorpyrifos alone and in combination, were studied on the central cholinergic system and sensorimotor performance in rats. Male Sprague-Dawley rats (250–300 g) were treated with nicotine (1 mg/kg s.c., in normal saline), chlorpyrifos (0.1 mg/kg dermally, in 0.1 ml 70% ethanol), or a combination of both, daily for 30 days. Control rats were treated with saline and dermally with ethanol. Sensorimotor behavior was evaluated 24 h following the last dose using a battery of tests. There was a significant deficit in incline plane performance, beam-walk score and beam-walk time following exposure to each chemical, alone or in combination. The deficit in incline plane performance was greater when the two chemicals were given in combination than with either compound alone. Biochemical analysis showed a decrease in cerebellar and an increase in midbrain acetylcholinesterase (AChE) activity following combined exposure. Exposure to nicotine alone resulted in a significant increase in AChE activity in brainstem and midbrain, whereas there was no significant change after exposure to chlorpyrifos, alone. A significant increase in ligand binding to nicotinic acetylcholine receptors (nAChR) was observed in brainstem and cortex following exposure to nicotine or chlorpyrifos. This was further augmented with combined exposure, which caused a modest but significant increase in m2 muscarinic acetylcholine receptors (m2-mAChR) ligand binding in the cortex. These data suggest that exposure to either nicotine or chlorpyrifos or a combination of the two may impair neurobehavioral performance and affect the central nervous system cholinergic pathways.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Despite well-known adverse health effects of cigarette smoking, approximately 20% of the US population continues to smoke (US DHHS 2000). In addition to individuals' exposure to nicotine through cigarette smoking, many are also exposed to this chemical through environmental smoke and transiently via various preparations used for smoking cessation (Benowitz 1996). The general population is also exposed to organophosphorus insecticides such as chlorpyrifos (Clegg and van Gemert 1999). Both chemicals exert their toxic effects primarily by affecting the cholinergic pathway (Slotkin 1999).
Nicotine (3-[1-methyl-2-pyrrolidinyl] pyridine) is a tertiary amine consisting of a pyridine and a pyrrolidine ring. Once absorbed, nicotine enters the circulation and distributes rapidly to different tissues including brain, where it is a direct cholinergic agonist at the nicotinic acetylcholine receptor (nAChR) site, resulting in behavioral (Martin and Becker 1970) and cellular effects (Slotkin 1999) consistent with cholinergic over-stimulation. Inhaled or dermally applied nicotine travels directly to the brain, bypassing the liver, and readily crosses the blood–brain barrier (Benowitz 1996). Several metabolites of nicotine are found in rat brain after peripheral administration (Miskys et al. 2000).
Behaviorally, a single dose of nicotine causes biphasic effects on locomotion that are characterized by an initial decrease in activity followed by a period of hyperactivity (Stolerman et al. 1995). Following repeated exposure, nicotine results in an increase in locomotor activity because of a locomotor desensitization that is mediated by nAChR (Stolerman et al. 1995; Booze et al. 1999).
Chlorpyrifos (O,O-diethyl-O-[3,5,6trichloro-2-pyridinyl]phosphorothioate) is one of the most commonly used insecticides in the USA today. Concerns about adverse health effects of chlorpyrifos exposure in infants have recently led to restriction of its domestic use (Davis and Ahmed 1998; Gurunathan et al. 1998). Chlorpyrifos is metabolized to chlorpyrifos oxon, which inhibits acetylcholinesterase (AChE) resulting in the accumulation of acetylcholine at acetylcholine receptors and subsequent development of cholinergic over-stimulation (Abou-Donia 1992). Chlorpyrifos also exhibits cholinergic agonist-like actions, opening and then desensitizing nicotinic cholinergic receptor/ion channels (Katz et al. 1997). It interacts with signaling components, such as G proteins and adenylate cyclase involving muscarinic acetylcholine receptors (Huff et al. 1994). Large doses of chlorpyrifos produce organophosphate-induced delayed neuropathy (OPIDN) in humans (Aiuto et al. 1993) and other sensitive animal species, e.g., in the hen (Fikes et al. 1992; Abou-Donia 1992). Recently, impairments in various neurological functions of termiticide applicators exposed to low dose of chlorpyrifos have been reported (Steenland et al. 2000).
Combined neurotoxicity can result from simultaneous exposure to different chemicals. Although nicotine has been linked to a variety of harmful health effects associated with cigarette smoking, environmental exposures to other cholinotoxic chemicals such as organophosphorus insecticides could also play a role in tobacco-induced illnesses.
In the present study, we evaluated sensorimotor performance, brain regional AChE activity, and nicotinic and m2 muscarininc acetylcholine receptor ligand binding following daily treatment with nicotine and chlorpyrifos, alone and in combination, in adult male Sprague-Dawley rats. The results suggest that exposure to nicotine and chlorpyrifos, alone and in combination, results in sensorimotor impairments and affects the central nervous system cholinergic function.
Materials and methods
Materials
Nicotine bitartrate, butyrylthiocholine iodide, and acetylthiocholine iodide were obtained from Sigma Chemical Co (St. Louis, Mo., USA). Chlorpyrifos (~99% purity) was purchased from Chem Service (West Chester, Pa., USA). [3H]Cytisine (specific activity 15 Ci/mmol) and [3H]AF-DX 384[2,3-dipropylamino-3H] (specific activity 100 Ci/mmol) was obtained from PerkinElmer Life Sciences formerly known as NEN (Boston, Ma., USA).
Procedures
Male Sprague-Dawley rats (200–250 g) were obtained from Zivic-Miller Laboratories (Allison Park, Pa., USA) and housed in Duke University Medical Center vivarium on 12 h dark–light cycle. The animals were allowed food and water ad libitum. The rats were treated between 7:30 and 11:00 a.m. daily. All treatments and procedures were carried out strictly according to Duke University Medical Center Institutional Animal Care and Use Committee guidelines.
Rats were randomly allocated into groups of five rats each. For dermal applications, the treatment was done at the back of the neck on a 1-square inch area preshaved with electric clippers. The rats were treated as follows:
- Control:
-
Animals received subcutaneously (s.c.) normal saline and dermal application of 70% ethanol, daily for 30 days
- Nicotine:
-
Animals received 1 mg/kg nicotine s.c. in normal saline, daily for 30 days
- Chlorpyrifos:
-
Animals received 0.1 mg/kg chlorpyrifos in 0.1 ml 70% ethanol by dermal application, daily for 30 days
- Nicotine and chlorpyrifos:
-
Animals received 1 mg/kg nicotine in normal saline s.c., and dermal application of chlorpyrifos (0.1 mg/kg) in 0.1 ml 70% ethanol, daily for 30 days
After behavioral testing had been performed, the animals were anesthetized with 0.2 ml of ketamine/xylazine (100 mg/kg ketamine, 15 mg/kg xylazine) and blood was withdrawn into heparinized syringes. Brains were removed and washed thoroughly with ice-cold normal saline. Cerebral cortex, midbrain, cerebellum and brainstem were dissected on ice and rapidly frozen in liquid nitrogen. Plasma was separated and frozen at −80°C for enzyme studies.
Behavioral evaluations
The behavioral tests employed in these studies evaluate sensorimotor reflexes, motor strength, and coordination. All behavioral testing was performed 24 h after the last dose by an observer blind to the animal's treatment status, and was carried out in a sound-proof room with subdued lighting (less than 10.76 lumens/m2, ambient light) between 7:00 and 11:30 a.m.
Incline plane
Rats were placed on a flat plane in the horizontal position, with the head facing the side of the board to be raised (Yonemori et al. 1998; Abou-Donia et al. 2001). The angle that the rat began to slip downward was recorded. The results of the two trials were averaged.
Forepaw grip time
The rats' forepaw strength was assessed by having them grip a 5-mm diameter wood dowel that was held horizontally and raised so that the rat supported its body weight as described by Andersen et al. (1991) and Abou-Donia et al. (2001). Time to release grip was recorded in seconds. The results of the two trials were averaged.
Beam-walking
The testing apparatus was a 2.5×122 cm wooden beam elevated 75.5 cm above the floor with wooden supports, as described by Goldstein (1993) and Abou-Donia et al. (2001). Beam-walking ability was measured with a seven point scoring system scale as previously described by Goldstein (1993): at score 1, the rat is unable to place the hindpaws on the horizontal surface of the beam; 2, the rat places the hindpaws on the horizontal surface of the beam and maintains balance for at least 5 s; 3, the rat traverses the beam while dragging the hindpaws; 4, the rat traverses the beam and at least once places a hindpaw on the horizontal surface of the beam; 5, the rat crosses the beam and places a hindpaw on the horizontal surface of the beam to aid less than half its steps; 6, the rat uses the hindpaws to aid more than half its steps and; 7, the rat traverses the beam with no more than two footslips. In addition, the latency until the animal's nose entered the goal box (up to 90 s) was recorded. Rats that fall off of the beam or did not enter the goal box were assigned latencies of 90 s.
Statistical analyses
Data for the behavioral tests were compared among groups by one-way analysis of variance (ANOVA). If a significant difference was found, Fisher's LSD tests were applied to permit post hoc, pair-wise comparisons. A two-tailed P-value of <0.05 was considered statistically significant.
Cholinesterase determination
AChE in brain regions and butyrylcholinesterase (BChE) activities in plasma were determined according to the method of Ellman et al. (1961) modified for assay in a UV Max Kinetic Microplate Reader (Molecular Devices, Sunnyvale, Calif., USA) as previously described (Abou-Donia et al. 1996; Khan et al. 2000). Protein concentration was determined by the BCA method according to Smith et al. (1985). The enzyme activities are expressed as micromoles substrate hydrolyzed per minute per milligram protein for brain regions and nanomoles substrate hydrolyzed per minute per milligram protein for plasma (percentage of control).
Nicotinic acetylcholine receptor (nAChR) ligand binding assay
[3H]Cytisine at saturating concentration was used as specific ligand for binding studies with nAChR (Slotkin et al. 1999; Abou-Donia et al. 2001). The results are expressed as specific binding (femtomoles per milligram protein, as percentage of control).
Muscarinic acetylcholine receptor (mAChR) binding assay
The m2-mAChR binding assay was carried out by using the m2-selective ligand, [3H]AF-DX 384 (Slotkin et al. 1999; Khan et al. 2000). The results are expressed as specific binding (femtomoles per milligram protein, as percentage of control).
Statistical analysis
The results were analyzed by one-way ANOVA, followed by Dunnett's multiple comparison test. A P-value <0.05 was considered significant.
Results
Clinical signs
The rats were observed daily for the development of clinical signs of toxicity throughout the treatment period. No rats died. All animals from control and treated groups gained weight. The rats treated with nicotine alone, or in combination with chlorpyrifos, had a higher but non-significant body weight gain compared with either those treated with chlorpyrifos alone or the controls. No sign of overt cholinergic toxicity was observed during the entire period of exposure in any group of animals.
Behavioral assessments
Except for incline plane performance (in which only the combination differed from control), each chemical or the combination severely impaired each behavioral measure (Fig. 1) (beam-walk score F (3,15)=90, P<0.00001, ANOVA; beam walk time F (3,15)=20, P<0.00002, ANOVA; incline plane F (3,15)=5, P=0.015, ANOVA; grip time F (3,15)=8, P=0.002, ANOVA).
Effect of treatment with chlorpyrifos and nicotine, alone or in combination on sensorimotor performance. Rats were treated with nicotine (CPF, 1 mg/kg s.c.) and chlorpyrifos (NIC, 0.1 mg/kg dermally), alone or in combination (CPF+NIC), for 30 days. Twenty-four hours after the last dose, the animals were tested for Beam-Walk Score, Beam-Walk Time, Incline Plane performance and Grip Time. At least one treatment on each behavioral test resulted in a significant impairment (P<0.05, ANOVA, see text) as compared with controls (Cont.). The data are presented as means ±SE, n=5. P-values represent post hoc tests by pair-wise Fisher LSD test (NS not significant)
Chlorpyrifos and nicotine alone impaired beam-walk scores and beam-walk times (all P<0.001 and P<0.005, respectively compared with controls). Exposure to chlorpyrifos alone and chlorpyrifos plus nicotine caused poorer performances on these tests than nicotine alone. There was no significant difference, however, in chlorpyrifos alone versus chlorpyrifos plus nicotine for these parameters. There was no significant change on incline plane performance induced by chlorpyrifos or nicotine treatment alone; however, rats that received both chemicals had poorer performances compared with either controls (P<0.003, Fisher LSD) or each chemical given alone (P<0.04). Grip time was severely impaired by each treatment, with no significant differences among those given chlorpyrifos alone, nicotine alone, and chlorpyrifos plus nicotine.
Effect of treatment with nicotine and chlorpyrifos, alone and in combination, on brain regional AChE and plasma BChE activity
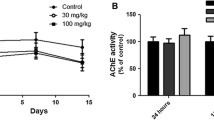
Given alone, chlorpyrifos exposure did not result in a significant change in the AChE activity in the various brain regions (Fig. 2). There was a significant increase in AChE activity in brainstem and midbrain (~141 and 129% of control, respectively, P<0.05) following treatment with nicotine alone. Co-exposure with chlorpyrifos and nicotine caused a significant increase (~142% of control, P<0.05) in midbrain and a decrease in AChE activity in cerebellum (~84% of control, P<0.05). Plasma BChE activity was not changed by any treatment.
Effect of treatment with chlorpyrifos and nicotine, alone or in combination, on acetylcholinesterase activity in different brain regions. Rats were treated with nicotine (NICOT, 1 mg/kg s.c.) and chlorpyrifos (CPF, 0.1 mg/kg dermally), alone and in combination (CPF/NIC), for 30 days. Twenty-four hours after the last dose, the animals were killed and enzyme activity in plasma and brain regions was determined. The control activity expressed as nanomoles acetylthiocholine hydrolyzed per minute per milligram protein was 860±180 for cortex, 618±70 for brainstem, 543±29 for midbrain, and 239±12 for cerebellum, and the plasma control activity was 21±2 nmol butyrylthiocholine hydrolyzed/min per mg protein. Data are presented as means ±SE (percentage of control), n=5. *Statistically significant compared with control
Effect of treatment with nicotine and chlorpyrifos, alone or in combination, on nAChR ligand binding in the cortex and brainstem
Exposure to nicotine or chlorpyrifos alone resulted in a significant increase in the ligand binding densities for nAChR in the brainstem (~183 and 178% of control, respectively, P<0.05; Fig. 3). Co-exposure to both agents resulted in a significant increase in the ligand binding in the cortex as well as brainstem (~123 and 223% of control, respectively, P<0.05).
Effect of treatment with chlorpyrifos and nicotine, alone or in combination, on nicotinic acetylcholine receptor ligand binding in the cortex and brainstem. Rats were treated with nicotine (NICOT, 1 mg/kg s.c.) and chlorpyrifos (CPF, 0.1 mg/kg dermally), alone and in combination (CPF/NIC), for 30 days. Twenty-four hours after the last dose, the animals were killed and ligand binding in the membrane fraction of brainstem and cortex was determined using [3H]cytisine. The control [3H]cytisine binding was 19.5±0.62 and 6.9±1.7 fmol/min per mg protein for cortex and brainstem, respectively. Data are presented as means ±SE (percentage of control), n=5. *Statistically significant compared with control
Effect of treatment with nicotine and chlorpyrifos, alone or in combination, on m2-mAChR ligand binding in the cortex and brainstem
Treatment with nicotine and chlorpyrifos, alone and in combination resulted in a significant increase in m2-mAChR ligand binding in the cortex (~127–135% of control, P<0.05) (Fig. 4). No significant changes were observed in the brainstem following any treatment.
Effect of treatment with chlorpyrifos and nicotine, alone or in combination, on m2 muscarinic acetylcholine receptor ligand binding in the cortex and brainstem. Rats were treated with nicotine (NICOT, 1 mg/kg s.c.) and chlorpyrifos (CPF, 0.1 mg/kg dermally), alone and in combination (CPF/NIC), for 30 days. Twenty-four hours after the last dose, the animals were killed and ligand binding was determined using [3H]AF-DX 384. The control [3H]AF-DX 384 binding was 855±92 and 36±9.1 fmol/min per mg protein for the cortex and brainstem, respectively. Data are presented as means ±SE (percentage of control), n=5. *Statistically significant compared with control
Discussion
Many individuals are exposed to a variety of chemicals at doses below the threshold that causes any acute toxicity. Tobacco smokers are directly exposed to nicotine. The same individuals may also be exposed to chlorpyrifos, either because of environmental contamination or occupational exposure. The present study was carried out to determine whether there is the possibility that neurotoxic effects following co-exposure to low doses of nicotine and chlorpyrifos could result in health consequences that are different from those caused by exposure to either chemical alone. Our results show that exposure to low doses of nicotine and chlorpyrifos given alone may cause neurobehavioral deficits as well as differential changes in the central cholinergic system, whereas combined exposure did not result in augmented deficits in most of the parameters studied.
We found that each chemical caused significant impairments of sensorimotor performance, presumably through the central cholinergic effects. However, previous work (Nostrandt et al. 1997) did not show a correlation between the extent of AChE inhibition and behavioral abnormalities. Although our studies were carried out using a low dose of chlorpyrifos (0.1 mg/kg for 30 days) by dermal application, the studies by Nostrandt et al. (1997) were carried out with oral dose of relatively higher doses of chlorpyrifos. High-dose nicotine exposure for 8–10 weeks has been shown to cause inhibition of AChE activity (Larsson et al. 1980). The low dose of nicotine (1 mg/kg s.c.) used in our studies may have caused metabolic changes leading to the increased activity. Repeated dosing with chlorpyrifos caused slow accumulation of AChE protein in rat brain (Chiappa, et al. 1995). Thus, it is possible that co-exposure to nicotine and chlorpyrifos might have resulted in neurobehavioral changes due to an increase in AChE. It has also been shown that increased AChE protein may reflect an increased axonal repair and synaptic modeling (Sternfeld et al. 1998). Therefore, it is also possible that an increased activity in midbrain following co-exposure to nicotine and chlorpyrifos may cause subtle changes that are reflected in increased synaptic modeling and repair.
Neuronal nicotinic receptors are large ligand-gated ion channels comprised of various combinations of α and β subunits (McGehee 1999). The predominant high affinity nicotinic receptor protein is composed of α4β2 receptor subtypes (Flores et al. 1992), and the subunit composition determines the rate of desensitization following low-level nicotine exposure (Fenster et al. 1997). Upregulation of this receptor subtype is initiated by receptor desensitization after chronic exposure to nicotine (Fenster et al. 1999). It is believed that both acute and chronic nicotine treatment cause differential regulation of various nAChRs that may be relevant for the nicotine-induced tolerance, dependence, and withdrawal symptoms (Dani and Heinmann 1996). Consistent with the published reports (Benwell et al. 1988; Slotkin et al. 1999), we also observed a significant increase in the ligand binding for α4β2 nAChR subtype (Fig. 3). It is also possible that other subtypes, such as α7 may also be upregulated, as reported earlier (Hellstrom-Lindahl et al. 2001), because [3H]cytisine has highest affinity for nicotinic acetylcholine receptors containing α4β2 subunits.
Our results also show that treatment with chlorpyrifos, alone or in combination with nicotine, resulted in an increased ligand binding for m2-mAChR in the cortex, whereas treatment with nicotine alone did not cause a significant change (Fig. 4). We have previously shown that chlorpyrifos and its bioactivated species (chlorpyrifos oxon) bind directly to m2-mAChR, and activate down stream signaling events (Huff et al. 1994). Studies by Ward et al. (1993) have also shown that organophosphate anticholinesterases selectively regulate m2-mAChR ligand binding. Chaudhuri et al. (1993) reported an increased m2-mAChR ligand binding in response to chlorpyrifos exposure. It is known that treatment with muscarinic antagonists induce receptor upregulation (Ben-Barak and Dudai 1980). Wang et al. (1996) reported an increase in muscarinic receptor ligand binding by repeated exposure to nicotine. Increased ligand binding for m2 muscarinic receptor results in inhibition of adenylate cyclase activity through a pertussis toxin-sensitive G protein, resulting in an inhibitory postsynaptic response (Wess 1996). Increased m2-mAChR receptor ligand binding density in the cortex in response to treatment with chlorpyrifos, alone and in combination with nicotine, could also reflect a compensatory mechanism for a reduced ability of these receptors to bind their respective ligands due to desensitiztion.
In summary, these results suggest that daily exposure for 30 days to low-dose chlorpyrifos and/or nicotine results in sensorimotor impairments with co-exposure causing a deficit in incline plane performance not present when each chemical is given alone. These changes may be related to the capacity of these chemicals to directly affect the cholinergic system in a brain region-specific manner. Furthermore, our data show that sub-chronic exposure to low doses of nicotine and chlorpyrifos, alone or in combination, may lead to a differential alteration in the cholinergic system that may be relevant in assessing the health risk associated with exposure to each of these agents in general population. Further work is necessary in order to elucidate the mechanism of neurobehavioral deficits following exposure to these compounds.
References
Abou-Donia MB (1992) Pesticides. In: Abou-Donia MB (ed.) Neurotoxicology. CRC Press, Boca Raton FL, pp 437–478
Abou-Donia MB, Wilmarth KR, Jensen KF, Oehme FM Kurt TL (1996) Neurotoxicity resulting from coexposure to pyridostigmine bromide, DEET, and permethrin. J Toxicol Environ Health 48:35–56
Abou-Donia MB, Goldstein LB, Jones KH, Abdel-Rehman AA, Damodran TV, Dechkovskaia AM, Bullman SL, Amir BE Khan WA (2001) Locomotor and sensorimotor performance deficit in rats following exposure to pyridostigmine bromide, DEET, and permethrin, alone and in combination. Toxicol Sci 60:305–314
Aiuto LA, Pavlakis SG, Boxer RA (1993) Life-threatening organophosphate-induced delayed polyneuropathy in a child after accidental chlorpyrifos ingestion. J Pediatr 122:658–660
Andersen CS, Andersen AB, Finger S (1991) Neurological correlates of unilateral and bilateral "strokes" of the middle cerebral artery in the rat. Physiol Behav 50:263–269
Ben-Barak J, Dudai Y (1980) Scopolamine induces an increase in muscarinic receptor level in rat hippocampus. Brain Res 193:309–313
Benowitz NL (1996) Pharmacology of nicotine: addiction and therapeutics. Annu Rev Pharmacol Toxicol 36:597–613
Benwell ME, Balfour DJ, Anderson JM (1988) Evidence that tobacco smoking increases the density of (−)-[3H]nicotine binding sites in human brain. J Neurochem 50:1243–1247
Booze RM, Welch MA Wood ML, Billings KA Apple SR, Mactutus CF (1999) Behavioral sensitization following repeated intravenous nicotine administration: gender differences and gonadal hormones. Pharmacol Biochem Behav 64:827–839
Chaudhuri J, Chakraborti TK, Chanda S, Pope C N (1993) Differential modulation of organophosphate-sensitive muscarinic receptors in rat brain by parathion and chlorpyrifos. J Biochem Toxicol 8:207–216
Chiappa S, Padilla S, Koenisberger C, Moser V, Brimijoin S (1995) Slow accumulation of acetylcholinesterase in rat brain during enzyme inhibition by repeated dosing with chlorpyrifos. Biochem Pharmacol 49:955–963
Clegg DJ and van Gemert M (1999) Expert panel report of human studies on chlorpyrifos and/or other organophosphate exposures. J Toxicol Environ Health B Crit Rev 2:257–279
Dani JA, Heinemann S (1996) Molecular and cellular aspects of nicotine abuse. Neuron 16:905–908
Davis DL, Ahmed AK (1998) Exposures from indoor spraying of chlorpyrifos pose greater health risk to children than currently estimated. Environ Health Perspect 106:299–301
Ellman GL, Courtney KD, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetycholinesterase activity. Biochem Pharmacol 7:88–95
Fenster, CP, Rains MF, Noeranger B, Quick MW, Lester RAJ (1997) Influence of subunit composition on desensitization of neuronal acetylcholine receptors at low concentrations of nicotine. J Neurosci 17:5747–5759
Fenster, CP, Whitworth TL, Sheffield EB, Quick MW, Lester RAJ (1999) Upregulation of surface α4β2 nicotinic receptors is initiated by receptor desensitization after chronic exposure to nicotine. J Neurosci 19:4804–4814
Fikes JD, Zachary JF, Parker AJ, Beasley VR (1992) Clinical, biochemical, electrophysiologic, and histologic assessment of chlorpyrifos-induced delayed neuropathy in the cat. Neurotoxicology 13:663–678
Flores CM, Rogers SW, Pabreza LA, Wolf BB, Keller KJ (1992) A subtype of nicotinic cholinergic receptor in rat brain is composed of α4 and β2 subunits and is upregulated by chronic nicotine treatment. Mol Pharmacol 41:31–37
Goldstein LB (1993) Beam-walking in rats: measurement of motor recovery after injury to the cerebral cortex. Neurosci Protocol 10:1–13
Gurunathan S, Robson M, Freeman N, Buckley B, Roy A, Meyer R, Bukowski J, Lioy PJ (1998) Accumulation of chlorpyrifos on residential surfaces and toys accessible to children. Environ Health Perspect 106:9–16
Hellstorm-Lindahl E, Seiger A, Kjeldgaard A, Nordberg A (2001) Nicotine-induced alterations in the expression of nicotinic receptors in primary cultures from human prenatal brain. Neuroscience 105:527–534
Huff RA, Corcoran JJ, Anderson JK, Abou-Donia MB (1994) Chlorpyrifos oxon binds directly to muscarinic receptors and inhibits cAMP accumulation in rat striatum. J Pharmacol Exp Ther 269:329–335
Katz EJ, Cortes VI, Eldefrawi ME, Eldefrawi AT (1997) Chlorpyrifos, parathion and their oxons bind to and desensitize a nicotinic acetylcholine receptor: relevance to their toxicities. Toxicol Appl Pharmacol 146:227–236
Khan WA, Dechkovskaia AM, Herrick EA, Jones KH, Abou-Donia MB (2000) Acute sarin exposure causes differential regulation of choline acetyltransferase, acetylcholinesterase, and acetylcholine receptors in the central nervous system of the rat. Toxicol Sci 57:112–120
Larsson PA, Dahlstrom A, Heiwall PO, Booj S (1980) The effect of chronic nicotine and withdrawal on intra-axonal transport of acetylcholine and related enzymes in sciatic nerve of the rat. J Neural Transm 47:243–251
Martin JC, Becker RF (1970) The effects of nicotine administration in utero upon activity in the rat. Psychon Sci 19:59–60
McGehee DS (1999) Molecular diversity of neuronal nicotinic acetylcholine receptors. Ann NY Acad Sci 868: 565–577
Miskys S, Hoffman E, Tyndale RF (2000) Regional and cellular induction of nicotine-metabolizing CYP2B1 in rat brain by chronic nicotine treatment. Biochem Pharmacol 59:1501–1511
Nostrandt AC, Padilla S, Moser VC (1997) The relationship of oral chlorpyrifos effects on behavior, cholinesterase inhibition, and muscarinic receptor density in rat. Pharmacol Biochem Behav 58:15–23
Slotkin TA (1999) Developmental cholinotoxicants: nicotine and chlorpyrifos. Environ Health Prespect 107:71–80
Slotkin TA, Epps TA, Stenger ML, Sawyer KJ, Seidler FJ (1999) Cholinergic receptors in heart and brainstem of rats exposed to nicotine during development: implications for hypoxia tolerance and prenatal mortality. Brain Res Dev Brain Res 113:1–12
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner LH, Provenzano MD, Fujimoto EK, Goete NM, Olson BJ Dlenk DD (1985) Measurement of protein using bicinchonic acid. Anal Biochem 150:76–85
Steenland K, Dick RB, Howell RJ, Chrislip DW, Hines CJ, Reid TM, Lehman E, Laber P. Kreig EF, Knott C (2000) Neurologic functions among termiticide applicators exposed to chlorpyrifos. Environ Health Perspect 108:293–300
Sternfeld M, Ming G, Song K, Sela R, Timberg M, Poo, Soreq H (1998) Acetylcholinesterase enhances neurite growth and synapse development through alternative contributions of its hydrolytic capacity, core protein, and variable C termini. J Neurosci 18:1240–1249
Stolerman IP, Garcha HS, Mirza NR (1995) Dissociations between the locomotor stimulant and depressant effects of nicotine agonists in rats. Psychopharmacology 11:430–437
US DHHS (Department of Health and Human Services) (2000) Reducing tobacco use: Report of the Surgeon General, Atlanta. US Department of Health and Human Services, Center for Disease Control and Prevention, Office on Smoking and Health, Washington DC
Wang H, Cui W-Y, Liu C-G (1996) Regulatory effects of acutely repeated nicotine treatment towards central muscarinic receptors. Life Sci 17:1415–1421
Ward TR, Ferris DJ, Tilson HA, Mundy WR (1993) Correlation of the anticholinesterase activity of a series of organophosphates with their ability to compete with agonist binding to muscarinic receptors. Toxicol Appl Pharmacol 122:300–307
Wess, J. (1996). Molecular biology of muscarinic acetylcholine receptors. Crit Rev Neurobiol 10:69–99
Yonemori F, Yamaguchi T, Yamada H, Tamura A (1998) Evaluation of a motor deficit after chronic focal cerebral ischemia in rats. J Cereb Blood Flow Metab 18:1099–1106
Acknowledgements
This study was supported in part by an US Environmental Protection Agency (EPA) grant R829399-01-0. The views, opinion and/or findings contained in this report are those of the authors and should not be construed as an official US EPA policy or decision unless so designated by other documents.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abou-Donia, M.B., Abdel-Rahman, A., Goldstein, L.B. et al. Sensorimotor deficits and increased brain nicotinic acetylcholine receptors following exposure to chlorpyrifos and/or nicotine in rats. Arch Toxicol 77, 452–458 (2003). https://doi.org/10.1007/s00204-003-0463-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-003-0463-x