Abstract
In the Western world, increased consumption of carbonated soft drinks combined with a decreasing intake of milk may increase the risk of osteoporosis. This study was designed to reflect the trend of replacing milk with carbonated beverages in a group of young men on a low-calcium diet and studies the effects of this replacement on calcium homeostasis and bone turnover. This controlled crossover intervention study included 11 healthy men (22–29 years) who were given a low-calcium basic diet in two 10-day intervention periods with an intervening 10-day washout. During one period, they drank 2.5 l of Coca Cola per day and during the other period 2.5 l of semi-skimmed milk. Serum concentrations of calcium, phosphate, 25-hydroxycholecalciferol, 1,25-dihydroxycholecalciferol (1,25(OH)2D), osteocalcin, bone-specific alkaline phosphatase (B-ALP) and cross-linked C-telopeptides (CTX), plasma intact parathyroid hormone (PTH) and urinary cross-linked N-telopeptides (NTX) were determined at baseline and endpoint of each intervention period. An increase in serum phosphate (P<0.001), 1,25(OH)2D (P<0.001), PTH (P=0.046) and osteocalcin (P<0.001) was observed in the cola period compared to the milk period. Also, bone resorption was significantly increased following the cola period, seen as increased serum CTX (P<0.001) and urinary NTX (P<0.001) compared to the milk period. No changes were observed in serum concentrations of calcium or B-ALP. This study demonstrates that over a 10-day period high intake of cola with a low-calcium diet induces increased bone turnover compared to a high intake of milk with a low-calcium diet. Thus, the trend towards a replacement of milk with cola and other soft drinks, which results in a low calcium intake, may negatively affect bone health as indicated by this short-term study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis is an increasing public health problem worldwide. Both men and women develop osteoporosis; however, women are more prone to develop osteoporosis due to a lower peak bone mass and estrogen depletion after menopause [1]. As the population ages and dietary patterns change, an increased prevalence of osteoporosis will likely occur in the future [1, 2], and osteoporosis is already of concern in the male population as well. Efforts to reverse osteoporosis once the disease has developed have not until recently been successful, where recombinant parathyroid hormone (PTH) has been shown to have an anabolic effect on bone [3]. However, an adequate diet throughout life is still important to prevent or delay the onset of the disease [2, 4, 5].
Adolescence is a critical period for bone mass accretion and maximizing peak bone mass in order to prevent development of osteoporosis later in life [1, 4]. A recent study demonstrated that intake of milk in childhood and adolescence is associated with increased bone mass and density in adulthood [5]. However, the trend in food consumption is currently moving towards a reduced milk intake coinciding with an increased consumption of soft drinks [6, 7, 8], of which the most popular are cola beverages.
Several researchers have investigated an association between the consumption of carbonated beverages and bone health [6, 9, 10, 11, 12, 13]. In two cross-sectional studies [10, 13], Wyshak and coworkers observed a positive relation between bone fractures and carbonated beverage consumption in American teenage girls, but not boys, and others have found an inverse association between carbonated soft drink consumption and bone mineral density (BMD) [11] and bone mineral content [6] in adolescent girls. Again, no significant association was observed in boys. Moreover, this association was most pronounced in physically active girls, which has been ascribed to their lower levels of endogenous estrogen or possibly the increased rehydration needs of active children [10, 13]. However, when a similar study was conducted in a group of women aged 44 to 98 years, no association between BMD and carbonated soft drink consumption could be established [12], nor could a relation between urinary calcium excretion and carbonated beverages as such be confirmed [9]. Suggestions have been made that different substances in cola beverages, i.e., phosphorus, sugar or caffeine, could be responsible for the observed association [6, 9, 10], but more likely, the displacement of milk is responsible. This clinical intervention study was carried out to elucidate the findings of the epidemiological studies described. The study is thought to reflect the trend of replacing milk with cola beverages in young men on a low-calcium diet in order to investigate its short-term effects on calciotrophic hormones and biochemical markers related to bone turnover. The amount of soft drinks varies a great deal among individuals, and the 2.5 l of cola consumed in the present study is an extreme amount, but may reflect intakes seen in a small percentage of the population, since previously high intakes of cola have been reported [6, 8]. Also, 2.5 l of milk per day is a large quantity of milk, which does not reflect the intake in the general population, although the milk consumption is high in Denmark and other Nordic countries.
Subjects and methods
Subjects
Subjects were healthy Caucasian males aged 22–29 years, all university students. Subjects who were smokers or elite athletes (>10 h/week), or who had taken dietary supplements or donated blood in the 3 months prior to the study were excluded. At entry of the study, each subject’s weight was measured to the nearest 0.1 kg by using an electronic digital scale and height was measured to the nearest 1 mm by using a wall-mounted stadiometer. The Municipal Ethical Committee of Copenhagen and Frederiksberg approved this research study (KF 01238/98).
Study design
Eleven men participated in this 30-day controlled intervention study. The study was arranged in a randomized crossover design consisting of two 10-day experimental periods with an intervening 10-day washout period. In the two intervention periods, subjects consumed a strictly controlled basic diet with 2.5 l of either Coca Cola or semi-skimmed milk (0.5% fat) per day contributing equally to the daily energy intake (4 MJ/d). In the washout period, the subjects consumed their habitual diet.
Diet
Subjects were given the same low-calcium basic diet in the two intervention periods in addition to the 2.5 l of cola or milk, and all foods and drinks were provided and served by the institute. The basic diet consisted of ordinary, low-calcium foods (Table 1). Two different lunch meals and three different dinner meals were served in the same order during the two intervention periods in the study; breakfast and snack meals were the same every day. The subjects were instructed to consume a minimum of 1.5 l of the provided cola or milk in combination with meals. Mineral water was provided ad libitum. The energy content of individual diets was adjusted according to the height, weight and physical activity of each subject to 9–14 MJ/d [14]. If subjects were hungry, the energy level was adjusted by adding bread equivalent to either 0.5 or 1.0 MJ from the 2nd day of the first intervention period. The energy content of cola and semi-skimmed milk is the same and provided 4 MJ/d and thus the energy intake was identical in the two periods for each subject, with total energy intake ranging from 13 to 18 MJ/d. To ensure compliance, the lunch meals were served at the institute on all weekdays, and researchers maintained daily contact with each subject when handing out the food and drinks for consumption at home. All subjects completed the two intervention periods. Self-recordings of daily mineral water consumption were carried out.
The phosphorus (P) and calcium (Ca) contents of the diets were calculated using a Danish computerized nutrient database (DanKost 2000, version 1.2). In the milk period, the average intake of calcium and phosphorus was 3,500 mg/day and 3,640 mg/day, respectively (Ca:P=0.96). Milk contributed 3,100 mg calcium and 2,375 mg phosphorus per day. The energy distribution was as follows in the milk period: 20.2% from protein, 51.8% from carbohydrates and 28.1% from fat. In the cola period, the average daily intake of calcium and phosphorus was 470 mg and 1,690 mg, respectively (Ca:P=0.28). Here, cola contributed 75 mg calcium and 425 mg phosphorus per day. In the cola period, the energy distribution was as follows: 9.4% from protein, 67.4% from carbohydrates and 24.1% from fat.
Analytical procedures
Serum and plasma samples were obtained from blood drawn under fasting conditions at baseline and on the last day of each intervention period. Subjects fasted for 12 h and were instructed to avoid severe physical activity before blood sampling. Urine samples were collected from the second morning urination after fast. Serum, plasma and urine samples were stored at −20°C until analysis. All measurements were carried out in a single batch after the second intervention period. Calcium, phosphate, 25-hydroxycholecalciferol (25(OH)D), 1,25-dihydroxycholecalciferol (1,25(OH)2D), osteocalcin, bone-specific alkaline phosphatase (B-ALP) and cross-linked C-telopeptides (CTX) were measured in serum, intact parathyroid hormone (PTH) was measured in plasma, and cross-linked N-telopeptides (NTX) in urine.
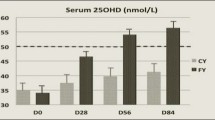
Calcium was measured by atomic-absorption spectroscopy (Spectra AA-200 Varian, Varian Techtron Pty. Limited, Victoria, Australia), and intra-assay precision was 4.2%. Inorganic phosphate was determined photometrically at 340 nm (Cobas Mira, Roche Diagnostics Systems, Basel, Switzerland) using a Roche Diagnostics kit (Roche Diagnostics GmbH, Annheim, Germany). Intra-assay precision was 1.4%. Levels of 25(OH)D and 1,25(OH)2D were determined by radioimmunoassays (RIA), and both cholecalciferol and ergocalciferol metabolites were measured. 25(OH)D was only measured in the first blood sample as an indicator of the subjects’ vitamin D status using a 125I RIA kit (DiaSorin, Stillwater, Minn.). 1,25(OH)2D was quantified using 125I IDS Gamma-B kit (Immune Diagnostic Systems, Boldon, UK). Intra-assay precisions were 10 and 13% for 25(OH)D and 1,25(OH)2D, respectively. Intact PTH was quantified with an Immulite intact PTH immunoassay kit (Diagnostic Products Corporation, Los Angeles, Calif.). Intra-assay precision was measured to 5.8%. Osteocalcin and B-ALP were determined with an N-MID Osteocalcin assay based on an electrochemiluminescence immunoassay (ECLIA) and a Roche B-ALP kit based on wheat-germ lectin precipitation (Roche Diagnostics GmbH, Annheim, Germany). The intra-assay precisions were 2.5 and 6.7% for osteocalcin and B-ALP, respectively. CTX was measured using a Serum Crosslaps One Step ECLIA (Osteometer Biotech, Herlev, Denmark), with an intra-assay of 4.3%. Urinary NTX was measured by a competitive enzyme immunoassay technique using Vitros NTX assay (Ortho-Clinical Diagnostics, Amersham, UK). Intra-assay precision was 3.3%, and NTX is expressed as nmol of bone collagen equivalents (BCE) per mmol of creatinine.
Statistical analysis
All statistical analyses and calculations were performed using the Statistical Analysis System software package, version 8.02 (SAS Institute inc., Cary, N.C.). Data were controlled for homogeneity of variance verified by residual plots, and the changes from baseline to endpoint were analyzed by paired t-test. Subject characteristics are presented as medians (range), and results are presented as means ± SE; the statistical significance was defined as P<0.05.
A mixed model analysis was performed using the MIXED procedure (proc mixed). First, baseline concentrations of the biochemical markers [calcium, phosphate, 25(OH)D, 1,25(OH)2D, PTH, osteocalcin, B-ALP, CTX and NTX] were modeled as the dependent variable in a mixed linear model to control for differences in baseline levels in the two intervention periods resulting from the treatment. Denoting the i’th observation (1,2...,n), the effect of treatment (milk or cola) and period and their interaction were modeled as a class variable.
In a mixed linear model, end point concentrations of the biochemical markers were modeled as the dependent variable to evaluate the effect of treatment. The same denotation and class variables were used as described above, and furthermore, the baseline level of the respective dependent variable was included in the model as covariate, resulting in the model:
where η(subjecti) is independently distributed subject-specific random effects (~N(0,ση2)), and εis are independently distributed random variables (~ N(0,σ2)).
Results
The subject characteristics including baseline serum 25(OH)D concentrations, within the normal range for all subjects, are presented in Table 2. There were no significant differences in baseline values in any of the biochemical markers, suggesting that the 10-day washout period was of sufficient length to avoid carry-over effects (all P>0.17). No difference between the two treatments was observed in serum concentration of calcium, but a treatment by period interaction was seen (P=0.002) (Table 3). The serum concentration of phosphate was significantly different after the milk period compared to the cola period (P<0.001). A significant difference between the two treatments was observed also in 1,25(OH)2D (P<0.001), resulting from a non-significant decline after milk consumption, but a significant increase in the cola period (P=0.019). Also, increases in the calciotrophic hormone PTH were observed in both intervention periods, but the change from baseline was only significant in the cola period (P=0.020), resulting in a significant difference between the two treatments (P=0.046). The serum concentration of osteocalcin was significantly increased after the cola period (P=0.014), whereas a decrease was observed after the milk period (P<0.001). Thus, a significant difference between the two treatments was found (P<0.001). No differences were observed in B-ALP. Bone resorption measured as serum CTX concentration and urinary NTX excretion differed between the two treatments (P<0.001) as these increased after the cola period, although not significantly, whereas decreases were seen in the milk period (P<0.001 and P=0.024 for CTX and NTX, respectively).
Moreover, the statistical analysis demonstrated a significant dependency of baseline on the endpoint concentration of osteocalcin (P<0.001), PTH (P<0.001), CTX (P<0.001) and B-ALP (P<0.001).
Discussion
This short-term intervention study was designed to reflect the current trend of replacing milk with carbonated beverages, leading to a low intake of calcium. The results strongly indicate that such a replacement over a short period adversely affects calcium homeostasis and bone turnover. When the subjects were placed on a low-calcium diet, consumption of 2.5 l cola per day for 10 days caused significant increases in the calciotrophic hormones and biochemical markers of bone turnover compared to an iso-energetic intake of 2.5 l milk with the same low-calcium diet.
Previous studies have found an association between cola consumption and increased secretion of calciotrophic hormones and bone resorption in adult women [15, 16]. Johansen and Eiken [15] describe two cases of secondary hyperparathyroidism caused by a yearlong massive cola consumption of ≥1 l/d together with a low calcium intake. In a case control study with postmenopausal women, higher consumption of carbonated soft drinks was significantly associated with hypocalcemia [16], which has also been found in a case control study with children [17]. Moreover, increased cola consumption has been linked to lower BMD in young girls [6, 11] as well as an increased occurrence of fractures in teenage girls [10, 13, 18]. Thus, it is likely that the responses obtained here after a 10-day period of high cola intake may, in fact, induce bone resorption if sustained over time and thereby increase the long-term risk for osteoporosis. It is likely that the amount of cola consumed in the present study does indeed reflect some extremes of the intake seen in the general population, particularly among teenagers, since high intakes of cola previously have been reported [6, 8]. No food recording was made prior to this study, so the habitual diet of the subjects is unknown. However, the intake of calcium in Denmark is rather high due to a large consumption of dairy products, and the mean calcium intake in young men (19–24 years) was 1,379 mg/d in the most recently published survey [19]. Thus, it is assumed that both treatments were preceded by a high calcium intake.
Maintaining the stability of the serum concentration of calcium is vital for optimal physiological function, and its regulation primarily involves the calciotrophic hormone PTH, in that calcium acts directly on the parathyroid glands in a feed back fashion to regulate the secretion of PTH [20]. In the present study, no changes were observed in serum calcium concentrations as was expected because of a functional regulation, but higher levels of the calciotrophic hormones PTH and 1,25(OH2)D were observed after the cola period than after the milk period. This disturbance in calcium homeostasis can result from a number of things: a reduced calcium intake, a change in the Ca:P ratio (0.96 vs. 0.28 in the milk and cola period, respectively) and the caffeine content of cola or a combination of these. The phosphorus content of the diet may be important if it is not balanced by dietary calcium because of its potential risk for elevation of PTH and bone loss [21].
It is an ongoing discussion whether a high phosphorus intake adversely affects bone mass and density by PTH stimulation. A sustained rise in PTH following the intake of phosphates has been observed in both animal and human studies [21, 22, 23], which is generally attributed to the formation of calcium phosphate complexes in the blood, resulting in a drop in free ionized serum calcium. Calvo et al. [21] observed elevated levels of PTH and 1,25(OH)2D in 16 men and women after 8 days on a high-phosphorus and low-calcium diet. Contrarily, in a study by Bizik et al. [23], PTH levels were not increased as a result of high dietary phosphorus intake when given together with adequate amounts of calcium, where the decrease in free ionized calcium is compensated for by the diet. Thus, an adverse effect on PTH secretion from high phosphorus intake was seen only when calcium intake was inadequate. In the present study, calcium intake was inadequately low in the cola period (470 mg Ca/d). The cola intake of 2.5 l/d provided 425 mg P/d in addition to the phosphorus content of the basic diet, whereas milk gave an additional 2,380 mg P/d. Hence, cola did not contribute to an excess amount of phosphorus; rather, a significant rise was observed in serum phosphate after the milk period. This also illustrates that the phosphate concentration in the blood is less tightly regulated than calcium. Thus, a low-calcium diet is capable of inducing increases in PTH and 1,25(OH)2D in order to normalize ionized calcium concentrations at the expense of bone mass. The phosphorus content of the diet seems to be of less significance than the molar ratio of calcium to phosphorus, substantiated by the finding that Ca:P is a better predictor of BMD than calcium or phosphorus alone in a cross-sectional study of 38 young women [24], as well as in a group of 510 perimenopausal women [25]. The recommended molar ratio of Ca:P is 1 [26], which was obtained in the milk period in the present study (0.96), whereas it was only about one fourth of that in the cola period (0.28). This is a significant change from the recommended Ca:P ratio. However, a similarly low ratio is not uncommonly seen in Western populations. The serum concentrations of osteocalcin exhibited a small but significant decrease after the milk period, whereas a significant increase was observed after the cola period. Thus, milk consumption slows down bone turnover, whereas cola together with a low-calcium diet results in increased bone turnover. This is supported by the decreases and increases observed in the bone resorption markers, serum CTX and urinary NTX following milk and cola consumption, respectively.
To the authors’ knowledge only one other controlled intervention study has previously investigated the effect of cola and other carbonated beverages on calcium homeostasis [9]. This study included 30 women aged 20–40 years and demonstrated an association between 5-h urinary excess calcium excretion and caffeine-containing carbonated beverages. Beverages without caffeine including those containing phosphoric acid did not affect urinary calcium excretion, but the available evidence concerning caffeine and bone health is conflicting [27, 28, 29]. Nonetheless, this study was not designed to reach conclusions about the role of caffeine in relation to cola consumption.
A strong aspect of this study is that the diets were identical in the two intervention periods and thus the only difference was the replacement of milk with cola, which reflects the food trend investigated here. However, this study also has some limitations.
Protein intake differed in the two intervention periods, in that 20.2% of the energy derived from protein in the milk period, whereas only 9.4% of the energy was protein-derived in the cola period. This may very well have a short-term calciuric effect, although there is a lack of evidence from long-term studies of continued urinary calcium loss and bone loss. The available evidence rather suggests that protein may have a biphasic effect on bone health as reviewed by Ginty [30], since both low and high protein intakes may adversely affect bone. However, no consensus has been reached at present.
Male subjects were chosen over females to avoid the variation in hormone levels resulting from the use of oral contraceptives, which could potentially influence the results. Had women instead been chosen, it is possible that the changes would have been more pronounced, since previous studies show a relation between bone health and soft drink consumption primarily in females [11, 13]. Most importantly, no conclusions on the effect of cola specifically can be obtained from this study, since neither a non-cola carbonated beverage nor a cola and high-calcium diet period were included in the study. Also, the habitual diet of the subjects could potentially have affected the results, but it is not possible to examine this, as no food recording was conducted as part of this study. However, the main object of this study was to investigate the effects of the current trend of replacing milk with cola. Finally, the intervention periods only lasted 10 days, and thus no long-term conclusions on cola consumption in relation to bone health can be made, since adaptation is only expected to occur to some degree after a 10-day period.
In conclusion, the present study illustrates a potential adverse effect of high cola consumption on bone turnover in individuals with low calcium intake. These findings confirm the existing epidemiological evidence that the health risk of soft drink consumption primarily seems to be due to the replacement of milk with cola, which results in an inadequately low calcium intake and a low Ca:P ratio. The current trend towards the replacement of milk with carbonated beverages results in a decreased intake of calcium. This is an upsetting aspect of the current nutritional habits, and it is necessary to give some consideration as to how soft drink consumption can be reduced and the intake of milk and other dairy product increased in order to obtain an adequate calcium intake and thereby improve bone health, particularly among adolescents.
References
Ettinger MP (2003) Aging bone and osteoporosis: strategies for preventing fractures in the elderly. Arch Intern Med 163:2237–2246
World Health Organization (1999) Osteoporosis: Both health organizations and individuals must act now to avoid an impeding epidemic. WHO, Geneva
Brixen KT, Christensen B, Ejersted C, Langdahl BL (2004) Teriparatide (biosynthetic human parathyroid hormone 1–34): a new paradigm in the treatment of osteoporosis. Pharmacol Toxicol 94:260–270
Whiting SJ, Vatanparast H, Baxter-Jones A, Faulkner RA, Mirwald R, Bailey DA (2004) Factors that affect bone mineral accrual in the adolescent growth spurt. J Nutr 134:696S-700
Kalkwarf HJ, Khoury JC, Lanphear BP (2003) Milk intake during childhood and adolescence, adult bone density, and osteoporotic fractures in US women. Am J Clin Nutr 77:257–265
Whiting SJ, Healey A, Psiuk S, Mirwald R, Kowalski K, Bailey DA (2001) Relationship between carbonated and other low nutrient dense beverages and bone mineral content of adolescents. Nutr Res 21:1107–1115
Mrdjenovic G, Levitsky DA (2003) Nutritional and energetic consequences of sweetened drink consumption in 6- to 13-year-old children. J Pediatr 142:604–610
Harnack L, Stang J, Story M (1999) Soft drink consumption among US children and adolescents: nutritional consequences. J Am Diet Assoc 99:436–441
Heaney RP, Rafferty K (2001) Carbonated beverages and urinary calcium excretion. Am J Clin Nutr 74:343–347
Wyshak G, Frisch RE (1994) Carbonated beverages, dietary calcium, the dietary calcium/phosphorus ratio, and bone fractures in girls and boys. J Adolesc Health 15:210–215
McGartland C, Robson PJ, Murray L et al (2003) Carbonated soft drink consumption and bone mineral density in adolescence: the Northern Ireland Young Hearts project. J Bone Miner Res 18:1563–1569
Kim SH, Morton DJ, Barrett-Connor EL (1997) Carbonated beverage consumption and bone mineral density among older women: The Rancho Bernado Study. Am J Public Health 87:276–279
Wyshak G (2000) Teenaged girls, carbonated beverage consumption, and bone fractures. Arch Pediatr Adolesc Med 154:610–613
Sandström B, Aro A, Becker W, Lyhne N, Pedersen JI, Pórsdóttir I (1996) Nordiska näringsrekommendationer 1996. 113–114 (The Nordic Dietary Recommendations)
Johansen DL, Eiken PA (2002) Cola as a possible cause of secondary hyperparathyroidism. Ugeskrift for laeger 164:4290–4291 (report of two case stories)
Guerrero-Romero F, Moran MR, Reyes E (1999) Consumption of soft drinks with phosphoric acid as a risk factor for the development of hypocalcemia in postmenopausal women. J Clin Epidemiol 52:1007–1010
Mazariegos-Ramos E, Guerrero-Romero F, Rodriguez-Moran M, Lazcano-Burciaga G, Paniagua R, Amato D (1995) Consumption of soft drinks with phosphoric acid as a risk factor for the development of hypocalcemia in children: A case-control study. J Pediatr 126:940–942
Petridou E, Karpathios T, Dessypris N, Simou E, Trichopoulos D (1997) The role of dairy products and non alcoholic beverages in bone fractures among school age children. Scand J Soc Med 25:119–125
Danish Food and Venterinary Administration (1006) Danskernes Kostvaner 1995. Hovedresultater. Danish Food and Veterinary Administration, Copenhagen, Denmark (survey on the dietary food patterns in the Danish population)
Houillier P, Nicolet-Barousse L, Maruani G, Paillard M (2003) What keeps serum calcium levels stable? Joint Bone Spine 70:407–413
Calvo MS, Kumar R, Heath H III (1988) Elevated secretion and action of serum parathyroid hormone in young adults consuming high phosphorus, low calcium diets assembled from common foods. J Clin Endocrinol Metab 66:823–829
Estepa JC, Aguilera-Tejero E, Lopez I, Almaden Y, Rodriguez M, Felsenfeld AJ (1999) Effect of phosphate on parathyroid hormone secretion in vivo. J Bone Miner Res 14:1848–54.
Bizik BK, Ding W, Cerklewski FL (1996) Evidence that bone resorption of young men is not increased by high dietary phosphorus obtained from milk and cheese. Nutr Res 16:1143–1146
Metz JA, Anderson JJ, Gallagher PN Jr (1993) Intakes of calcium, phosphorus, and protein, and physical-activity level are related to radial bone mass in young adult women. Am J Clin Nutr 58:537–542
Brot C, Jorgensen N, Madsen OR, Jensen LB, Sorensen OH (1999) Relationships between bone mineral density, serum vitamin D metabolites and calcium:phosphorus intake in healthy perimenopausal women. J Int Med 245:509–516
United States Institute of Medicine, Food and Nutrition Board (1997) Dietary reference intakes: calcium, phosphorus, magnesium, vitamin D and flouride. National Academy Press, Washington, DC
Barger-Lux MJ, Heaney RP, Stegman MR (1990) Effects of moderate caffeine intake on the calcium economy of premenopausal women [published erratum appears in Am J Clin Nutr 1991 Jan;53(1):182]. Am J Clin Nutr 52:722–725
Kynast-Gales SA, Massey LK (1994) Effect of caffeine on circardian excretion of urinary calcium and magnesium. J Am Coll Nutr 13:467–472
Wise KJ, Bergman EA, Sherrard DJ, Massey LK (1996) Interactions between dietary calcium and caffeine consumption on calcium metabolism in hypertensive humans. Am J Hypertens 9:223–229
Ginty F (2003) Dietary protein and bone health. Proc.Nutr Soc 62:867–876
Acknowledgement
We would like to thank H. Jensen, H.L. Petersen, K. Ebbesen, B. Hoielt, K.H.B. Larsen and K.G. Rossen for their help with this study. Also, we thank Arla Foods for sponsoring the milk.
Author information
Authors and Affiliations
Corresponding author
Additional information
All authors were involved in the design of the study. M.K., M.J. and J.K. were responsible for the collection of data, analysis of data and writing of the manuscript. All authors participated in the discussion of the results and revision of the manuscript. None of the authors has any financial or personal interest in Arla Foods.
Rights and permissions
About this article
Cite this article
Kristensen, M., Jensen, M., Kudsk, J. et al. Short-term effects on bone turnover of replacing milk with cola beverages: a 10-day interventional study in young men. Osteoporos Int 16, 1803–1808 (2005). https://doi.org/10.1007/s00198-005-1935-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-005-1935-z