Abstract:
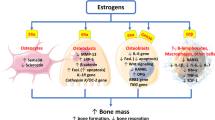
A total of 277 early postmenopausal women were enrolled in this placebo-controlled 2-year study to examine the efficacy of a matrix transdermal 17β-estradiol system, at three different dosages (25, 50 and 75 mg/day) combined with sequential oral dydrogesterone 20 mg/day, in preventing bone loss. At 2 years, the difference from placebo in percentage change from baseline of L1–4 lumbar spine bone mineral density (BMD) (assessed by dual-energy X-ray absorptiometry) was 4.7%± 0.7% with estradiol 25 mg/day, 7.3%± 0.7% with estradiol 50 mg/day and 8.7%± 0.7% with estradiol 75 mg/day (all values mean ± SEM). There were also significant increases in femoral neck, trochanter and total hip BMD with all doses of estradiol compared with placebo. Additionally, most patients had a significant gain (increase greater than 2.08%) in lumbar spine bone mass compared with placebo. Patients who received estradiol also experienced clinically significant and dose-related decreases in total serum osteocalcin, serum bone alkaline phosphatase and urinary C-telopeptide, with all three markers of bone turnover returning to premenopausal levels. Estradiol was well tolerated during the 2-year treatment period. Transdermal estradiol is effective and well tolerated at dosages between 25–75 mg/day in the prevention of bone loss in postmenopausal women; 25 mg/day offers an effective option for those women who cannot tolerate higher doses.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Consortia
Additional information
Received: 30 June 1998 / Accepted: 22 September 1998
Rights and permissions
About this article
Cite this article
Cooper, C., Stakkestad, J., Radowicki, S. et al. Matrix Delivery Transdermal 17β-Estradiol for the Prevention of Bone Loss in Postmenopausal Women . Osteoporos Int 9, 358–366 (1999). https://doi.org/10.1007/s001980050159
Issue Date:
DOI: https://doi.org/10.1007/s001980050159