Abstract
Zwittermicin A (ZwA) is a novel, broad-spectrum linear aminopolyol antibiotic produced by some Bacillus cereus and Bacillus thuringiensis. However, only part of its biosynthesis cluster has been identified and characterized from B. cereus UW85. To better understand the biosynthesis cluster of ZwA, a bacterial artificial chromosome (BAC) library of B. thuringiensis subsp. kurstaki strain YBT-1520, a ZwA-producing strain, was constructed. Two BAC clones, 1F8 and 5E2, were obtained by PCR, which overlap the known ZwA biosynthesis cluster of B. cereus UW85. This ZwA biosynthesis cluster is at least 38.6 kb and is located on the chromosome, instead of the plasmid. Partial DNA sequencing revealed both BAC clones carry three new ZwA biosynthesis-related genes, zwa6, zwa5A and zwa5B, which were found at the corresponding location of B. cereus UW85. Putative amino acid sequences of these genes shown that ZWA6 is homologous to a typical carbamoyltransferase from Streptomyces avermitilis, while ZWA5A and ZWA5B are homologs of cysteine synthetase and ornithine cyclodeaminase which jointly synthesize 2,3-diaminopropionate in the viomycin biosynthesis pathway, respectively. The identification of these three genes further supports the hypothesized ZwA biosynthesis pathway.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Zwittermicin A (ZwA) is a novel aminopolyol antibiotic produced by some Bacillus cereus and Bacillus thuringiensis (He et al. 1994; Silo-Suh et al. 1994; Stabb et al. 1994; Raffel et al. 1996). It is highly active against Oomycetes and their relatives, and has moderate activity against diverse Gram-negative bacteria and certain Gram-positive bacteria, as well as activity against a wide range of plant pathogenic fungi (Silo-Suh 1994, 1998). On the other hand, ZwA also facilitates the insecticidal activity of the protein toxin produced by B. thuringiensis, increasing mortality of insects that are typically resistant to this toxin (Broderick et al. 2000, 2003).
Zwittermicin A is different from polyketide and peptide antibiotics in chemical structure with a molecular formula of C13H28N6O8 (He et al. 1994; Stabb and Handelsman 1998). The hydroxyl groups on the linear carbon backbone are similar to a partially reduced polyketide structure, but the nitrogen-rich end of ZwA may be derived from an amino acid, like some peptide antibiotics (Stohl et al. 1999a).
A 16-kb DNA fragment (zwa16Bc) covering zmaR (ZwA resistance gene) was cloned from B. cereus strain UW85 and its DNA sequence revealed a potential ZwA biosynthesis cluster, including nine genes and a partial gene (Stohl et al. 1999a; Emmert et al. 2004). Based on the structural and genetic information, Handelsman’s group hypothesized that the premature ZwA is synthesized by a nonribosomal peptide synthetase and polyketide synthetase (PKS) hybrid pathway from five kinds of precursors: l-serine, malonyl-CoA, aminomalonyl-ACP, hydroxymalonyl-ACP and 2,3-diaminopropionate (Ansari et al. 2004; Emmert et al. 2004; Fig. 1a). Then, a hypothetic carbamoyltransferase catalyzes the premature ZwA into ZwA (Emmert et al. 2004; Fig. 1b). However, neither the carbamoyltransferase gene nor the 2,3-diaminopropionate synthetase gene has been identified in zwa16Bc.
Chemical structure and proposed biosynthesis pathway of ZwA (modified from Thomas et al. 2003; Emmert et al. 2004). a Structure of ZwA and proposed biosynthesis precursors. b Hypothesized function of ZWA5A, ZWA5B and ZWA6 in the biosynthesis of ZwA. The premature ZwA is synthesized by a NRPS and PKS hybrid pathway from five kinds of precursors: l-serine, malonyl-CoA, aminomalonyl-ACP, hydroxymalonyl-ACP and 2,3-diaminopropionate. ZWA5A and ZWA5B synthesize 2,3-diaminopropionate by their concerted actions. Then ZWA6, a typical carbamoyltransferase, catalyzes the premature ZwA into ZwA. A adenylation domain, ACP acyl carrier domain, C condensation domain, KR ketoreductase, KS ketosynthase, NRPS nonribosomal peptide synthetase, PCP peptidyl carrier protein, PKS polyketide synthase, PLP pyridoxal 5′-phosphate, ZwA zwittermicin A
In this study, a bacterial artificial chromosome (BAC) library was constructed with the total DNA of B. thuringiensis subsp. kurstaki strain YBT-1520 (briefly, B. thuringiensis YBT-1520), a ZwA-producing bacterium with high toxicity against Helicoverpa arrnigera larvae (Sun et al. 2000). From this library, nine clones overlapping zwa16Bc were identified and two of them, 1F8 and 5E2, were found carry the carbamoyltransferase gene and the 2,3-diaminopropionate synthetase gene.
Materials and methods
Bacterial strains and primers
The bacterial strains and primers used in this study are listed in Tables 1 and 2, respectively.
Purification and identification of ZwA produced by Bacillus thuringiensis YBT-1520
Zwittermicin A was initially purified from B. thuringiensis YBT-1520 with amberlite IRC-50 cation exchange resin (SIGMA) according to the purification method described previously (Silo-Suh et al. 1994, 1998). The eluants were concentrated in a rotary evaporator. Resuspended sample was analyzed by high-performance liquid chromatography-mass spectrometry (HPLC-MS) with a Waters Alliance 2695 instrument. Fractions were collected and assayed for inhibition of Erwinia herbicola LS005 by the method reported earlier (Silo-Suh et al. 1994; Stabb et al. 1994).
Construction of BAC library
Bacillus thuringiensis YBT-1520 was cultured for 5 h at 28°C in Luria Bertani medium to a density of 3 × 108 cells/ml. Cells were harvested by centrifugation and agarose plugs were prepared as described (Rondon et al. 1999). Genomic DNA embedded in the agarose plugs was partially digested with HindIII (TAKARA) and then separated by pulse field gel electrophoresis (PFGE). High molecular weight genomic DNA was recovered and ligated into cloning-ready BAC vector pBeloBAC11 (GenBank accession no. U51113) digested with HindIII. The ligation mix was transformed into Escherichia coli strain DH10B by electroporation with a Bio-Rad GenePulser instrument. To estimate the insert sizes of BAC clones, BAC clones were digested with NotI and HindIII, and then separated by PFGE with a Bio-Rad CHEF III instrument.
PCR screening and DNA sequencing
In order to identify those BAC clones related to ZwA synthesis by PCR, two primer pairs were designed according to the sequence of the 5′- and 3′-terminals of zwa16Bc (Emmert et al. 2004). Primer pair up-F and up-R, were designed to amplify the 5′-terminal, while primer pair down-F and down-R for the 3′-terminal.
Both ends of target BAC clones were sequenced with T7 and SP6 universal primers, respectively. A 1.2-kb DNA sequence located on the 3′-terminal of BAC clone 1F8 was found to encode a putative protein ZWA6. The 2-kb DNA sequence following it was tested by means of primer walking with BAC clone 5E2 as a template. Another two potential proteins ZWA5A and ZWA5B were found. All of these three genes, zwa6, zwa5A and zwa5B were deposited to GenBank under accession no. DQ120781.
Southern hybridization
Southern hybridization was performed to determine the location of the ZwA biosynthesis cluster. A 1,058-bp DNA fragment encoding 2,3-diaminopropionate synthetase was amplified from strain YBT-1520 with primer 5BU and primer 5BD and labeled by (α-32P)-dGTP with hexanucleotide random primer amplification. Intact total DNA from bacteria was separated by PFGE and then transferred to a nylon membrane. Southern hybridization was performed according to a standard protocol (Sambrook and Russell 2001).
Results and discussion
Identification of ZwA produced by Bacillus thuringiensis YBT-1520
The initial purified ZwA from strain YBT-1520 was analyzed by HPLC. There is a main peak distinguish from the others obviously (Fig. 2a). The fraction eluted from 5.5 to 6.0 min was collected to give pure ZwA. This purified sample showed activity to inhibit Erwinia herbicola LS005 (data not shown), which is sensitive to ZwA. The mass spectrogram (Fig. 2b) showed that the molecular weight of this sample is 397 Da, the molecular weight of ZwA determined by Handelsman’s group (Silo-Suh et al. 1994). Previous studies also showed that many B. thuringiensis strains could produce ZwA, especially B. thuringiensis subsp. kurstaki (Nair et al. 2004). In addition, ZwA-resistant gene zmaR, which is necessary for ZwA production (Milner et al. 1996; Stohl et al. 1999b), was detected from YBT-1520 by PCR and sequence analysis (unpublished data).
HPLC-MS analysis of ZwA produced by B. thuringiensis. a The chromatogram (C18 column was used, 40% methanol–water used as mobile phase, monitored at 220 nm wavelength) of initial purified antibiotic. b The mass spectrogram (electron ionization, 70 eV were used) of the fraction collected from 5.5 to 6.0 min in HPLC analysis
BAC library of Bacillus thuringiensis YBT-1520
To isolate the ZwA biosynthesis cluster, a BAC library of B. thuringiensis YBT-1520 was constructed. This library consists of 960 clones, with an average insert size of 50 kb and a distribution of insert size ranging from 24.5 to 72.5 kb. Assuming the size of the YBT-1520 genome is 5 Mb, the library would have a 9.6-fold coverage of the YBT-1520 genome and the probability of the library covering any particular 1 kb gene is 99.999%.
Identification of ZwA biosynthesis-related BAC clones
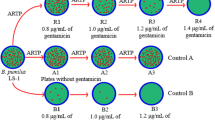
PCR amplification showed that there were nine BAC clones overlapping zwa16Bc. Among them, clone 1G7, 4B1 and 5C8 overlap 671 bp region of the 5′-terminal (nt54–nt724), clone 1F8, 4C5 and 5E2 overlap 1,005 bp region of the 3′-terminal (nt14847–nt15851), while clone 1H4, 3D7 and 4B4 overlap both.
As for clone 1F8, its 5′-terminal DNA analysis showed 676-bp DNA sequence shares 98% nucleotide identities with zmaR in zwa16Bc (nt621–nt1296). Another pair of primers named PKS-U and PKS-D was designed to check whether clone 1F8 contains the pks gene, which is present at the 3′-terminal of zwa16Bc. A 1,069-bp DNA fragment was amplified from clone 1F8 with this primer pair. DNA sequence of PCR product has a 97% nucleotide identity to the corresponding region in pks gene (nt11201–nt12269). This implied that clone 1F8 covers a counterpart of zwa16Bc (Fig. 3).
Schematic show of the putative alignment of BAC clones 1F8 and 5E2 relative to the ZwA biosynthesis cluster of B. cereus strain UW85. The diagonal frame represents part of ZwA biosynthesis cluster, zwa16Bc, identified from B. cereus UW85. All of the DNA fragments in the dashed frames were sequenced and analyzed, DNA identity percentages of corresponding sequence are as shown in the middle of the frames
The 3′-terminal DNA sequence of clone 1F8 was determined and PCR primers were then designed according to the determined sequence. PCR amplification found that clone 5E2 overlaps the 3′-terminal of clone 1F8. Furthermore, PCR amplification (with primer pair down-F/R and primer pair PKS-U/D) and DNA analysis showed that clone 5E2 overlaps the 3′-terminal and pks gene of zwa16Bc as well as 1F8 (Fig. 3).
These results indicated that the gene organization of ZwA biosynthesis cluster of B. thuringiensis YBT-1520 is most likely similar to that of B. cereus UW85.
Identification and characterization of zwa6, zwa5A and zwa5B genes
In the proposed ZwA biosynthesis pathway, neither the carbamoyltransferase gene nor the 2,3-diaminopropionate synthetase gene has been included in zwa16Bc (Emmert et al. 2004).
Interestingly, end DNA sequencing found that the 5′-terminal of clone 1F8 shares high similarity to the carbamoyltransferase gene. An intact open reading frame (ORF) zwa6 was identified from clone 1F8 by walking sequencing with primer A. Based on the sequence of zwa6, three ORFs (zwa6–zwa5A–zwa5B) were identified from clone 5E2 by primer walking with five sequencing primers (primer B, C, D, E and F) step by step. At the upstream of zwa5B, no obvious promoter could be recognized. Therefore, most likely these genes are in the same operon with zwa5A and share a common promoter.
Gene zwa6 encodes a putative protein sharing 56% amino acid identity with ornithine carbamoyltransferase from Streptomyces avermitilis (Omura et al. 2001). According to the proposed ZwA biosynthesis hypothesis (Emmert et al. 2004), ZWA6 would be the candidate enzyme catalyzing the premature ZwA into ZwA in the last step of ZwA biosynthesis (Fig. 1b). Further work is in progress to test this hypothesis.
Meanwhile, ZWA5A and ZWA5B share 39% identity and 33% identity to VioB and VioK, which are homologs of cysteine synthetase and ornithine cyclodeaminase and involved in viomycin biosynthesis by concerted actions to synthesize the precursor—2,3-diaminopropionate (Thomas et al. 2003), respectively. Therefore, it is reasonable to draw a conclusion that ZWA5A and ZWA5B synthesize 2,3-diaminopropionate by their concerted actions in ZwA biosynthesis (Fig. 1b) and that 2,3-diaminopropionate is actually one of the precursors of ZwA. PCR amplification showed zwa6, zwa5A and zwa5B could be amplified from strain UW85 with primer pairs 6U/6D, 5AU/5AD and 5BU/5BD, respectively. PCR product shares 97% DNA identity with zwa6–zwa5A–zwa5B from B. thuringiensis YBT-1520. All these three genes cloned from B. cereus are registered in GenBank as DQ336618.
Furthermore, according to the insert size in BAC clone 1F8 (36 kb) and its alignment with zwa16Bc (Fig. 3), the minimal size of ZwA biosynthesis cluster should be 38.6 kb (36 kb plus the 2 kb fragment of zwa5A and zwa5B, and 0.6 kb of zmaR upstream). However, it is necessary to determine whether the 38.6-kb DNA fragment is sufficient for the biosynthesis of ZwA. Long-range PCR with down-F and 5BD as primers showed that the distance between 3′-terminal of zwa16Bc and zwa6 gene is 19.6 kb (subtracting 1 kb of zwa16Bc 3′-terminal and 3 kb of zwa6–zwa5A–zwa5B from the total 23.6 kb PCR product) in clone 5E2, B. cereus UW85 and B. thuringiensis YBT-1520 (Fig. 4). The 23.6-kb DNA fragment amplified from B. thuringiensis YBT-1520 and B. cereus UW85 showed similar restriction fragment length polymorphisms except that B. thuringiensis YBT-1520 has two more HindIII sites, while B. cereus UW85 has one more EcoRI site. It indicates that ZwA biosynthesis clusters from YBT-1520 and UW85 have a similar organization.
Restriction fragment length polymorphisms of the 23.6-kb DNA fragment amplified from B. thuringiensis YBT-1520, B. cereus UW85 and BAC clone 5E2. Lane 1 UW85/HindIII, lane 2 YBT-1520/HindIII, lane 3 5E2/HindIII, lane 4 UW85/EcoRI, lane 5 YBT-1520/EcoRI, lane 6 5E2/EcoRI, lane 7 UW85, lane 8 YBT-1520, lane 9 5E2, lane 10 lambda DNA/HindIII (size in kb)
Localization of the ZwA biosynthesis cluster
As far as we know, several antibiotic biosynthesis gene clusters have been found on the chromosome of the host, even though in many Streptomyces and other bacteria, gene(s)/gene cluster responsible for antibiotics biosynthesis are located on plasmids, instead of chromosome. Some B. thuringiensis and B. cereus can produce ZwA, but not all of them (Silo-Suh et al. 1994; Nair et al. 2004). No ZwA biosynthesis cluster was found from the genome sequence of B. cereus group (Ivanova et al. 2003; Read et al. 2003). This tells us that ZwA biosynthesis cluster is not a constitutive component of their chromosome.
Pulse field gel electrophoresis showed that ZwA producing strains (e.g., B. thuringiensis strain YBT-1520, HD1 and B. cereus strain UW85 and UW56) have gigantic plasmids (Fig. 5). To determine the location of the ZwA biosynthesis genes of these strains, the gigantic plasmids were separated from their chromosomes by PFGE and southern hybridization was performed with a 1,058-bp DNA fragment containing zwa5B as the probe. Results showed that zwa5B locates on the chromosome, but not on the plasmid (Fig. 5). Antibiotic biosynthesis genes are usually organized in a gene cluster (Du et al. 2000; Wu et al. 2000; Ahlert et al. 2002). If it is also true in these strains we tested, the ZwA biosynthesis clusters should locate on their chromosomes.
Southern blot analysis of four ZwA-producing strains. Intact total DNA of four ZwA-producing strains prepared from plugs were separated by PFGE (left panel; 1% agarose gel, 6 V/cm, 14°C for 18 h; switch times ramped from 1 to 25 s), transferred onto a nylon membrane and followed by hybridization with a 1,029-bp DNA fragment containing zwa5B as probe. The autoradiograph is as shown on the right panel. Lane 1 MidRange II PFG markers (size in kb), lane 2 B. thuringiensis HD1, lane 3 B. cereus UW56, lane 4 B. cereus UW85, lane 5 B. thuringiensis YBT-1520
Abbreviations
- A:
-
Adenylation domain
- ACP:
-
Acyl carrier domain
- BGSC:
-
Bacillus Genetic Stock Center
- C:
-
Condensation domain
- KS:
-
Ketosynthase
- KR:
-
Ketoreductase
- NRPS:
-
Nonribosomal peptide synthetase
- PCP:
-
Peptidyl carrier protein
- PKS:
-
Polyketide synthase
- PLP:
-
Pyridoxal 5′-phosphate
- ZwA:
-
Zwittermicin A
References
Ahlert J, Shepard E, Lomovskaya N, et al (2002) The calicheamicin gene cluster and its iterative type I enediyne PKS. Science 297:1173–1176
Ansari MZ, Yadav G, Gokhale RS, Mohanty D (2004) NRPS–PKS: a knowledge-based resource for analysis of NRPS/PKS megasynthases. Nucleic Acids Res 32(web server issue):W405–W413
Broderick NA, Goodman RM, Raffa KF, Handelsman J (2000) Synergy between zwittermicin A and Bacillus thuringiensis subsp. kurstaki against gypsy moth (Lepidoptera: Lymantriidae). Environ Entomol 29:101–107
Broderick NA, Goodman RM, Handelsman J, Raffa KF (2003) Effect of host diet and insect source on synergy of gypsy moth (Lepidoptera: Lymantriidae) mortality to Bacillus thuringiensis subsp. kurstaki by zwittermicin A. Environ Entomol 32:387–391
Du LC, Sánchez C, Chen M, Edwards DJ, Shen B (2000) The biosynthetic gene cluster for the antitumor drug bleomycin from Streptomyces verticillus ATCC15003 supporting functional interactions between nonribosomal peptide synthetases and a polyketide synthase. Chem Biol 7:623–642
Emmert EA, Klimowicz AK, Thomas MG, Handelsman J (2004) Genetics of zwittermicin A production by Bacillus cereus. Appl Environ Microbiol 70(1):104–113
He HY, Silo-Suh LA, Handelsman J, Clardy J (1994) Zwittermicin A, an antifungal and plant protection agent from Bacillus cereus. Tetrahedron Lett 35(16):2499–2502
Ivanova N, Sorokin A, Anderson I, et al (2003) Genome sequence of Bacillus cereus and comparative analysis with Bacillus anthracis. Nature 423(6935):87–91
Luo MZ, Wing RA (2003) An improved method for plant BAC library construction. In: Grotewold E (ed) Methods in molecular biology. Humana, Totowa
Milner JL, Stohl EA, Handelsman J (1996) Zwittermicin A resistance gene from Bacillus cereus. J Bacteriol 78:4266–4272
Nair JR, Narasimman G, Sekar V (2004) Cloning and partial characterization of zwittermicin A resistance gene cluster from Bacillus thuringiensis subsp. kurstaki strain HD1. J Appl Microbiol 97:495–503
Omura S, Ikeda H, Ishikawa J, et al (2001) Genome sequence of an industrial microorganism Streptomyces avermitilis: deducing the ability of producing secondary metabolites. Proc Natl Acad Sci USA 98(21):12215–12220
Read T, Peterson S, Tourasse N, et al (2003) The genome sequence of Bacillus anthracis Ames and comparison to closely related bacteria. Nature 423(6935):81–86
Rondon MR, Raffel SJ, Goodman RM, Handelsman J (1999) Toward functional genomics in bacteria: analysis of gene expression in Escherichia coli from a bacterial artificial chromosome library of Bacillus cereus. Proc Natl Acad Sci USA 25.96:6451–6455
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Schnepf E, Crickmore N, Van Rie J, et al (1998) Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol Mol Biol Rev 62:775–806
Silo-Suh LA, Lethbridge BJ, Raffel SJ, He HY, Clardy J, Handelsman J (1994) Biological activities of two fungistatic antibiotics produced by Bacillus cereus UW85. Appl Environ Microbiol 60(6):2023–2030
Silo-Suh LA, Stabb EV, Raffel SJ, Handelsman J (1998) Target range of zwittermicinA, an aminopolyol antibiotic from Bacillus cereus. Curr Microbiol 37:6–11
Stabb EV, Handelsman J (1998) Genetic analysis of zwittermicin A resistance in Escherichia coli: effects on membrane potential and RNA polymerase. Mol Microbiol 27(2):311–322
Stabb EV, Jacobson LM, Handelsman J (1994) Zwittermicin A-producing strains of Bacillus cereus from diverse soils. Appl Environ Microbiol 60(12):4404–4412
Stohl EA, Milner JL, Handelsman J (1999a) Zwittermicin A biosynthetic cluster. Gene 237:403–411
Stohl EA, Brady SF, Clardy J, Handelsman J (1999b) ZmaR, a novel and widespread antibiotic resistance determinant that acetylates zwittermicin A. J Bacteriol 181:5455–5460
Sun M, Liu Z, Yu Z (2000) Characterization of the insecticidal crystal protein genes of Bacillus thuringiensis YBT-1520. Wei Sheng Wu Xue Bao 40(4):365–371
Thomas MG, Chan YA, Ozanick SG (2003) Deciphering tuberactinomycin biosynthesis: isolation, sequencing and annotation of the viomycin biosynthetic gene cluster. Antimicrob Agents Chemother 9:2823–2830
Wu K, Chung L, Revill WP, Katz L, Reeves CD (2000) The FK520 gene cluster of Streptomyces hygroscopicus var. ascomyceticus (ATCC 14891) contains genes for biosynthesis of unusual polyketide extender units. Gene 251:81–90
Acknowledgments
We thank Meizhong Luo for his helpful advice on library construction and Jo Handelsman for providing B. cereus strains UW85 and UW56. This project was supported by the National High Technology Research & Development Program (863 Program) of China (2006AA02Z174) and the National Basic Research Program (973 Program) of China (2003CB114201).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, C., Luo, Y., Song, C. et al. Identification of three Zwittermicin A biosynthesis-related genes from Bacillus thuringiensis subsp. kurstaki strain YBT-1520. Arch Microbiol 187, 313–319 (2007). https://doi.org/10.1007/s00203-006-0196-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-006-0196-3