Abstract
Only a few studies have examined the risk of individual chronic health disorders on perimenopausal (i.e., around menopause) fractures in a single study. We evaluated the effect of chronic illnesses on fracture rate in a prospective cohort study of 3,078 women. These women were a stratified sample from the population base of 14,220 women aged 47–56 years and residing in the province of Kuopio in eastern Finland in 1989. Data on physician-diagnosed chronic diseases were collected by a baseline questionnaire in 1989. For certain diseases, questionnaire information of self-reported chronic disorders were compared with drug reimbursement data provided by the Social Insurance Institution of Finland. Axial bone mineral density (BMD) measurements from the femoral neck and lumbar spine were performed in 1989–91. Two hundred sixty-five (265) women experienced at least one fracture during the follow-up period of 3.6 years (SD±0.78). The first fracture during the follow-up period was taken to be the end-point event. The risk of follow-up fracture for an individual health disorder was estimated with the Cox’s proportional hazards model. Several chronic health disorders increased the fracture risk in perimenopausal women. However, hypertension was a statistically significant ( p=0.018) risk factor for fracture (adjusted hazard ratio [HR], 1.4; 95% confidence interval [CI], 1.1–1.9), especially in overweight and obese (body mass index ≥28) women (HR, 2.0; 95% CI, 1.4–3.0). In addition, coronary heart disease (adjusted HR, 1.76; 95% CI, 1.13–2.76), hyperthyroidism (adjusted HR, 1.7; 95% CI, 1.0–2.9), epilepsy (adjusted HR, 2.0; 95% CI, 1.1–3.6), alcoholism (adjusted HR, 3.5; 95% CI, 1.3–9.5) and chronic hepatic disease (adjusted HR, 5.2; 95% CI, 1.7–16.4) predicted fracture. BMD was either normal or even elevated in disease groups. However, women with a fracture during the follow-up usually had decreased bone density, although the difference was statistically significant only in women with hypertension and hyperthyroidism. We conclude that hypertension, coronary heart disease, alcoholism, epilepsy and hyperthyroidism can markedly increase the risk of fracture in perimenopausal women and should be taken into account when assessing the risk of future fracture in an individual patient. Furthermore, in contrast to previous data, obesity alone does not increase the risk of perimenopausal fracture, but in association with hypertension the risk seems to be markedly elevated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Almost 40% of women will experience a fracture during their postmenopausal period [1]. Osteoporosis is one of the major reasons for fracture, but risk factors for falls also modulate the overall fracture risk [1]. Important risk factors for fractures and osteoporosis are low bone mineral density, ageing, lack of hormone replacement therapy (HRT) and positive fracture history [2,3].
The prevalence of chronic health disorders increases with advancing age. Certain diseases, usually those affecting calcium metabolism, have been shown to increase the risk of osteoporosis [4,5]. Moreover, high morbidity is associated with an increased risk of falls in the elderly [6,7]. Morbidity in the elderly might elevate the fracture risk by affecting normal gait or balance, thus predisposing to falls, or by affecting the quality of bone [8]. In contrast, in perimenopausal women, i.e., in women around the menopause, risk factors for falls have been attributed to increased weight [9] and the absence of hormone replacement therapy [10]. Furthermore, there is evidence that increased morbidity might also independently predispose for fractures in perimenopausal women, as shown in our previous report from the same study cohort [11]. However, there are no studies that have examined the risk of individual chronic health disorders for perimenopausal fractures in a single population-based study.
The aim of our present study was to evaluate the impact of a specific chronic health disorders on the fracture rate in a prospective study. Our previous study [11] suggested that the number of chronic health disorders can be regarded as an independent risk factor for fracture. This study focuses on the effect of selected disorders on fracture risk, and thus the study is an extension of our previous work.
Materials and methods
The study group of 3,078 women was a stratified sample from the population base of all 14,220 women aged 47–56 years who resided in the province of Kuopio in eastern Finland in 1989. The baseline postal enquiry was sent in 1989, to which 13,100 (92.8%) women responded. Respondents were given a list of chronic diseases to check off, and an additional open space was provided for diseases not mentioned on the list. Respondents were advised to mark the diseases that were—according to respondent’s recollection—diagnosed by a physician they had at the time of baseline enquiry. Furthermore, respondent’s willingness to take part in axial bone mineral density measurements was asked.
A total of 11,055 (84.4%) respondents were willing to participate in bone densitometry. Of those, 3,686 women (33.3%) were selected and 3,222 (87.4%) were finally measured, belonging to either the random sample (n=2,025) or those selected based on certain risk factors (stratified sample =1,197). The selection of the stratified sample was made on the basis of previous knowledge of certain physical conditions, diseases, or treatments. The stratified sample included 633 women who had experienced menopause within 2 years before baseline BMD measurement, 193 with certain diseases or drugs affecting bone metabolism (insulin-dependent diabetes, hyperparathyroidism, ovariectomy before age 45 without HRT, corticosteroids, phenytoin, carbamazepine, phenobarbital), 95 with multiple behavioral risk factors (three of the following five factors: dairy calcium intake of less than 500 mg/day, current smoking of more than four cigarettes per day, light work and no regular exercise, body mass index below 22 or weight below 56 kg), 224 eligible for a randomized 5-year clinical hormone trial (generally healthy women who fulfilled the selection criteria and were willing to participate in a study) and 52 women with rheumatoid arthritis [12].
The baseline bone mineral density (BMD) was determined using dual X-ray absorptiometry (DXA) (Lunar DPX, Madison, WI, USA) of the spine (L2–L4) and left femoral neck during 1990–91 at Kuopio University Hospital by trained personnel. The women undergoing BMD measurements also filled out an additional questionnaire in which they reported their current consumption of dairy products. Both the baseline and the additional questionnaire were confirmed by a trained nurse with the participant at the time of the densitometry [11].
At the end of May 1994, a follow-up questionnaire was sent to the entire cohort. Of the densitometry sample (n=3,222), 3,078 women (96%) responded to this follow-up enquiry, and they form the present study population in this paper. The enquiry included questions about whether the respondent had suffered a fracture after the baseline bone densitometry. If so, questions about the type, mechanism, circumstances and treatment of the fracture were asked. Furthermore, the respondents were asked about the occurrence of maternal hip fracture. Only hip fractures occurring after the age of 60 years were included in the analyses. All self-reported fractures were validated by cross-checking radiological reports from medical records. However, rib fractures were accepted without radiological evidence, if the clinical diagnosis in the medical records was a rib fracture. All fractures, also high-energy fractures, were included in the analyses. The sensitivity of self-reported fractures was 78.4% and the specificity was 95.7% [13].
Postmenopausal status was defined as a natural or surgical cessation of menstruation occurring more than 6 months previously. Hormone replacement therapy was considered to be represented by the continuous use of estrogen (with or without progestin) replacement therapy at the time of the BMD measurement. An estimate of dietary calcium intake was derived from the reported consumption of dairy products (1 dl milk=120 mg calcium; 1 slice cheese=87 mg calcium). A positive response to a smoking enquiry meant the respondent was currently smoking in 1989. The study was approved by the local ethics committee [11].
The question about chronic diseases was formulated as: “Have you any of the following diseases diagnosed by a physician?” The respondents were advised to circle all the diseases they had from the following list of chronic diseases: hypertension, hypercholesterolemia, coronary heart disease, chronic heart failure, stroke, deep venous thrombosis, pulmonary embolism, insulin-dependent diabetes, chronic renal failure, chronic hepatic failure, rheumatoid arthritis, epilepsy, asthma or other chronic pulmonary disease, lactose intolerance, chronic mental disorder, hyperthyroidism, hyperparathyroidism, alcoholism and cancer. Moreover, an additional empty space was provided for other diseases not mentioned on the list. There was no specific order or gradation for diseases to enter in the final analyses.
In order to validate self-reported diseases, all self-reported chronic disorders were compared, if available, with the statistics provided by the Social Insurance Institution of Finland. These statistics include information on diseases with medical expenses reimbursed by the state. Before reimbursement, a medical certificate on the diagnosis and severity of disease is required. Thus, we were able to compare the information of self-reported hypertension, coronary heart disease, chronic heart failure, epilepsy, hypothyroidism and thrombocytopenia with the reimbursement statistics. Sensitivity and specificity, respectively, and agreement estimated as kappa values for self-reported chronic disorders were 89% and 98% for hypertension (kappa 0.9), 72% and 98% for coronary heart disease (kappa 0.6), 77% and 95% for heart failure (kappa 0.2), 86% and 100% for epilepsy (kappa 0.9), 100% and 100% (kappa 0.4) for thrombocytopenia, 74% and 99% (kappa 0.7) for hypothyroidism.
Statistical analysis
Statistical analyses were performed using SPSS for Windows software, version 11.0 (Statistical Package for Social Sciences, SPSS). All self-reported chronic health disorders were tested to assess which disorders were associated ( p<0.10) with an elevated risk of fracture during follow-up. In the analyses, a single disease was regarded as a variable to examine the risk of fracture. We compared diseased women against all the others and, when appropriate, against women without self-reported disorders or without disease information in the database of the Social Insurance Institution of Finland. The first fracture during the follow-up period was taken to be the end-point event. Univariate analyses were performed by two-tailed, unpaired Student’s t -test for continuous variables and by chi-square statistics or Fisher’s exact test, when appropriate, for categorical variables. Sensitivity, specificity and kappa values for self-reported illnesses were computed from cross-tabulations. Backward stepwise Cox regression analyses (unadjusted and adjusted models) were performed to investigate the relative risk of fracture for each disease that had a significant or nearly significant ( p<0.10) association with follow-up fractures in crude analyses. The total study cohort was included in the Cox’s analyses, without separating the random and stratified samples. The covariates in the adjusted Cox’s model were: age, weight, height, BMD, history of previous fracture and use of HRT. The results are expressed as relative risk (hazard ratio, HR) estimates with 95% confidence intervals (CI). The results for the Cox analyses were considered to be statistically significant if the p value was less than 0.05.
Results
In all, 2,061 women (67%) reported having at least one chronic disease at the baseline. The list and number of self-reported health disorders are provided in Table 1. A total of 265 women experienced one or more fractures during the follow-up period of 3.6 years (SD, ±0.78). The total number of fractures was 306. The most common fractures were in the distal forearm (n=78), ankle (n=49) and ribs (n=45). Table 2 summarizes the characteristics of the study groups according to reported chronic disease (i.e., those reporting at least one chronic health disorder on the questionnaire). Women who reported chronic diseases were slightly older (mean difference, 0.7 years, p< 0.001), heavier (mean difference, 2.7 kg, p< 0.001) and did not use HRT as much as those without disorders (24% vs. 29%, p=0.001). Moreover, the proportion of postmenopausal women was higher in the disease group (73% vs 63%). Interestingly, maternal hip fracture was more prevalent in the healthy group (8% vs 5%). Bone mineral density (BMD) in the femoral neck or lumbar spine did not differ between healthy and non-healthy groups. The prevalence of previous fractures and the total frequency of fractures during follow-up were also similar.
In the subgroup of women without self-reported chronic health disorders (Table 3), the women experiencing a fracture during follow-up were slightly but significantly ( p=0.031) younger (mean age 52.3 years) than women without a follow-up fracture (mean age, 53.0 years). Femoral neck BMD, spinal BMD and daily calcium consumption were also significantly lower in healthy women who experienced a follow-up fracture ( p values<0.001, 0.022 and 0.041, respectively) than other healthy women. HRT use and maternal hip fracture were similarly distributed in both groups. In the non-healthy cohort (i.e., those reporting at least one chronic disorder) women who suffered from fracture during follow-up had significantly lower spinal and femoral neck BMD ( p values <0.001 for both), used HRT less frequently (14% vs 25%, p=0.001) and more often had a positive history of maternal hip fracture (36% vs 22%, p<0.001) at baseline than those without a follow-up fracture. Dietary calcium intake was, however, not different between the fracture or non-fracture groups in women with at least one chronic disorder.
All self-reported diseases, except varicose vein disease and hypertension, either increased or decreased significantly ( p<0.05) the risk of follow-up fracture in unadjusted models (Table 4). In the adjusted model, all variables—except degenerative arthritis, heart failure and varicose vein disease—showed a significant ( p<0.05) association with follow-up fractures. Furthermore, hypertension, per se, and also in combination with other common cardiovascular diseases, appeared to significantly predict ( p=0.018) a follow-up fracture in the adjusted model. If a woman was suffering from hypertension, coronary heart disease and heart failure at the same time, the risk of a follow-up fracture was increased almost five-fold (HR, 4.92; 95% CI, 1.82–13.27). The reason for choosing these combinations of circulatory disease only was that they are commonly encountered clinically. Other combinations were also examined, but the number of women with combinations of diseases was small, limiting the statistical power.
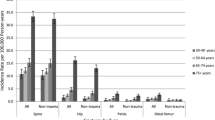
Further analyses revealed that the risk of fracture in women with hypertension was enhanced by the co-presence of obesity (Fig. 1). Ankle fracture was the most common fracture in this cohort. Furthermore, if ankle fractures were excluded from the analysis, the effect of hypertension on fracture risk was no longer statistically significant ( p=0.432, Cox’s model). The prevalence of diabetes in the group of obese women with hypertension as compared with obese women without hypertension was 6% and 1%, respectively ( p<0.001 between groups).
Percentage of follow-up fractures in groups according to hypertension and body mass index (BMI) and in the total study cohort (* p=0.129 between groups I and II; p= 0.002 between groups I and III; p= 0.032 between groups I and IV; ** p= 0.177 between groups III and II; p= 0.065 between groups III and IV)
Femoral neck BMD was usually lower in women who had a fracture during the follow-up in different disease groups (Table 5). However, the difference was statistically significant only for hypertension, hyperthyroidism and varicose vein disease.
Discussion
We have previously shown that the presence of three or more chronic diseases is a predictor of follow-up fracture [11], but the present study affirms, as a novel finding, that even the presence of a single common chronic disease can increase the risk of perimenopausal fracture. In the elderly, chronic health disorders have been associated with increased risk of hip fracture [14,15], but also the risk of other non-spinal fracture is elevated [16]. However, according to our knowledge, no prospective studies on perimenopausal populations have been published earlier. Fall-related fracture mechanisms were somewhat more common in the chronic disorder group than in women reporting no chronic health disorders in our study. The mechanism of how a single chronic health disorder could increase the fracture risk might be due to a direct effect on bone quality, such as BMD, or, on the other hand, due to an increment in the risk of falling. We suggest that a chronic disorder may affect the equilibrium, proprioceptive or locomotive systems, thus leading to an elevated risk of falling. For example, it has been suggested that diabetes could increase the risk of fracture because of an increased risk of falling due to diabetic neuropathy [17].
Another noteworthy finding in the present study was that obese women with hypertension had significantly increased risk of fracture. The high prevalence of ankle fractures in this cohort could explain this, in part because obesity has previously been shown to be associated with ankle fractures [18]. However, we found that obese patients without hypertension seemed to sustain fewer fractures than the average. Higher body mass index has been associated with increased risk of ankle fracture [16,18] and falls [9] in perimenopausal women in previous studies, but our finding suggests that obesity per se might not be the only reason for that. Furthermore, in our study we found that diabetes was more frequent among obese women with hypertension than among those without hypertension. We may therefore speculate that the prevalence of excess morbidity, including the metabolic syndrome, of obese patients is increased, which can lead to a higher frequency of falling, and an increased risk of fractures. Unfortunately, we were not able to study the possible increase in falling frequency of these women. There can also be differences in the medication of hypertension in these obese and non-obese patients. For example, thiazides have been shown to decrease fracture risk [19]. In contrast to our findings, hypertension per se has been found [20] to have a protective effect on distal forearm fractures in Rochester women 35–94 years old (unadjusted odds ratio, 0.64). Our study cohort had a narrower age range, and we included all fractures, not only distal forearm fractures, in analyses.
Interestingly, femoral neck BMD, overall a strong predictor of follow-up fracture in this study, was not statistically significantly different between women with and without fractures in individual disease groups, except among women with hypertension, hyperthyroidism and varicose vein disease. The lack of association of BMD with fracture emphasizes the importance of chronic diseases as a predictor of fracture, but might also be because of insufficient study power inside the groups.
Hyperthyroidism has been shown to be associated with elevated risk of hip fractures [21] and other types of fractures [22]. BMD has been found to be lower in patients with hyperthyroidism [23]. Our present study is in agreement with the results of previous studies. Also, epilepsy was found to be an independent risk factor for fracture. The adjusted relative risk for a follow-up fracture was 2.0 in women who reported epilepsy. Interestingly, we could not find any difference in femoral neck BMD between women with and without fractures among women with epilepsy, possibly indicating an increased risk of falls or other traumatic events in these patients. Our finding supports a recent study that showed a similar risk ratio, especially in epileptic women over 45 years of age [24]. The risk of fracture in epileptic patients has been suggested to be mostly seizure-related, and less related to bone status [24]. Moreover, a recent study has suggested that certain anti-epilepsy medications, e.g., phenytoin, might increase the risk of fracture by affecting vitamin D metabolism [25].
Varicose vein disease was found to be a protective factor, even though femoral neck BMD was significantly lower in those women who sustained a fracture in the varicose vein group. This is explained by the fact that there were only two women with varicose vein disease who experienced a fracture, indicating that a type II error is likely to play a role.
It has been shown [26] that alcoholic women have significantly lower spinal and femoral neck BMD than do non-alcoholic women. In our study, those women giving a positive history of alcoholism at baseline showed a marked increase in the fracture rate during the follow-up (unadjusted HR, 5.14; adjusted HR, 3.49). However, we did not find any difference in BMD between alcoholic women with and without fractures ( p=0.275), suggesting that these fractures are more trauma-related. Nevertheless, it has to be understood that we simply asked whether or not an individual was alcoholic. This kind of approach might underestimate the true number of alcoholics. In addition, we also asked about the amount of alcohol consumed. According to those data, almost 59% of women did not use alcohol at all, including 25% of alcoholic women. We suggest, however, that the amount of alcohol consumed is an important risk facture for fracture, because the average alcohol consumption was significantly higher in the fracture group than in the non-fracture group (61 g vs 50 g, p= 0.045).
There are certain limitations to our study. First of all, the selection of women to participate in BMD baseline measurements was not totally random. Approximately one-third of the women were selected as the stratified sample, while two-thirds of women made up the random sample. However, over half of the stratified sample was selected on the basis of recent menopause, in order to achieve an early postmenopausal group large enough, and the rest of the sample was a weighted sample of certain diseases and risk factors. Thus, our selection criteria overestimate the prevalence of certain diseases in this group (Table 1). Nevertheless, our selection criteria do not bias the fracture incidence in these disease groups, even though the selection criteria may decrease the generalizability of the results.
Another noteworthy limitation is that we did not exclude women who had several health disorders at the same time, which could lead to somewhat biased results for individual disorders and increase the probability of a type I error. Furthermore, the study power for many diseases and conditions might have been too low because of the small number of cases (e.g., alcoholism, pulmonary embolism, thrombocytopenia, etc.). The observational nature of this study has to be recognized. However, there were some diseases frequent enough (e.g., hypertension, asthma, coronary heart disease, heart failure, diabetes, hypothyroidism, hyperthyroidism, hypercholesterolemia and rheumatoid arthritis) to give sufficient power for the analyses, thus making generalization of the results to the whole population possible. Moreover, the limitations of this study include the potential bias from self-reported diseases. The sensitivity of our questionnaire data compared with the national register was excellent or good for thrombocytopenia (100%), coronary heart disease (98%), hypertension (89%) and epilepsy (86%), respectively, but only moderate for heart failure (77%), and hypothyroidism (74%). The agreement between self-reports and national register information was excellent for hypertension and epilepsy (both kappa 0.9) and good for hypothyroidism (kappa 0.7) and coronary heart disease (kappa 0.6). However, the agreement was poor for heart failure (kappa 0.2) due to a large number with a mild form of the disease. Nevertheless, these numbers appear to be acceptable compared with other studies [27–29]. On the other hand, if only validated data from the Social Insurance Institution of Finland had been used for the analyses, there might have been an underestimation of actual prevalence of diseases, because the national register includes only patients with established disease and ignores mild forms of disease.
Our study does not provide an answer to the question of whether it is the disease or the treatment that is potentially contributing to falls and fractures. This is particularly important when one considers hypertension, because some treatments, e.g., thiazide therapy, can increase BMD [30,31] and potentially decrease the risk of fracture [19]. On the other hand, untreated hypertension could increase the risk of postprandial or orthostatic hypotension, thus elevating the risk of fall [32]. This study shows that common health disorders can increase the risk of fracture, but the reason for that remains unclear.
We conclude that even a single chronic health disorder can elevate fracture risk in perimenopausal women, by increasing the risk of falls and predisposing to osteoporosis. Obese women with hypertension seem to be especially at risk. This is a novel finding and in contrast with the previous conception that obese women have a lower risk of fractures. Our finding suggests that obesity might be considered a risk factor when coexisting with hypertension. The appropriate treatment of chronic disease provides the basis for fracture prevention in these women. Therefore, perimenopausal women with chronic health disorders should be informed of their elevated risk of fracture and instructed on how to avoid falls.
References
Lips P (1997) Epidemiology and predictors of fractures associated with osteoporosis. Am J Med 103:3S–8S
Wu F, Mason B, Horne A, Ames R, Clearwater J, Liu M, Evans MC, Gamble GD, Reid IR (2002) Fractures between the ages of 20 and 50 years increase women’s risk of subsequent fractures. Arch Intern Med 162:33–36
Cauley JA, Robbins J, Chen Z, Cummings SR, Jackson RD, LaCroix AZ, LeBoff M, Lewis CE, McGowan J, Neuner J, Pettinger M, Stefanick ML, Wactawski-Wende J, Watts NB, Women’s Health Initiative Investigators (2003) Effects of estrogen plus progestin on risk of fracture and bone mineral density: the Women’s Health Initiative randomized trial. JAMA 290:1729–1738
Nordin BE (1997) Calcium and osteoporosis. Nutrition 13:664–686
Stein E, Shane E (2003) Secondary osteoporosis. Endocrinol Metab Clin North Am 32:115–134, vii
Luukinen H, Koski K, Laippala P, Kivela SL (1995) Predictors for recurrent falls among the home-dwelling elderly. Scand J Prim Health Care 13:294–299
Rubenstein LZ, Josephson KR (2002) The epidemiology of falls and syncope. Clin Geriatr Med 18:141–158
Ensrud KE, Nevitt MC, Yunis C, Cauley JA, Seeley DG, Fox KM, Cummings SR (1994) Correlates of impaired function in older women. Am Geriatr Soc 42:481–489
Torgerson DJ, Garton MJ, Reid DM (1993) Falling and perimenopausal women. Age Ageing 22:59–64
Randell KM, Honkanen RJ, Komulainen MH, Tuppurainen MT, Kroger H, Saarikoski S (2001) Hormone replacement therapy and risk of falling in early postmenopausal women—a population-based study. Clin Endocrinol (Oxf) 54:769–774
Huopio J, Kroger H, Honkanen R, Saarikoski S, Alhava E (2000) Risk factors for perimenopausal fractures: a prospective study. Osteoporos Int 11:219–227
Kroger H, Tuppurainen M, Honkanen R, Alhava E, Saarikoski S (1994) Bone mineral density and risk factors for osteoporosis—a population-based study of 1600 perimenopausal women. Calcif Tissue Int 55:1–7
Honkanen K, Honkanen R, Heikkinen L, Kroger H, Saarikoski S (1999) Validity of self-reports of fractures in perimenopausal women. Am J Epidemiol 1 150(5):511–516
Cummings SR, Nevitt MC, Browner WS, Stone K, Fox KM, Ensrud KE, Cauley J, Black D, Vogt TM (1995) Risk factors for hip fracture in white women. Study of Osteoporotic Fractures Research Group. N Engl J Med 332:767–773
Willig R, Luukinen H, Jalovaara P (2003) Factors related to occurrence of hip fracture during a fall on the hip. Public Health 117:25–30
Hasselman CT, Vogt MT, Stone KL, Cauley JA, Conti SF (2003) Foot and ankle fractures in elderly white women. Incidence and risk factors. J Bone Joint Surg Am 85:820–824
Nicodemus KK, Folsom AR; Iowa Women’s Health Study (2001) Type 1 and type 2 diabetes and incident hip fractures in postmenopausal women. Diabetes Care 24:1192–1197
Valtola A, Honkanen R, Kroger H, Tuppurainen M, Saarikoski S, Alhava E (2002) Lifestyle and other factors predict ankle fractures in perimenopausal women: a population-based prospective cohort study. Bone 30:238–242
Feskanich D, Willett WC, Stampfer MJ, Colditz GA (1997) A prospective study of thiazide use and fractures in women. Osteoporos Int 7(1):79–84
Melton LJ 3rd, Achenbach SJ, O’Fallon WM, Khosla S (2002) Secondary osteoporosis and the risk of distal forearm fractures in men and women. Bone 31:119–125
Bauer DC, Ettinger B, Nevitt MC, Stone KL; Study of Osteoporotic Fractures Research Group (2001) Risk for fracture in women with low serum levels of thyroid-stimulating hormone. Ann Intern Med 134:561–568
Solomon BL, Wartofsky L, Burman KD (1993) Prevalence of fractures in postmenopausal women with thyroid disease. Thyroid 3:17–23
Vestergaard P, Mosekilde L (2003) Hyperthyroidism, bone mineral, and fracture risk—a meta-analysis. Thyroid 13:585–593
Persson HB, Alberts KA, Farahmand BY, Tomson T (2002) Risk of extremity fractures in adult outpatients with epilepsy. Epilepsia 43:768–772
Vestergaard P, Rejnmark L, Mosekilde L (2004) Fracture risk associated with use of antiepileptic drugs. Epilepsia 45(11):1330–1337
Clark MK, Sowers MF, Dekordi F, Nichols S (2003) Bone mineral density and fractures among alcohol-dependent women in treatment and in recovery. Osteoporos Int 14:396–403
Colditz GA, Martin P, Stampfer MJ, Willett WC, Sampson L, Rosner B, Hennekens CH, Speizer FE (1986). Validation of questionnaire information on risk factors and disease outcomes in a prospective study of women. Am J Epidemiol 123:894–900
Klungel OH, de Boer A, Paes AH, Seidell JC, Bakker A (1999) Cardiovascular diseases and risk factors in a population-based study in the Netherlands: agreement between questionnaire information and medical records. Neth J Med 55:177–183
Haapanen N, Miilunpalo S, Pasanen M, Oja P, Vuori I (1997) Agreement between questionnaire data and medical records of chronic diseases in middle-aged and elderly Finnish men and women . Am J Epidemiol (15)145:762–769
Sowers MR, Clark MK, Jannausch ML, Wallace RB (1993) Body size, estrogen use and thiazide diuretic use affect 5-year radial bone loss in postmenopausal women. Osteoporos Int 3(6):314–321
Reid IR, Ames RW, Orr-Walker BJ, Clearwater JM, Horne AM, Evans MC, Murray MA, McNeil AR, Gamble GD (2000) Hydrochlorothiazide reduces loss of cortical bone in normal postmenopausal women: a randomized controlled trial. Am J Med 109(5):362–70
Le Couteur DG, Fisher AA, Davis MW, McLean AJ (2003) Postprandial systolic blood pressure responses of older people in residential care: Association with risk of falling. Gerontology 49:260–264
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huopio, J., Honkanen, R., Jurvelin, J. et al. Role of chronic health disorders in perimenopausal fractures. Osteoporos Int 16, 1404–1411 (2005). https://doi.org/10.1007/s00198-005-1851-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-005-1851-2