Abstract
Vitamin D supplements have been used to prevent fractures. The effect may be mediated through increased bone mass, but also through reduced falling propensity. The aim of this study was to evaluate the association between 25-hydroxy vitamin D levels (25OHD), fall-associated variables (including tests of functional performance), and fracture in ambulatory women. At baseline 25OHD was measured in 986 women. Fall-associated variables were investigated at baseline. Fractures were recorded during a 3-year follow-up. Four percent of the women had 25OHD levels below 20 ng/ml (50 nmol/l), and 26% had 25OHD levels below 30 ng/ml (75 nmol/l). 25OHD correlated with gait speed ( r =0.17, P <0.001), the Romberg balance test ( r =0.14, P <0.001), self-estimated activity level ( r =0.15, P <0.001), and thigh muscle strength ( r =0.08, P =0.02). During the 3-year follow-up, 119 out of the 986 women sustained at least one fracture. The Cox proportional hazard ratio (HR) (95% confidence interval) for sustaining a fracture during the follow-up was 2.04 (1.04–4.04) for the group of women with 25OHD below 20 ng/ml, in which 9 out of 43 women sustained a fracture. Thirty-two of the 256 women with 25OHD levels below 30 ng/ml sustained a fracture during the follow-up, with a non-significant HR of 1.07 (1.07–1.61). This cohort of elderly, ambulatory women had a high mean 25OHD. A low 25OHD was associated with inferior physical activity level, gait speed and balance. A 25OHD level below 30 ng/ml was not associated with an increased risk of fractures in this study. However, a subgroup of women with 25OHD levels below 20 ng/ml had a tendency to an increased risk of fractures, which may be associated with an inferior physical activity and postural stability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With advancing age, the risk of sustaining a low-energy fracture increases rapidly [1]. The risk increase is due to a higher incidence of osteoporosis, but also to a higher incidence of falls with higher age [1, 2]. The importance of vitamin D for skeletal health is well known [3, 4], but its possible association with fall-associated variables and falls is less clear [4, 5].
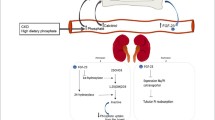
Vitamin D deficiency has been suggested to play a role in the development of osteoporosis because of the induction of a secondary hyperparathyroidism, which mobilizes calcium from the skeleton [4]. Vitamin D deficiency has been shown to be associated with myopathy in subjects of various ages [6, 7, 8, 9], with body sway in osteoporotic [10] and fall-prone subjects [11], and with falls in elderly, institutionalized subjects [12, 13].
Conversely, vitamin D supplementation has been shown to prevent fractures, but contradictory results exist [14]. It has been suggested that the anti-fracture efficacy is primarily mediated through the effect on bone mass that results from the vitamin D-mediated increase in calcium absorption, which reverses the secondary hyperparathyroidism and decreases a high bone turnover rate [4]. However, in a recent meta-analysis concerning randomized studies of vitamin D supplementation in older community-dwelling or institutionalized persons, falls were reduced in number [15], and this result could be related to improved muscle function and reduced body sway because of vitamin D supplementation [10, 12, 15].
The prevalence of vitamin D deficiency varies depending on the population studied, vitamin D fortification of foods, geographic location (latitude) and season [3, 16]. However, consensus has not been reached regarding the exact threshold level for vitamin D deficiency (measured as serum 25-hydroxy vitamin D, 25OHD) in relation to bone status [3, 4].
At present, there are few studies of the association between 25OHD levels and risk factors for falls and fractures, also evaluating the outcome, of fractures [17]. Our aim was to study the relation between 25OHD and fall-associated variables at baseline and the relation between 25OHD and fractures during a 3-year follow-up of elderly ambulatory women in Malmö, Sweden. We hypothesized that low 25OHD levels (1) correlate with poor balance, muscle strength and activity level and (2) are associated with a risk for future fractures.
Subjects and methods
Individuals
From December 1995 to May 1999, 1,604 women, picked randomly from the city files of Malmö, Sweden (latitude 56° north), were invited to participate in the Malmö Osteoporosis Prospective Risk Assessment (OPRA) study. Of the 1,604 women initially invited, 1,044 (65%) participated in the baseline investigation, which took place all year round. All women were 75 years old (range 75.0–75.9) at the initial visit. The reasons for non-participation have been described elsewhere [18]. Basically, all the women attending were ambulatory and living independently.
At baseline, all women answered an extensive questionnaire on their health and lifestyle. They were also examined for balance, muscle strength and gait speed. Venous blood samples were obtained at baseline, and the sera stored at –80° C until analysis. The women were followed for a mean of 3.0 years (range 2.9–3.4) after baseline examination until a mean age of 78.2 years (range 78.0–79.0). Fifty-five women died during the follow-up period. The Ethics Committee of Lund University approved the study, and informed consent was obtained from all subjects.
Methods
Vitamin D
The serum concentration of 25-hydroxy vitamin D (25OHD) was used as a measure of vitamin D status. Baseline serum samples were available for 986 women. Sampling times were recorded. All samples were taken, non-fasting, between 8:00 a.m. and 12:45 p.m. The 25OHD analysis was done with the Nichols Advantage assay (Nichols Institute Diagnostics, Calif.), which utilizes chemiluminescence detection. All samples were analyzed at the same time. According to the manufacturer, the reference range is 10–68 ng/ml (25–170 nmol/l), and the intra-assay coefficient of variation (CV) is less than 10% for 25OHD levels between 8.4 and 75.1 ng/ml (21 and 188 nmol/l). The intra-assay CV was 8.7% in this specific cohort as determined by duplicate measurements in 37 individuals.
Parathyroid hormone
Serum concentration of parathyroid hormone (PTH) was measured with the Elecsys PTH immunoassay (Roche Diagnostics, Mannheim Germany) with a reference range of 1.6–6.9 pmol/l. Intra- and inter-assay CVs were 1.6 and 5.7%, respectively.
Balance
A modified Romberg balance test was performed. It consisted of four parts (standing on the right or left foot, eyes open or closed) [19]. The time until balance was lost was recorded (or maximum 30 s). A result less than 5 s was considered a failed test and recorded as 0. Three trials per test were allowed, the best was registered, and all results were added to produce a score (minimum 0, maximum 120 s).
Gait
The women were asked to walk as fast as they could in their ordinary shoes for 30 m with one turn. The time taken was recorded in seconds (S) [19]. The gait speed test was performed once.
Muscle strength
The thigh muscle strength was tested with a computerized isokinetic dynamometer (Biodex Medical Systems, Version 4.5.0, Biodex Corporation, Shirley, New York). The best of three maximal isometric contractions in each direction (knee flexion at 45° and knee extension at 90°) of the right knee was recorded in Newton meter seconds (NmS) [19].
Areal bone mineral density
Areal bone mineral density (aBMD) of the femoral neck and lumbar spine (L II–L IV) was assessed by DXA technique (Lunar DPX-L, Madison, Wis.). The precision of this DXA equipment as assessed by duplicate measurements on healthy persons after repositioning has been determined to be 1.6% at the femoral neck and 0.5% at the spine.
Questionnaire
The self-assessment questionnaire used at baseline included questions on activity level, time spent outdoors, medication, and fractures. The activity level was self-assessed on a scale from one (bed rest only) to eight (still working, no limitation of mobility) and has been described elsewhere [20].
Fractures
The Malmö University Hospital is the only hospital in the city where fractures are treated. All X-rays and X-ray films are saved at the archive of the Radiology Department, which was searched to identify any symptomatic fractures sustained during the course of the study. Fracture data were searched for all participants in the OPRA study cohort using the Swedish personal identification number, thereby capturing all women with fractures examined radio-graphically at our hospital. In addition, at the 1- and 3-year follow-ups (data available for 994 and 907 of the participants, respectively) of the OPRA study, the participants were asked to record any incident fractures. Fractures not caused by low-energy trauma, such as traffic accidents, fall from heights or malignancies, were excluded from the analysis ( n =10).
Statistics
Spearman rank tests were used for correlations. A multiple regression was used to determine the combined influence of fall-associated and anthropometric variables on 25OHD. In addition, two different thresholds were used to define 25OHD deficiency, 20 or 30 ng/ml (50 and 75 nmol/l), as suggested by others [4, 21, 22, 23]. Differences between groups were determined with t -tests, Mann-Whitney U-tests or analysis of variance (ANOVA). The Cox proportional hazard ratio and the Kaplan-Meier survival analysis with log rank testing of differences between groups above and below the 25OHD thresholds were applied with the objective of comparing the association between 25OHD levels and the occurrence of at least one low-energy fracture during the follow-up. The level of significance was set to P <0.05.
Results
Mean (±SD) 25OHD for all women was 38.0±12.0 ng/ml (95±30 nmol/l). Forty-three (4.4%) women had a 25OHD level of less than 20 ng/ml (50 nmol/l), and 256 (26%) women had a 25OHD of less than 30 ng/ml (75 nmol/l) (Fig. 1).
Fall-associated variables
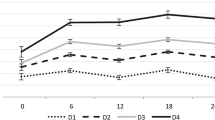
Women with 25OHD below either of the two 25OHD thresholds had a significantly lower self-assessed activity level, inferior gait speed, poorer balance, and spent less time outdoors than women with 25OHD above the thresholds (Table 1). Lower 25OHD was correlated with few hours spent outdoors/day ( r =0.16, P <0.001), inferior gait speed ( r =0.17, P <0.001), inferior Romberg balance test results ( r =0.15, P <0.001), lower self-estimated activity level ( r =0.15, P <0.001), lower knee extension strength ( r =0.08, P =0.020) and flexion strength ( r =0.07, P =0.032). There was a significant correlation between the 25OHD levels and self-assessed activity level in the ANOVA test (Fig. 2). A possible threshold in the vicinity of 35 ng/ml (87.5 nmol/l) is suggested by Fig. 2. Lower 25OHD was also correlated with high BMI ( r =–0.15, P <0.001) and high body weight ( r =–0.13, P <0.001), but not with body height ( r =0.04, P =0.24). In a multiple regression model, 5% of the variability in 25OHD was explained by the fall-associated variables and anthropometric variables combined. There was no correlation between sampling time of the day and 25OHD levels ( r =–0.06, P =0.056).
A significant association ( P <0.001; ANOVA) between the self-assessed physical activity level and 25OHD levels was seen ( n = number of women). As suggested by the figure, a threshold may exist between the 25OHD level and activity level in the vicinity of 35 ng/ml (87.5 nmol/l). To convert ng/ml to nmol/l, multiply by 2.5. 1 Bed rest only, cannot walk; 2 walking possible with human support only; 3 walks with walking aid, only indoors; 4 walks with walking aid, in- and outdoors; 5 no walking aid, walks independently without limitation; 6 takes frequent or regular walks; 7 participates in physical activity more strenuous than walking only; 8 still working, no limitation in mobility
PTH correlated to hours spent outdoors/day ( r =–0.06, P =0.044), gait speed ( r =–0.07, P =0.023), Romberg balance test result ( r =–0.12, P <0.001) and self-estimated activity level ( r =–0.11, P <0.001), but not to knee extension strength ( r =–0.03, P =0.26) and flexion strength ( r =–0.03, P =0.27).
Fractures
After 3 years, 12% (119 out of 986 women) had sustained a total of 159 low-energy fractures (29 hip, 28 wrist, 12 proximal humerus, 43 vertebral, and 47 other fractures). Nine out of the 43 women (21%) with 25OHD below 20 ng/ml (50 nmol/l) sustained at least one fracture of any type vs. 110 out of the 943 women (12%) with 25OHD levels above 20 ng/ml. Women with 25OHD levels below 20 ng/ml had an increased risk of sustaining at least one fracture during the follow-up with a Cox proportional hazard ratio (HR) (95% confidence interval) of 2.04 (1.04–4.04) and a significant Kaplan-Meier and log rank test (Fig. 3). Thirty-two out of the 256 women (12%) with 25OHD levels below 30 ng/ml (75 nmol/l) sustained at least one fracture of any type vs. 87 out of 730 (12%) women with a 25OHD level above 30 ng/ml. The HR for at least one fracture for women with 25OHD levels below 30 ng/ml was 1.01 (0.71–1.61), a result that was non-significant also in the Kaplan-Meier statistics (log rank test P =0.70).
Mean levels of 25OHD were highest in September (40.4 ng/ml; 101 nmol/l) and lowest in February (35.9 ng/ml; 89.8 nmol/l). The largest half-year difference was found between the periods November to April and May to October (37.1 vs. 39.5 ng/ml; 92.8 vs. 98.8 nmol/l; P =0.002). The fracture association was independent of the season of baseline investigation (data not shown).
Women taking vitamin D supplementation at baseline ( n =55) had higher mean 25OHD than women without supplementation (44.4 vs. 37.7 ng/ml; 111 vs. 94.2 nmol/l; P <0.001). Women taking vitamin D supplementation had a significantly lower activity level ( P =0.012), lower body weight ( P =0.003) and BMI ( P =0.003) than the other women. The women with vitamin D supplementation had a higher fracture incidence than women without supplementation (21 vs. 12%; log rank test P =0.03).
Exclusion of the women with vitamin D supplementation did not significantly change the correlations between 25OHD and the fall-associated variables or between PTH and the fall-associated variables (data not shown).
When women with vitamin D supplementation were excluded from the fracture calculations, women with 25OHD below 20 ng/ml (50 nmol/l) had a HR of 1.99 (0.97–4.02) for sustaining a fracture during the follow-up when compared to the other women. The corresponding HR for women with 25OHD below 30 ng/ml (75 nmol/l) was 1.10 (0.72–1.69).
Discussion
In this population-based cohort of Swedish elderly ambulatory women, we found (1) that low 25OHD levels were correlated with inferior balance, gait speed and activity level and (2) that 25OHD levels below 20 ng/ml (75 nmol/l), but not below 30 ng/ml (75 nmol/l), were associated with an increased risk of fracture.
Women below either of the two 25OHD thresholds (20 or 30 ng/ml) had significantly inferior gait speed, lower physical activity level and balance than women above the thresholds, even though the correlations were low. Low activity level and poor gait and balance are important predictors for falls [24] and fractures [25]. Whether a low functional performance level is caused by a low 25OHD level was not possible to elucidate in this cross-sectional study. Time spent outdoors differed significantly between the groups and may explain part of the association. However, an association between low activity and low 25OHD or low 1,25(OH)2 D have been reported from earlier cross-sectional and case-control studies [26, 27, 28, 29]. Furthermore, supplementation with vitamin D has been reported to increase the functional ability in subjects with very low baseline 25OHD levels (less than 15 ng/ml or 38 nmol/l) and 2.8 ng/ml (7.1 nmol/l), respectively [9, 30]; however, contradictory results exist [31]. A recent meta-analysis has shown a preventive effect on falls with vitamin D supplementation [15], but these studies may have concerned individuals with lower baseline 25OHD levels than in the present study [10, 12, 15].
The question whether hypovitaminosis D itself or a secondary hyperparathyroidism is the cause of an effect on muscle tissue still has not been answered [4, 5, 13]. 25OHD and PTH are (negatively) correlated, and it is difficult to distinguish any separate effects on muscle function. In this study, the correlations between PTH and the fall-associated variables were slightly lower or in the same magnitude as between 25OHD and the fall-associated variables.
Others have reported an association between a low dietary vitamin D intake and hip fracture [32, 33]. Low baseline levels of 1,25(OH)2 D, but not of 25OHD, have been reported to be associated with prospective hip fractures [17]. In this study, women with 25OHD levels below 30 ng/ml (75 nmol/l) had inferior gait speed, poorer balance and less physical activity than women above this threshold, but their fracture risk was not increased. A clinically important effect on fracture risk at this threshold would have been possible to detect. Considering the 25OHD threshold of 30 ng/ml (75 nmol/l) and the group sizes, a relative risk of fracture of 1.6 would have been required to achieve a power of 80% at a level of significance of 0.05.
In women with 25OHD levels below 20 ng/ml (50 nmol/l), albeit limited in number, a significantly higher incidence of fractures was seen during the follow-up compared to women with 25OHD levels above 20 ng/ml (hazard ratio 2.04). In this study, we did not find an association between baseline 25OHD and bone quality (as measured by aBMD). It can therefore be speculated that an increased fracture risk may be due to the association between low 25OHD and fall-associated variables. We wanted to examine the effect of vitamin D levels irrespective of source. However, after exclusion of women with vitamin D supplementation, the fracture risk in this group was no longer significantly higher (hazard ratio 1.99). The explanation for this is that the exclusion of women in an already small group affects the power of the study.
Women with poor results on neuromuscular function tests who spend little time outdoors are likely to have low 25OHD levels. These women may benefit from vitamin D supplementation. Fracture preventive effects of vitamin D supplementation have been shown for community-dwelling subjects with lower mean 25OHD than in this study [34, 35]. Whether supplementation in general in this group of ambulatory women would be of value is therefore not clear. The finding that the group of women with vitamin D supplementation had a high fracture risk is somewhat contradictory, but it is likely that treatment had been started in fracture prone subjects. Whether fracture risk was reduced or not in these subjects cannot be determined from this study.
25OHD levels may differ depending on the assay used and have to be taken into account when thresholds are discussed and studies are compared [36, 37, 38]. The specific assay used in this study has previously been reported to have a good correlation with 25OHD levels measured with high performance liquid chromatography ( r =0.91) and a radioimmunoassay ( r =0.90) [39].
The definition of threshold levels for vitamin D deficiency remains controversial. A threshold for 25OHD deficiency of 20 ng/ml has been based on the treatment effect of vitamin D on PTH-levels [21]. A threshold for 25OHD deficiency of approximately 30 ng/ml (75 nmol/l) has been suggested on the basis of evidence of secondary hyperparathyroidism [22, 23]. A reduction in fracture risk [34] and improvement in calcium absorption have been reported when 25OHD was increased from approximately 20 ng/ml (50 nmol/l) to 30 ng/ml (75 nmol/l) [40]. Nevertheless, the existence of a definite threshold level for vitamin D in relation to PTH has been questioned [29].
The mean 25OHD was high in comparison to other studies [4, 16, 29]. There are some possible explanations for this, besides assay differences. The women in the study may represent a healthy section of the population since 25% of the non-attendees (at baseline) refrained from participating due to illness [18]. Of the attendees, 98% were regularly outdoors for more than 30 min per day. Oil and low-fat milk products are commonly fortified with cholecalciferol in Sweden (between 3.8 and 10 microgram cholecalciferol/l); 74% consumed more than 1 l of milk weekly, and 68% had a regular intake of buttermilk or yogurt (at least once a week). In North America and Scandinavia, higher 25OHD values have been reported for healthy elderly populations compared to non-Scandinavian European countries, in which individuals also have a lower intake of vitamin D [16, 22].
Women with 25OHD levels below 20 ng/ml (50 nmol/l) most likely represent a frailer section of the population with less time spent outdoors, poorer results on neuromuscular function tests, and a higher fracture rate than other women. This group of women had a high mortality rate, where 21% (9 women out of 43) died during the 3-year follow-up, vs. only 5% among women with 25OHD levels above 20 ng/ml (50 nmol/l). Whether the low 25OHD is the primary cause of the results in this study is not possible to elucidate. Because of co-morbidities, frail women may be vitamin D deficient, for example, due to less time spent outdoors. The co-morbidities may contribute importantly to poorer results on indices of physical activity and neuromuscular function, and subsequently an increased risk of falls, fracture and mortality. Nevertheless, low 25OHD levels are associated with a higher incidence of both fall-related variables and fractures, suggesting a causal pathway.
References
Kanis JA (2002) Diagnosis of osteoporosis and assessment of fracture risk. Lancet 359:1929–1936
Gillespie LD, Gillespie WJ, Robertson MC, Lamb SE, Cumming RG, Rowe BH (2002) Interventions for preventing falls in elderly people (Cochrane Review). Cochrane Library, www.cochrane.org
Holick MF (2002) Vitamin D: the underappreciated D-lightful hormone that is important for skeletal and cellular health. Curr Opin Endocrinol Diabetes:87–98
Lips P (2001) Vitamin D deficiency and secondary hyperparathyroidism in the elderly: consequences for bone loss and fractures and therapeutic implications. Endocr Rev 22:477–501
Pfeifer M, Begerow B, Minne HW (2002) Vitamin D and muscle function. Osteoporos Int 13:187–194
Bischoff HA, Stahelin HB, Urscheler N, Ehrsam R, Vonthein R, Perrig-Chiello P, Tyndall A, Theiler R (1999) Muscle strength in the elderly: its relation to vitamin D metabolites. Arch Phys Med Rehabil 80:54–58
Mowe M, Haug E, Bohmer T (1999) Low serum calcidiol concentration in older adults with reduced muscular function. J Am Geriatr Soc 47:220–226
Schott GD, Wills MR (1976) Muscle weakness in osteomalacia. Lancet 1:626–629
Glerup H, Mikkelsen K, Poulsen L, Hass E, Overbeck S, Andersen H, Charles P, Eriksen EF (2000) Hypovitaminosis D myopathy without biochemical signs of osteomalacic bone involvement. Calcif Tissue Int 66:419–424
Pfeifer M, Begerow B, Minne HW, Abrams C, Nachtigall D, Hansen C (2000) Effects of a short-term vitamin D and calcium supplementation on body sway and secondary hyperparathyroidism in elderly women. J Bone Miner Res 15:1113–1118
Dhesi JK, Bearne LM, Moniz C, Hurley MV, Jackson SH, Swift CG, Allain TJ (2002) Neuromuscular and psychomotor function in elderly subjects who fall and the relationship with vitamin D status. J Bone Miner Res 17:891–897
Bischoff HA, Stahelin HB, Dick W, Akos R, Knecht M, Salis C, Nebiker M, Theiler R, Pfeifer M, Begerow B, Lew RA, Conzelmann M (2003) Effects of vitamin D and calcium supplementation on falls: a randomized controlled trial. J Bone Miner Res 18:343–351
Stein MS, Wark JD, Scherer SC, Walton SL, Chick P, Di Carlantonio M, Zajac JD, Flicker L (1999) Falls relate to vitamin D and parathyroid hormone in an Australian nursing home and hostel. J Am Geriatr Soc 47:1195–1201
Gillespie WJ, Avenell A, Henry DA, O’Connell DL, Robertson J (2001) Vitamin D and vitamin D analogues for preventing fractures associated with involutional and post-menopausal osteoporosis. Cochrane Database Syst Rev:CD000227
Bischoff-Ferrari HA, Dawson-Hughes B, Willett WC, Staehelin HB, Bazemore MG, Zee RY, Wong JB (2004) Effect of Vitamin D on falls: a meta-analysis. JAMA 291:1999–2006
McKenna MJ (1992) Differences in vitamin D status between countries in young adults and the elderly. Am J Med 93:69–77
Cummings SR, Browner WS, Bauer D, Stone K, Ensrud K, Jamal S, Ettinger B (1998) Endogenous hormones and the risk of hip and vertebral fractures among older women. Study of Osteoporotic Fractures Research Group. N Engl J Med 339:733–738
Gerdhem P, Ringsberg KA, Magnusson H, Obrant KJ, Akesson K (2003) Bone mass cannot be predicted by estimations of frailty in elderly ambulatory women. Gerontology 49:168–172
Ringsberg K, Gerdhem P, Johansson J, Obrant KJ (1999) Is there a relationship between balance, gait performance and muscular strength in 75-year-old women? Age Ageing 28:289–293
Gerdhem P, Akesson K, Obrant KJ (2003) Effect of previous and present physical activity on bone mass in elderly women. Osteoporos Int 14:208–212
Malabanan A, Veronikis IE, Holick MF (1998) Redefining vitamin D insufficiency. Lancet 351:805–806
Chapuy MC, Preziosi P, Maamer M, Arnaud S, Galan P, Hercberg S, Meunier PJ (1997) Prevalence of vitamin D insufficiency in an adult normal population. Osteoporos Int 7:439–443
Melin AL, Wilske J, Ringertz H, Saaf M (1999) Vitamin D status, parathyroid function and femoral bone density in an elderly Swedish population living at home. Aging (Milano) 11:200–207
Moreland J, Richardson J, Chan DH, O’Neill J, Bellissimo A, Grum RM, Shanks L (2003) Evidence-based guidelines for the secondary prevention of falls in older adults. Gerontology 49:93–116
Cummings SR, Nevitt MC, Browner WS, Stone K, Fox KM, Ensrud KE, Cauley J, Black D, Vogt TM (1995) Risk factors for hip fracture in white women. Study of Osteoporotic Fractures Research Group. N Engl J Med 332:767–773
Nelson ME, Meredith CN, Dawson-Hughes B, Evans WJ (1988) Hormone and bone mineral status in endurance-trained and sedentary postmenopausal women. J Clin Endocrinol Metab 66:927–933
Bell NH, Godsen RN, Henry DP, Shary J, Epstein S (1988) The effects of muscle-building exercise on vitamin D and mineral metabolism. J Bone Miner Res 3:369–373
Bischoff HA, Dietrich T, Orav EJ, Zhang Y, W. KE, Dawson-Hughes B (2003) Higher 25-hydroxyvitamin D levels are associated with better lower extremity function in active and inactive ambulatory elderly in the US (abstract). J Bone Miner Res 18:S52
Bates CJ, Carter GD, Mishra GD, O’Shea D, Jones J, Prentice A (2003) In a population study, can parathyroid hormone aid the definition of adequate vitamin D status? A study of people aged 65 years and over from the British National Diet and Nutrition Survey. Osteoporos Int 14:152–159
Gloth FM, 3rd, Smith CE, Hollis BW, Tobin JD (1995) Functional improvement with vitamin D replenishment in a cohort of frail, vitamin D-deficient older people. J Am Geriatr Soc 43:1269–1271
Corless D, Dawson E, Fraser F, Ellis M, Evans SJ, Perry JD, Reisner C, Silver CP, Beer M, Boucher BJ (1985) Do vitamin D supplements improve the physical capabilities of elderly hospital patients? Age Ageing 14:76–84
Meyer HE, Henriksen C, Falch JA, Pedersen JI, Tverdal A (1995) Risk factors for hip fracture in a high incidence area: a case-control study from Oslo, Norway. Osteoporos Int 5:239–246
Feskanich D, Willett WC, Colditz GA (2003) Calcium, vitamin D, milk consumption, and hip fractures: a prospective study among postmenopausal women. Am J Clin Nutr 77:504–511
Trivedi DP, Doll R, Khaw KT (2003) Effect of four monthly oral vitamin D3 (cholecalciferol) supplementation on fractures and mortality in men and women living in the community: randomised double blind controlled trial. BMJ 326:469
Dawson-Hughes B, Harris SS, Krall EA, Dallal GE (1997) Effect of calcium and vitamin D supplementation on bone density in men and women 65 years of age or older. N Engl J Med 337:670–676
Lips P, Chapuy MC, Dawson-Hughes B, Pols HA, Holick MF (1999) An international comparison of serum 25-hydroxyvitamin D measurements. Osteoporos Int 9:394–397
Binkley N, Krueger D, Cowgill CS, Plum L, Lake E, Hansen KE, DeLuca HF, Drezner MK (2004) Assay variation confounds the diagnosis of hypovitaminosis D: a call for standardization. J Clin Endocrinol Metab 89:3152–3157
Hollis BW (2004) Editorial: the determination of circulating 25-hydroxyvitamin D: no easy task. J Clin Endocrinol Metab 89:3149–3151
Roth HJ, Zahn I, Alkier R, Schmidt H (2001) Validation of the first automated chemiluminescence protein-binding assay for the detection of 25-hydroxycalciferol. Clin Lab 47:357–365
Heaney RP, Dowell MS, Hale CA, Bendich A (2003) Calcium absorption varies within the reference range for serum 25-hydroxyvitamin D. J Am Coll Nutr 22:142–146
Acknowledgements
Funds for this study were received from the Albert Påhlsson Foundation, the Alfred Österlund Foundation and the Swedish Research Council. The Nichols Institute Diagnostics, Calif., and ElectraBox, Sweden, provided assay kits and supported the work for the analysis of 25-hydroxy vitamin D.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gerdhem, P., Ringsberg, K.A.M., Obrant, K.J. et al. Association between 25-hydroxy vitamin D levels, physical activity, muscle strength and fractures in the prospective population-based OPRA Study of Elderly Women. Osteoporos Int 16, 1425–1431 (2005). https://doi.org/10.1007/s00198-005-1860-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-005-1860-1