Abstract
High physical activity level has been associated with high bone mass and low fracture risk and is therefore recommended to reduce fractures in old age. The aim of this study was to estimate the effect of potentially modifiable variables, such as physical activity, muscle strength, muscle mass and weight, on bone mass in elderly women. The influence of isometric thigh muscle strength, self-estimated activity level, body composition and weight on bone mineral density (dual energy X-ray absorptiometry; DXA) in total body, hip and spine was investigated. Subjects were 1004 women, all 75 years old, taking part in the Malmö Osteoporosis Prospective Risk Assessment (OPRA) study. Physical activity and muscle strength accounted for 1–6% of the variability in bone mass, whereas weight, and its closely associated variables lean mass and fat mass, to a much greater extent explained the bone mass variability. We found current body weight to be the variable with the most substantial influence on the total variability in bone mass (15–32% depending on skeletal site) in a forward stepwise regression model. Our findings suggest that in elderly women, the major fracture-preventive effect of physical activity is unlikely to be mediated through increased bone mass. Retaining or even increasing body weight is likely to be beneficial to the skeleton, but an excess body weight increase may have negative effects on health. Nevertheless, training in elderly women may have advantages by improving balance, co-ordination and mobility and therefore decreasing the risk of fractures.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Physical activity is recommended to increase bone mass based on retrospective and prospective studies [1,2,3,4,5,6]. Randomized controlled trials of the effect of physical activity on the clinically relevant end-point, fracture, require several years, or maybe decades of follow-up and seem very difficult or impossible to do. Available retrospective and cross-sectional data that exists of lifetime physical activity on fracture rate in old age are contradictory [2,3,7,8]. How much of the variability in bone mass is influenced by the potentially modifiable variables physical activity and muscle strength is not well studied in elderly women. Studies often rely on subjective activity scores rather than an objective assessment of muscle strength. Another potentially modifiable variable, body weight, and its closely related variables lean and fat mass, have well-known effects on bone mass [9,10,11,12,13].
The objective of this study was to determine the effect of potentially modifiable variables, such as physical activity, muscle strength, muscle mass, fat mass, and body weight, on bone mass in a cross-sectional sample of ordinary, elderly women. The study design could give an indication of the effect of increased physical activity, muscle strength and weight change on bone mass in old age.
Materials and methods
Subjects
The Malmö Osteoporosis Prospective Risk Assessment (OPRA) study consists of 1044 women, all 75 years of age, randomly selected from the population files of the city of Malmö, Sweden. Inclusions were made during 1995–1999 by sending out letters to 1604 women one week after their 75th birthday. This sample represents 33% of all women in this age group living in the city during the study period. The reasons for non-participation have been described in detail elsewhere [14]. This study consists of the 1004 women who attended our research department and excludes 40 women examined at home, since muscle strength and bone mineral density (BMD) were not measured in these women. Informed consent was obtained for all subjects in the study, which also was approved by the local ethics committee.
Methods
Voluntary maximal, isometric muscle strength of the right knee (knee extension at 90° and knee flexion at 45°) was measured using a Biodex computerised dynamometer. Three trials for each direction, each trial lasting 5 s, were performed and the best attempt was recorded as work in Newton meter seconds (Nms). Two physiotherapists measured all participants. Sixty women could not manage the muscle strength test due to disability or unwillingness.
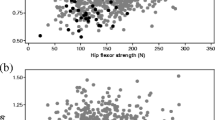
A self-assessment questionnaire modified after Jonsson [15] was used to estimate the present activity level from "bed rest only, cannot walk" to "still working, no limitation of mobility" (Fig. 1), as presented earlier [16].
. a, b Association between muscle strength (knee extension) and self-reported activity level (P<0.001, ANOVA). No significant association between activity level and bone mineral density (BMD) at any site was found (P=0.12–0.33, ANOVA) (n=number of women in each activity level). None of the subjects reported in this study were in activity level 1. None of the three subjects in activity level 2 could manage knee extension. Activity levels: 1) bed rest only, cannot walk, 2) walking possible with human support only, 3) walking with walking aid, only indoors, 4) walking with walking aid, in- and outdoors, 5) no walking aid, walks independently without limitation, 6) take frequent or regular walks, 7) participates in physical activity more strenuous than walking only, 8) still working, no limitation of mobility
A total of 995 women had their BMD assessed in at least one site with Lunar DPX-L-equipment (Lunar, Madison, Wisc., USA). Nine women could not be measured at all with DXA due to technical errors, high body weight, disability not allowing supine position for the time required for measuring or prior surgery interfering with the measurement. Assessment of total body, lumbar spine (L2–L4) and hip (femoral neck and trochanter) BMD was performed in 931, 974 and 951 women, respectively. From the total body scan mode, lean mass and fat mass were calculated. DXA scans were made with software version 1.33 (774 women) or 1.35 (221 women). The mean bone mass did not differ between women scanned with either one of the two scan software versions (data not shown). The precision of our equipment was 0.5% for total body, 1.2% at the spine, 3.9% at the femoral neck, 5.0% at the trochanter, 2.9% for fat mass and 1.4% for lean mass. Precision was calculated from duplicate measurements after repositioning in 15 female volunteers, all 80 years old. All bone mass measurements were made with the same densitometer and performed by the same two technicians during the whole study period. Analysis of the scans was made without knowledge of the results from the muscle strength and physical activity assessments.
Body weight and height were measured in a standardised way. Body mass index (BMI; kg/m2) was calculated.
Statistics
Data are presented as mean (±SD) unless otherwise stated. Pearson correlation was used for parametric variables and Spearman rank correlation for the non-parametric variable physical activity. Forward stepwise multiple regression was used to study the influence of the different variables, including physical activity, on BMD. Analysis of variance (ANOVA) was used to study differences between groups. To compare the effect of a hypothetical increase of the different variables on BMD a forward stepwise multiple regression was used. The beta-value achieved in this model shows the change of the dependent variable BMD with each change of the independent variable, which here was set to 1 SD to allow comparison between different variables. The significance level was set to P<0.05.
Results
All women were 75 years old (75.2±0.1; age range 75.01–75.99). Mean values was for body weight 67.7 (±11.5) kg, height 1.61 (±0.06) m, BMI 26.3 (±4.2) kg/m2, fat mass 26.1 (±7.9) kg and lean mass 37.2 (±3.9) kg. The muscle strength was for knee extension and knee flexion 268 (±80) and 130 (±44) Nms, respectively. The mean self-estimated activity was level 6 (±1), which represents "take frequent or regular walks" (Fig. 1). Bone mineral density values were 1.01 (±0.10) g/cm2 for total body, 0.75 (±0.13) g/cm2 at the femoral neck, 0.70 (±0.14) g/cm2 at the trochanter, and 0.99 (±0.19) g/cm2 at the spine. Mean T-score was –1.9 at the femoral neck, −0.8 at the trochanter and –1.7 at the spine.
There was a significant difference in muscle strength between women in the different physical activity categories (P<0.001, ANOVA, Fig. 1a). Muscle strength, lean mass, fat mass, body weight and height were all correlated to each other (Table 1). A high physical activity level was correlated to low fat mass and body weight, but not to lean mass and body height (Table 1). The highest correlations were seen between weight, lean and fat mass. The relationship between BMD and all other variables in the Pearson correlation are shown in Table 2. Muscle strength correlated to BMD (r-values 0.11–0.24, P<0.01). Physical activity did not correlate to BMD (Table 2, Fig. 1b). The 60 women who could not manage the muscle strength test due to disability or unwillingness did not differ in bone mass results compared to all other women (P=0.13 to P=0.43). The correlations between BMD and body weight, lean mass and fat mass were higher than the correlation with muscle strength (Table 2).
Using forward stepwise multiple regression, the variable with the most pronounced effect on BMD was determined. All variables together explained 16–34% (r 2) of the total variation in BMD depending on site. Physical activity or muscle strength explained 1% or less of the BMD variability in this model. Body weight explained almost all the variability in BMD (15–32%) (Table 3).
The effect on BMD by the independent variables was compared by using a hypothetical 1 SD change of the independent variables. The largest change of BMD was seen for 1 SD (11.5 kg) change in weight (0.058, 0.065, 0.076 and 0.079 g/cm2 in total body, femoral neck, trochanter and spine BMD, respectively, P<0.001), corresponding to a T-score change of 0.54–0.69. The BMD change for 1 SD change in fat mass was between 0.055 and 0.071 g/cm2, for lean mass between 0.035 and 0.047 g/cm2, for knee extension strength between 0.018 and 0.021 g/cm2 and for knee flexion strength between 0.022 and 0.034 g/cm2, all P<0.001. For physical activity a 1 SD change in the physical activity score did not significantly change BMD.
When body weight and height were exchanged to BMI in the forward stepwise multiple regression analysis, BMI, lean or fat mass were the variables with the most pronounced effect (18–29%) on BMD, and muscle strength or physical activity explained 2% or less of BMD.
The multiple regression model was also tested for co-linearity. In subjects with similar body weights, no other variables had a significant effect on bone mass.
Discussion
When comparing the effect on BMD from potentially modifiable variables, such as muscle strength, physical activity, body weight, muscle and fat mass in this population-based sample of elderly women, we found that muscle strength and physical activity only explained a small proportion of the variation in BMD.
All participants were women and of identical age, 75 years, at the time of the investigation. This has the advantage of allowing us to disregard the influence of gender and age on bone mass. In addition, all women lived in the same city and were all of Caucasian background, reducing environmental and ethnical differences. The research staff and the equipment remained the same throughout the entire inclusion period. To our knowledge, no large population-based studies exist concerning the relationship between subjectively measured physical activity, objectively measured isometric muscle strength, lean mass and bone mass in elderly women.
There is no universally accepted measurement of physical activity, and different studies use different criteria for its determination. In this study, current physical activity was assessed using an 8-level scale. The range of the score probably includes the level of activity for most elderly women and was normally distributed. Together with other questions, it has been used to assess differences in physical activity, fracture pattern and bone mass in an urban and a rural population [15]. Physical activity in the present study was closely linked to isometric muscle strength, but muscle strength had a stronger correlation to bone mass.
We chose to measure isometric muscle strength instead of isokinetic muscle strength. Our intention was to obtain a maximum value of knee extension and flexion strength. Functional differences in an elderly population can influence the results of isokinetic tests. Isometric tests are less dependent on range of joint motion, prolonged reaction time, cognitive impairment or knee pain than isokinetic tests.
Muscle strength could be seen as a functional description of the lean mass acquired in the DXA measurement. The effect of muscle strength and physical activity on bone mass, when included in the multiple regression analysis, was small or non-significant (Tables 2 and 3). In prospective studies (up to 5 years) of elderly women, a positive effect on bone mass of high physical activity has been reported, but contradictory results have been reported [5,10,12,17].
In a recent meta-analysis of randomized controlled studies of exercise programs in peri- and post-menopausal women, impact as well as non-impact exercise programs only resulted in about 1% increase in bone mass at the spine. In the hip, impact exercise increased bone mass; however, too few studies existed to draw conclusions about the effect of non-impact activities [6]. Impact-loading activities are not likely to resemble the average normal life of a 75-year-old woman. The small influence of muscle strength and physical activity on BMD in this study raises the question about whether an increase in ordinary activity could significantly increase BMD in the elderly. It is possible that even a small increase in BMD may have a fracture preventive effect. On the other hand, a clearly positive effect of training on muscle strength has been seen in several studies in elderly [8]. Increased physical activity may therefore prevent fractures by preventing falls and limiting the deleterious effects of falling.
The present data indicate a more pronounced effect of body weight on BMD versus any of the other variables included. It is not possible from this cross-sectional study to state definitely that a body weight change will influence BMD. Several earlier studies have shown the importance of body weight on BMD [9,10,11,12,13]. Using BMI instead of weight and height did not substantially alter the relation between muscle strength, physical activity and BMD. There is no need for a statistical calculation to state that body weight is highly correlated with either fat mass or lean body mass. It is therefore questionable whether these variables should be included in the multiple regression analysis. The data should, because of the risk of co-linearity (due to high correlations between closely related variables), be judged accordingly. If, for instance, fat or lean mass had been more important for the variance in bone mineral density, weight would have been of minor importance. The independent effect of lean mass or fat mass on BMD may be related to how bone mass is expressed, for example, bone mineral density or content [18,19]. It has been suggested that fat mass is more important in sedentary women and muscle mass more important in active women for BMD [20].
In this population-based study, including randomly selected subjects and no exclusion criteria applied, the participation rate was as high as 63% (1004/1604). Nevertheless, the women who took part were all ambulant, therefore there is reason to believe that those attending were healthier than those not willing to attend, as supported by the fact that 27% (152/560) of the non-participants reported illness or died shortly after receiving the invitation [14]. Subsequently, it is likely that the non-participant group includes proportionally more women with low activity levels (activity score 1–2). The present data may therefore not be valid in women with very low activity levels.
We conclude that current physical activity, both objectively and subjectively measured, accounts for a minor proportion of the variability in BMD. This leaves limited room for the effect of physical activity as a potential bone mass modifier in elderly women. Retaining or even increasing body weight is likely to be the most important predictor for maintaining bone mass in old age, but an excess body weight increase may have negative effects on health.
References
NOF (2002) NOF physicians´s guide. National Osteoporosis Foundation
Greendale GA, Barrett-Connor E, Edelstein S, Ingles S, Haile R (1995) Lifetime leisure exercise and osteoporosis. The Rancho Bernardo study. Am J Epidemiol 141:951–959
Farahmand BY, Persson PG, Michaelsson K, Baron JA, Alberts A, Moradi T, Ljunghall S (2000) Physical activity and hip fracture: a population-based case-control study. Swedish Hip Fracture Study Group. Int J Epidemiol 29:308–314
Puntila E, Kroger H, Lakka T, Honkanen R, Tuppurainen M (1997) Physical activity in adolescence and bone density in peri- and postmenopausal women: a population-based study. Bone 21:363–367
Puntila E, Kroger H, Lakka T, Tuppurainen M, Jurvelin J, Honkanen R (2001) Leisure-time physical activity and rate of bone loss among peri- and postmenopausal women: a longitudinal study. Bone 29:442–446
Wallace BA, Cumming RG (2000) Systematic review of randomized trials of the effect of exercise on bone mass in pre- and postmenopausal women. Calcif Tissue Int 67:10–18
Karlsson MK, Linden C, Karlsson C, Johnell O, Obrant K, Seeman E (2000) Exercise during growth and bone mineral density and fractures in old age. Lancet 355:469–470
Karlsson M, Bass S, Seeman E (2001) The evidence that exercise during growth or adulthood reduces the risk of fragility fractures is weak. Best Pract Res Clin Rheumatol 15:429–450
Felson DT, Zhang Y, Hannan MT, Anderson JJ (1993) Effects of weight and body mass index on bone mineral density in men and women: the Framingham study. J Bone Miner Res 8:567–573
Hannan MT, Felson DT, Dawson-Hughes B, Tucker KL, Cupples LA, Wilson PW, Kiel DP (2000) Risk factors for longitudinal bone loss in elderly men and women: the Framingham Osteoporosis Study. J Bone Miner Res 15:710–720
Michaelsson K, Bergstrom R, Mallmin H, Holmberg L, Wolk A, Ljunghall S (1996) Screening for osteopenia and osteoporosis: selection by body composition. Osteoporos Int 6:120–126
Nguyen TV, Sambrook PN, Eisman JA (1998) Bone loss, physical activity, and weight change in elderly women: the Dubbo Osteoporosis Epidemiology Study. J Bone Miner Res 13:1458–1467
Compston JE, Bhambhani M, Laskey MA, Murphy S, Khaw KT (1992) Body composition and bone mass in post-menopausal women. Clin Endocrinol (Oxf) 37:426–431
Gerdhem P, Magnusson H, Karlsson MK, Akesson K (2002) Ultrasound of the phalanges is not related to a previous fracture. A comparison between ultrasound of the phalanges, calcaneus, and DXA of the spine and hip in 75-year-old women. J Clin Densitom 5:159–166
Jonsson B (1993) Life style and fracture risk. Department of Orthopaedics, Malmö University Hospital, Lund University, Malmö, Sweden
Gerdhem P, Åkesson K, Obrant KJ (2003) Effect of previous and present physical activity on bone mass in elderly women. Osteoporos Int 14:208–212
Dennison E, Eastell R, Fall CH, Kellingray S, Wood PJ, Cooper C (1999) Determinants of bone loss in elderly men and women: a prospective population-based study. Osteoporos Int 10:384–391
Reid IR, Ames R, Evans MC, Sharpe S, Gamble G, France JT, Lim TM, Cundy TF (1992) Determinants of total body and regional bone mineral density in normal postmenopausal women—a key role for fat mass. J Clin Endocrinol Metab 75:45–51
Khosla S, Atkinson EJ, Riggs BL, Melton LJ 3rd (1996) Relationship between body composition and bone mass in women. J Bone Miner Res 11:857–863
Reid IR, Legge M, Stapleton JP, Evans MC, Grey AB (1995) Regular exercise dissociates fat mass and bone density in premenopausal women. J Clin Endocrinol Metab 80:1764–1768
Acknowledgements
Financial support was obtained from the Swedish Medical Research Council, Trygg-Hansa Research Fund, Malmö University Hospital Research Funds and the Greta and Johan Kock Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gerdhem, P., Ringsberg, K.A.M., Åkesson, K. et al. Influence of muscle strength, physical activity and weight on bone mass in a population-based sample of 1004 elderly women. Osteoporos Int 14, 768–772 (2003). https://doi.org/10.1007/s00198-003-1444-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-003-1444-x