Abstract
Soil microbes play important roles in plant growth and in the biogeochemical cycling of earth’s elements. However, the structure and functions of the microbial community associated with the growth of second-generation energy crops, such as Miscanthus, remain unclear. Thus, in this study, the composition and function of the bacterial and fungal communities associated with Miscanthus cultivation were analyzed by MiSeq sequencing combined with PICRUSt and FUNGUIld analyses. The results of community composition and diversity index analyses showed that Miscanthus cultivation significantly altered the bacterial and fungal community composition and reduced bacterial and fungal diversity. In addition, Miscanthus cultivation increased the soil organic matter (SOM) and total nitrogen (TN) contents. The correlation analysis between microbial community composition and environmental factors indicated that SOM and TN were the most important factors affecting bacterial and fungal communities. Miscanthus cultivation could enrich the abundances of Pseudomonas, Rhizobium, Luteibacter, Bradyrhizobium, Phenylobacterium and other common plant-promoting bacteria, while also increasing Cladophialophora, Hymenula, Magnaporthe, Mariannaea, etc., which predicted corresponded to the saprotrophic, plant pathogenic, and pathotrophic trophic modes. The PICRUSt predictive analysis indicated that Miscanthus cultivation altered the metabolic capabilities of bacterial communities, including the metabolism of carbon, nitrogen, and phosphorus cycle. In addition, FUNGUIld analysis indicated that Miscanthus cultivation altered the fungal trophic mode. The effects of Miscanthus on the communities and function of bacteria and fungi varied among Miscanthus species. Miscanthus specie Xiangdi NO 1 had the greatest impact on soil bacterial and fungal communities, whereas Miscanthus specie Wujiemang NO 1 had the greatest impact on soil bacteria and fungi functions. The results of this study provide a reference for the composition and function of microbial communities during the growth of Miscanthus.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
As an environmentally friendly and renewable energy source, biomass energy is considered to be the best alternative energy source for fossil fuels (Hoogwijk et al. 2003). As an important component of biomass energy, fuel ethanol has been vigorously developed in various countries. Furthermore, 12 provinces in China currently use ethanol gasoline, with fuel ethanol consumption reaching 2.22 million tons in 2015 (Heaton et al. 2008; Li and Chan-Halbrendt 2009; Gupta and Verma 2015).
As the raw material for fuel ethanol, energy crops grow in the soil for long periods of time and can affect the physicochemical properties of soil and the composition and function of soil microbial communities through the production of root exudates and sheddings (Bais et al. 2006; Sánchez-Cañizares et al. 2017). Miscanthus, a representative second generation of energy crops, has become a popular research area because of its many advantages, including high biomass, high cellulose content, strong adaptability and low production cost (Heaton et al. 2008; Brosse et al. 2012). Currently, numerous studies have investigated the effects of Miscanthus cultivation on soil nutrient cycling and greenhouse gas emission (Davis et al. 2010; Case et al. 2014; Dufossé et al. 2014; Zatta et al. 2014; Masters et al. 2016; Thompson et al. 2018; Chen et al. 2019). There are many types of soil microorganisms that exhibit numerous biological activities, playing important roles in plant growth (Chauhan et al. 2015). Farrar et al. (2014) analyzed the composition of endophytic bacteria in energy crops, including Miscanthus, and noted that the effective promotion of the associations between plants and beneficial microorganisms is conducive to the growth of energy crops in various habitats. Currently, few studies have investigated the impact of Miscanthus cultivation on soil microbial community composition and function, and relevant investigations need to be continued (Mao et al. 2013; Li et al. 2016). Because the majority of microorganisms cannot be cultured isolation (> 99%), the use of modern culture-free based molecular biology techniques has become an important means of assessing microbial diversity. Li et al. (2016) studied the bacterial communities of miscanthus rhizome and rhizosphere soil using automated ribosomal intergenic spacer analysis (ARISA) and terminal restriction fragment length polymorphism (TRFLP), observing that total bacterial and diazotroph communities were influenced by different Miscanthus plants and soil conditions. Compared with ARISA and TRFLP, high-throughput sequencing (HTS) can generate a large amount of microbial 16S rRNA gene-related population information, which can be used to more comprehensively understand the characteristics of microbiome (van Dijk et al. 2014). HTS has been used to study the bacterial community composition in the rhizosphere soil of Miscanthus in different habitats. Pham et al. (2018) demonstrated that metal pollution can affect the bacterial composition and structure of the Miscanthus giganteus rhizosphere, while Zhang et al. (2017) observed significant differences in the composition of bacterial communities with different soil depth in biofuel cropping systems, such as Miscanthus and switchgrass. In addition, Bourgeois et al. (2015) showed that Miscanthus can enrich bacterial and fungal communities in wastewater irrigated farmland. These results provide a basis for investigating the ecological function of microbes with respect to the growth of Miscanthus and provide a reference for Miscanthus cultivation in marginal soils, such as those polluted with heavy metals (Nebeská et al. 2018).
As a major energy consumer, China urgently needs to develop biomass energy, and as an abundant natural resource in China, Miscanthus is an excellent biomass resource for use in energy production (Yan et al. 2012). Soil microbes play an important role in plant growth. However, few studies have investigated the effect of the microbial community composition and function of the rhizosphere during Miscanthus cultivation. Therefore, the primary goals of this paper were as follows: (1) investigate the impact of the 5-year cultivation of three Miscanthus species suitable for growth in China on soil physicochemical properties, with uncultivated soil serving as a control; (2) analyze the effect of Miscanthus cultivation on soil bacterial and fungal communities through HTS; and (3) predict the effects of Miscanthus cultivation on soil bacterial and fungal functions by PICRUSt and FUNGuild analyses.
Materials and methods
Study site and sampling
Miscanthus sinensis Xiangmang NO 1 (XM), Miscanthus sacchariflorus specie Xiangdi NO 1 (XD) and Miscanthus floridulus specie Wujiemang NO 1 (WJM), were bred by Chinese scientists, suitable for growth in China. Miscanthus species XM, XD and WJM were planted in May 2013 in Nanyang city, Henan province, China (32° 56′ 45.34″ N, 112° 24′ 56.28″ E). The characteristics of the soil samples were as follows: yellow cinnamon soil; soil pH 5.79, soil organic matter (SOM) 23.57 g kg−1, cation exchange capacity (CEC) 17.6 cmol kg−1 and total nitrogen (TN) 1.33 g kg−1, total phosphorus (TP) 0.67 mg kg−1, and total potassium (TK) 15.98 mg kg−1. The fields were divided into plots for Miscanthus planting. Each plot was 52 m2 (13 m × 4 m) in area, with plants growing in a row spacing of 0.5 m × 0.5 m. The field plot experiments were conducted using a randomized arrangement with three replications. Rhizosphere soil samples were collected on June 10, 2018 from Miscanthus species XM, XD and WJM (three biological replicates for each sample), which was grown for 5 years. Rhizosphere soil was obtained by firstly gently shaking off the loosely bound soil, while the rhizosphere soil adhering to the root system was isolated by more vigorous shaking or by hand. Uncultivated soil were collected from non-plant sites serving as a control (CK). Each sample was divided into two parts. One part was immediately sieved through a 2-mm mesh and then stored at 4 °C until soil Nitrate-Nitrogen (NO3-N) could be analysed, the other section was air dried for analysis of physicochemical soil properties. Soil pH was determined with a pH meter (PHS-3C, Leizi, China) in 1:2.5 (soil:water, weight/volume, air-dried soil) suspensions. Soil organic matter (SOM) and total nitrogen (TN) were analyzed with an N/C Soil Analyzer (Flash, EA, 1112 Series, Italy). Exchangeable ammonium nitrogen (NH4-N) and NO3-N were determined with a spectrophotometer using the indophenol blue colorimetric method and phenol disulfonic acid colorimetry, respectively. Soil total phosphorus (TP) and total potassium (TK) were determined by the molybdenum (Mo)-antimony (Sb) colorimetric method and flame atomic absorption spectrophotometry. Available phosphorus (AP) and potassium (AK) in the soil were extracted by sodium bicarbonate and ammonium acetate, respectively.
DNA extraction and sequencing
Genomic DNA was extracted from 0.5 g of fresh soil using the Fast DNA® SPIN for Soil Kit (MP Biochemicals, Solon, OH, USA) according to the manufacturer’s instructions. Electrophoresis and Nano Drop ND 2000 (Thermo Scientific, USA) were used to examine the quantity of extracted DNA. The V3–V4 region of the bacterial 16S rRNA gene was amplified using 338F (5′-ACTCCTACGGGAGGCAG CA-3′) and 806R (5′-GGACTACHVGGGTWTCTAAT-3′) with sample-identifying barcodes. The PCR assays were performed in 20 μL mixture containing 4 μL of 5× FastPfu buffer, 2 μL of 2.5 mM dNTPs, 0.8 μL of each primer (5 μM), 0.4 μL of FastPfu Polymerase, 10 ng of template DNA, and Milli-Q water. The PCR conditions were as follows: 95 °C for 3 min; followed by 27 cycles at 95 °C for 30 s, 55 °C for 30 s, and 72 °C for 45 s; and a final extension at 72 °C for 10 min. The fungal ITS rRNA genes were amplified using the primers ITS1F (5′-CTTGGTCATTTAGAGGAAGTAA-3′) and ITS2R (5′-GCTGCGTTCTTCATC GATGC-3′). The PCR reactions were conducted using the following program: 3 min of denaturation at 95 °C, 35 cycles of 30 s at 95 °C, 30 s for annealing at 55 °C, and 45 s for elongation at 72 °C, and a final extension at 72 °C for 10 min. PCR was performed in triplicate for each sample, and the products were purified using the AxyPrepDNA Gel Extraction Kit (Axygen, USA) and re-quantified with QuantiFluor™ ST (Promega, USA). The sequencing was performed by Shanghai Majorbio Bio-Pharm Technology Co., Ltd. (Shanghai, China) with an Illumina MiSeq PE300 platform.
Bioinformatics analysis
After pyrosequencing, the raw data was filtered according to barcode and primer sequences using the software of Trimmomatic and FLASH as follows: bases lower than 20 on the reads tail were filtered; the minimum overlap was 10 bp when merging the paired reads; the maximum mismatch ratio was 0.2 in the overlap and the maximum barcode and primer mismatch number were set as 0 and 2, respectively. Then the high-quality sequences were processed using the QIIME Pipeline (Version 1.7.0 http://qiime.org/tutorials/tutorial.html) (Caporaso et al. 2010). The taxonomy of each 16S rRNA gene was assigned to taxonomic classification by Ribosomal Database Project Classifier (http://rdp.cme.msu.edu/) with a confidence threshold of 70% (Wang et al. 2007). The taxonomy of each ITS gene sequence was analyzed using the RDP Classifier against the UNITE fungal database at 75% similarity (Abarenkov et al. 2010). The operational taxonomic units (OTUs) at 97% similarity level were clustered using Usearch (version 7.1 http://drive5.com/uparse/) (Edgar 2010). The OTU number of each sample was used to represent species richness. Rarefaction curves and Shannon–Wiener indices were generated, and the ACE, Shannon, and Chao1 estimators were calculated to compare the bacterial and fungal richness and diversity. Linear discriminant analysis (LDA) effect size (LEfSe) was used to elucidate the biomarkers in each treatment (Segata et al. 2011). Those with an LDA score ≥ 3.0 were considered to be important biomarkers in each treatment. The metagenomes were predicted from 16 S data using Phylogenetic Investigation of Communities by Reconstruction of Unobserved States (PICRUSt) (Langille et al. 2013). The functional genes were identified from Kyoto Encyclopedia of Genes and Genomes (KEGG) database (Kanehisa and Goto 2000). Heat map were generated from the gene copy number of the functional genes using the program Heml heat map illustrator (Deng et al. 2014). FUNGuild (https://github.com/UMNFuN/FUNGuild) database was used to annotate fungal functions (Nguyen et al. 2016). The FUNGuild software annotates taxonomic data within the OTU table with corresponding data on its online database, the annotations include the guild, trophic mode and growth morphology; only confidence scores of ‘Probable’ and ‘Highly Probable’ were used. Prediction for functionality was based on assessments given in existing researches. The bacterial and fungal sequencing data are uploaded into the Sequence Read Archive (SRA) of NCBI (http://www.ncbi.nlm.nih.gov/sra) and can be accessed through Accession number PRJNA578501 and PRJNA578556, respectively.
Statistical analyses
The data of the treatments were compared by analysis of variance and the Tukey’s test at 5% significance level (P < 0.05) in SPSS V. 19.0 for Windows.
Results
Physicochemical properties of soil
The physicochemical properties of the collected uncultivated and Miscanthus rhizosphere soil were measured, the results of which are shown in Table 1. Compared with the uncultivated soil, the TN, TP, and SOM contents in the rhizosphere soil of the Miscanthus plants all increased, with significant increases in the TN and SOM contents in the XD samples observed (P < 0.05). The trend in the variation of the NH4-N and NO3-N contents was consistent, with the contents observed in the WJM samples being higher than that observed in the uncultivated soil samples, while those in the XD and XM samples were lower than that observed in the uncultivated soil samples. The potassium content results showed that the TK content in the WJM and XM samples was significantly higher than that detected in the uncultivated soil samples, whereas the AK values in the WJM and XD samples were lower than those observed in the uncultivated soil samples. The phosphorus content results showed that the TP of the Miscanthus sample was higher than that of the uncultivated soil sample but AP was lower than that of the control.
High-throughput sequencing results and assessment of diversity
The HTS results showed that an average of 23,032 and 50,554 sequence reads were obtained for the bacterial and fungal samples, respectively, with the library coverage of the samples being higher than 99.05% (Table 2). The rarefaction curve of the samples based on the number of species observed (Sobs) became stable (Fig. 1). Subsequently, the community richness, Chao1, ACE, Shannon, and Simpson indices were used to evaluate the bacterial and fungal communities. The results showed that the diversity indices of the Miscanthus cultivation samples were different than those observed for the uncultivated samples (Table 2). The Sobs, Shannon, ACE, and Simpson indices of the bacterial and fungal communities of the Miscanthus cultivation samples were all lower than those of the uncultivated samples, with bacterial and fungal diversity trends of CK > WJM > XM > XD, indicating that different Miscanthus cultivation species decreased soil bacterial and fungal community diversity.
Analysis of the bacterial and fungal community composition
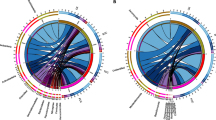
The HTS results showed that the bacteria in the soil samples from the uncultivated and Miscanthus samples comprised 20 phyla and 468 genera of bacteria, of which 74.67–81.66% of the total bacteria were from the phyla Proteobacteria, Acidobacteria, Bacteroidetes, and Actinobacteria, representing the dominant species (Fig. 2a). The fungal community analysis showed the presence of 7 phyla and 206 genera, with Ascomycota (31.10–73.12%), Glomeromycota (1.19–26.04%), and Basidiomycota (6.97–53.81%) being the dominant phyla (Fig. 2b), with the differences in fungal composition among different samples being greater than those observed for bacteria.
Unweighted pair group method using arithmetic averages (UPGMA) clustering and nonmetric multidimensional scaling (NMDS) were used to analyze the community differences between different samples, the results of which are shown in Figs. 3 and 4. The UPGMA clustering results of bacterial communities showed that at a similarity level of 0.20, the samples were divided into three groups, where the uncultivated and WJM samples clustered together and the community structure was the most similar, while the distance between the XD sample and other samples was the greatest, and the structural difference was the greatest (Fig. 3a). The UPGMA clustering results of fungal communities showed that at the similarity level of 0.35, the WJM and XM samples clustered together, exhibiting a relatively similar community structure. Similar to the bacterial community, the greatest distance was observed between the XD sample and other samples, while the structural difference was the largest (Fig. 3b). The results of the NMDS analysis of bacterial and fungal communities were similar to those of UPGMA cluster analysis. In the NMDS images, the uncultivated bacterial and fungal samples were distributed in the lower left corner of the figure and were separated from other samples. The ANOSIM and Adonis results indicated that Miscanthus cultivation significantly altered the community composition of soil bacteria (Anosim, P = 0.001, Adonis, P = 0.001) and fungi (Anosim, P = 0.001, Adonis, P = 0.002). In addition, the XD samples were distributed on the right side of the NMDS map alone, indicating that the effect of on the soil bacterial and fungal communities for the XD samples was greater than that observed for the WJM and XM samples (Fig. 4a, b).
Differential microbial analysis of different samples
To study the effect of Miscanthus cultivation on the soil bacterial and fungal community composition, we used LEfSe to search for biomarkers with significant differences. The LEfSe analysis (LDA > 3.0) results identified 43 bacterial genera with proportions of abundance in the different samples of 21.44–31.19% (Fig. S1). These bacterial genera were primarily members of the phyla Actinobacteria and Proteobacteria, of which RB41, Candidatus_Solibacter, Bryobacter, Gemmatimonas, Rhodanobacter, Rhizomicrobium, Haliangium, H16 had relative abundances of greater than 1%. A comparison of the bacterial abundances between the Miscanthus cultivation and uncultivated samples showed that for the Actinobacteria genera Acidicapsa, Lysinimonas, RB41, as well as Proteobacteria genera Bradyrhizobium, Labrys, Luteibacter, Phenylobacterium, Pseudomonas, Rhizobium, the proportions of these genera in the Miscanthus cultivation samples were all greater than those of uncultivated samples (Fig. 5). RB41 accounted for 12.70, 4.98, and 0.92% of the relative abundances in the XM, XD, and CK samples, respectively, exhibiting the greatest variation among the identified genera. For the genera Acidothermus, Actinospica, Crossiella of the phylum Actinobacteria, as well as Azoarcus and Haliangium of the phylum Proteobacteria, Nitrolancea of the phylum Chloroflexi, Phormidium of the phylum the Cyanobacteria, proportions in the Miscanthus cultivation samples were all lower than those observed in the uncultivated samples (Fig. 5).
Extended error bar plots showing statistically significant differences in the bacterial community composition at the genus levels between Miscanthus cultivation samples and the uncultivated control. Error bars indicate within-group standard deviations. Presented categories passed a corrected P value of < 0.05 in Welch’s t test
The LEfSe analysis (LDA > 3.0) results showed that 32 genera of fungi exhibited different abundances, relative abundances accounting for 11.37–34.65% of the total genera (Fig. S2). These fungi were primarily distributed in the phyla Ascomycota and Basidiomycota, with the genera Cistella, Cladophialophora, Fusarium, Hymenula, Lophiostoma, Magnaporthe, Phoma, Sarocladium, Stachybotrys, Trichoderma, Mrakiella, Psathyrella and Paraglomus exhibiting relative abundances of greater than 1%, being major components of the fungal community. The relative abundances of Trichoderma (18.57%) in the XD samples and Paraglomus (25.99%) in the WJM samples were greater than those observed in the uncultivated samples (0.63 and 3.69%, respectively), exhibiting the greatest variations. Comparative analysis of fungal abundances showed that the proportions of the Ascomycota genera Cladophialophora, Hymenula, Magnaporthe, Mariannaea, Myxocephala, Ramichloridium and Sarocladium, the Basidiomycota genera Entoloma, Mrakiella and Psathyrella were higher in the Miscanthus cultivation samples than in the uncultivated samples (Fig. 6). The Ascomycota genera Boeremia, Fusarium, Lecanicillium, Lophiostoma, Monocillium, Monosporascus, Nectria, Phialemonium, Phoma, Stachybotrys, the Basidiomycota genera Eocronartium, Rhodosporidium, the Basidiomycota genera Glomeromycota Entrophospora were lower in the Miscanthus cultivation samples than in the uncultivated samples (Fig. 6).
Extended error bar plots showing statistically significant differences in the fungal community composition at the genus levels between Miscanthus cultivation samples and the uncultivated control. Error bars indicate within-group standard deviations. Presented categories passed a corrected P value of < 0.05 in Welch’s t test
Correlation analysis of microbial community and environmental factors
Canoco 4.5 was used for redundancy analysis (RDA) and canonical correspondence analysis (CCA) of the bacterial and fungal communities at the OTU level. The RDA analysis results are shown in Fig. 7a. The physicochemical properties with a high correlation with the first ordination axis were TN (R = 0.9944), SOM (R = 0.9961), and pH (R = 0.8334), while TK exhibited a high correlation with the second ordination axis (R = − 0.9400). These environmental factors had a significant correlation with the bacterial community composition (P < 0.05). The CCA analysis results are shown in Fig. 7b. The physicochemical properties with high correlation with the first ordination axis were TN (R = 0.7218) and SOM (R = 0.8364), while TK (R = 0.9966) and AK (R = 0.7699) were highly correlated with the second ordination axis. These environmental factors had a significant correlation with the fungal community composition (P < 0.05).
PICRUSt and FUNGuild functional prediction analysis
To study the effect of Miscanthus cultivation on soil bacterial function, we used PICRUSt to perform bacterial function prediction analysis. Through comparisons with the Kyoto Encyclopedia of Genes and Genomes (KEGG) database, 6 categories of biological metabolic pathways (primary functional level) were obtained, including metabolism, genetic information processing, environmental information processing, cellular processes, organ systems, and human diseases. Among these pathways, metabolism, genetic information processing, and environmental information processing were the primary components, accounting for 51.61–51.75%, 15.60–168.3%, and 11.79–12.97%, respectively. The comparison of soil bacterial community function predictions in different samples showed that the copy number sequence of predicted genes in six primary functional layers followed the pattern WJM > CK > XM > XD. In addition, the analysis of the secondary functional layer of the predicted genes showed that it consisted of 34 subfunctions, including amino acid metabolism, carbohydrate metabolism, membrane transport, replication and repair, and energy metabolism (Fig. 8). A heat map of the secondary functional layer gene copy number was plotted (Deng et al. 2014). From the clustering analysis results of the gene copy number shown in Fig. 8, the biological replicates of different samples clustered together, indicating good repeatability. XM samples was close to XD samples, whereas uncultivated samples and WJM samples were far away from other samples, indicating that Miscanthus cultivation alters soil bacterial community function and that there are differences among different varieties. Previous studies have shown that PICRUSt can accurately predict the presence and abundance of genes related to carbon (C), nitrogen (N) and phosphorus (P) cycles (Hartman et al. 2017; LeBrun and Kang 2018; Ribeiro et al. 2018; Hu et al. 2019). In the present study, PICRUSt predicted the relative abundance of key genes involved in the C, N and P cycles, primarily those involved in C fixation and methane metabolism in the C cycle (Fig. S3); N fixation, nitrification and denitrification in the N cycle (Fig. S4); and the key genes K00655 (plsC), K01507 (ppa), K02036 (pstB), and K02037 (pstC) of the P cycle (Fig. S5). The cluster analysis results regarding the copy numbers of genes in the C and P cycles (Fig. S3, S5) show that the WJM and XM samples clustered together, while those of uncultivated samples were separated other samples. The copy number clustering analysis results of the N cycling genes indicate that WJM and the XD samples clustered together, while those of uncultivated samples were separated other samples. The above results indicated that Miscanthus cultivation altered the metabolic capacities of microbes with respect to the soil C, N and P cycles.
FUNGuild was used to predict the nutritional and functional groups of the fungal communities with different treatments. The results showed that six trophic mode groups could be classified, with pathotrophs, saprotrophs, and symbiotrophs being the major components, with the percentages of OTUs in these three major functional categories being 1.75–5.22%, 4.71–16.06% and 1.62–26.36%, respectively (Fig. 9a). For the uncultivated, XD and XM samples, saprotrophy was the primary trophic mode, accounting for 9.22%, 16.06% and 8.89% of OTUs, respectively. The WJM samples primarily harbored symbiotrophs, accounting for 26.36% of OTUs. The saprotroph composition analysis showed that the proportions of dung saprotrophs in the XM and XD samples were 8.75% and 3.28%, respectively, higher than that observed in the control (1.31%) (Fig. 9c). In addition, the proportions of plant saprotrophs in the WJM, XM and XD samples were all greater than those observed in the uncultivated control. The symbiotrophic mode groups primarily consisted of arbuscular mycorrhizal, endophyte, and ectomycorrhizal. The proportion of arbuscular mycorrhizal OTUs in the WJM samples was 25.97%, which was significantly greater than that observed in the uncultivated control group (4.24%) (Fig. 9d). The pathotrophic mode was used in the detection of uncultivated and Miscanthus rhizosphere soil samples. The ratios of plant pathogens in the WJM and XM samples were higher than those observed in the uncultivated control, while animal pathogens in the Miscanthus samples were significantly lower than that observed in the control samples (Fig. 9b).
Discussion
Soil microbes are the primary drivers of earth’s biogeochemical cycles of elements in soil, directly or indirectly affecting plant growth (Falkowski et al. 2008; Pii et al. 2015). Because few studies have been performed on the rhizosphere microbial community and function of Miscanthus, in this study the effects of Miscanthus cultivated for 5 years on soil bacterial and fungal community and function was assessed.
The MiSeq sequencing results indicated that the rhizospheric bacteria of Miscanthus comprised 468 genera from 20 phyla, including proteobacteria, acidobacteria, bacteroidetes, actinobacteria, etc. The fungal community consisted of 206 genera from 7 phyla, including ascomycota, glomeromycota, basidiomycota, etc. Previous studies have shown that these bacterial community components also appeared to be dominant in different soil depths studied by Zhang et al. (2017) and heavy metal-contaminated soils studied by Pham et al. (2018). The composition of the dominant fungal communities were observed in wastewater-contaminated farmland studied by Bourgeois et al. (2015), but the composition ratio exhibited large variations, indicating that different soil types and Miscanthus cultivars can affect the compositions of rhizospheric bacterial and fungal communities.
As a plant with high yield potentials, Miscanthus absorb nutrients from the soil during the growth (Cadoux et al. 2012). Therefore, the contents of available elements (NH4-N, NO3-N, AK and AP) in Miscanthus cultivation samples all showed a decreasing trend compared with the control sample (Table 1). Similar to the results of Dufossé et al. (2014), due to the enrichment of sheddings and plant roots, as well as exemption from tillage, the SOM content in the rhizosphere soil of Miscanthus was higher than that of the control, with a significant difference observed in the XD samples. Plant roots provide effective carbon and nitrogen sources for soil microbes in the rhizosphere by producing exudates and sheddings, resulting in significantly different rhizospheric biological characteristics from uncultivated soil or nonrhizosphere soil, exhibiting rhizospheric effects (Bais et al. 2006; Sánchez-Cañizares et al. 2017). UPGMA cluster analysis and NMDS analysis of the bacterial and fungal community compositions in this study showed that Miscanthus cultivation significantly altered the composition of the soil bacterial and fungal communities, with the fungal communities showing greater variation than bacterial communities (Figs. 3, 4). This result is similar to that reported by Bourgeois et al. (2015), which showed that Miscanthus cultivation can influence nutrient-cycler bacteria and fungi in wastewater-contaminated farmland, in which the changes in the fungal community were more significant. The diversity index of Miscanthus cultivation samples was lower than that of uncultivated samples, indicating that the growth of Miscanthus roots reduced the diversity of bacteria and fungi in the rhizosphere (Table 2). Studies have shown that the plant rhizosphere has a specific selectivity for bacteria and fungi colonizing the rhizosphere, which alters the richness and reduces the homogeneity of species, leading to a decrease in α-diversity (Shi et al. 2015).
To investigate the factors that affect the compositions of bacterial and fungal communities, the physicochemical properties of soil and the bacterial and fungal community compositions were analyzed by RDA and CCA, respectively. The results showed that SOM and TN could significantly affect the bacterial and fungal community compositions (P < 0.05) (Fig. 7). Previous studies have shown that different plant species have different root exudates, which can affect microbial community composition (Pérez-Jaramillo et al. 2016). Analysis of the physicochemical properties showed differences among the evaluated Miscanthus cultivars. In particular, the SOM and TN values of the XD samples were significantly higher than those of the other samples. The UPGMA cluster analysis and NMDS analysis results showed that the XD samples bacterial and fungal communities had the greatest differences from other Miscanthus cultivars. In addition, the XD samples diversity index value was the lowest, indicating that different varieties of Miscanthus promoted differences in soil physicochemical properties, affecting bacterial and fungal community composition. To specifically analyze the differences in the composition of the Miscanthus rhizospheric bacterial and fungal communities, LEfSe was used to identify the differential species. The results indicated that Miscanthus cultivation can enrich and increase the proportions of Acidicapsa, Lysinimonas, RB41, Bradyrhizobium, Labrys, Luteibacter, Phenylobacterium, Pseudomonas and Rhizobium, where Pseudomonas (Ren et al. 2019), Rhizobium and Luteibacteria (Abdelkrim et al. 2018), Bradyrhizobium (Masciarelli et al. 2014), and Phenylobacterium (Yang et al. 2017) are common plant growth-promoting bacterial species. Miscanthus cultivation enriched these bacterial populations and may play a role in their growth and environmental adaptation (Bourgeois et al. 2015; Gopalakrishnan et al. 2015).
PICRUSt analysis can predict the metabolic function of bacterial communities with high reliability (Langille et al. 2013) and has been used to study bacterial functions in different plant soils, promoting the study of bacterial ecological functions (Jiang et al. 2016; Pii et al. 2016; Yuan et al. 2016; Luo et al. 2017). We performed PICRUSt functional predictive analysis using the MiSeq HTS results. The results showed that the rhizospheric bacteria of Miscanthus primarily comprised 34 secondary functional layers, such as metabolism and carbohydrate metabolism, showing functional abundance. To study the effect of Miscanthus cultivation on rhizospheric bacterial function, cluster analysis was performed on the predicted gene copy numbers of the secondary functional layer genes (Fig. 8). The results showed that Miscanthus cultivation altered the metabolic functions of bacteria, with the WJM samples exhibiting the largest variations. This result is similar to those obtained in a study of the function of soybean rhizosphere bacteria by Mendes et al. (2014), where soybean planting was shown to alter the gene expression levels of functional groups such as ‘membrane transport’, ‘nitrogen metabolism’ and ‘phosphorus metabolism’. PICRUSt analysis can predict the presence or absence of functional genes and their abundances. Therefore, this technique has been used in the analysis of bacterial genes related to C, N, and the P cycles in soil (Hartman et al. 2017; LeBrun and Kang 2018; Ribeiro et al. 2018; Hu et al. 2019). Pii et al. (2016) used PICRUSt to predict the function of rhizospheric bacterial communities in barley and tomato, observing that metabolism is the primary component of their functional modules. They also observed that some OTUs are closely related to the functions of C fixation, N and S metabolism, and their analysis suggested that plants affected the bacterial community and function through root exudates as C and N sources. In the present study, PICRUSt was used to predict the relative abundances of key genes in the C, N and P cycles. Cluster analysis of the predicted gene copy number of the relevant genes indicated that Miscanthus cultivation altered the metabolism capacities of soil C, N and P cycles (Figs. S3–S5). Because of the limitations of PICRUSt functional prediction analysis, this study only preliminarily predicted the functions of related bacteria. Further validation should be performed in future studies using methods such as metagenomics to better understand the function of the Miscanthus rhizosphere bacterial community.
FUNGUIld is a database used for comparisons of fungal functions and perform specific functional classifications of fungi. Thus, it has been widely used in fungal community research (Nguyen et al. 2016; Martínez-Diz et al. 2019). The FUNGUIld prediction results of Miscanthus rhizosphere fungi were consistent with the Grapevine bulk, rhizosphere and endorhizosphere functional groups studied by Martínez-Diz et al. (2019) and primarily consisted of three trophic modes, pathotrophic, saprotrophic and symbiotrophic (Fig. 9). The XD and XM samples were dominated by saprotroph, similar to the uncultivated samples, although the proportions were different. The WJM samples were dominated by symbiotrophs. LEfSe analysis of the composition of Miscanthus rhizosphere fungal communities showed that Miscanthus cultivation could simultaneously enrich and increase proportions of Cladophialophora, Hymenula, Magnaporthe, Mariannaea, Myxocephala, Ramichloridium, Sarocladium, Entoloma, Mrakiella, and Psathyrella (Fig. 6). The FUNGuild results predicted that these fungi primarily corresponded to trophic mode, including saprotrophs, plant pathogens, pathotrophs, and the pathotrophic–saprotrophic–symbiotrophic Entoloma. Studies have shown that saprophytic fungi are the primary decomposers of dead or aged plants in the soil and play important roles in decomposing organic matter and nutrient cycling. Therefore, the increase of the proportions of the saprotrophic fungi Cladophialophora, Mariannaea and Myxocephala in the rhizosphere soil of Miscanthus promotes the formation of soil organic matter and the rate of nutrition cycling (Phillips et al. 2014; Zhang et al. 2019). The proportions of the plant pathogenic or pathotrophic fungi Hymenula, Magnaporthe, and Ramichloridium were significantly higher than those observed in the control, which was consistent with the proportion of the plant pathogenic trophic mode in the WJM and XM samples being higher than that observed in the uncultivated control, indicating that the long-term Miscanthus cultivation increases the presence of potential pathogens (Martínez-Diz et al. 2019). As a dominant trophic mode in the WJM samples, arbuscular mycorrhizal fungi (AMF) accounted for the majority of symbiotrophs. Studies have shown that AMF can form a symbiotic relationship with more than 80% of terrestrial plants, enhancing the absorption capacity of root systems and providing increasing the levels of nutrients for plant growth, such as phosphorus and nitrogen (Berruti et al. 2016). The proportion of AMF in the WJM samples was 68.48%, significantly higher than that observed in the control (22.76%), suggesting that AMF may have promoted growth in the WJM samples. Although FUNGuild has been used to analyze the functions of fungi to a some extent, this method has some limitations due to it being based on preexisting literature and data. Thus, additional in-depth studies on soil fungal classification and functional groups are needed to further investigate the function of the rhizosphere fungal community of Miscanthus.
Conclusion
The rhizospheric microbial communities of 5-year-old XM, XD, and WJM plants were determined and comprised 20 phyla and 468 genera of bacteria and 7 phyla and 206 genera of fungi. Miscanthus cultivation can alter the physicochemical properties of soil and increase the contents of TN, TP, and SOM. Miscanthus cultivation significantly altered the composition of bacterial and fungal communities and reduced the diversity of bacteria and fungi, with SOM and TN being the most important factors affecting the composition of the bacterial and fungal communities. Miscanthus cultivation enriched the abundances of Pseudomonas, Rhizobium, Luteibacter, Bradyrhizobium, and Phenylobacterium and other common plant-promoting bacteria, while also increasing the presence of fungal genera, including Cladophialophora, Hymenula, Magnaporthe, and Mariannaea. The PICRUSt predictive analysis results showed that Miscanthus cultivation altered the total bacterial function and C, N, and P cycle and metabolic functions. In addition, the FUNGuild analysis results indicated that Miscanthus cultivation altered the trophic mode groups. The effect of Miscanthus on the structure and function of the bacterial and fungal communities varied with the species of Miscanthus. Due to the limitations of PICRUSt and FUNGuild prediction analysis, subsequent studies need to be performed in combination with metagenomic sequencing and analysis of functional genes associated with C, N, and P cycling to better understand the functions of the Miscanthus bacterial and fungal rhizospheric communities.
References
Abarenkov K, Henrik Nilsson R, Larsson K-H, Alexander IJ, Eberhardt U, Erland S, Høiland K, Kjøller R, Larsson E, Pennanen T, Sen R, Taylor AFS, Tedersoo L, Ursing BM, Vrålstad T, Liimatainen K, Peintner U, Kõljalg U (2010) The UNITE database for molecular identification of fungi—recent updates and future perspectives. New Phytol 186:281–285
Abdelkrim S, Jebara SH, Saadani O, Jebara M (2018) Potential of efficient and resistant plant growth-promoting rhizobacteria in lead uptake and plant defence stimulation in Lathyrus sativus under lead stress. Plant Biol 20:857–869
Bais HP, Weir TL, Perry LG, Gilroy S, Vivanco JM (2006) The role of root exudates in rhizosphere interactions with plants and other organisms. Annu Rev Plant Biol 57:233–266
Berruti A, Lumini E, Balestrini R, Bianciotto V (2016) Arbuscular mycorrhizal fungi as natural biofertilizers: let’s benefit from past successes. Front Microbiol 6:1559
Bourgeois E, Dequiedt S, Lelièvre M, Van Oort F, Lamy I, Ranjard L, Maron P (2015) Miscanthus bioenergy crop stimulates nutrient-cycler bacteria and fungi in wastewater-contaminated agricultural soil. Environ Chem Lett 13:503–511
Brosse N, Dufour A, Meng X, Sun Q, Ragauskas A (2012) Miscanthus: a fast-growing crop for biofuels and chemicals production. Biofuel Bioprod Biorefin 6:580–598
Cadoux S, Riche AB, Yates NE, Machet J-M (2012) Nutrient requirements of Miscanthus × giganteus: conclusions from a review of published studies. Biomass Bioenergy 38:14–22
Caporaso J, Kuczynski J, Stombaugh J, Bittinger K, Bushman F, Costello E (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Case SDC, Mcnamara NP, Reay DS, Whitaker J (2014) Can biochar reduce soil greenhouse gas emissions from a Miscanthus bioenergy crop? GCB Bioenergy 6:76–89
Chauhan H, Bagyaraj DJ, Selvakumar G, Sundaram SP (2015) Novel plant growth promoting rhizobacteria—prospects and potential. Appl Soil Ecol 95:38–53
Chen H, Dai Z, Jager HI, Wullschleger SD, Xu J, Schadt CW (2019) Influences of nitrogen fertilization and climate regime on the above-ground biomass yields of miscanthus and switchgrass: a meta-analysis. Renew Sustain Energy Rev 108:303–311
Davis SC, Parton WJ, Dohleman FG, Smith CM, Grosso SD, Kent AD, Delucia EH (2010) Comparative biogeochemical cycles of bioenergy crops reveal nitrogen-fixation and low greenhouse gas emissions in a Miscanthus × giganteus agro-ecosystem. Ecosystems 13:144–156
Deng W, Wang Y, Liu Z, Cheng H, Xue Y (2014) HemI: a toolkit for illustrating heatmaps. PLoS ONE 9:e111988
Dufossé K, Drewer J, Gabrielle B, Drouet JL (2014) Effects of a 20-year old Miscanthus × giganteus stand and its removal on soil characteristics and greenhouse gas emissions. Biomass Bioenergy 69:198–210
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461
Falkowski PG, Fenchel T, Delong EF (2008) The microbial engines that drive earth’s biogeochemical cycles. Science 320:1034–1039
Farrar K, Bryant D, Cope-Selby N (2014) Understanding and engineering beneficial plant–microbe interactions: plant growth promotion in energy crops. Plant Biotechnol J 12:1193–1206
Gopalakrishnan S, Sathya A, Vijayabharathi R, Varshney RK, Gowda CLL, Krishnamurthy L (2015) Plant growth promoting rhizobia: challenges and opportunities. 3 Biotech 5:355–377
Gupta A, Verma JP (2015) Sustainable bio-ethanol production from agro-residues: a review. Renew Sustain Energy Rev 41:550–567
Hartman WH, Ye R, Horwath WR, Tringe SG (2017) A genomic perspective on stoichiometric regulation of soil carbon cycling. ISME J 11:2652–2665
Heaton EA, Dohleman FG, Long SP (2008) Meeting US biofuel goals with less land: the potential of Miscanthus. Glob Change Biol 14:2000–2014
Hoogwijk M, Faaij A, Van Den Broek R, Berndes G, Gielen D, Turkenburg W (2003) Exploration of the ranges of the global potential of biomass for energy. Biomass Bioenergy 25:119–133
Hu Y, Zhang Z, Huang L, Qi Q, Liu L, Zhao Y, Wang Z, Zhou H, Lv X, Mao Z, Yang Y, Zhou J, Kardol P (2019) Shifts in soil microbial community functional gene structure across a 61-year desert revegetation chronosequence. Geoderma 347:126–134
Jiang L, Song M, Yang L, Zhang D, Sun Y, Shen Z, Luo C, Zhang G (2016) Exploring the influence of environmental factors on bacterial communities within the rhizosphere of the Cu-tolerant plant, Elsholtzia splendens. Sci Rep 6:36302
Kanehisa M, Goto S (2000) KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28:27–30
Langille MGI, Zaneveld J, Caporaso JG, Mcdonald D, Knights D, Reyes JA, Clemente JC, Burkepile DE, Vega Thurber RL, Knight R, Beiko RG, Huttenhower C (2013) Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat Biotechnol 31:814–821
Lebrun E, Kang S (2018) A comparison of computationally predicted functional metagenomes and microarray analysis for microbial P cycle genes in a unique basalt-soil forest. F1000 Research 7:179
Li S-Z, Chan-Halbrendt C (2009) Ethanol production in (the) People’s Republic of China: potential and technologies. Appl Energy 86:S162–S169
Li D-F, Voigt TB, Kent AD (2016) Plant and soil effects on bacterial communities associated with Miscanthus × giganteus rhizosphere and rhizomes. GCB Bioenergy 8:183–193
Luo J, Tao Q, Wu K, Li J, Qian J, Liang Y, Yang X, Li T (2017) Structural and functional variability in root-associated bacterial microbiomes of Cd/Zn hyperaccumulator Sedum alfredii. Appl Microbiol Biotechnol 101:7961–7976
Mao Y, Yannarell AC, Davis SC, Mackie RI (2013) Impact of different bioenergy crops on N-cycling bacterial and archaeal communities in soil. Environ Microbiol 15:928–942
Martínez-Diz MDP, Andrés-Sodupe M, Bujanda R, Díaz-Losada E, Eichmeier A, Gramaje D (2019) Soil-plant compartments affect fungal microbiome diversity and composition in grapevine. Fungal Ecol 41:234–244
Masciarelli O, Llanes A, Luna V (2014) A new PGPR co-inoculated with Bradyrhizobium japonicum enhances soybean nodulation. Microbiol Res 169:609–615
Masters MD, Black CK, Kantola IB, Woli KP, Voigt T, David MB, Delucia EH (2016) Soil nutrient removal by four potential bioenergy crops: Zea mays, Panicum virgatum, Miscanthus × giganteus, and prairie. Agric Ecosyst Environ 216:51–60
Mendes LW, Kuramae EE, Navarrete AA, Van Veen JA, Tsai SM (2014) Taxonomical and functional microbial community selection in soybean rhizosphere. ISME J 8:1577–1587
Nebeská D, Trögl J, Pidlisnyuk V, Popelka J, Veronesi Dáňová P, Usťak S, Honzík R (2018) Effect of growing Miscanthus × giganteus on soil microbial communities in post-military soil. Sustainability 10:4021
Nguyen NH, Song Z, Bates ST, Branco S, Tedersoo L, Menke J, Schilling JS, Kennedy PG (2016) FUNGuild: an open annotation tool for parsing fungal community datasets by ecological guild. Fungal Ecol 20:241–248
Pérez-Jaramillo JE, Mendes R, Raaijmakers JM (2016) Impact of plant domestication on rhizosphere microbiome assembly and functions. Plant Mol Biol 90:635–644
Pham HN, Pham PA, Nguyen TTH, Meiffren G, Brothier E, Lamy I, Michalet S, Dijoux-Franca M-G, Nazaret S (2018) Influence of metal contamination in soil on metabolic profiles of Miscanthus × giganteus belowground parts and associated bacterial communities. Appl Soil Ecol 125:240–249
Phillips LA, Ward V, Jones MD (2014) Ectomycorrhizal fungi contribute to soil organic matter cycling in sub-boreal forests. ISME J 8:699–713
Pii Y, Mimmo T, Tomasi N, Terzano R, Cesco S, Crecchio C (2015) Microbial interactions in the rhizosphere: beneficial influences of plant growth-promoting rhizobacteria on nutrient acquisition process. A review. Biol Fertil Soils 51:403–415
Pii Y, Borruso L, Brusetti L, Crecchio C, Cesco S, Mimmo T (2016) The interaction between iron nutrition, plant species and soil type shapes the rhizosphere microbiome. Plant Physiol Biochem 99:39–48
Ren X-M, Guo S-J, Tian W, Chen Y, Han H, Chen E, Li B-L, Li Y-Y, Chen Z-J (2019) Effects of plant growth-promoting bacteria (PGPB) inoculation on the growth, antioxidant activity, cu uptake, and bacterial community structure of rape (Brassica napus L.) grown in Cu-contaminated agricultural soil. Front Microbiol 10:1455
Ribeiro H, De Sousa T, Santos JP, Sousa AGG, Teixeira C, Monteiro MR, Salgado P, Mucha AP, Almeida CMR, Torgo L, Magalhães C (2018) Potential of dissimilatory nitrate reduction pathways in polycyclic aromatic hydrocarbon degradation. Chemosphere 199:54–67
Sánchez-Cañizares C, Jorrín B, Poole PS, Tkacz A (2017) Understanding the holobiont: the interdependence of plants and their microbiome. Curr Opin Microbiol 38:188–196
Segata N, Izard J, Waldron L, Gevers D, Miropolsky L, Garrett WS, Huttenhower C (2011) Metagenomic biomarker discovery and explanation. Genome Biol 12:1–18
Shi S, Nuccio E, Herman DJ, Rijkers R, Estera K, Li J, Da Rocha UN, He Z, Pett-Ridge J, Brodie EL, Zhou J, Firestone M (2015) Successional trajectories of rhizosphere bacterial communities over consecutive seasons. mBio 6:e00746-00715
Thompson KA, Deen B, Dunfield KE (2018) Impacts of surface-applied residues on N-cycling soil microbial communities in miscanthus and switchgrass cropping systems. Appl Soil Ecol 130:79–83
Van Dijk EL, Auger H, Jaszczyszyn Y, Thermes C (2014) Ten years of next-generation sequencing technology. Trends Genet 30:418–426
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naïve Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267
Yan J, Chen W, Luo F, Ma H, Meng A, Li X, Zhu M, Li S, Zhou H, Zhu W, Han B, Ge S, Li J, Sang T (2012) Variability and adaptability of Miscanthus species evaluated for energy crop domestication. GCB Bioenergy 4:49–60
Yang Y, Wang N, Guo X, Zhang Y, Ye B (2017) Comparative analysis of bacterial community structure in the rhizosphere of maize by high-throughput pyrosequencing. PLoS ONE 12:e0178425
Yuan Z-L, Druzhinina IS, Labbé J, Redman R, Qin Y, Rodriguez R, Zhang C, Tuskan GA, Lin F (2016) Specialized microbiome of a halophyte and its role in helping non-host plants to withstand salinity. Sci Rep 6:32467
Zatta A, Clifton-Brown J, Robson P, Hastings A, Monti A (2014) Land use change from C3 grassland to C4 Miscanthus: effects on soil carbon content and estimated mitigation benefit after six years. GCB Bioenergy 6:360–370
Zhang B, Penton CR, Xue C, Quensen JF, Roley SS, Guo J, Garoutte A, Zheng T, Tiedje JM (2017) Soil depth and crop determinants of bacterial communities under ten biofuel cropping systems. Soil Biol Biochem 112:140–152
Zhang W-H, Sun R-B, Xu L, Liang J-N, Zhou J (2019) Assessment of bacterial communities in Cu-contaminated soil immobilized by a one-time application of micro-/nano-hydroxyapatite and phytoremediation for 3 years. Chemosphere 223:240–249
Acknowledgements
This research was supported by the National Natural Science Foundation of China (Grant No. 41601332), the Key Research Project of Colleges and Universities of Henan Province Education Department (Grant No. 16A210012, 17A180032) and the Central Public-interest Scientific Institution Basal Research Fund: Study on the mechanism of cadmium accumulation in soil by rice controlled by heavy metal immobilized microorganism and organic materials.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, Y., Tian, W., Shao, Y. et al. Miscanthus cultivation shapes rhizosphere microbial community structure and function as assessed by Illumina MiSeq sequencing combined with PICRUSt and FUNGUIld analyses. Arch Microbiol 202, 1157–1171 (2020). https://doi.org/10.1007/s00203-020-01830-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-020-01830-1