Abstract
Summary
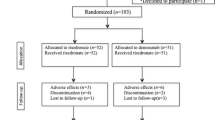
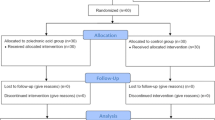
Forty-three patients who had undergone cementless THA were randomly assigned to receive no osteoactive drug or oral risedronate for 6 months. Postoperative decrease of BMD in the risedronate group was significantly lower than that seen in the control group in zones 1, 2, 3, 6, and 7.
Introduction
Proximal bone resorption around the femoral stem often has been observed after total hip arthroplasty (THA), could lead to late stem loosening. We previously reported the efficacy of etidronate on periprosthetic bone resorption after cementless THA. Recently risedronate is suggested to be effective for the prevention and treatment of for osteoporosis. The purpose of the present study was to evaluate the effects of risedronate on periprosthetic bone loss after cementless THA.
Methods
Forty-three patients who had undergone cementless THA were randomly assigned to receive no osteoactive drug (21 patients) or oral risedronate 2.5 mg/day (22 patients) for 6 months. Three patients were eliminated from the risedronate group because of dyspepsia. Periprosthetic bone mineral density (BMD) in seven regions of interest based on the zones of Gruen et al. was measured with dual energy X-ray absorptiometry at 3 weeks and 6 months postoperatively.
Results
At 6 months after surgery, postoperative decrease of BMD in the risedronate group was significantly lower than that seen in the control group in zones 1, 2, 3, 6, and 7 (p < 0.05, p < 0.01, p < 0.01, p < 0.05, and p < 0.05, respectively).
Conclusion
These outcomes suggested that risedronate might reduce the periprosthetic bone resorption after cementless THA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Total hip arthroplasty (THA) has become one of the most reliable treatments to reduce groin pain and to improve the quality of life of the patients [1, 2]. Cementless THA has been widely accepted to obtain the biological bone fixation of the implant but proximal bone resorption around the stem occurs frequently with this procedure [3–5]. After the implantation of the femoral component, the stress that would normally be borne by the bone alone is reduced, which leads to proximal femoral bone resorption. The stiff stem implant has a significantly higher incidence of proximal femoral bone resorption due to “stress shielding” than does the flexible implant [6]. The greater periprosthetic bone resorption could lead to progressive stem loosening and could cause the negative influences on the implant durability. Administration using osteoactive drugs could reduce the stress shielding and bone resorption in proximal prosthetic femoral bone.
Bisphosphonates inhibit bone resorption and have been used for the treatment of the postmenopausal and glucocorticoid-induced osteoporosis [7–12]. We previously reported that cyclic therapy with etidronate was effective in preventing proximal bone resorption after cementless THA [13–15]. A positive effect of alendronate on the prevention of periprosthetic postoperative bone resorption has also been described [16]. However, to our knowledge, the effects of risedronate on postoperative bone loss have not been published to date. The purpose of the present study was to clarify the effect of risedronate in the treatment of periprosthetic bone resorption after cementless THA.
Materials and methods
Study design
The present randomized and placebo controlled study was designed to evaluate whether risedronate could prevent the early postoperative periprosthetic bone loss after cementless THA. Forty-six patients who underwent cementless THA between February 2004 and January 2005 were included. The criteria for inclusion were primary cementless THA, no perioperative complications, no metabolic changes causing a reduction of BMD, and no previous use of drugs affecting bone metabolism such as steroids, calcitonin, or bisphosphonates. All patients were randomized into two groups (risedronate group and control group). Patients were treated with either risedronate (Actonel®, Eisai Co., Ltd., Tokyo, Japan) (2.5 mg/day orally; 22 patients) or calcium lactate (3 g/day orally; 21 patients) starting 1 week postoperatively. As risedronate only 2.5 mg once a day is solely approved by the Ministry of Health, Labour and Welfare in Japan as prevention and treatment of osteoporosis, this dosage was administered. Three patients in the risedronate group were eliminated from the study because of side effects (dyspepsia). Administration of risedronate was stopped by 2 months after postoperatively in three cases and the symptom had entirely recovered. The three cases were excluded from this study because the duration of non-administration of risedronate was 4 months. These patients were not included in the final analyses, and results are presented for only the 40 patients (19: risedronate, 21: control) who completed this study (Table 1). Thirty-six patients were postmenopausal women and 4 patients were men. All patients had osteoarthritis secondary to acetabular dysplasia. The local hospital ethics committee approved the present study, and informed consent was obtained from each patient.
Surgery
The Spongiosa Metal II Hip Prosthesis (S&G; ESKA, Lübeck, Germany) was used. The implant was made of cobalt-chrome-molybdenum alloy with a metal cancellous structure. In the operation, either extensively coated stems or proximally coated stems were used. The proximally coated stems had a porous surface applied circumferentially to the proximal 60%. The stem size used for the patients ranged from 100 to 130 mm in length with 10-mm intervals. The choice between two different types of prostheses depended on the femoral bone geometry. The operation was performed by posterolateral approach under spinal anesthesia using standardized techniques by a single hip surgeon. Partial weight-bearing was started at 1 week postoperatively, and full weight-bearing was performed at 3 weeks.
Roentgenographic evaluation
Anteroposterior radiographs were obtained by a standard technique immediately and 6 months after the surgery. Femoral migration was defined as subsidence or a change in the height of the stem, as measured from the tip of the greater trochanter, or any change in the varus or valgus orientation of the stem within the medullary cavity [17, 18].
Periprosthetic BMD measurement
The periprosthetic BMD in seven regions of interest based on the zones of Gruen et al. [19] was measured 3 weeks and 6 months postoperatively by dual-energy X-ray absorptiometry (DXA) with a Hologic QDR 2000 (Hologic, Waltham, MA, USA) (Fig. 1). During scanning, the patients were placed supine on the scan table with standard knee and foot supports to place the femur in a neutral position. The computer software used (prosthetic scanning software version 5.73; Hologic Inc.) was designed to measure the periprosthetic bone mineral content and density. The precision error of the calculation by dual-energy x-ray absorptiometry was tested. In our institution, the margin of error ranged from 1.0% in zone 6 to 3.8% in zone 7 [13]. Ten patients with a fully coated stem and ten patients with a proximally coated stem were scanned three times in succession. The periprosthetic BMD in seven zones was measured at 3 weeks and 6 months postoperatively.
The BMD ratio was calculated by dividing the BMD value at 3 weeks by the value at 6 months for each of the seven zones separately. The difference in BMD ratio between the risedronate group and the control group was examined for each zone at 6 months after surgery to evaluate the effects of risedronate administration on periprosthetic bone resorption after THA.
Biochemical bone markers
Biochemical markers of bone turnover were measured from serum-separated blood and a second-void urine collection after an overnight fast. Blood samples were collected between 8:00 and 9:00 AM. Urine samples were collected between 9:00 and 11:00 AM and urine creatinine was measured at the time of sampling.
Serum bone alkaline phosphatase (BAP, enzyme immunoassay; SRL, Inc., Osaka, Japan) and urinary type I collagen N-telopeptide breakdown products (NTX, enzyme-linked immunosorbent assay; SRL, Inc.) were measured preoperatively and 6 months postoperatively.
Statistical analysis
All data are expressed as group means ± SD. Student’s t-test was applied to test for differences between the two groups. To compare the changes between different time points during the follow-up, the Wilcoxon signed rank test was used. Differences were considered statistically significant at p < 0.05 on a two-tailed test. All statistical analyses were performed using SPSS software (version 11.0J; SPSS Japan Inc., Tokyo, Japan).
Results
Patient age and weight, stem size and the proportion of extensively coated stems to proximally coated stems, and preoperative values of biochemical bone markers did not differ significantly between the two groups (Table 1). The preoperative serum concentrations of BAP and the urinary concentrations of creatinine-corrected NTX did not differ between the two groups. No significant differences in the initial postoperative BMD at 3 weeks at any zone were observed (Table 2). No perioperative complications including intraoperative fracture and postoperative dislocation was observed, There was no stem subsidence in any patient.
The BMD ratio at 6 months in each zone is summarized in Table 3. At 6 months, BMD ratios in the risedronate group were significantly higher than those in the control group in zones 1, 2, 3, 6, and 7 (p < 0.05, p < 0.01, p < 0.01, p < 0.05, and p < 0.05, respectively). No significant differences of BMD ratios were found in zones 4 or 5. The greatest decrease of BMD was observed in zone 7 during 6 months in the two groups (20.2% for control group and 11.9% for risedronate group).
At 6 months, only the risedronate group showed significant decreases from preoperative values in serum BAP (p < 0.01) and urinary NTX (p < 0.01) (Table 4). In addition, the risedronate group had significantly greater decreases than the control group in both serum BAP (p < 0.01) and urinary NTX (p < 0.01). None of patients in either group did the BMD ratio in any zone at 6 months correlate significantly with the stem size or initial BMD at 3 weeks in the 7 zones.
Discussion
Maximal bone remodeling after cementless total hip arthroplasty has been observed in the first 6 months after surgery [20]. Our previous studies using the Spongiosa Metal system have supported this outcome and shown that BMD appears to stabilize in all zones by 12 months after prosthesis implantation [13, 14, 21, 22]. It is important to maintain high BMD to avoid the late stem loosening that has been seen after cementless THA. Therefore, we evaluated the therapeutic effect of risedronate on the local osteoporosis of the proximal femur at 6 months postoperatively.
Bisphosphonates inhibit bone resorption and have been widely used for treatment and prevention of osteoporosis [7–12]. We previously described that cyclic therapy of etidronate was effective in reducing periprosthetic bone resorption after cementless THA [13–15]. The suppressive effect of etidronate on stress shielding was confirmed to be dose-dependent (200 mg–400 mg) [15]. In our previous study, the BMD ratios at 6 months in the etidronate group and control group were 95.9% vs. 88.6% in zone 1 and 85.9% vs. 75.9% in zone 7 (Table 5) [13]. In the present study, the BMD ratio in the risedronate group and the control group was 100.9% vs. 91.1% in zone 1 and 88.1% vs.79.8% in zone 7. Patient profiles in the two studies could be considered relatively compatible and the BMD ratio of the control group did not differ significantly between the studies. Biochemical markers of bone turnover revealed similar tendency after administration risedronate and etidronate (Table 5). We suggest that risedronate could offer treatment as potent as etidronate to reduce periprosthetic bone resorption after cementless THA.
To achieve the stable bone-ingrown fixation, it is desirable to maintain a close apposition of the bone to the implant. However, there is a concern that bisphosphonates might retard bone mineralization and have a negative influence upon the longevity of the implant. In a canine model, Frenkel et al. examined the effectiveness of subcutaneous alendronate treatment over 24 weeks postoperatively on new bone formation in test channels coated with implant material [23]. Using histomorphometry, scanning electron microscopy and material testing, the authors demonstrated that alendronate did not adversely affect bone ingrowth into test channels. Peter CP et al. investigated the effect of alendronate on fracture healing and the slow callus bone remodeling was observed in mature beagle dogs that received alendronate [24]. The callus formation phase of fracture healing appeared to be unaffected by treatment, but the resorptive phase of callus remodeling processed at a slow rate. Bone histomorphometry revealed that treatment with alendronate before or during fracture healing, or both, resulted in no adverse effects on the union, strength, or mineralization of bone. A high dosed bisphosphonate treatment with ibandronate is potent to improve osteointegrated implant surface and to enhance periprosthetic bone volume [25]. According to these findings, bisphosphonates do not have the adverse effects on the initial bone ingrowth to the implant.
Alendronate and risedronate have been shown in several clinical trials to reduce the risk of fractures in postmenopausal women with osteoporosis [7–12]. Regarding the effect of periprosthetic bone resorption, Venesmaa et al. described that alendronate treatment significantly prevented periprosthetic BMD loss in the proximal femur after cementless THA at 6 months [16]. Arabmotlagh et al. reported that alendronate treatment was able to prevent bone loss in zone 1, 2, and 6, while moderate bone loss was observed in these regions in placebo controls [26]. Although, Scott et al. reported that risedronate reduced proximal bone resorption after cementless THA at 6 months in the American Society for Bone and Mineral Research 25th Annual Meeting in 2003, there have been no public reports for the efficacy of risedronate after cementless THA. Kinov et al. reported that BMD of the nonsurgical hip with risedronate reached 1.01% increase at 6 months after cementless THA [27]. Although BMD on the non-operated femoral neck was evaluated, periprosthetic BMD was not measured in this study. In the present study, periprosthetic BMD was evaluated and risedronate had a therapeutic effect on periprosthetic bone resorption after cementless THA.
Duration of risedronate treatment after cementless THA is controversial. In our previous study using the Spongiosa Metal system, maximum bone resorption was observed in the first 6 months and BMD appears to stabilize by 12 months after surgery [13, 14, 21, 22]. Arabmotlagh et al. demonstrated that BMD treated for 6 months with alendronate was significantly higher than that for 4 months at 6 and 12 postoperative months [26]. Accordingly, risedronate could be administered for longer than 6 months after cementless THA to maintain the periprosthetic BMD.
Regarding biochemical bone markers, both serum BAP and urinary NTX in the risedronate group were significantly lower than those in the control group at 6 months after surgery. Significant decrease in serum BAP and urinary NTX was observed in the etidronate cyclic therapy during the first 12 months [13–15]. Bisphosphonates reduce bone resorption through the inhibition of osteoclastic activity [28]. The level of biochemical bone markers represents the bone turnover activity in the whole body. Regarding the BMD after cementless THA, no significant alteration was observed in the nonoperated femur compared with preoperative BMD [29]. The changes in biochemical bone markers could be considered to reflect mainly the changes in the periprosthetic bone remodeling.
Although bisphosphonates are widely used for the prevention and treatment of osteoporosis, upper gastrointestinal injury associated with some compounds has caused concern [30, 31]. Gastrointestinal side effects resulted in withdrawal of three of 22 patients who received risedronate in the present study but all patients recovered promptly following discontinuation of risedronate. Significantly lower rates of gastric ulcer were observed in healthy postmenopausal women who received risedronate 5 mg/day (4.1%) than those who received alendronate 10 mg/day (13.2%) in a 2-week study [31]. No excessive incidence of upper gastrointestinal adverse events relative to placebo has been observed in patients who received risedronate 2.5 or 5 mg/day for up to 3 years [32, 33]. Bisphosphonates are an orally administered drug that was initially approved for once-daily dosing at least 30 minutes prior to food or drink. Patients should not lie down more than 30 minutes after taking the medication. Risedronate once a week administration is an effective anti-osteoporotic therapy in osteoporotic postmenopausal women [34, 35]. Risedronate 35 mg given weekly has the same therapeutic effect and safety compared with daily 5 mg administration [36, 37] To reduce gastrointestinal side effects and to improve the compliance of the patients, once-weekly risedronate might be indicated for the treatment of stress shielding after cementless THA, if the therapeutic effect of once-weekly risedronate would be as potent as that of daily risedronate. Cyclic therapy with etidronate also may offer an alternative therapy for patients who cannot comply with continuous and daily administration, because gastrointestinal side effects are rarely seen with etidronate.
Some limitations in the present study need to be considered. First, the long-term influence on the implant by administration of risedronate. Our previous studies using Spongiosa Metal system demonstrated the BMD appears to stabilize by 12 months after surgery and the BMD ratio has been maintained about 80% on the proximal region after 3 years postoperatively. Although periprosthetic bone resorption of this system until 3 years postoperatively was evaluated, the results of longer than 3 years are unclear. In addition, bisphosphonates have been widely used for the treatment and prevention of osteoporosis. It is difficult to secure a number of patients as a control group without administration of bisphosphonates after cementless THA. Further research would be necessary to clarify the long-term influence on the implant.
Second, the delay in the administration of risedronate remains an unsettled point. In the present study, patients were treated with risedronate starting 1 week postoperatively. The effect of delaying for several weeks might not interfere with the ingrowth and local osteoporosis around the prosthesis. Further investigation would be necessary to clarify the role starting point of administration.
The last limitation of the present study is whether the therapeutic effects by risedronate on proximal femoral osteoporosis could be applied to any other implants than the Spongiosa Metal system. Spongiosa Metal stem has the anatomical curved shape and the relatively short length in comparison with other types of the implants. Based on various biomechanical studies, much less stress shielding could be expected in Spongiosa Metal stem than other designs of the stems [13, 14, 21, 22]. The long and straight stem could possibly have the greater periprosthetic bone resorption than that of the stems in the Spongiosa Metal system. In the cases of the revision surgery, the long stems are commonly used to achieve the rigid stabilization of the implant; therefore, strong stress shielding might occur. The effect of the risedronate on the revisional cases remains unidentified.
In conclusion, risedronate had a therapeutic effect on periprosthetic bone resorption with significantly less post-operative decrease of BMD in the risedronate group than that in the control group in zone 1, 2, 3, 6, and 7 at 6 months after cementless THA. Further investigations with prolonged follow-up are needed to clarify the efficacy of risedronate on proximal femoral bone resorption for periods of more than 6 months.
References
Engh CA Jr, Claus AM, Hopper RH Jr et al (2001) Long-term results using the anatomic medullary locking hip prosthesis. Clin Orthop 393:137–146
Xenos JS, Callaghan JJ, Heekin RD et al (1999) The porous-coated anatomic total hip prosthesis, inserted without cement. A prospective study with a minimum of ten years of follow-up. J Bone Jt Surg (Am) 81:74–82
Engh CA, Bobyn JD (1988) The influence of stem size and extent of porous coating on femoral bone resorption after primary cementless hip arthroplasty. Clin Orthop 231:7–28
Lord G, Marotte JH, Blanchard JP et al (1988) Cementless madreporic polarised total hip prostheses. A ten-year review of 2688 cases. Rev Chir Orthop Reparatrice Appar Mot 74:3–14
Bobyn JD, Mortimer ES, Glassman AH et al (1992) Producing and avoiding stress shielding. Laboratory and clinical observations of noncemented total hip arthroplasty. Clin Orthop 274:79–96
Engh CA, Massin P (1989) Cementless total hip arthroplasty using the anatomic medullary locking stem. Results using a survivorship analysis. Clin Orthop Relat Res 249:141–158
Black DM, Cummings SR, Karpf DB et al (1996) Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet 348:1535–1541
Saag KG, Emkey R, Schnitzer TJ et al (1998) Alendronate for the prevention and treatment of glucocorticoid-induced osteoporosis. Glucocorticoid-Induced Osteoporosis Intervention Study Group. N Engl J Med 39:292–299
Harris ST, Watts NB, Genant HK et al (1999) Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group. JAMA 282:1344–1352
Reginster J, Minne HW, Sorensen OH et al (2000) Randomized trial of the effects of risedronate on vertebral fractures in women with established postmenopausal osteoporosis. Vertebral Efficacy with Risedronate Therapy (VERT) Study Group. Osteoporosis Int 11:83–91
Reid DM, Hughes RA, Laan RF et al (2000) Efficacy and safety of daily risedronate in the treatment of corticosteroid-induced osteoporosis in men and women: a randomized trial. European Corticosteroid-Induced Osteoporosis Treatment Study. J Bone Miner Res 15:1006–1013
McClung MR, Geusens P, Miller PD et al (2001) Effect of risedronate on the risk of hip fracture in elderly women. Hip Intervention Program Study Group. N Engl J Med 344:333–340
Yamaguchi K, Masuhara K, Yamasaki S et al (2003) Cyclic therapy with etidronate has a therapeutic effect against local osteoporosis after cementless total hip arthroplasty. Bone 33:144–149
Yamaguchi K, Masuhara K, Yamasaki S et al (2004) Effects of discontinuation as well as intervention of cyclic therapy with etidronate on bone remodeling after cementless total hip arthroplasty. Bone 35:217–223
Yamaguchi K, Masuhara K, Yamasaki S et al (2005) Efficacy of different dosing schedules of etidronate for stress shielding after cementless total hip arthroplasty. J Orthop Sci 10:32–36
Venesmaa PK, Kroger HP, Miettinen HJ et al (2001) Alendronate reduces periprosthetic bone loss after uncemented primary total hip arthroplasty; a prospective randomized study. J Bone Miner Res 16:2126–2131
Kim YH, Kim VE (1992) Results of the Harris-Galante cementless hip prosthesis. J Bone Jt Surg Br 74:83–87
Wixson RL, Stulberg SD, Mehlhoff M (1991) Total hip replacement with cemented, uncemented, and hybrid prostheses. A comparison of clinical and radiographic results at two to four years. J Bone Jt Surg (Am) 73:257–270
Gruen TA, McNeice GM, Amstutz HC (1979) “Mode of failure” of cemented stem-type femoral components. A radiographic analysis of loosening. Clin Orthop 141:17–27
Kiratli BJ, Checovich MM, McBeath AA et al (1996) Measurement of bone mineral density by dual-energy X-ray absorptiometry in patients with the Wisconsin hip, an uncemented femoral stem. J Arthroplasty 11:184–193
Nishii T, Sugano N, Masuhara K et al (1997) Longitudinal evaluation of time related bone remodeling after cementless total hip arthroplasty. Clin Orthop 339:121–131
Yamaguchi K, Masuhara K, Ohzono K et al (2000) Evaluation of periprosthetic bone-remodeling after cementless total hip arthroplasty. J Bone Jt Surg 82-A:1426–1431
Frenkel SR, Jaffe WL, Valle CD et al (2001) The effect of alendronate (Fosamax) and implant surface on bone integration and remodeling in a canine model. J Biomed Mater Res 58:645–650
Peter CP, Cook WO, Nunamaker DM et al (1996) Effect of alendronate on fracture healing and bone remodeling in dogs. J Orthop Res 14:74–79
Eberhardt C, Raussen W, Thiemann S et al (2005) Improved osseointegration and periprosthetic bone volume around cementless metal implants under bisphosphonate treatment. Z Orthop Ihre Grenzgeb 143:645–651
Arabmotlagh M, Rittmeister M, Hennigs T (2006) Alendronate prevents femoral periprosthetic bone loss following total hip arthroplasty: prospective randomized double-blind study. J Orthop Res 24:1336–1341
Kinov P, Tivchev P, Doukova P et al (2006) Effect of risedronate on bone metabolism after total hip arthroplasty: a prospective randomised study. Acta Orthop Belg 72:44–50
Fisher JE, Rogers MJ, Halasy JM et al (1999) Alendronate mechanism of action: geranylgeraniol, an intermediate in the mevalonate pathway, prevents inhibition of osteoclast formation, bone resorption, and kinase activation in vitro. Proc Natl Acad Sci USA 96:133–138
Wilkinson JM, Stockley I, Peel NF et al (2001) Effect of pamidronate in preventing local bone loss after total hip arthroplasty: a randomized, double-blind, controlled trial. J Bone Miner Res 16:556–564
Lichtenberger LM, Romero JJ, Gibson GW et al (2000) Effect of bisphosphonates on the surface hydrophobicity and phosphatidylcholine concentration of rodent gastric mucosa. Dig Dis Sci 45:1792–1801
Lanza F, Schwartz H, Sahba B et al (2000) An endoscopic comparison of the effects of alendronate and risedronate on upper gastrointestinal mucosae. Am J Gastroenterol 95:3112–3117
Fogelman I, Moreland L, Woodson G et al (2000) Gastrointestinal side effects and endoscopic findings similar between risedronate and placebo-treated patients. Osteoporos Int 11(Suppl 2):S179
Hosking D, Beckman R, Glowinski J et al (2000) Similar gastrointestinal side effects and endoscopic findings between risedronate and placebo-treated patients. J Bone Miner Res 15:S428
Delaney MF, Hurwitz S, Shaw J et al (2003) Bone density changes with once weekly risedronate in postmenopausal women. J Clin Densitom 6:45–50
Palomba S, Orio F Jr, Manguso F et al (2005) Efficacy of risedronate administration in osteoporotic postmenopausal women affected by inflammatory bowel disease. Osteoporos Int 14:1141–1149
Brown JP, Kendler DL, McClung MR et al (2002) The efficacy and tolerability of risedronate once a week for the treatment of postmenopausal osteoporosis. Calcif Tissue Int 71:103–111
Ilter E, Karalok H, Tufekci EC et al (2006) Efficacy and acceptability of risedronate 5 mg daily compared with 35 mg once weekly for the treatment of postmenopausal osteoporosis. Climacteric 9:129–134
Acknowledgements
The authors wish to gratefully acknowledge Masazumi Notou for technical assistance.
This study is based on data collected in Osaka Kosei-Nenkin Hospital.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamasaki, S., Masuhara, K., Yamaguchi, K. et al. Risedronate reduces postoperative bone resorption after cementless total hip arthroplasty. Osteoporos Int 18, 1009–1015 (2007). https://doi.org/10.1007/s00198-007-0339-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-007-0339-7