Abstract
Introduction and hypothesis
Enhanced recovery programs (ERPs) are evidence-based protocols designed to improve functional rehabilitation after surgery. ERPs have gained widespread acceptance in many surgical disciplines, and their use leads to significant improvements in patient outcomes while reducing hospital length of stay (LOS). There remains a paucity of data on the use of ERPs in benign gynecologic surgery. The purpose of this review was to evaluate current literature on the use of ERP concepts in benign gynecologic surgery.
Methods
A systematic search of PubMed, CINAHL, Web of Science, and the Cochrane databases was conducted, cross-referencing search terms related to gynecologic surgery and ERP concepts. The search was limited to publications available in English. Studies published prior to 2000, and those involving gynecologic oncology, nonadult patients, and outpatient surgery were excluded.
Results
Nine studies were included in the analysis. Due to heterogeneity of the included studies, no statistical pooling was possible and comparison between studies was limited to their respective themes. Primary study outcomes included LOS, postoperative nausea and vomiting (PONV), pain management, patient satisfaction, and hospital costs. Five studies investigated ERPs, two evaluated measures to improve PONV, and four focused on unique aspects of pain management. Across the studies, ERPs that focused on the patients’ basic symptoms and recovery were found to have equal, if not better, outcomes than standard practice.
Conclusions
This integrative review supports the implementation of ERPs in benign gynecologic surgery. The results showed that the use of ERPs decreased LOS, improved pain scores, and reduced hospital costs, without increasing perioperative complications. We suggest additional randomized controlled trials of ERP concepts in benign gynecologic surgery to support their more widespread use and application.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the past several decades, advances in minimally invasive surgical techniques and technology have led to significantly improved patient outcomes and reduced morbidity after gynecologic surgery [1]. Limited data exist to support conventional perioperative interventions such as preoperative caloric restriction, use of bowel preparations, liberal administration of intravenous fluids and opioids, prolonged immobilization, and use of drains and catheters. Kehlet introduced the “enhanced recovery” concept in the 1990s as a comprehensive, multimodal approach to minimizing the effects of surgical trauma by maintaining normal physiology perioperatively and encouraging early mobilization postoperatively [2]. A variety of enhanced recovery programs (ERPs) have since been developed to standardize perioperative care, limit postoperative organ dysfunction, and expedite recovery to baseline functional status and health after surgery.
In both Europe and the USA, ERPs have gained widespread acceptance in many surgical specialties with benefits to both patients and healthcare systems [3, 4]. There is a large body of evidence in the colorectal and gynecologic oncology surgery literature indicating that the widespread adoption of ERP protocols has led to significant reductions in hospital length of stay (LOS), postoperative complications and healthcare costs, and increases in patient satisfaction [5,6,7,8]. Unfortunately, implementation of ERPs in nonmalignant gynecologic surgery has been less rapid, and evidence on patient outcomes and cost-savings is less clear. Creating standardized surgical protocols has been shown to improve patient safety [9]. Thus, ERPs exemplify a multidisciplinary approach to standard work that adheres to best practices. The purpose of this article is two-fold: to review the existing literature evaluating the use of ERPs in benign gynecologic surgery and to encourage an expedited integration of ERP concepts into benign gynecologic surgical programs.
Materials and methods
We conducted an integrative review based on the following:
-
Population: Adult female patients undergoing benign gynecologic surgery.
-
Intervention: Standardized, nontraditional preoperative interventions including prophylactic pain medications, preoperative fasting/hydration (or lack thereof), and preoperative prophylaxis for postoperative nausea and vomiting (PONV).
-
Comparison: Traditional preoperative standard of care.
-
Outcomes: Postoperative recovery time measured in terms of LOS (days or hours). Secondary outcome measures included occurrence of PONV, pain management, patient satisfaction, and hospital costs.
Search strategy
We conducted a systematic search and integrative review of the literature using the PubMed, CINAHL, Web of Science and the Cochrane Library databases. Initial keywords are presented in grid form in Appendix 1. Search terms were tailored to each database as necessary and an example search strategy is presented in Appendix 2. The search was limited to publications available in English. Studies published prior to 2000 were excluded due to a clear gap in substantive literature between the early 1990s and 2000. The database search was assisted by a professional scientific librarian.
Study selection
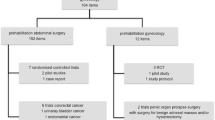
Two members of the research team independently reviewed the titles of the identified articles. Studies were included if they met the eligibility criteria of using a standardized, nontraditional preoperative intervention method. Studies were excluded if they pertained to gynecologic oncology patients, mixed gynecologic and nongynecologic patients, nonadult populations, and surgery performed in the outpatient setting. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Fig. 1) were followed for the selection of studies.
Data extraction
Data were extracted based on research design, outcome measures, and the aspect of ERPs included in the study. Due to the integrative nature of this review, the studies represent a wide range of research methodology and selected outcomes. Not all of the studies included in this review represent each anticipated outcome from the authorship team.
Results
The comprehensive search identified 6,350 articles in PubMed, CINAHL, Web of Science and the Cochrane Library. Upon removal of irrelevant articles and duplicate publications, 41 studies were available for inclusion. After screening by abstract, 31 articles were excluded as they did not meet the inclusion criteria. One additional article was excluded as it was found to be a review. Nine studies met the eligibility criteria and were included in the review (Table 1). Studies were assessed using the appropriate standardized critical appraisal tool from the Joanna Briggs Institute (JBI) based on study design [19].
Length of stay
Decreased LOS has been an important outcome resulting from the implementation of comprehensive and standardized ERPs. In one of the first published studies on ERPs in 2002, 79% of the study population in a Danish university hospital were discharged 24 h postoperatively [17]. Specifically, this study evaluated patients undergoing vaginal hysterectomy and vaginal reconstruction for pelvic organ prolapse after implementation of a fast-track/multimodal rehabilitation model. Prior to implementation of the fast-track protocol, the median LOS was 4 days following vaginal hysterectomy [17]. Similarly, a 2014 case-control study evaluated patients undergoing vaginal hysterectomy before and after implementation of an ERP. ERP implementation led to a 51.6% reduction in the median LOS (22.0 h vs. 45.5 h; p < 0.01) [16]. In addition, in this study the percentage of patients discharged within 24 h was increased fivefold (78% vs. 16%; p < 0.05) [16].
In a descriptive prospective study evaluating the effects of standardized anesthesia and analgesia protocols on LOS, 97% of patients were discharged on postoperative day 1 following laparoscopic hysterectomy for benign indications [14]. Due to the more invasive nature of abdominal surgery, gynecologic ERPs have also sought to decrease LOS following total abdominal hysterectomy for benign indications.
The aim of more recent studies of gynecologic ERPs has been to identify the components of fast-track recovery that would further accelerate recovery and decrease LOS. A large randomized controlled study in 2010 investigated the use of intrathecally administered morphine combined with a low-dose mode of total intravenous anesthesia (TIVA) after abdominal hysterectomy in comparison with patient-controlled analgesia (PCA) combined with general endotracheal anesthesia with volatile anesthetics [18]. Patients with TIVA had a shorter LOS (2 days, range 1–2 days) than patients with PCA (3 days, range 1–6 days; p < 0.001) [18]. To support the evidence that ERP implementation leads to a decrease in LOS, a retrospective case-control study in a university-based hospital compared LOS before and after ERP implementation in women undergoing abdominal hysterectomy. The final analysis of 400 charts showed a significant decrease in the median LOS from 3 days (range 1–12 days) before implementation to 1 day (range 1–17 days) after implementation (p < 0.001) [15]. Because of concerns regarding global changes in medical and surgical care during the study period, the investigators used a hospital of similar size as the control group (no ERPs) and confirmed that LOS at the ‘control’ institution remained unchanged at 3 days during the study period.
These studies and their findings are summarized in Table 2.
Postoperative nausea and vomiting
Prompt and adequate management of PONV is critical in gynecologic surgery because 70% of women undergoing major abdominal and pelvic surgery can experience PONV [20]. While this is a core value in enhanced recovery after surgery (ERAS) programs, only one study included in this review assessed PONV as a primary outcome. ERPs include both prophylactic pharmacologic and nonpharmacologic multimodal methods for the management of PONV. Dexamethasone, a potent corticosteroid, is generally used with other antiemetics. Specifically, dexamethasone is thought to synergistically act with a serotonin receptor antagonist (e.g. ondansetron) to reduce nausea, and also has analgesic benefits. For this reason, most ERPs include dexamethasone in their perioperative protocols.
A double-blind randomized controlled trial in women undergoing vaginal reconstruction investigated the use of dexamethasone (8 mg) given 60 min prior to surgery in comparison with placebo [10]. PONV was assessed in terms of the Postoperative Nausea and Vomiting Intensity score (score greater than 50 indicates intense or clinically important symptoms) and visual analog scales (VAS) for nausea and pain on postoperative day 1. Following surgery, the placebo group required rescue antiemetic (promethazine) and though PONV scores did not differ between the groups, four patients in the placebo group but only two patients in the intervention group had scores greater than 50. PONV VAS scores were lower in those receiving dexamethasone. Interestingly, patients in the placebo group were more likely to fail a voiding trial and to be discharged with a catheter (p = 0.01). This study highlights the importance of dexamethasone in the ERP clinical pathway.
These studies and their findings are summarized in Table 3.
Pain management
Of the studies selected for this review, three focused on the unique aspects of pain management in fast-track pathways. A recent 2016 randomized double-blind placebo-controlled trial evaluated the effectiveness of preemptive local anesthesia before incision in reducing pain as compared to its use at the time of wound closure in patients undergoing elective gynecologic laparoscopic surgery for benign indications [11]. Specifically, the aim of the study was to determine if local anesthesia could reduce movement-evoked pain after laparoscopic gynecologic surgery. A significant reduction in movement-evoked pain 5 h after surgery (p < 0.044) was found [11]. The authors concluded that preemptive local anesthesia could reduce postoperative pain in an alternative nonopioid pathway.
A prospective randomized trial in 92 women undergoing minimally invasive myomectomy evaluated preemptive analgesia and multimodal analgesia in comparison with traditional analgesic protocols (no preemptive or intraoperative medication) [13]. The multimodal analgesia protocol included a combination of dexamethasone 8 mg intravenously and trocar infiltration of local anesthetic (0.26% ropivacaine) prior to skin incision [13]. Ten minutes before completion of surgery, 4 mg ondansetron was administered intravenously and a mixture of 600 mg paracetamol and 75 mg diclofenac was administered intramuscularly. The comparison group received only diclofenac with no multimodal agent. Both groups underwent general anesthesia. Postoperative pain scores at 2 h and 8 h were assessed using a VAS. The multimodal analgesia group had significantly lower VAS scores at 2 h (4.7 vs. 7.1; p < 0.001) and 8 h (2.0 vs. 4.5; p < 0.001) hours after surgery [13]. In addition, earlier return of bowel function and shorter hospitalization was also observed in the intervention group [13]. This specific pathway offers effective nonopioid pain management in surgical patients.
Lastly, a secondary data analysis of a multicenter, open, prospective randomized controlled trial in women undergoing total abdominal hysterectomy for benign indications in five Swedish hospitals where fast-track protocols had been previously implemented compared postoperative symptoms between women receiving general anesthesia or spinal anesthesia with intrathecal morphine (no intubation) [12]. A total of 162 women were included between March 2007 and June 2009, 82 in the spinal anesthesia group and 80 in the general anesthesia group. The primary parent study included an ERP which stipulated intravenous fluid restriction, decreased sedatives, and an emphasis on nonopioid pain medication, early antiemetic therapy, mobilization and early nutrition. Women in the spinal anesthesia group had overall less postoperative discomfort, and there was no difference in symptoms reported between the groups [12]. The study showed significant advantages of spinal anesthesia compared with general anesthesia with regard to postoperative pain and opioid use on postoperative day 0. The authors concluded that spinal anesthesia is beneficial in terms of the incidence and intensity of symptoms.
These studies and their findings are summarized in Table 4.
Hospital cost
Two of the articles included in this review reported data on hospital costs or savings associated with implementation of ERPs. One of these studies evaluated costs before and after ERP implementation as a follow-up from a case-control study of patients undergoing vaginal hysterectomy [16]. Although the analysis did not include start-up costs and the costs of additional hiring needed for program implementation, this study did show a cost saving of 9.25% per patient in the treatment group [16]. The cost savings were largely due to a decrease in LOS from 43.5 h to 22 h [16]. The other study using a similar method showed a reduction in hospital stay from 3 to 2 days that could represent a substantial saving of at least 500 euros [18].
These studies and their findings are summarized in Table 5.
Patient satisfaction
Data on patient satisfaction with ERPs following gynecologic surgery for benign indications are limited and only three of the studies in this review evaluated patient satisfaction. Ottensen et al. evaluated patient satisfaction with an ERP and its acceptability following vaginal surgery for uterine prolapse. Of 41 patients included, 92% found the postoperative hospital stay ‘as expected’, ‘easier than expected’ or ‘much easier than expected’. The median score for ‘how acceptable was the program, treatment and advice’ was 10 (on a scale of 0–10, with 10 indicating the highest acceptability) [17]. In a study of patients undergoing total laparoscopic hysterectomy in a fast-track protocol, 97% reported being satisfied with the procedure and would recommend it to a close relative [14]. Yoong et al. compared patients undergoing vaginal hysterectomy before and after implementation of an ERP. The median patient satisfaction score 4 weeks after surgery on a ten-point VAS was 8, and 65% of the patients in the ERP had a median score of >9. A few patients (7%) reported some dissatisfaction with the protocol with a median score of 1 [16].
Discussion
The current inpatient environment, and the surgical arena in particular, is one of the most complex settings in healthcare. The many clinical and economic headwinds in medicine, such as cost constraints, increasing surgical specialization, and new technologies, contribute to this increasing level of complexity. Academic medical centers face additional complexities with a myriad of learners in various stages of their training and development. The literature supports ERPs as an effective means to address some of these challenges.
While the literature is not replete with robust, prospective evidence of the value of full ERP implementation in benign gynecologic surgery, this integrative literature review supports several aspects of ERP care in comparison with traditional perioperative benign gynecologic practice. The following interventions decrease LOS. and improve patient satisfaction, pain, and nausea/vomiting scores: employing preoperative hydration (rather than traditional fasting and bowel preparations), providing preoperative prophylaxis for PONV (rather than only postoperative antiemetics), and using perioperative multimodal analgesia (rather than only postoperative narcotics and pain medication). While cost–benefit analyses have yet to be performed in this setting, based on other work, decreased LOS translates into decreased hospital costs.
We acknowledge some limitations of this integrative review, including heterogeneous populations, settings, and outcomes, as well as the inclusion of single-institution studies with a consequent lack of generalizability. In addition, benign gynecologic surgery encompasses a much broader scope of practice than other surgical subspecialties for which ERPs have been more readily crafted, adopted and studied (e.g. gynecologic oncology and colorectal surgery). While the gold standard of scientific investigation includes the performance of large, prospective, randomized trials, such trials would be impractical, unwieldy and ultimately impossible to perform across the entire spectrum of benign gynecologic surgery.
Future research efforts could be aimed at evaluating the effects of ERP preoperative patient education/preparedness on patient satisfaction. ERPs may also help mitigate the national epidemic in opioid abuse given the reduction in inpatient narcotic use [21]. National, prospective multicenter trials investigating the value of ERPs could potentially be done in other benign gynecologic surgical subspecialties through their research networks (e.g. Reproductive Endocrinology or Female Pelvic Medicine and Reconstructive Surgery). In addition, the impact of ERPs on provider and nurse satisfaction has yet to be elucidated. Finally, it would be useful to evaluate the benefits of ERP development and implementation in fostering a culture of safety, since multidisciplinary stakeholders are critical to ERP creation and sustainability.
The studies included in this review together cover the basic clinical aspects of the preoperative protocols necessary for fast-track program implementation across various settings. Previous studies have consistently shown that programs incorporating alternative methods of anesthesia with an emphasis on early ambulation, early oral nutrition and alternative pain management have positive outcomes in terms of earlier discharge and patient satisfaction.
Based on the best available evidence, as well as our institutional experience, we strongly advocate broad acceptance and adaptation of ERPs for benign gynecologic surgery, including preoperative hydration, multimodal pain management, and preoperative prophylaxis for PONV. The use of ERPs appears to have a positive (or at least not negative) impact on outcomes including decreased LOS, improved patient satisfaction and pain scores, and decreased hospital costs. In summary, the risk/benefit profile highly favors the use of ERPs in current benign gynecologic surgical practice.
References
American College of Obstetricians and Gynecologists. Choosing the route of hysterectomy for benign disease. Committee Opinion No. 701. Obstet Gynecol. 2017;129:e155–9
Kehlet H. Multimodal approach to control postoperative pathophysiology and rehabilitation. Br J Anaesth. 1997;78:606–17.
Chapman J, Roddy E, Ueda S, Brooks R, Chen L, Chen L. Enhanced recovery pathways for improving outcomes after minimally invasive gynecologic oncology surgery. Obstet Gynecol. 2016;128(1):138–44.
Thiele R, Rea K, Turrentine F, Friel C, Hassinger T, Goudreau B, et al. Standardization of care: impact of an enhanced recovery protocol on length of stay, complications, and direct costs after colorectal surgery. J Am Coll Surg. 2015;220(4):430–43.
de Groot J, Maesen J, Slangen B, Winkens B, Dirksen C, van der Weijden T. A stepped strategy that aims at the nationwide implementation of the Enhanced Recovery After Surgery programme in major gynaecological surgery: study protocol of a cluster randomised controlled trial. Implement Sci. 2015;10:106.
Kalogera E, Bakkum-Gamez J, Jankowski C, Trabuco E, Lovely J, Dhanorker S, et al. Enhanced recovery in gynecologic surgery. Obstet Gynecol. 2013;122(2 Part 1):319–28.
Nelson G, Altman A, Nick A, Meyer L, Ramirez P, Achtari C, et al. Guidelines for pre- and intra-operative care in gynecologic/oncology surgery: Enhanced Recovery After Surgery (ERAS®) Society recommendations – Part I. Gynecol Oncol. 2016;140:313–22.
Pedziwiatr M, Kisialeuski M, Wierdak M, Stanek M, Natkaniec M, Matłok M, et al. Early implementation of Enhanced Recovery After Surgery (ERAS®) protocol – compliance improves outcomes: a prospective cohort study. Int J Surg. 2015;21:75–81.
Raman J, Leveson N, Samost AL, Dobrilovic N, Oldham M, Dekker S, et al. When a checklist is not enough: how to improve them and what else is needed. J Thorac Cardiovasc Surg. 2016;152(2):585–92. doi:10.1016/j.jtcvs.2016.01.022.
Pauls R, Crisp C, Oakley S, Westermann L, Mazloomdoost D, Kleeman S, et al. Effects of dexamethasone on quality of recovery following vaginal surgery: a randomized trial. Am J Obstet Gynecol. 2015;718:1–7.
Ravndal C, Vandrevala T. Preemptive local anesthetic in gynecologic laparoscopy and postoperative movement-evoked pain: a randomized trial. J Minim Invasive Gynecol. 2016;23:775–80.
Wodlin N, Nilsson L, Arestedt K, Kjolhede P. Mode of anesthesia and postoperative symptoms following abdominal hysterectomy in a fast-track setting. Acta Obstet Gynecol Scand. 2011;90:369–79.
Xiromeritis P, Kalogiannidis I, Papadopoulos E, Prapas N, Prapas Y. Improved recovery using multimodal perioperative analgesia in minimally invasive myomectomy: a randomised study. Aust N Z J Obstet Gynaecol. 2011;51:301–6.
de Lapasse C, Rabischong B, Bolandard F, Canis M, Botchorischvili R, Jardon K, et al. Total laparoscopic hysterectomy and early discharge: satisfaction and feasibility study. J Minim Invasive Gynecol. 2008;15:20–5.
Dickson E, Argenta P, Reichert J. Results of introducing a rapid recovery program from total abdominal hysterectomy. Gynecol Obstet Invest. 2012;73:21–5. doi:10.1159/000328713.
Yoong W, Sivashanmugarajan V, Relph S, Bell A, Fajemirokun E, Davies T, et al. Can enhanced recovery pathways improve outcomes of vaginal hysterectomy? Cohort control study. J Minim Invasive Gynecol. 2014;21(1):83–9.
Ottesen M, Sorensen M, Rasmussen Y, Smidt-Jensen S, Kehlet H, Ottesen B. Fast track vaginal surgery. Acta Obstet Gynecol Scand. 2002;81:138–46.
Kroon U, Radstromb M, Hjelthea C, Dahlinc C, Kroon L. Fast-track hysterectomy: a randomised, controlled study. Eur J Obstet Gynecol Reprod Biol. 2010;151:203–7.
The Joanna Briggs Institute. Reviewers’ Manual, 2014 edition. Adelaide: Joanna Briggs Institute, University of Adelaide; 2014.
Chatterjee S, Rudra A, Sengupta S. Current concepts in the Management of Postoperative Nausea and Vomiting. Anesthesiol Res Pract. 2011;2011:748031. doi:10.1155/2011/748031.
Smith G, Durieux M, Bhiken N. Trends of intraoperative opioid and non-opioid analgesic use at an academic tertiary care hospital over a four-year period. Poster session presented at the PostGraduate Assembly in Anesthesiology (PGA70), 2016, New York, NY.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Appendices
Appendix 1
Appendix 2: Example search – PubMed
(“Gynecologic Surgical Procedures”[MeSH] OR “Gynecologic Surgeries”[All Fields] OR “gynecologic surgery”[All Fields] OR “Gynaecologic surgical procedures”[All Fields] OR “hysterectomy”[MeSH] OR “hysterectomy”[All Fields] OR “uterine myomectomy”[MeSH Terms] OR “uterine myomectomy”[All Fields] OR “myomectomy”[All Fields] OR “sterilization reversal”[All Fields] OR “sterilization reversal”[MeSH Terms] OR (“sterilization”[All Fields] AND “reversal”[All Fields]) OR “sterilization reversal”[All Fields] OR “ovariectomy”[MeSH Terms] OR “ovariectomy”[All Fields] OR “oophorectomy”[All Fields] OR sacrocolpopexy[All Fields] OR “vesicovaginal fistula”[MeSH Terms] OR “vesicovaginal”[All Fields] OR “menorrhagia”[MeSH Terms] OR “menorrhagia”[All Fields] OR “metrorrhagia”[MeSH Terms] OR “metrorrhagia”[All Fields] OR “leiomyoma”[MeSH Terms] OR “leiomyoma”[All Fields] OR “fibroid”[All Fields] OR fibroid’s[All Fields] OR “endometriosis”[MeSH Terms] OR “endometriosis”[All Fields] OR “adenomyosis”[MeSH Terms] OR “adenomyosis”[All Fields] OR ((“uterus”[MeSH Terms] OR “uterus”[All Fields] OR “vagina”[MeSH Terms] OR “vagina”[All Fields] OR “vaginal”[All Fields]) AND (“prolapse”[MeSH Terms] OR “prolapse”[All Fields])) OR ((“pelvis”[MeSH Terms] OR “pelvis”[All Fields] OR “pelvic”[All Fields]) AND (“pain”[MeSH Terms] OR “pain”[All Fields])) OR ((“ovary”[MeSH Terms] OR “ovary”[All Fields] OR ovarian[All Fields]) AND (“cysts”[MeSH Terms] OR “cysts”[All Fields])) OR ((tubal[All Fields] OR tubular[All Fields] OR tube[All Fields]) AND reanastomosis[All Fields]) OR ((“Gynecology”[Mesh] OR “gynecology”[All Fields] OR “gynecology”[All Fields] OR gynecologic[All Fields] OR “gynecological”[All Fields] OR gynaecologic[All Fields] OR gynecological[All Fields]) AND (“disease”[MeSH Terms] OR “disease”[All Fields] OR “diseases”[All Fields] OR “disorder”[All Fields] OR “disorders”[All Fields] AND benign[All Fields]))) AND ((ERAS[tiab] OR “enhanced recovery after surgery”[All Fields] OR “enhanced recovery”[All Fields]) OR ((enhanced[All Fields] OR early[All Fields] OR accelerated[All Fields] OR rapid[All Fields] OR “fast track”[All Fields] OR “fast-track”[All Fields]) AND (recovery[All Fields] OR recovers[All Fields] OR recovering[All Fields] OR “rehabilitation”[MeSH Terms] OR “rehabilitation”[All Fields] OR “convalescence”[MeSH Terms] OR “convalescence”[All Fields] OR “convalesced”[All Fields] OR “convalescent”[All Fields] OR “convalescents”[All Fields] OR “mobile”[All Fields] OR “mobilization”[All Fields] OR “ambulate”[All Fields] OR “ambulation”[All Fields] OR “ambulatory”[All Fields] OR “ambulating”[All Fields] OR “walking”[MeSH Terms] OR “walking”[All Fields] OR “walk”[All Fields] OR “walks”[All Fields] OR “feeding behavior”[MeSH Terms] OR “feeding”[All Fields] OR “feed”[All Fields] OR “feeds”[All Fields] OR “nutrition”[All Fields] OR “eat”[All Fields] OR “eats”[All Fields] OR “eating”[All Fields])) AND ((((“surgery”[Subheading] OR “surgery”[All Fields] OR “surgical procedures, operative”[MeSH Terms]) OR protocol[All Fields]) OR pathway[All Fields] OR “Critical Pathways”[MeSH] OR “clinical protocols”[MeSH]) OR ((((“perioperative care”[MeSH Terms] OR “Perioperative Care”[All Fields]) OR “preoperative care”[MeSH Terms]) OR “Preoperative Care”[All Fields]) OR “Length of Stay”[MeSH] OR “Length of Stay”[All Fields]))) AND English[lang].
Rights and permissions
About this article
Cite this article
Trowbridge, E.R., Dreisbach, C.N., Sarosiek, B.M. et al. Review of enhanced recovery programs in benign gynecologic surgery. Int Urogynecol J 29, 3–11 (2018). https://doi.org/10.1007/s00192-017-3442-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-017-3442-0