Abstract
Summary
We investigated the timeline of functional recovery after hip fracture over 12 months in adults age ≥ 65 years using objective lower extremity function tests and subjective physical functioning. Objective functional recovery was largely complete in the first 6 months, whereas subjective recovery improved up to 9 months after hip fracture.
Introduction
Hip fractures are a major cause of loss of function among seniors. We assessed the timeline of objective and subjective functional recovery after hip fracture.
Methods
We conducted a prospective observational secondary analysis of a 1-year clinical trial on vitamin D and home exercise treatment and complications after hip fracture among 173 patients age ≥ 65 years (mean age 84 years; 79.2% women; 77.4% community-dwelling) conducted from January 2005 through December 2007. Lower extremity function (Timed Up and Go test (TUG), knee extensor and flexor strength) and grip strength was assessed at baseline and at 6 and 12 months follow-up. Subjective physical functioning was assessed using the SF-36 questionnaire also at 3 and 9 months follow-up. Multivariable-adjusted repeated-measures models were used to assess the timeline of functional recovery in the total population and in subgroups of patients.
Results
Lower extremity function including TUG (− 61.1%), knee extensor (+ 17.6%), and knee flexor (+ 11.6%) strength improved significantly in the first 6 months (P < 0.001). However, between 6 and 12 months, there was no further significant improvement for any of the functional tests. Grip strength decreased from baseline to 6 months (− 7.9%; P < 0.001) and from 6 to 12 months (− 10.8%; P < 0.001). Subjective physical functioning improved from 3 to 9 months (+ 15.2%, P < 0.001), but no longer thereafter.
Conclusions
Functional recovery after hip fracture may be largely complete in the first 6 months for objective functional tests, whereas may extend up to 9 months for subjective recovery, with oldest-old, female, institutionalized, and cognitively impaired patients recovering most poorly.
Clinical trials registry (original trial)
NCT00133640.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hip fractures are a major cause of functional impairment [1] as well as of loss of autonomy and mortality [2, 3] in senior adults. Consequently, hip fractures constitute a major public health and socio-economic burden [4, 5], with associated cost estimates ranging from $19,000 to $66,000 per fracture [2]. About 1.6 million hip fractures occur worldwide each year [6], and due to the imminent demographic change, the global volume of hip fractures has been estimated to reach about 4.5 [7] up to 6.3 [8] million per year by 2050.
In the first year after a hip fracture, reported mortality rates range up to 36% [3, 9, 10] and a greater risk of mortality may persist for at least 5 years after the index fracture [11]. Moreover, about 18–40% of patients are readmitted to acute care within the first 12 months after their hip fracture [10, 12, 13]. Hospital readmissions and prolonged hospitalizations after hip fracture are often associated with a delay in the recovery process, incidence of complications, and mortality [13,14,15]. Further, the frequent loss of function and decrease in mobility among hip fracture survivors may provoke reduction in quality of life [16, 17] and may lead to loss of autonomy and nursing home admission in up to 30% of cases [9]. Notably, less than half of hip fracture patients regain their pre-fracture level of function [18, 19], and about 60% have been reported to require assistance even at 12 months after their hip fracture [16]. Therefore, next to prevention, rehabilitation efforts after hip fracture have become a public health priority [20] to maintain autonomy and reduce the enormous health economic burden of hip fractures [5].
Although several studies have investigated patterns of recovery after hip fracture [10, 21,22,23,24,25], most studies used subjective self-report data from questionnaires only [10, 22, 24,25,26,27] and findings about when to measure meaningful functional outcomes and when to initiate rehabilitation after the acute hip fracture are inconsistent [28]. Moreover, it has been recommended to use both performance-based and self-reported functional measures to assess functional recovery after hip fracture because both measures impart distinct but complementary information on functional status [29]. While objective measures are specific for the respective function measured as well as independent of personal perception and experience, subjective measures are influenced by a person’s perception and experience and thus do not only reflect the quantity, but also the quality of recovery, and may also not be independent from other similar recovery measures but part of a more complex psychological construct [30]. Consequently, objective and subjective measures of functional recovery together can be seen as a complementary continuum ranging from highly objective (e.g., clinical test parameters) to highly subjective (e.g., integrated measures of a physical functioning construct). Which objective or subjective aspects of functional recovery finally should be measured depends on the specific research question and whether objective or subjective measures, or both, are considered important for the research question [30].
To our knowledge, besides one study [10] that, however, did not use muscle strength tests, no other study has used objective lower and upper extremity function and strength tests combined with self-reported subjective physical functioning (SPF) to assess the timeline of functional recovery after hip fracture and to get an overall picture of the functional recovery process as a continuum of highly objective and subjective functional recovery measures.
Therefore, the aim of the present study was to determine the overall timeline of functional recovery after hip fracture utilizing data on objective lower extremity function tests and SPF measures in senior men and women sustaining an acute hip fracture at age 65 and older that was originally collected as part of a clinical trial. A second aim was to investigate whether subjective and objective functional recovery over time may differ between subgroups of senior adults by gender, age, body mass index (BMI), number of comorbidities, baseline vitamin D status, living condition prior to the hip fracture (home vs. assisted living/nursing home), and baseline cognitive function.
Methods
Study design and participants
The present study is a prospective observational secondary analysis utilizing data of a 1-year randomized controlled clinical trial conducted from January 2005 through December 2007 (ClinicalTrials.gov Identifier: NCT00133640) by pooling the data of the individual treatment arms of the trial. The original trial investigated the effect of vitamin D (2000 vs. 800 IU/d cholecalciferol) and/or a home exercise program (simple home exercise program in addition to standard physiotherapy vs. no home exercise program in addition to standard physiotherapy) on complications after hip fracture surgery among 173 patients age 65 years and older (range 65 to 99 years, mean age 84 years; 79.2% women; 77.4% community-dwelling) using a 2 × 2 factorial design; i.e., there were 4 treatment arms of all possible combinations of the two treatments (dose of cholecalciferol and/or home exercise program yes/no), with the treatment 800 IU/day cholecalciferol and standard physiotherapy but no home exercise program being considered the standard of care [12].
During the first year after hip fracture, repeated measures of objective functional tests were obtained at baseline (day 1–12 after hip fracture surgery) and at 6 and 12 months by the same trained physiotherapist, and for SPF repeated measures were taken at baseline and at 3, 6, 9, and 12 months after hip fracture surgery by a trained study nurse. The results of this trial in terms of the treatment effect on complications after hip fracture [12] and functional recovery after hip fracture [31] have been published recently. For the present observational analysis, we re-utilized the dataset collected during the original trial by pooling all treatment arms, and we applied multivariable modeling with adjustment for treatment and possible confounders to investigate the timeline of functional recovery after hip fracture in all participants of the original trial.
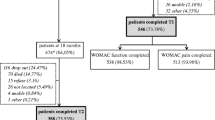
During the 12-month observational period, of the 173 participants, a total of 45 participants dropped out of the follow-up (20 died, 10 stopped for personal reasons, 6 withdrew because of illness and overall decline, and 9 withdrew because they wanted to discontinue the study medication therapy). Due to these drop outs, the analytical sample size at 6 months and 12 months was n = 129 and n = 128, respectively. In 2018, we published a similar prospective observational secondary analysis as the present study on the association between caregiver role and functional recovery after hip fracture using the same dataset and the same pooling and methodological approach of data analysis [32].
Assessment of physical functioning
Objective functional tests
Objective functional tests included the Timed Up and Go test (TUG), knee extensor (KES), knee flexor (KFS), and hand grip (GS) strength [33]. All tests were performed at baseline (day 1–12 after hip fracture surgery) and at the 6- and 12-month follow-up and had been validated in a prior study among frail seniors [33].
TUG
The TUG is a standardized objective functional test that measures the time (in seconds) to rise from a chair, walk 3 m, turn around, go back, and sit down on the chair again by stopwatch [34]. The smaller the TUG values (less seconds needed to perform the test), the better the test performance. Values below 12 s reflect normal mobility among community-dwelling adults [35].
KES and KFS
A pull gauge (Hand-Held Dynamometer, Typ Mecmesin, BFG 1000) was used to assess KES and KFS. The measurements (kilogram force (kgf), with1 kgf = 9.80665 Newton) were conducted on the not fractured leg [36]. The position of the patient was predetermined for each measurement in such a way that gravitational forces did not influence the assessment. The angle of the joint involved was predefined and was usually neutral or at 90° if in flexion. The patient was instructed to increase its isometric muscle strength continuously and gradually until the maximum strength plateau was reached. For both assessments, knee joint was flexed at 90° and hands were relaxed or folded. The band of the pull gauge was placed on the lower leg, with a distal border to the ankle joint as close as possible. The upper legs were fixed using a belt close to the knee joint, and the gauge was stabilized by being leaned against the frame of the examination table. For KES, the patient had to press the lower leg slowly forward against the band, whereas for KFS, the patient had to press the lower leg slowly backward against the band [37].
GS
A Martin Vigorimeter was used to assess the muscular strength (Bar, 1 Bar = 100 kPa) of the dominant hand and forearm. The patient was seated on a chair with the elbow flexed at 90°. The rubber ball of the Martin Vigorimeter was in the palm, between fingers 1–4 and the thumb, and the patient had to squeeze the ball slowly [38].
Self-reported physical functioning
SPF was assessed using the RAND Short-Form 36 Health Survey (SF-36) questionnaire (version 1.0) [39] to record self-reported pre-fracture as well as 3, 6, 9-month and 12-month follow-up mobility. Of the eight health concepts included in the SF-36, we used the 10-item section on physical functioning that describes limitations in physical performance concerning their presence and extent due to health restriction [39]. These 10 items assess vigorous and moderate activities including climbing one or several flights, lift and carry groceries, bend knee, walk one or several blocks, walking a mile, and grooming (bathing, dressing) independently [39]. Each item was rated on a three-point scale (yes, limited a lot; yes, limited a little; and no, not limited at all). Answers to each question were transformed to a 0–100 scale, summed, and divided by 10.
Assessment of covariates
Age, gender, Charlson comorbidity index (CCI, score 0–37) [40], living situation (at home vs. assisted living/nursing home), and global cognitive function measured by the total score of the Folstein mini-mental state examination (MMSE, score 0–30) [41] were assessed at baseline during acute care. Baseline weight (kg) and height (cm) were measured with subjects dressed in light clothing without shoes, and BMI (in kg/m2) was calculated as weight divided by height squared. Baseline 25-hydroxyvitamin D (25(OH)D) status was measured using collected venous blood and a radioimmunoassay (Diasorin, Inc., Stillwater, Minnesota). Treatment of the 2 × 2 factorial design of the original trial comprised extended physiotherapy (supervised 60 min/day during acute care plus an unsupervised home program) versus standard physiotherapy (supervised 30 min/day during acute care plus no home program) and cholecalciferol therapy (2000 versus 800 IU/day).
Statistical analysis
Statistical analysis was performed using SAS, version 9.4 (SAS Institute, Inc., Cary, North Carolina, USA). Differences in subject characteristics between men and women were analyzed by using a χ2 test for categorical and a Student’s t test for continuous variables.
Associations between repeated measurements of objective or subjective physical performance variables (response variables) and time (predictor variable) were analyzed using multivariable linear mixed models with random intercept and random slope treating time as a random variable and subject as identifier. Given the specific dataset of the original trial, to correct our analysis for the treatment of the original trial, a possible interaction between treatment and time, and other possible confounders (i.e., any variable that may bias the association between the recovery time and the functional outcomes of interest because this variable may also influence the functional recovery after hip fracture), our models were adjusted for the treatment group of the original intervention trial and an interaction term for treatment by time, as well as for baseline age, gender, and BMI (basic adjusted model 1) or additionally for baseline CCI (score 0–37), living situation (at home vs. assisted living/nursing home), total score of the MMSE (score 0–30), and baseline blood 25(OH)D concentration (fully adjusted model 2). To justify the pooled analysis of the trial data, we tested whether the associations observed between the objective or subjective physical performance variables and time were modified by the assignment of the subjects to different treatment arms by testing for interaction effects between the treatment arms and time. However, since the interaction terms in the models were not statistically significant (P > 0.5) and the results did not change, we were able to use the pooled dataset for our observational analysis.
Moreover, first-, second-, or third-order polynomials of time, respectively, were used in the linear mixed models to test for a linear, quadratic, or cubic trend of change in the physical performance variables across the repeated measures.
In subgroup analysis by baseline age (< 85 vs. ≥ 85 year), gender, BMI (< 25 vs. ≥ 25 kg/m2), number of comorbidities (< 3 vs. ≥ 3), 25(OH)D status (< 10 vs. ≥ 10 ng/mL), living condition prior to the hip fracture (home vs. assisted living/nursing home), and baseline cognitive function measured by MMSE total score (< 25 vs. ≥ 25), models were adjusted for treatment of the original intervention trial only. Differences in recovery between subgroups over time were tested by using the change of the repeated measurements over time as outcome variable and adjusting for the respective baseline values of the measurements. Statistical significance was set at P < 0.05; reported P values are two-sided.
Results
Subject characteristics
Subject characteristics are presented by gender (Table 1). The ratio of men to women was 1:3.8. Men had a higher CCI (P = 0.005) and higher KES, KFS, and GS (P < 0.001) than women. There were no significant differences in the distribution of subgroups of age, BMI, CCI, living condition, total score of the MMSE, or baseline 25(OH)D status between men and women.
Repeated measurements of objective and subjective functional recovery after hip fracture
In fully adjusted models, adjusted mean values (least square means (LSM)) of the TUG (− 61.1%), KES (+17.6%), and KFS (+ 11.6%) improved in the first 6 months after hip fracture surgery (P < 0.001), but leveled off between 6 and 12 months (Fig. 1a–c; Table 2), following a positive or negative quadratic trend in time (P < 0.001), respectively. By contrast, GS decreased linearly from baseline to 12 months (− 18.2%, P < 0.001) (Fig. 1d; Table 2). Ratings of SPF decreased (− 27.8%) from pre- to postoperative state (P < 0.001), whereas improved equally from 3 to 9 months (+ 15.2%, P < 0.001), and did not improve any longer, but even deteriorated between 9 and 12 months (− 3.2%, P = 0.03) (Fig. 2; Table 2), following a negative cubic trend in time (P < 0.001). In Table 2, the corresponding exact mean values (LSM) for the fully adjusted models (model 2) as depicted in Figs. 1 and 2 are shown for all objective and subjective functional measures. In addition, Table 2 also presents the exact values (LSM) of the basic adjusted models (model 1) for all objective and subjective functional measures. Moreover, Table 2 provides the exact values for the mean changes (i.e., ∆ LSM) in all objective and subjective functional measures between individual follow-up time points, as well as for the changes (i.e., ∆ LSMBL) between the baseline measurement and individual follow-up time points over the 12-month follow-up period. Finally, Table 2 provides information (i.e., Ptrend linear, Ptrend quadratic, or Ptrend cubic) whether the changes across the repeated measurements of the individual objective and subjective functional measures over the 12 months followed a linear, quadratic, or cubic trend in the pattern of functional recovery after hip fracture.
Temporal profiles of performance in the a Timed Up and Go test, b knee extensor strength (kgf, 1 kgf = 9.81 Newton), c knee flexor strength (kgf, 1 kgf = 9.81 Newton), and d hand grip strength, in older adults ≥65 years after hip fracture (n = 173). Values are adjusted mean values (LSM ± SE) from multivariable linear mixed models adjusted for the treatment group of the original intervention trial and an interaction term for treatment by time, as well as for age, gender, and body mass index (kg/m2), Charlson comorbidity index (score 0–37), living situation (at home vs. assisted living/nursing home), total score of the mini-mental state examination (score 0–30), and blood 25-OH vitamin D concentration at baseline. Baseline measurements (month 0) were performed on days 1–12 after hip fracture surgery. LSM, least square mean (an adjusted mean value of an outcome variable; i.e., the originally observed mean value of an outcome variable was adjusted for the influence of possible confounders stated in the description of the model adjustments above); SE, standard error
Temporal profiles of ratings of subjective physical functioning in older adults ≥ 65 years after hip fracture (n = 173). Values are adjusted mean values (LSM ± SE) from multivariable linear mixed models adjusted for treatment group of the original intervention trial and an interaction term for treatment by time, as well as for age, gender, and body mass index (kg/m2), Charlson comorbidity index (score 0–37), living situation (at home vs. assisted living/nursing home), total score of the mini-mental state examination (score 0–30), and blood 25-OH vitamin D concentration at baseline. Baseline measurements (month − 1) refer to the month preceding the hip fracture surgery. LSM, least square mean (an adjusted mean value of an outcome variable; i.e., the originally observed mean value of an outcome variable was adjusted for the influence of possible confounders stated in the description of the model adjustments above); SE, standard error
Differences in physical recovery after hip fracture between subgroups over time
Overall, subgroup analysis (Supplementary Figs. 1–5) by baseline gender, age (< 85 vs. ≥ 85 years), BMI (< 25 vs. ≥ 25 kg/m2), number of comorbidities (< 3 vs. ≥ 3), living condition prior to the hip fracture (home vs. assisted living/nursing home), cognitive function measured by MMSE (< 25 vs. ≥ 25), and 25(OH)D status (< 10 vs. ≥ 10 ng/mL), revealed a similar pattern of change for individual subgroups across the repeated measurements as observed for the total population, i.e., an improvement in physical recovery in the first 6 months (objective performance tests) or 3 to 9 months (SPF ratings), with no further improvement thereafter. As for the total population, the only exception was GS that consistently decreased with time.
Regarding the overall absolute differences in performance between subgroups over 12 months, as expected men, patients < 85 years, and patients with BMI < 25 kg/m2 (except for KES and KFS), number of comorbidities < 3 (except for KES, KFS, and GS), baseline 25(OH)D status ≥ 10 ng/mL, living at home prior to the hip fracture, and MMSE ≥ 25 performed significantly or nominally better in all objective and subjective physical functioning measures than patients of the respective other subgroup. Also, regarding the relative recovery, i.e., the change in performance between subgroups over time, independent of the baseline performance, for both objective (TUG) and subjective (SPF) lower extremity function, patients < 85 years, those living at home prior to the hip fracture, and those with MMSE ≥ 25 recovered better over 12 months than patients of the respective other subgroup. For the three muscular strength tests KES, KFS, and GS, men and patients < 85 years recovered better within 12 months than women and patients ≥ 85 years.
Objective functional recovery
TUG (Supplementary Fig. 1). At 6 and 12 months, patients < 85 years (P < 0.01) and patients living at home prior to the hip fracture (P < 0.01), performed absolutely better in the TUG than patients ≥ 85 years and patients living in assisted facilities or a nursing home. Over time, patients ≥ 85 years recovered better in the TUG than patients < 85 years between baseline and 6 months (Page x time = 0.0002), whereas between 6 and 12 months patients < 85 years still improved while patients ≥ 85 years got worse (Page x time = 0.0006). Moreover, patients living at home prior to the hip fracture recovered better in the TUG between baseline and 12 months than patients living in assisted facilities or a nursing home (Pliving x time < 0.0001), and patients with MMSE ≥ 25 recovered better between baseline and 6 months than patients with MMSE < 25 (PMMSE × time < 0.03). Finally, patients with a 25(OH)D status ≥ 10 ng/mL recovered better in the TUG than patients with < 10 ng/mL 25(OH)D status between 6 and 12 months (Pinteraction = 0.02).
KES (Supplementary Fig. 2). At 6 and/or 12 months, men (P < 0.0001), patients < 85 year (P < 0.01), patients with BMI ≥ 25 (P < 0.01), patients living at home (P < 0.05), or patients with MMSE ≥ 25 (P < 0.001) had absolutely higher KES than women and patients ≥ 85 years, patients with BMI < 25, patients living in assisted facilities or a nursing home, and patients with MMSE < 25. Over time, recovery in KES between subgroups was only significantly better among men between baseline and 6 months compared to women (Pgender x time = 0.005).
KFS (Supplementary Fig. 3). At 6 and 12 months, men (P < 0.001) and patients < 85 years (P < 0.0001), patients living at home (P < 0.001), or patients with MMSE ≥ 25 (P < 0.001) had absolutely higher KFS than women and patients ≥ 85 years, patients living in assisted facilities or a nursing home, or patients with MMSE < 25. Over time, recovery in KFS between subgroups was only significantly better among men between baseline and 6 months compared to women (Pgender x time < 0.0001) as well as for patients < 85 years between 6 and 12 months compared to patients ≥ 85 years (Page x time = 0.02), and for patients with MMSE ≥ 25 between baseline and 12 months compared to patients with MMSE < 25 (PMMSE × time = 0.05).
GS (Supplementary Fig. 4). At 6 and 12 months, men (P < 0.0001) and patients < 85 years (P < 0.0001), patients living at home (P < 0.001), or patients with MMSE ≥ 25 (P < 0.05) had absolutely higher GS than women, patients ≥ 85 years, patients living in assisted facilities or a nursing home, or patients with MMSE < 25. Over time, GS worsened over the 12 months, however, with the decrease in GS between subgroups being only significantly larger among women compared to men (Pgender x time = 0.007) and patients ≥ 85 years compared to patients < 85 years (Page x time = 0.03) between 6 and 12 months.
Subjective functional recovery
SPF (Supplementary Fig. 5). At 3, 6, 9, and 12 months (for age subgroups at 3 and 12 months only), men (P < 0.05), patients < 85 years (P < 0.01), patients living at home (P < 0.0001), or patients with MMSE ≥ 25 (P < 0.05) had absolutely higher ratings of SPF than women, patients ≥ 85 years, patients living in assisted facilities or nursing homes, or patients with MMSE < 25. Over time, recovery in SPF between subgroups over 12 months was only significantly better among patients < 85 years (Page × time = 0.01), those living at home prior to the hip fracture (Pliving x time = 0.005), and those with MMSE ≥ 25 (PMMSE × time = 0.005) than among patients ≥ 85 years, patients living in assisted facilities or a nursing home, or patients with MMSE < 25.
Discussion
In this prospective study after acute hip fracture, objective lower extremity function measured by the TUG, KES, and KFS improved significantly in the first 6 months after hip fracture, but no more thereafter. Similarly, SPF measured by the SF-36 improved significantly from 3 to 9 months after hip fracture, but no more thereafter. By contrast, objective upper extremity function measured by GS decreased continuously from baseline to 12 months. In subgroup analysis, younger age (< 85 years), living at home prior to the hip fracture, and having a better cognitive function (MMSE ≥ 25) was advantageous for better recovery after hip fracture for both objective (TUG) and subjective (SF-36) lower extremity function. Men and younger patients (age < 85 years) also recovered better with regard to muscle strength including GS as well as KES and KFS.
In line with our findings, the majority of previous studies reported the time frame of significant improvement in lower extremity function after hip fracture to be within the first 6 months of follow-up [21, 23, 42]. Depending on the specific area of function, however, this time frame has also been reported to range from 3 months [43] or 4 months [10, 22], and up to 9 months [23] and 12 months [10] after hip fracture. Based on our study of a sample of 173 acute hip fracture patients, functional recovery of objective measures of lower extremity function after hip fracture may be complete by 6 months, while recovery of SPF appeared to extend up to 9 months, or may take up to 12 months or even longer based on other studies [24, 44].
Regarding the individual objective functional tests applied in our study, we found improvement after hip fracture for all objective functional tests but GS. This finding is in contrast to previous studies that reported GS as a good indicator for the ability to walk and an excellent correlate of lower limb strength [44,45,46] among hip fracture patients. In support of our finding, however, also Felicio et al. [47] did not find important correlations between GS and KES or KFS in community-dwelling older women unselected for hip fracture.
To date, no consensus has been reached regarding the frequency and timing of physiotherapy to maximize functional recovery after hip fracture surgery [28]. While early physiotherapy may be most beneficial due to its immediate prevention of functional decline [28], a strength training program may be more effective and better tolerated several weeks after the fracture when patients are past frequent physical (i.e., infections) and cognitive (i.e., delirium) postoperative complications [48]. Moreover, for high-intensity strength exercise where certain functional recovery has to occur first, patients may also benefit if applied after the first 6 months of recovery. Our study suggests that objective recovery of lower extremity function may already be complete within the first 6 months after fracture. Thus, based on these findings for objective functional recovery, interventions to improve recovery after hip fracture may be most effective if initiated early and before 6 months after the hip fracture repair, and interventions that target a timeline after the first 6 months may not be recommended. Our additional information on SPF, however, suggests that initiation of interventions may still be useful between 6 and 9 months after hip fracture repair, as SPF improved from 3 to 6 months and 6 to 9 months follow-up, and may also indicate that not all aspects of subjectively perceived functional recovery are captured by individual objective functional recovery measures.
Conceptually, if, e.g., a specific objective physical function measure reaches its peak recovery earlier than a self-reported physical function measure, this may indicate that the perception of the recovery of the specific objective function starts only with a time delay and/or that also other objective physical functions reaching their peak later may have contributed to the overall subjectively perceived progress in functional recovery. On the other hand, if a subjectively perceived function measure reaches its peak earlier than a specific objective function measure, this may indicate that the further improvement in the objective function was not relevant for the subjectively perceived functional recovery. Dependent on the specific research questions, it may therefore be appropriate to (1) measure specific objective recovery measures only (if the recovery of specific objective measures is considered more important than the subjectively perceived recovery), (2) measure subjective recovery only (if the perceived recovery and thus well-being of the person is considered more important than the actual objective physical recovery), or (3) measure both (if the aim is to get an overall picture of the objective and subjective recovery process as an overall continuum) [30].
Our study has several strengths. First, we were able to take advantage of a clinical trial that assessed both objective and subjective measures of functional recovery in a highly standardized way. Second, our results show high consistency between standardized tests of lower extremity function, as well as with our measure of subjective lower extremity function. Third, our study population with a mean age of 84 years and an enrollment of both community-dwelling (70%) and institutionalized (30%) patients is likely representative of an older hip fracture population.
Our study also has limitations. As we have no assessment of objective lower extremity function between baseline (during acute care) and before 6 months follow-up, we lack further details on the early phase of function recovery. However, from our additional assessments of SPF (SF-36) at 3 and 9 months follow-up, we do have an indication that patients may improve significantly between 3 to 6 and 6 to 9 months follow-up. Thus, training programs initiated between 3 and 9 months after the hip fracture may be beneficial. Another limitation is that baseline SPF had to be assessed retrospectively as we recruited participants during acute care of their hip fracture.
In summary, our study provides a consistent signal across three objective measures of lower extremity function (TUG, KES, and KFS) that objective recovery of lower extremity function after hip fracture may be complete after 6 months follow-up, while recovery of SPF may extend up to 9 months. Our study also suggests that younger patients (< 85 year), those living at home prior to their hip fracture, and those with better cognitive function will have better recovery of their lower extremity function, both objectively (TUG) and subjectively (SF-36). Additionally, men may have an advantage over women to regain their upper and lower extremity strength.
Conclusions
Objective functional recovery after hip fracture may be largely complete in the first 6 months, whereas subjective functional recovery may extent up to 9 months after fracture. Rehabilitation of lower extremity function may therefore be most effective if initiated in the first 6 to 9 months after hip fracture, while a later initiation may miss the early window of opportunity where most changes in function can be expected. This may be most important for the oldest-old, women, those living in institutions prior to their hip fracture, and those with reduced cognitive function, as they are prone to recover most poorly from their hip fracture.
References
da Costa JA, Ribeiro A, Bogas M, Costa L, Varino C, Lucas R, Rodrigues A, Araújo D (2009) Mortality and functional impairment after hip fracture—a prospective study in a Portuguese population. Acta Reumatol Port 34:618–626
Braithwaite RS, Col NF, Wong JB (2003) Estimating hip fracture morbidity, mortality and costs. J Am Geriatr Soc 51:364–370
Abrahamsen B, van Staa T, Ariely R, Olson M, Cooper C (2009) Excess mortality following hip fracture: a systematic epidemiological review. Osteoporos Int 20:1633–1650
Barrett-Connor E (1995) The economic and human costs of osteoporotic fracture. Am J Med 98:3S–8S
Leal J, Gray AM, Prieto-Alhambra D, Arden NK, Cooper C, Javaid MK, Judge A, group Rs (2016) Impact of hip fracture on hospital care costs: a population-based study. Osteoporos Int 27:549–558
Johnell O, Kanis JA (2006) An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int 17:1726–1733
Gullberg B, Johnell O, Kanis JA (1997) World-wide projections for hip fracture. Osteoporos Int 7:407–413
Cooper C, Campion G, Melton LJ (1992) Hip fractures in the elderly: a world-wide projection. Osteoporos Int 2:285–289
Leibson CL, Tosteson AN, Gabriel SE, Ransom JE, Melton LJ (2002) Mortality, disability, and nursing home use for persons with and without hip fracture: a population-based study. J Am Geriatr Soc 50:1644–1650
Magaziner J, Hawkes W, Hebel JR, Zimmerman SI, Fox KM, Dolan M, Felsenthal G, Kenzora J (2000) Recovery from hip fracture in eight areas of function. J Gerontol A Biol Sci Med Sci 55:M498–M507
Magaziner J, Lydick E, Hawkes W, Fox KM, Zimmerman SI, Epstein RS, Hebel JR (1997) Excess mortality attributable to hip fracture in white women aged 70 years and older. Am J Public Health 87:1630–1636
Bischoff-Ferrari HA, Dawson-Hughes B, Platz A, Orav EJ, Stähelin HB, Willett WC, Can U, Egli A, Mueller NJ, Looser S, Bretscher B, Minder E, Vergopoulos A, Theiler R (2010) Effect of high-dosage cholecalciferol and extended physiotherapy on complications after hip fracture: a randomized controlled trial. Arch Intern Med 170:813–820
Paula Fde L, da Cunha GM, Leite Ida C, Pinheiro RS, Valente JG (2015) Elderly readmission and death after discharge from treatment of hip fracture, occurred in public hospitals from 2008 to 2010, Rio de Janeiro. Rev Bras Epidemiol 18:439–453
Kates SL, Behrend C, Mendelson DA, Cram P, Friedman SM (2015) Hospital readmission after hip fracture. Arch Orthop Trauma Surg 135:329–337
Boockvar KS, Halm EA, Litke A, Silberzweig SB, McLaughlin M, Penrod JD, Magaziner J, Koval K, Strauss E, Siu AL (2003) Hospital readmissions after hospital discharge for hip fracture: surgical and nonsurgical causes and effect on outcomes. J Am Geriatr Soc 51:399–403
Magaziner J, Simonsick EM, Kashner TM, Hebel JR, Kenzora JE (1990) Predictors of functional recovery one year following hospital discharge for hip fracture: a prospective study. J Gerontol 45:M101–M107
Griffin XL, Parsons N, Achten J, Fernandez M, Costa ML (2015) Recovery of health-related quality of life in a United Kingdom hip fracture population. The Warwick hip trauma evaluation—a prospective cohort study. Bone Joint J 97-B:372–382
Keene GS, Parker MJ, Pryor GA (1993) Mortality and morbidity after hip fractures. BMJ 307:1248–1250
Vochteloo AJ, Moerman S, Tuinebreijer WE, Maier AB, de Vries MR, Bloem RM, Nelissen RG, Pilot P (2013) More than half of hip fracture patients do not regain mobility in the first postoperative year. Geriatr Gerontol Int 13:334–341
Colón-Emeric CS, Saag KG (2006) Osteoporotic fractures in older adults. Best Pract Res Clin Rheumatol 20:695–706
Hannan EL, Magaziner J, Wang JJ, Eastwood EA, Silberzweig SB, Gilbert M, Morrison RS, McLaughlin MA, Orosz GM, Siu AL (2001) Mortality and locomotion 6 months after hospitalization for hip fracture: risk factors and risk-adjusted hospital outcomes. JAMA 285:2736–2742
Heikkinen T, Jalovaara P (2005) Four or twelve months’ follow-up in the evaluation of functional outcome after hip fracture surgery? Scand J Surg 94:59–66
Latham NK, Harris BA, Bean JF, Heeren T, Goodyear C, Zawacki S, Heislein DM, Mustafa J, Pardasaney P, Giorgetti M, Holt N, Goehring L, Jette AM (2014) Effect of a home-based exercise program on functional recovery following rehabilitation after hip fracture: a randomized clinical trial. JAMA 311:700–708
Córcoles-Jiménez MP, Villada-Munera A, Del Egido-Fernández M, Candel-Parra E, Moreno-Moreno M, Jiménez-Sánchez MD, Piña-Martínez A (2015) Recovery of activities of daily living among older people one year after hip fracture. Clin Nurs Res 24:604–623
Vochteloo AJ, van Vliet-Koppert ST, Maier AB, Tuinebreijer WE, Röling ML, de Vries MR, Bloem RM, Nelissen RG, Pilot P (2012) Risk factors for failure to return to the pre-fracture place of residence after hip fracture: a prospective longitudinal study of 444 patients. Arch Orthop Trauma Surg 132:823–830
Mariconda M, Costa GG, Cerbasi S, Recano P, Aitanti E, Gambacorta M, Misasi M (2015) The determinants of mortality and morbidity during the year following fracture of the hip: a prospective study. Bone Joint J 97-B:383–390
Givens JL, Sanft TB, Marcantonio ER (2008) Functional recovery after hip fracture: the combined effects of depressive symptoms, cognitive impairment, and delirium. J Am Geriatr Soc 56:1075–1079
Penrod JD, Boockvar KS, Litke A, Magaziner J, Hannan EL, Halm EA, Silberzweig SB, Sean Morrison R, Orosz GM, Koval KJ, Siu AL (2004) Physical therapy and mobility 2 and 6 months after hip fracture. J Am Geriatr Soc 52:1114–1120
Latham NK, Mehta V, Nguyen AM, Jette AM, Olarsch S, Papanicolaou D, Chandler J (2008) Performance-based or self-report measures of physical function: which should be used in clinical trials of hip fracture patients? Arch Phys Med Rehabil 89:2146–2155
Cleary PD (1997) Subjective and objective measures of health: which is better when? J Health Serv Res Policy 2:3–4
Stemmle J, Marzel A, Chocano-Bedoya PO, Orav EJ, Dawson-Hughes B, Freystaetter G, Egli A, Theiler R, Staehelin HB, Bischoff-Ferrari HA (2018) Effect of 800 IU versus 2000 IU vitamin D3 with or without a simple home exercise program on functional recovery after hip fracture: a randomized controlled trial. J Am Med Dir Assoc
Nardi M, Fischer K, Dawson-Hughes B, Orav EJ, Meyer OW, Meyer U, Beck S, Simmen HP, Pape HC, Egli A, Willett WC, Theiler R, Bischoff-Ferrari HA (2018) Association between caregiver role and short- and long-term functional recovery after hip fracture: a prospective study. J Am Med Dir Assoc 19:122–129
Bischoff HA, Stahelin HB, Dick W et al (2003) Effects of vitamin D and calcium supplementation on falls: a randomized controlled trial. J Bone Miner Res 18:343–351
Podsiadlo D, Richardson S (1991) The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc 39:142–148
Bischoff HA, Stahelin HB, Monsch AU, Iversen MD, Weyh A, von Dechend M, Akos R, Conzelmann M, Dick W, Theiler R (2003) Identifying a cut-off point for normal mobility: a comparison of the timed 'up and go' test in community-dwelling and institutionalised elderly women. Age Ageing 32:315–320
Madsen OR, Lauridsen UB (1995) Knee extensor and flexor strength in elderly women after recent hip fracture: assessment by the Cybex 6000 dynamometer of intra-rater inter-test reliability. Scand J Rehabil Med 27:219–226
Stoll T (2002) Isometric muscle strength measurement. Thieme
Desrosiers J, Bravo G, Hébert R, Dutil E (1995) Normative data for grip strength of elderly men and women. Am J Occup Ther 49:637–644
Ware JE, Sherbourne CD (1992) The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care 30:473–483
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373–383
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Tseng MY, Shyu YI, Liang J (2012) Functional recovery of older hip-fracture patients after interdisciplinary intervention follows three distinct trajectories. Gerontologist 52:833–842
Beaupre LA, Jones CA, Johnston DW, Wilson DM, Majumdar SR (2012) Recovery of function following a hip fracture in geriatric ambulatory persons living in nursing homes: prospective cohort study. J Am Geriatr Soc 60:1268–1273
Visser M, Harris TB, Fox KM, Hawkes W, Hebel JR, Yahiro JY, Michael R, Zimmerman SI, Magaziner J (2000) Change in muscle mass and muscle strength after a hip fracture: relationship to mobility recovery. J Gerontol A Biol Sci Med Sci 55:M434–M440
Beseler MR, Rubio C, Duarte E, Hervás D, Guevara MC, Giner-Pascual M, Viosca E (2014) Clinical effectiveness of grip strength in predicting ambulation of elderly inpatients. Clin Interv Aging 9:1873–1877
Savino E, Martini E, Lauretani F, Pioli G, Zagatti AM, Frondini C, Pellicciotti F, Giordano A, Ferrari A, Nardelli A, Davoli ML, Zurlo A, Lunardelli ML, Volpato S (2013) Handgrip strength predicts persistent walking recovery after hip fracture surgery. Am J Med 126:1068–1075.e1061
Felicio DC, Pereira DS, Assumpção AM, de Jesus-Moraleida FR, de Queiroz BZ, da Silva JP, de Brito Rosa NM, Dias JM, Pereira LS (2014) Poor correlation between handgrip strength and isokinetic performance of knee flexor and extensor muscles in community-dwelling elderly women. Geriatr Gerontol Int 14:185–189
Muangpaisan W, Wongprikron A, Srinonprasert V, Suwanpatoomlerd S, Sutipornpalangkul W, Assantchai P (2015) Incidence and risk factors of acute delirium in older patients with hip fracture in Siriraj Hospital. J Med Assoc Thail 98:423–430
Funding
This project was funded by the Baugarten Foundation Centre Grant for the Centre on Aging and Mobility (Dr. Bischoff-Ferrari). The original trial was supported by Swiss National Foundations (NFP-53) (Drs Bischoff-Ferrari and Theiler); Vontobel Foundation (Dr Bischoff-Ferrari; and Swiss National Foundations professorship grant PP00B-114864 (Dr Bischoff-Ferrari).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
K. Fischer and M. Trombik are shared first authorship.
Electronic supplementary material
ESM 1
(PDF 5819 kb)
Rights and permissions
About this article
Cite this article
Fischer, K., Trombik, M., Freystätter, G. et al. Timeline of functional recovery after hip fracture in seniors aged 65 and older: a prospective observational analysis. Osteoporos Int 30, 1371–1381 (2019). https://doi.org/10.1007/s00198-019-04944-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-019-04944-5