Abstract
Clinical evaluation and management of anterior cruciate ligament (ACL) injury is one of the most widely researched topics in orthopedic sports medicine, giving providers ample data on which to base their practices. The ACL is also the most commonly treated knee ligament. This study reports on current topics and research in clinical management of ACL injury, starting with evaluation, operative versus nonoperative management, and considerations in unique populations. Discussion of graft selection and associated procedures follows. Areas of uncertainty, rehabilitation, and prevention are the final topics before a reflection on the current state of ACL research and clinical management of ACL injury.
Level of evidence V.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Management of anterior cruciate ligament (ACL) injury has evolved significantly in the past several decades. Prior fundamental questions, such as anatomic versus nonanatomic reconstruction, anteromedial versus transtibial drilling, and use of allografts in young patients, have largely been answered. Nevertheless, countless other questions have been investigated over the past several years. This study seeks to summarize the recent literature on the clinical care of ACL injury and highlight topics of particular interest.
Evaluation
Physical examination
Physical examination is an essential element in the setting of suspected ACL injury and involves the anterior drawer, Lachman, and pivot-shift tests (Fig. 1). The anterior drawer test has been shown to have poor sensitivity (49%) and specificity (58%) in the acute setting [16]. The anterior drawer test performs substantially better in the chronic setting and when secondary restraints are concomitantly injured [16, 145]. The Lachman test has much higher sensitivity (86%) and specificity (91%) [145]. The Lachman test only assesses the anteromedial bundle of the ACL and does not evaluate rotatory knee laxity [48]. The pivot-shift test is useful to assess rotatory knee laxity by applying a valgus stress and axial load while internally rotating the tibia. Because it tests lateral compartment rotatory laxity related to ACL insufficiency, the pivot shift is able to assess the posterolateral bundle of the ACL more directly than both the anterior drawer and Lachman tests [62]. The pivot shift demonstrates very high specificity (98%), but low sensitivity (32%) [16]. Because of the relative complexity of the pivot-shift test, examiner experience and technique play a major role in testing accuracy [121, 156]. Computer tablets recording the pivot-shift test have been shown to be reproducible and valid in quantifying tibial translation and thus the degree of rotatory knee laxity [82, 83].
Assessment of ACL injury with the Lachman and pivot-shift tests. A The Lachman test evaluates the amount of anterior tibial translation and firmness of the endpoint relative to the contralateral side, with increased translation and a soft endpoint in a positive test. B The pivot-shift test involves the examiner providing a valgus (1) and internal rotation (2) force with the proximal hand while axially loading the ankle with the distal hand (3) to flex the knee. C In a positive test, as the knee flexes to roughly 30 degrees, the anteriorly subluxated tibia reduces with a visible glide or clunk posteriorly (4)
Imaging
Magnetic Resonance Imaging (MRI) can confirm equivocal clinical diagnoses, identify associated injuries, provide information about prognosis via detection of intra-articular damage, and assist during preoperative planning. The presence and location of a bone bruise can provide insight into the injury mechanism and associated injuries [39]. For example, a lateral bone bruise has been associated with a higher prevalence of lateral meniscal tears [39, 176, 190]. A medial bone bruise is associated with medial meniscus tears, with medial meniscus injury present in 66% of a series of ACL injuries with both lateral and medial bone contusions on MRI [18, 190]. Bone bruises have been shown to be associated with increased risk of chondral damage and subsequent development of osteoarthritis (OA) [59, 166]. At 12-month follow-up after ACL reconstruction (ACL-R), MRI has been shown to detect cartilage alterations including fibrillation, thinning, and areas of cartilage loss overlying areas of bone bruising seen on preoperative MRI [166]. Imaging findings of cartilage destruction have been supported by histological data and joint fluid analysis that suggest chondral damage [170].
Beyond identification of intra-articular pathology, MRI allows the surgeon to estimate autograft size, ACL insertion site size, and notch width [8]. MRI evaluation of the size of the patellar tendon, quadriceps tendon, and hamstring tendon is reliable with moderate-to-good accuracy when compared with intraoperative graft measurement [162, 192]. The size of the ACL insertion site on the tibia, as well as the femur, can be useful if the surgeon considers double bundle ACL-R in the setting of a large (> 18 mm) insertion site to better replicate the insertion [67, 146]. Notch width can also factor into graft size, with narrower notches better accommodating a smaller graft [174].
Table 1 summarizes the key facts on evaluation of ACL injury.
Consideration of operative or nonoperative management
ACL-R is generally the preferred treatment for restoration of knee stability in young and active patients, particularly those hoping to return to sports that require pivoting or change of direction [122]. In older and less active patients, and/or those participating in linear activities alone, nonoperative management may be more strongly considered [48]. Evaluating the data on the merits of operative or nonoperative management requires evaluation of objective knee laxity, subsequent injuries, and information about patient-reported outcomes (PROs).
The most consistent data on the topic are related to the evaluation of objective knee laxity. There is substantial evidence that ACL-R significantly decreases anterior tibial translation and pivot shift [60, 114, 153, 172]. These findings are the basis for concerns about possible subsequent injuries and worse outcomes related to nonoperative management.
Without the primary restraint of the ACL to anteroposterior translation and rotation, increased load is placed on the secondary stabilizers to these motions, often leading to injury. This is demonstrated in a recent systematic review of long-term outcomes of nonoperative and operative management of over 2200 ACL injuries, showing significantly higher rates of subsequent meniscus surgery in the nonoperative group (29.4 vs 13.9%) [29]. This in part contributed to significantly higher rates of repeat knee surgery in the nonoperative group (24.9 vs 12.4%). Similarly, a database study of nearly 1400 ACL injuries found that long-term (mean 8–14 year follow-up) rates of secondary meniscal tears were 7% in the acute ACL-R group, 19% in the nonoperative group, and 33% in the delayed ACL-R group, with significant differences between the groups [69]. On the other hand, a Level 1 study of 121 patients at 5-year follow-up showed no difference in subsequent meniscus surgery between operative and nonoperative groups, though there was significant crossover from the nonoperative to operative group [60].
Progression of OA is an important indicator of the success of operative and nonoperative management. Many studies have found no differences in terms of short-to-long-term rates of OA [29, 69, 114, 172, 181]. Increased rates of OA were found in a systematic review of 1,397 patients in studies with greater than 10-year follow-up (OR 1.56), which was not seen in studies with shorter follow-up, i.e., 5–10 years [153]. A study of nonoperatively-managed ACL-injured patients at 20-year follow-up showed arthritic changes in 82.4% [64]. Comparative high-level long-term studies are needed to evaluate the risk of OA with nonoperative versus operative management of ACL injuries.
In terms of patient outcomes, the literature is mixed. The first randomized controlled trial on nonoperative versus operative management of ACL tears showed no difference in KOOS, SF-36, or Tegner scores among 121 ACL-injured patients at 5 years [60]. The study was limited by 51% of the nonoperative cohort crossing over into the operative cohort, but their “as-treated analysis” showed the same findings. A smaller randomized study with 10-year follow-up found significantly higher IKDC Subjective Knee Form (SKF) Scores in the operative group (86.6 vs 77.5) [172]. A Level 2 study of 105 combined operative and nonoperative ACL-injured patients showed that at 5 years, there were no differences in performance in single-leg hop tests, return to preinjury activity level, IKDC SKF, and most KOOS subscales [180].
Screening tests to identify potential copers (able to return to sport without ACL-R) incorporate objective knee function testing, PROs, and episodes of functional instability [86]. Outcomes of these tests are mixed and suggest that a minority of ACL-injured patients are able to return to cutting and pivoting sports without ACL-R [131]. With nonoperative treatment of the torn ACL, most patients will become ‘adapters’ (willing to reduce activity levels to prevent instability) and ‘non-copers’ (unable to avoid instability or return to prior level without ACL-R) [119].
Amidst at times conflicting data, an international group of experts on ACL management evaluated the literature and published consensus statements on the management of ACL injuries in 2019 (Fig. 2) [48]. Notably, there was unanimous agreement in the following: operative and nonoperative treatments are both acceptable treatment options; in patients wishing to return to jumping/cutting/pivoting sports, operative treatment is preferred; and in patients wishing to return to straight plane activities, nonoperative treatment is an option, however it is less appropriate if the patient experiences recurrent functional instability in daily life.

Reproduced from: Treatment after anterior cruciate ligament injury: Panther Symposium ACL Treatment Consensus Group. Diermeier T, et al. Knee Surgery, Sports Traumatology, Arthroscopy 28(8):2390–2402©2020 with permission from BMJ Publishing Group Ltd
Consensus statements of the Panther Symposium on nonoperative and operative management of ACL injury.
In the pediatric population, nonoperative management shows consistently inferior outcomes with significantly higher recurrent functional instability (75 vs 14%), higher subsequent medial meniscus tears (35 vs 4%), and lower rates of return to previous levels of sport (0 vs 86%) [140]. A more recent systematic review confirmed widely disparate return to play rates [22].
Table 2 summarizes the key facts on considerations of operative versus nonoperative management of ACL injury.
Considerations in unique populations
Literature on ACL-R outcomes largely focuses on patients from skeletal maturity up to middle age. However, increased appreciation of the importance of the ACL has led to more research on ACL-R in younger and older populations that may also benefit from the procedure. Additionally, as female participation in sports continues to increase, there has been a strong push to evaluate the role of sex on outcomes after ACL-R. These three subsets of ACL-R patient populations warrant focused discussion.
The prevailing dogma of the twentieth century was that pediatric ACL rupture was an uncommon injury, owing mainly to pediatric skeletal immaturity and generalized joint laxity [42]. However, pediatric competitive athletics are increasing, and in turn, the incidence of ACL-Rs in patients aged 3–20 years increased from 17.6 to 50.9 per 100,000 over the past 20 years [50]. This paradigm shift in the management of pediatric ACL injuries has been substantiated by studies demonstrating that opting for nonoperative or delayed management of pediatric ACL injury is associated with subsequent meniscal and chondral injuries [7, 52, 103], worse PROs, increased rates of knee laxity and concomitant ligamentous injuries, and lower rates of return to preinjury activity level [53].
It is now widely accepted that early autograft reconstruction is the standard of care in pediatric and adolescent ACL injuries, with allografts having up to three times the failure rate as autografts [55, 140]. The techniques for creating the tunnels for pediatric ACL-R are more widely debated [135]. There are three commonly accepted techniques: physeal-sparing reconstruction with the tunnel(s) avoiding the physis of both the tibia and femur, partial transphyseal tunnels crossing the tibial physis but not the femoral physis, and transphyseal reconstruction with tunnels through both the tibial and femoral physes [135]. To date, clinical and biomechanical studies comparing these techniques are scarce, equivocal, and lack the power to guide clinical practice [135]. Additionally, there are no known randomized clinical trials comparing these techniques, likely due to the ethical dilemmas surrounding randomization of the pediatric population to surgical techniques. However, given the increased incidence of these injuries in the setting of increased pediatric participation in sports, it is expected that this topic will be at the forefront of pediatric ACL-R research in the coming years. Collaboration via multi-center studies and registries is of great benefit to pediatric ACL-R research by allowing for increased sample sizes and thus power.
The middle-aged population is another unique demographic group for ACL-R. Prior literature set a threshold of 40 years of age for an “older” population [14, 20, 34, 100]. With physical activity becoming more important to the middle-aged population, recent studies have pushed the ‘older’ threshold to 50 years of age and found that patients in this age group benefit biomechanically, clinically, and functionally after ACL-R [19, 34, 38, 175]. A recent systematic review evaluating 16 studies of 470 ACL-Rs in patients aged 50–75 found that surgical intervention significantly improved clinical and functional outcomes in all of the reviewed studies, concluding that increased age is not a contraindication to ACL-R [36]. These findings have expanded the indications for ACL-R and have opened the door for larger scale clinical studies that will continue to shape indications and techniques to optimize ACL-R in a population that, two decades ago, was unlikely to have been offered surgery.
Sex has also become an increasingly studied factor in ACL injury management over the past two decades, particularly in terms of rehabilitation and prognosis. It is widely accepted that females are at an increased risk for ACL injury [3, 77, 138]. Recent studies have demonstrated that females have inferior outcomes after ACL-R, with a recent systematic review and meta-analysis evaluating 135 publications and over 120,000 patients showing inferior PROs in females [163]. Males have greater strength recovery of the hamstrings and quadriceps after ACL-R [65, 95, 195]. One study evaluating 320 patients who underwent primary ACL-R with soft tissue quadriceps tendon found that female patients had greater deficits in quadriceps strength and extension range of motion up to six months after surgery compared with males [85], while another found significantly greater hamstring strength deficits in females after ACL-R with a hamstring tendon graft up to one year postoperatively [40]. Lastly, females have consistently been shown to have lower rates of return to sport relative to their male counterparts after ACL-R [11, 163]. There may be a higher rate of “psychological readiness” that allows males to return to their preinjury level of play earlier than females after ACL-R [179].
Table 3 summarizes the key facts on ACL injury management considerations in unique populations.
Graft selection
ACL graft selection must consider not only the biological and biomechanical properties of the graft itself, but also the clinical demands, characteristics, and expectations of the patient.
Bone-patellar tendon-bone (BTB) autograft
The most commonly utilized ACL-R graft historically was the BTB autograft [88]. A key advantage is more robust and rapid initial fixation compared with soft tissue grafts due to the retention of a native tendon-bone interface [129]. Patellar tendon grafts create a pure bony interface that has been demonstrated to be stronger than the fibrovascular scarring after soft tissue-to-bone healing [182]. Static laxity testing (instrumented laxity testing, Lachman, pivot shift) has consistently been superior in BTB compared with hamstring grafts [115, 116]. In a recent large registry study, risk of revision for graft rupture was twice as high among patients treated with hamstring compared with BTB [134]. PROs, patient satisfaction, and time to return to sport are similar between BTB and hamstring [81, 107, 110, 115, 116, 168]. Disadvantages with BTB autograft include graft-tunnel mismatch, donor-site morbidity with anterior knee pain (up to 32%) [188], and patella fracture (rare) [127, 161].
Quadriceps autograft
Quadriceps tendon autograft with or without bone block has recently gained popularity [152]. Comparing all soft tissue versus bone block, there is no difference in graft rupture, similar PROs, and less rotatory laxity with all soft tissue quadriceps autograft [37]. Quadriceps autograft has performed well compared with other graft options in terms of laxity (instrumented laxity testing, Lachman, pivot shift), range of motion, PROs, and overall patient satisfaction [152]. There is less donor-site morbidity, decreased rate of anterior knee pain, and consistently larger graft cross-sectional area compared with BTB [70, 96]. Comparing quadriceps autograft versus hamstring shows superior performance of quadriceps tendon on PROs, restoration of laxity as measured with the KT-1000 knee arthrometer, and higher likelihood of a negative Lachman test [27].
Hamstring autograft
Hamstring autograft is the most frequently used graft choice, though there are some signs of decline in its use [12]. Hamstring autograft has greater ultimate tensile load, stiffness, and cross-sectional area compared with both BTB and the native ACL [113, 182]. Hamstring harvest is faster than BTB and does not disrupt bone but requires tendon-to-bone healing after implantation. Tendon-to-bone healing has demonstrated slower in-growth and lower initial pullout strength compared with BTB [171]. Slower graft healing may be responsible for increased early re-rupture rate after hamstring autograft ACL-R compared with BTB [111, 134, 139], though there is high-level data that they have similar failure rates overall [33]. An increased early re-rupture rate with hamstring autograft compared with BTB is especially concerning for higher risk patients, particularly young women and those that play sports requiring cutting and pivoting [144].
A large study from the Swedish National Knee Ligament Registry demonstrated that for every 0.5 mm increase in hamstring graft diameter, the risk of revision surgery decreased by 0.86 times [155]. Grafts 8.5 mm or greater in diameter have significantly lower risk of revision than grafts less than 8 mm [154]. The risk associated with the use of smaller diameter hamstring grafts is of particular concern in patients younger than 20 years old [35]. Hamstring weakness after graft harvest can be present even at 5 year follow-up after ACL-R [102]. For this reason, some researchers recommend avoiding hamstring autograft in high-level athletes [2]. Tunnel widening is seen with use of hamstring autografts at a higher rate than after reconstruction with BTB autograft [58, 113], however, no long-term differences are found in terms of PROs or risk of OA [107, 168].
Allograft
Allograft is a relatively common graft choice, with use in 30% of primary ACL-Rs in the Multicenter Orthopaedic Outcomes Network (MOON) from 2002 to 2008 [84]. Allograft options for ACL-R include hamstring, quadriceps, BTB, tibialis anterior, tibialis posterior, and Achilles tendons. Among these choices, looped tibialis anterior tendon has shown the highest load to failure, while quadriceps tendon has shown the highest stiffness [5, 101]. There is very limited data on the clinical difference between allografts, with one large registry study showing BTB allograft had a higher rate of revision surgery (HR 1.79) compared with soft tissue allografts [164]. The primary concern with allograft is an increased re-rupture rate in young patients (up to 30%) [55, 89, 125]. This discrepancy narrows with increased patient age until around age 40, when re-rupture rates are similar for autografts and allografts (Fig. 3) [89]. Graft rupture risk is up to 3–5 times higher for BTB allograft compared with BTB autograft [97, 99] and roughly twice as high with allografts overall compared with autografts [91, 194].

Reproduced from: Kaeding CC, et al. Sports Health 3(1):73–81. Allograft versus autograft anterior cruciate ligament reconstruction: predictors of failure from a MOON prospective longitudinal cohort. ©2011 by SAGE Publications
Risk of graft failure in the Multicenter Orthopaedic Outcomes Network (MOON) cohort by age and autograft versus allograft.
A limiting factor of allograft ACL-R is graft processing. There is a dose-dependent relationship between gamma irradiation processing and increased risk of graft failure [47, 148]. Non-irradiating chemical processing techniques have been shown to similarly negatively affect both allograft load to failure and stiffness [101]. There is general consensus that sterilization techniques, particularly irradiation, negatively impact biomechanical properties of allograft and have the potential to lead to lower PROs, failure to restore normal stability, and increased revision rate [130, 169]. The variability in irradiation complicates the literature on autograft versus allograft. A large meta-analysis showed significant differences between autografts and irradiated allografts, but no such differences between autografts and non-irradiated allografts [194]. There is a need for larger scale studies on non-irradiated allografts.
Table 4 summarizes the key facts on graft selection in ACL-R.
Associated procedures
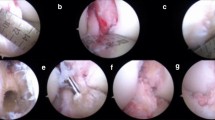
Whether in isolated ACL injuries or ACL tears with concurrent pathology, some surgeons consider associated procedures alongside ACL-R to increase the likelihood of successful ACL-R. The anterolateral complex and reconstruction of its components are the most notable in recent literature. The anterolateral ligament (ALL) has gained extensive traction in the orthopedic sports surgery community in the past several years, though there is evidence questioning if it is a true anatomic structure (Fig. 4) [68, 73, 120]. In recent years, several biomechanical studies investigated the biomechanical properties of the anterolateral complex and its kinematic function in association with ACL injuries [72, 94]. Biomechanical studies have shown that the ALL acts merely as a secondary stabilizer to anterior tibial translation with the knee in flexion and to pivot shift in the ACL-injured knee [160, 165].
Anatomic dissection of the anterolateral complex of the knee. A The superficial iliotibial band (sITB) is reflected, exposing the lateral joint capsule and the ITB insertion on the distal femur (Kaplan fibers, KF). The deep ITB (black arrow demonstrates its course) merges with the sITB. The capsulo-osseous layer of the ITB is also seen (black triangle). B Separation of the deep ITB (black arrow) and capsulo-osseous layer (black triangle) show the convergence of multiple layers of the ITB distally before inserting upon Gerdy’s tubercle (GT). Reproduced by permission from: Springer Nature. Knee Surgery, Sports Traumatology, Arthroscopy. The anterolateral complex of the knee: a pictorial essay. Herbst E, et al. ©2017
ACL-R with combined ALL reconstruction may decrease ACL-R failure rate. Minimum 2-year follow-up of 92 patients who underwent associated ACL-R with hamstring autograft and ALL reconstruction reported good subjective and objective outcomes, 1.1% failure rate, and 8.4% grade 1 pivot shift [159]. In a retrospective cohort study at a mean 9-year follow-up with a propensity matched isolated ACL-R group, a similar patient cohort reported decreased rates of revision ACL-R (3.5% with ALL reconstruction vs 17.4% without) and no differences in clinical outcomes between ACL-R with and without ALL reconstruction [158].
Lateral extra-articular tenodesis (LET) has been used for decades and received increased attention recently [6, 104]. Despite encouraging early postoperative results of isolated LET for ACL injury historically, some authors reported progressive knee laxity, clinical failures, and evidence of OA at longer-term follow-up [6, 141]. Following those reports and the progression of arthroscopic ACL-R, surgeons shifted towards isolated intra-articular ACL-R, and therefore most LET procedures were abandoned for years.
The re-emergence of LET has led to recent data on biomechanical and clinical outcomes of combined ACL-R and LET, with mixed results. LET leads to no difference in rotatory knee laxity or lateral compartment translation when added to ACL-R in vivo at time zero [149], and LET has also been shown to stretch out by one year postoperatively [30]. In a randomized study of BTB autograft ACL-R with and without additional LET, there were no differences in terms of clinical outcomes or failure rate at an average follow-up of 20 years [26]. However, LET had an increased risk of lateral compartment OA (59 vs 22%). In a prospective study of hamstring autograft over-the-top ACL-R with LET with minimum 20 years of follow-up, 86% of patients reported good or excellent clinical outcomes [191]. There was a positive pivot shift in 12% of patients, graft re-rupture in 2%, and increased medial OA compared with the contralateral knee only in patients who underwent concurrent medial meniscectomy.
The multi-center randomized controlled STABILITY trial investigated the effect of the LET (modified Lemaire technique) on a cohort of 618 high risk patients undergoing hamstring autograft ACL-R [63]. At 2-year follow-up, the LET group showed a relative risk reduction of 0.38 in clinical failure and 0.67 in graft rupture when compared with isolated ACL-R. A follow-up multi-center study, the STABILITY 2 Trial, is underway comparing clinical outcomes and ACL-R failure rates in 1,200 patients in four groups—ACL-R with BTB versus quadriceps tendon, with or without a LET (ClinicalTrials.gov Identifier: NCT03935750).
Beyond soft tissue procedures, osteotomies play a role in ACL-R in the setting of bony morphology that predisposes patients to graft failure or osteoarthritis. Increased posterior tibial slope is a risk factor for failure of ACL-R [31, 66]. Slope-reducing tibial osteotomy, or deflexion osteotomy, biomechanically decreases graft forces [87, 189] and has been shown to clinically improve knee stability and PROs in patients with increased posterior tibial slope following revision [4, 41] and more recently even primary ACL-R [157].
Table 5 summarizes the key facts on associated procedures with ACL-R.
Areas of uncertainty
As knee surgeons and researchers continue to learn more about the ACL and outcomes after reconstruction, there has been increased interest in developing new surgical techniques to further restore native function, return patients to preinjury activity, and reduce the incidence of knee OA. Of the wide variety of new techniques, ACL repair and ACL augmentation have received the most interest and scrutiny from the orthopedic community.
ACL repair, initially described in 1903 [143], gained popularity in the 1970’s. However, poor long-term outcomes were reported in the 1980’s and early 1990’s that stifled its use, in part due to simple operative techniques, wide array of clinical indications, and a precursory understanding of ACL healing [57, 92]. Amidst renewed interest in repair, various new ACL repair techniques have demonstrated promising short-term outcomes in tightly selected groups of patients. ACL repair techniques include suture anchor fixation into the femoral footprint [49], independent suture reinforcement [74, 185], dynamic intraligamentary stabilization [54], and bridge-enhanced ACL repair (BEAR) [117]. Each ACL repair technique has been reported in small case series or short-term outcome studies, and the literature has demonstrated mixed results, with reported failure rates from 0 to 60% [1, 80, 118]. A study of suture ligament augmentation, involving repair of femoral-sided ACL avulsions with sufficient length and quality of the remnant ACL with bridging suture tape, found graft failure for ACL repair to be 11 times greater than that of ACL-R (49 vs 5%) in adolescent patients [61]. A recent systematic review demonstrated ACL repair survivorship to be as low as 60%, with a reoperation rate over 50% [123]. Further large-scale long-term outcomes studies are warranted before ACL repair can be advocated in the general population.
Table 6 summarizes the key facts on areas of uncertainty in management of ACL injury.
Rehabilitation after ACL reconstruction
Rigid rehabilitation protocols that centered around time intervals following ACL-R have gradually been replaced by criteria-based guidelines [28]. One of the first key criteria after ACL-R is achieving full passive and active knee extension. Loss of knee extension leads to abnormal joint biomechanics and subsequently abnormal articular cartilage contact pressures and inhibition of quadriceps activation [71, 150]. Loss of 3–5 degrees of knee extension, including loss of hyperextension, has an adverse effect on PROs, leads to increased risk of osteoarthritic changes [151], and is a common cause of repeat surgery [187]. Full knee extension should be achieved preoperatively, given that preoperative extension loss is associated with postoperative extension loss [142]. Postoperatively, restoration of full passive and active knee extension symmetrical to the contralateral normal knee should be restored within the first several weeks after ACL-R. Strategies to achieve full passive knee extension include prolonged stretching under low load and sleeping in a postoperative brace locked in full extension [28]. Use of patellofemoral joint mobilization to restore normal superior translation of the patella is critical to achieving full active knee extension without a quadriceps lag. Exercises to regain flexion, including wall slides and stationary bicycle, should begin shortly after ACL-R.
Although controversy remains in terms of the precise timing of re-initiation of weight-bearing following isolated ACL-R, clinical practice guidelines recommend either early full weight-bearing exercises or immediate postoperative weight-bearing as tolerated [108, 173, 186]. Many studies have failed to demonstrate any advantage to the use of postoperative knee braces [17, 79, 112].
Quadriceps activation and strengthening are a major focus of ACL-R rehabilitation. Managing an effusion promotes quadriceps activation and function since joint effusion is sensed by capsule mechanoreceptors with subsequent inhibitory signals to the quadriceps muscle [126]. Early quadriceps strengthening is key because quadriceps atrophy is associated with deficits in performance-based functional tests and PROs [105] and contributes to long-term deficits in extension strength [167, 184]. Deficits in quadriceps strength, defined as a quadriceps limb symmetry index < 85% at the time of return to sports, has been associated with elevated MRI chondral T2 relaxation times five years after return to sports, suggesting that quadriceps strength deficits may contribute to the future development of knee cartilage degeneration and OA [21].
Rehabilitation and psychological/neurological factors
Beyond physical factors, psychological factors such as motivation and self-efficacy have been shown to play an important role in outcomes following ACL-R [32]. Psychological readiness to return to sport has shown the strongest association with return to preinjury activity level [9], suggesting a benefit to focusing on the patient’s psychological state during rehabilitation [10].
The systemic neurologic response to injury, and the relative failure of traditional neuromuscular rehabilitation to address changes after injury, may help explain part of the greater relative risk of ipsilateral ACL re-injury of patients after ACL-R [132, 133, 183]. Deficient ligament mechanoreceptors and downstream effects of inflammation and joint effusion may impact the central nervous system [93, 128, 147]. In addition to afferent disruptions, patient experience-driven factors such as pain, compensatory mechanical patterns, and postoperative rehabilitation can lead to neuroplastic alterations, including decreased neural excitability [105, 136, 137]. Decreased excitability of the motor cortex after ACL-R increases the required stimulus in the motor cortex needed to activate the quadriceps and to control the knee in space [15, 105].
Table 7 summarizes the key facts on rehabilitation after ACL reconstruction.
ACL injury prevention
There has been a progressive increase in interest in primary ACL injury prevention. As complete prevention is not possible, a more accurate and updated term is ACL injury reduction strategies. Given that most ACL injuries are noncontact or indirect contact, including 88% of football/soccer ACL injuries, some, or even many, of these injuries may be prevented [43, 109]. Well-known neuromuscular and biomechanical factors associated with ACL injuries, such as dynamic knee valgus loading, shallow knee flexion, and homolateral/ipsilateral trunk tilt [43, 109], are the target of neuromuscular interventions in ACL injury reduction programs [75]. Neuromuscular training (NMT) programs, such as the FIFA 11 + , have been shown to be effective in reducing primary ACL injuries, with a 50% reduction of all ACL injuries and a 67% reduction of noncontact ACL injuries in female athletes [178]. The effectiveness of the programs is correlated with athlete compliance and frequency.
Given the demonstrated effectiveness of injury reduction programs, there is increased focus on removing barriers to implementation. A common approach is to implement NMT in early- or preadolescent athletes to engrain it in their athletic routine. Preventative training may be targeted based on risk profile [76]. As specific biomechanical variables [78, 106, 193] are correlated with increased risk of ACL injury, the adoption of movement analysis of jumping [78, 124] and cutting tasks [44, 51] has been suggested to target NMT on the athlete’s movement profile. Low external knee abduction moment (KAM) tasks are preferred to limit ACL loading. Technique training aims to reduce high KAM (Fig. 5A) to low KAM (Fig. 5B) at 90° change of direction. While literature theoretically supports this approach, its effectiveness has yet to be proven [46]. There is no consensus on the utility of screening, with particularly low utility seen when limiting testing to jumping tasks [98].
Secondary ACL injury reduction strategies are important because subsequent ACL injury risk is high, ranging from 7.9% in the MOON cohort [90] to up to 42% in the young female population that returns to play football/soccer [56, 177]. Of particular focus are young active patients [177] and those who have sustained noncontact injuries [45]. Optimization of mid- and late-stage rehabilitation after ACL-R targets neuromuscular function, starting from the recovery of isolated muscle strength and finishing with complete sport specific reconditioning [23]. A targeted NMT program should start as soon as the patient achieves adequate strength (isokinetic deficit < 20% for knee extensors and flexors) [24, 25], which can lead to low re-injury rate [13].
Table 8 summarizes the key facts on prevention of ACL injury.
Conclusion
The ACL is perhaps the most studied musculoskeletal structure in the human body. The explosion of literature on the ACL over the past two decades has shed light on how important a biomechanical role it serves both in isolation as well as in concert with other bony and soft tissue structures of the knee. Since the concept of individualized ACL-R in the early 2000’s, investigations have informed orthopedic surgeons worldwide about the importance of understanding the bony and ligamentous anatomy of the knee, proper graft selection for ACL-R, and postoperative rehabilitation strategies to optimize clinical outcomes and patients’ return to their previous activities.
Amidst this influx of knowledge, this is an exciting time for ACL research. The years to come will continue to explore more established ACL-R techniques but also produce valuable data on outcomes of newer techniques. To truly understand the merits of new technology and techniques in ACL reconstruction, researchers must strive for high quality clinical studies with long-term clinical follow-up.
References
Achtnich A, Herbst E, Forkel P, Metzlaff S, Sprenker F, Imhoff AB et al (2016) Acute proximal anterior cruciate ligament tears: outcomes after arthroscopic suture anchor repair versus anatomic single-bundle reconstruction. Arthroscopy 32(12):2562–2569
Adachi N, Ochi M, Uchio Y, Sakai Y, Kuriwaka M, Fujihara A (2003) Harvesting hamstring tendons for ACL reconstruction influences postoperative hamstring muscle performance. Arch Orthop Trauma Surg 123(9):460–465
Ageberg E, Forssblad M, Herbertsson P, Roos EM (2010) Sex differences in patient-reported outcomes after anterior cruciate ligament reconstruction: data from the Swedish knee ligament register. Am J Sports Med 38(7):1334–1342
Akoto R, Alm L, Drenck TC, Frings J, Krause M, Frosch KH (2020) Slope-correction osteotomy with lateral extra-articular tenodesis and revision anterior cruciate ligament reconstruction is highly effective in treating high-grade anterior knee laxity. Am J Sports Med 48(14):3478–3485
Almqvist KF, Jan H, Vercruysse C, Verbeeck R, Verdonk R (2007) The tibialis tendon as a valuable anterior cruciate ligament allograft substitute: biomechanical properties. Knee Surg Sports Traumatol Arthrosc 15(11):1326–1330
Amirault JD, Cameron JC, MacIntosh DL, Marks P (1988) Chronic anterior cruciate ligament deficiency. Long-term results of MacIntosh’s lateral substitution reconstruction. J Bone Joint Surg Br 70(4):622–624
Anderson AF, Anderson CN (2015) Correlation of meniscal and articular cartilage injuries in children and adolescents with timing of anterior cruciate ligament reconstruction. Am J Sports Med 43(2):275–281
Araujo P, van Eck CF, Torabi M, Fu FH (2013) How to optimize the use of MRI in anatomic ACL reconstruction. Knee Surg Sports Traumatol Arthrosc 21(7):1495–1501
Ardern CL, Österberg A, Tagesson S, Gauffin H, Webster KE, Kvist J (2014) The impact of psychological readiness to return to sport and recreational activities after anterior cruciate ligament reconstruction. Br J Sports Med 48(22):1613–1619
Ardern CL, Taylor NF, Feller JA, Whitehead TS, Webster KE (2013) Psychological responses matter in returning to preinjury level of sport after anterior cruciate ligament reconstruction surgery. Am J Sports Med 41(7):1549–1558
Ardern CL, Webster KE, Taylor NF, Feller JA (2011) Return to the preinjury level of competitive sport after anterior cruciate ligament reconstruction surgery: two-thirds of patients have not returned by 12 months after surgery. Am J Sports Med 39(3):538–543
Arnold MP, Calcei JG, Vogel N, Magnussen RA, Clatworthy M, Spalding T et al (2021) ACL Study Group survey reveals the evolution of anterior cruciate ligament reconstruction graft choice over the past three decades. Knee Surg Sports Traumatol Arthrosc 29(11):3871–3876
Arundale AJH, Capin JJ, Zarzycki R, Smith AH, Snyder-Mackler L (2018) Two year ACL reinjury rate of 2.5%: outcomes report of the men in a secondary ACL injury prevention program (ACL-SPORTS). Int J Sports Phys Ther 13(3):422–431
Barrett G, Stokes D, White M (2005) Anterior cruciate ligament reconstruction in patients older than 40 years: allograft versus autograft patellar tendon. Am J Sports Med 33(10):1505–1512
Baumeister J, Reinecke K, Schubert M, Weiss M (2011) Altered electrocortical brain activity after ACL reconstruction during force control. J Orthop Res 29(9):1383–1389
Benjaminse A, Gokeler A, van der Schans CP (2006) Clinical diagnosis of an anterior cruciate ligament rupture: a meta-analysis. J Orthop Sports Phys Ther 36(5):267–288
Birmingham TB, Bryant DM, Giffin JR, Litchfield RB, Kramer JF, Donner A et al (2008) A randomized controlled trial comparing the effectiveness of functional knee brace and neoprene sleeve use after anterior cruciate ligament reconstruction. Am J Sports Med 36(4):648–655
Bisson LJ, Kluczynski MA, Hagstrom LS, Marzo JM (2013) A prospective study of the association between bone contusion and intra-articular injuries associated with acute anterior cruciate ligament tear. Am J Sports Med 41(8):1801–1807
Blyth MJ, Gosal HS, Peake WM, Bartlett RJ (2003) Anterior cruciate ligament reconstruction in patients over the age of 50 years: 2- to 8-year follow-up. Knee Surg Sports Traumatol Arthrosc 11(4):204–211
Brandsson S, Kartus J, Larsson J, Eriksson BI, Karlsson J (2000) A comparison of results in middle-aged and young patients after anterior cruciate ligament reconstruction. Arthroscopy 16(2):178–182
Brunst C, Ithurburn MP, Zbojniewicz AM, Paterno MV, Schmitt LC (2021) Return-to-sport quadriceps strength symmetry impacts 5-year cartilage integrity after anterior cruciate ligament reconstruction: a preliminary analysis. J Orthop Res. https://doi.org/10.1002/jor.25029
Brusalis CM, Lakomkin N, Suryavanshi JR, Cruz AI Jr, Green DW, Jones KJ et al (2017) Clinical outcome reporting in youth ACL literature is widely variable. Orthop J Sports Med 5(8):2325967117724431
Buckthorpe M, Della Villa F (2020) Optimising the ‘mid-stage’ training and testing process after ACL reconstruction. Sports Med 50(4):657–678
Buckthorpe M, Della Villa F, Della Villa S, Roi GS (2019) On-field rehabilitation part 1: 4 pillars of high-quality on-field rehabilitation are restoring movement quality, physical conditioning, restoring sport-specific skills, and progressively developing chronic training load. J Orthop Sports Phys Ther 49(8):565–569
Buckthorpe M, Della Villa F, Della Villa S, Roi GS (2019) On-field rehabilitation part 2: a 5-stage program for the soccer player focused on linear movements, multidirectional movements, soccer-specific skills, soccer-specific movements, and modified practice. J Orthop Sports Phys Ther 49(8):570–575
Castoldi M, Magnussen RA, Gunst S, Batailler C, Neyret P, Lustig S et al (2020) A randomized controlled trial of bone-patellar tendon-bone anterior cruciate ligament reconstruction with and without lateral extra-articular tenodesis: 19-year clinical and radiological follow-up. Am J Sports Med 48(7):1665–1672
Cavaignac E, Coulin B, Tscholl P, Nik Mohd Fatmy N, Duthon V, Menetrey J (2017) Is quadriceps tendon autograft a better choice than hamstring autograft for anterior cruciate ligament reconstruction? A comparative study with a mean follow-up of 3.6 years. Am J Sports Med 45(6):1326–1332
Cavanaugh JT, Powers M (2017) ACL rehabilitation progression: where are we now? Curr Rev Musculoskelet Med 10(3):289–296
Chalmers PN, Mall NA, Moric M, Sherman SL, Paletta GP, Cole BJ et al (2014) Does ACL reconstruction alter natural history?: A systematic literature review of long-term outcomes. J Bone Joint Surg Am 96(4):292–300
Chiba D, Gale T, Nishida K, Suntaxi F, Lesniak BP, Fu FH et al (2021) Lateral extra-articular tenodesis contributes little to change in vivo kinematics after anterior cruciate ligament reconstruction: a randomized controlled trial. Am J Sports Med 49(7):1803–1812
Christensen JJ, Krych AJ, Engasser WM, Vanhees MK, Collins MS, Dahm DL (2015) Lateral tibial posterior slope is increased in patients with early graft failure after anterior cruciate ligament reconstruction. Am J Sports Med 43(10):2510–2514
Christino MA, Fantry AJ, Vopat BG (2015) Psychological aspects of recovery following anterior cruciate ligament reconstruction. J Am Acad Orthop Surg 23(8):501–509
Ciccotti MC, Secrist E, Tjoumakaris F, Ciccotti MG, Freedman KB (2017) Anatomic anterior cruciate ligament reconstruction via independent tunnel drilling: a systematic review of randomized controlled trials comparing patellar tendon and hamstring autografts. Arthroscopy 33(5):1062–1071
Cinque ME, Chahla J, Moatshe G, DePhillipo NN, Kennedy NI, Godin JA et al (2017) Outcomes and complication rates after primary anterior cruciate ligament reconstruction are similar in younger and older patients. Orthop J Sports Med 5(10):2325967117729659
Conte EJ, Hyatt AE, Gatt CJ Jr, Dhawan A (2014) Hamstring autograft size can be predicted and is a potential risk factor for anterior cruciate ligament reconstruction failure. Arthroscopy 30(7):882–890
Costa GG, Grassi A, Perelli S, Agrò G, Bozzi F, Lo Presti M et al (2019) Age over 50 years is not a contraindication for anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 27(11):3679–3691
Crum RJ, Kay J, Lesniak BP, Getgood A, Musahl V, de Sa D (2021) Bone versus all soft tissue quadriceps tendon autografts for anterior cruciate ligament reconstruction: a systematic review. Arthroscopy 37(3):1040–1052
Dahm DL, Wulf CA, Dajani KA, Dobbs RE, Levy BA, Stuart MA (2008) Reconstruction of the anterior cruciate ligament in patients over 50 years. J Bone Joint Surg Br 90(11):1446–1450
Davies NH, Niall D, King LJ, Lavelle J, Healy JC (2004) Magnetic resonance imaging of bone bruising in the acutely injured knee–short-term outcome. Clin Radiol 59(5):439–445
de Jong SN, van Caspel DR, van Haeff MJ, Saris DB (2007) Functional assessment and muscle strength before and after reconstruction of chronic anterior cruciate ligament lesions. Arthroscopy 23(1):21.e21-21.e11
Dejour D, Saffarini M, Demey G, Baverel L (2015) Tibial slope correction combined with second revision ACL produces good knee stability and prevents graft rupture. Knee Surg Sports Traumatol Arthrosc 23(10):2846–2852
DeLee JC, Curtis R (1983) Anterior cruciate ligament insufficiency in children. Clin Orthop Relat Res 172:112–118
Della Villa F, Buckthorpe M, Grassi A, Nabiuzzi A, Tosarelli F, Zaffagnini S et al (2020) Systematic video analysis of ACL injuries in professional male football (soccer): injury mechanisms, situational patterns and biomechanics study on 134 consecutive cases. Br J Sports Med 54(23):1423–1432
Della Villa F, Di Paolo S, Santagati D, Della Croce E, Lopomo NF, Grassi A et al (2021) A 2D video-analysis scoring system of 90° change of direction technique identifies football players with high knee abduction moment. Knee Surg Sports Traumatol Arthrosc. https://doi.org/10.1007/s00167-021-06571-2
Della Villa F, Hägglund M, Della Villa S, Ekstrand J, Waldén M (2021) High rate of second ACL injury following ACL reconstruction in male professional footballers: an updated longitudinal analysis from 118 players in the UEFA Elite Club Injury Study. Br J Sports Med. https://doi.org/10.1136/bjsports-2020-103555
Dempsey AR, Lloyd DG, Elliott BC, Steele JR, Munro BJ (2009) Changing sidestep cutting technique reduces knee valgus loading. Am J Sports Med 37(11):2194–2200
DiBartola AC, Everhart JS, Kaeding CC, Magnussen RA, Flanigan DC (2016) Maximum load to failure of high dose versus low dose gamma irradiation of anterior cruciate ligament allografts: a meta-analysis. Knee 23(5):755–762
Diermeier T, Rothrauff BB, Engebretsen L, Lynch AD, Ayeni OR, Paterno MV et al (2020) Treatment after anterior cruciate ligament injury: Panther symposium ACL treatment consensus group. Knee Surg Sports Traumatol Arthrosc 28(8):2390–2402
DiFelice GS, Villegas C, Taylor S (2015) Anterior cruciate ligament preservation: early results of a novel arthroscopic technique for suture anchor primary anterior cruciate ligament repair. Arthroscopy 31(11):2162–2171
Dodwell ER, Lamont LE, Green DW, Pan TJ, Marx RG, Lyman S (2014) 20 years of pediatric anterior cruciate ligament reconstruction in New York state. Am J Sports Med 42(3):675–680
Dos’Santos T, McBurnie A, Donelon T, Thomas C, Comfort P, Jones PA (2019) A qualitative screening tool to identify athletes with ‘high-risk’ movement mechanics during cutting: the cutting movement assessment score (CMAS). Phys Ther Sport 38:152–161
Dumont GD, Hogue GD, Padalecki JR, Okoro N, Wilson PL (2012) Meniscal and chondral injuries associated with pediatric anterior cruciate ligament tears: relationship of treatment time and patient-specific factors. Am J Sports Med 40(9):2128–2133
Dunn KL, Lam KC, Valovich McLeod TC (2016) Early operative versus delayed or nonoperative treatment of anterior cruciate ligament injuries in pediatric patients. J Athl Train 51(5):425–427
Eggli S, Kohlhof H, Zumstein M, Henle P, Hartel M, Evangelopoulos DS et al (2015) Dynamic intraligamentary stabilization: novel technique for preserving the ruptured ACL. Knee Surg Sports Traumatol Arthrosc 23(4):1215–1221
Engelman GH, Carry PM, Hitt KG, Polousky JD, Vidal AF (2014) Comparison of allograft versus autograft anterior cruciate ligament reconstruction graft survival in an active adolescent cohort. Am J Sports Med 42(10):2311–2318
Fältström A, Kvist J, Hägglund M (2021) High risk of new knee injuries in female soccer players after primary anterior cruciate ligament reconstruction at 5- to 10-year follow-up. Am J Sports Med 49(13):3479–3487
Feagin JA Jr, Curl WW (1976) Isolated tear of the anterior cruciate ligament: 5-year follow-up study. Am J Sports Med 4(3):95–100
Feller JA, Webster KE (2003) A randomized comparison of patellar tendon and hamstring tendon anterior cruciate ligament reconstruction. Am J Sports Med 31(4):564–573
Filardo G, de Caro F, Andriolo L, Kon E, Zaffagnini S, Marcacci M (2017) Do cartilage lesions affect the clinical outcome of anterior cruciate ligament reconstruction? A systematic review. Knee Surg Sports Traumatol Arthrosc 25(10):3061–3075
Frobell RB, Roos HP, Roos EM, Roemer FW, Ranstam J, Lohmander LS (2013) Treatment for acute anterior cruciate ligament tear: five year outcome of randomised trial. BMJ 346:f232
Gagliardi AG, Carry PM, Parikh HB, Traver JL, Howell DR, Albright JC (2019) ACL repair with suture ligament augmentation is associated with a high failure rate among adolescent patients. Am J Sports Med 47(3):560–566
Galway HR, MacIntosh DL (1980) The lateral pivot shift: a symptom and sign of anterior cruciate ligament insufficiency. Clin Orthop Relat Res 147:45–50
Getgood AMJ, Bryant DM, Litchfield R, Heard M, McCormack RG, Rezansoff A et al (2020) Lateral extra-articular tenodesis reduces failure of hamstring tendon autograft anterior cruciate ligament reconstruction: 2-year outcomes from the stability study randomized clinical trial. Am J Sports Med 48(2):285–297
Gföller P, Abermann E, Runer A, Hoser C, Pflüglmayer M, Wierer G et al (2019) Non-operative treatment of ACL injury is associated with opposing subjective and objective outcomes over 20 years of follow-up. Knee Surg Sports Traumatol Arthrosc 27(8):2665–2671
Gobbi A, Domzalski M, Pascual J (2004) Comparison of anterior cruciate ligament reconstruction in male and female athletes using the patellar tendon and hamstring autografts. Knee Surg Sports Traumatol Arthrosc 12(6):534–539
Grassi A, Macchiarola L, Urrizola Barrientos F, Zicaro JP, Costa Paz M, Adravanti P et al (2019) Steep posterior tibial slope, anterior tibial subluxation, deep posterior lateral femoral condyle, and meniscal deficiency are common findings in multiple anterior cruciate ligament failures: an MRI case-control study. Am J Sports Med 47(2):285–295
Guenther D, Irarrázaval S, Albers M, Vernacchia C, Irrgang JJ, Musahl V et al (2017) Area of the tibial insertion site of the anterior cruciate ligament as a predictor for graft size. Knee Surg Sports Traumatol Arthrosc 25(5):1576–1582
Guenther D, Rahnemai-Azar AA, Bell KM, Irarrázaval S, Fu FH, Musahl V et al (2017) The anterolateral capsule of the knee behaves like a sheet of fibrous tissue. Am J Sports Med 45(4):849–855
Hagmeijer MH, Hevesi M, Desai VS, Sanders TL, Camp CL, Hewett TE et al (2019) Secondary meniscal tears in patients with anterior cruciate ligament injury: relationship among operative management, osteoarthritis, and arthroplasty at 18-year mean follow-up. Am J Sports Med 47(7):1583–1590
Han HS, Seong SC, Lee S, Lee MC (2008) Anterior cruciate ligament reconstruction: quadriceps versus patellar autograft. Clin Orthop Relat Res 466(1):198–204
Harner CD, Irrgang JJ, Paul J, Dearwater S, Fu FH (1992) Loss of motion after anterior cruciate ligament reconstruction. Am J Sports Med 20(5):499–506
Helito CP, Bonadio MB, Rozas JS, Wey JM, Pereira CA, Cardoso TP et al (2016) Biomechanical study of strength and stiffness of the knee anterolateral ligament. BMC Musculoskelet Disord 17:193
Herbst E, Albers M, Burnham JM, Shaikh HS, Naendrup JH, Fu FH et al (2017) The anterolateral complex of the knee: a pictorial essay. Knee Surg Sports Traumatol Arthrosc 25(4):1009–1014
Heusdens CHW, Hopper GP, Dossche L, Roelant E, Mackay GM (2019) Anterior cruciate ligament repair with independent suture tape reinforcement: a case series with 2-year follow-up. Knee Surg Sports Traumatol Arthrosc 27(1):60–67
Hewett TE, Ford KR, Hoogenboom BJ, Myer GD (2010) Understanding and preventing ACL injuries: current biomechanical and epidemiologic considerations—update 2010. N Am J Sports Phys Ther 5(4):234–251
Hewett TE, Ford KR, Xu YY, Khoury J, Myer GD (2017) Effectiveness of neuromuscular training based on the neuromuscular risk profile. Am J Sports Med 45(9):2142–2147
Hewett TE, Myer GD, Ford KR (2006) Anterior cruciate ligament injuries in female athletes: part 1, mechanisms and risk factors. Am J Sports Med 34(2):299–311
Hewett TE, Myer GD, Ford KR, Heidt RS Jr, Colosimo AJ, McLean SG et al (2005) Biomechanical measures of neuromuscular control and valgus loading of the knee predict anterior cruciate ligament injury risk in female athletes: a prospective study. Am J Sports Med 33(4):492–501
Hiemstra LA, Heard SM, Sasyniuk TM, Buchko GL, Reed JG, Monteleone BJ (2009) Knee immobilization for pain control after a hamstring tendon anterior cruciate ligament reconstruction: a randomized clinical trial. Am J Sports Med 37(1):56–64
Hoffmann C, Friederichs J, von Rüden C, Schaller C, Bühren V, Moessmer C (2017) Primary single suture anchor re-fixation of anterior cruciate ligament proximal avulsion tears leads to good functional mid-term results: a preliminary study in 12 patients. J Orthop Surg Res 12(1):171
Holm I, Oiestad BE, Risberg MA, Gunderson R, Aune AK (2012) No differences in prevalence of osteoarthritis or function after open versus endoscopic technique for anterior cruciate ligament reconstruction: 12-year follow-up report of a randomized controlled trial. Am J Sports Med 40(11):2492–2498
Horvath A, Meredith SJ, Nishida K, Hoshino Y, Musahl V (2020) Objectifying the pivot shift test. Sports Med Arthrosc Rev 28(2):36–40
Hoshino Y, Araujo P, Ahldén M, Samuelsson K, Muller B, Hofbauer M et al (2013) Quantitative evaluation of the pivot shift by image analysis using the iPad. Knee Surg Sports Traumatol Arthrosc 21(4):975–980
Houck DA, Kraeutler MJ, Vidal AF, McCarty EC, Bravman JT, Wolcott ML (2018) Variance in anterior cruciate ligament reconstruction graft selection based on patient demographics and location within the multicenter orthopaedic outcomes network cohort. J Knee Surg 31(5):472–478
Hunnicutt JL, Xerogeanes JW, Tsai LC, Sprague PA, Newsome M, Slone HS et al (2021) Terminal knee extension deficit and female sex predict poorer quadriceps strength following ACL reconstruction using all-soft tissue quadriceps tendon autografts. Knee Surg Sports Traumatol Arthrosc 29(9):3085–3095
Hurd WJ, Axe MJ, Snyder-Mackler L (2008) A 10-year prospective trial of a patient management algorithm and screening examination for highly active individuals with anterior cruciate ligament injury: part 2, determinants of dynamic knee stability. Am J Sports Med 36(1):48–56
Imhoff FB, Mehl J, Comer BJ, Obopilwe E, Cote MP, Feucht MJ et al (2019) Slope-reducing tibial osteotomy decreases ACL-graft forces and anterior tibial translation under axial load. Knee Surg Sports Traumatol Arthrosc 27(10):3381–3389
Jones KG (1963) Reconstruction of the anterior cruciate ligament. A techinque using the central one-third of the patellar ligament. J Bone Jt Surg Am 45:925–932
Kaeding CC, Aros B, Pedroza A, Pifel E, Amendola A, Andrish JT et al (2011) Allograft versus autograft anterior cruciate ligament reconstruction: predictors of failure from a MOON prospective longitudinal cohort. Sports Health 3(1):73–81
Kaeding CC, Pedroza AD, Reinke EK, Huston LJ, Spindler KP (2015) Risk factors and predictors of subsequent ACL injury in either knee after ACL reconstruction: prospective analysis of 2488 primary ACL reconstructions from the MOON cohort. Am J Sports Med 43(7):1583–1590
Kan SL, Yuan ZF, Ning GZ, Yang B, Li HL, Sun JC et al (2016) Autograft versus allograft in anterior cruciate ligament reconstruction: a meta-analysis with trial sequential analysis. Medicine 95(38):e4936
Kaplan N, Wickiewicz TL, Warren RF (1990) Primary surgical treatment of anterior cruciate ligament ruptures. A long-term follow-up study. Am J Sports Med 18(4):354–358
Kapreli E, Athanasopoulos S, Gliatis J, Papathanasiou M, Peeters R, Strimpakos N et al (2009) Anterior cruciate ligament deficiency causes brain plasticity: a functional MRI study. Am J Sports Med 37(12):2419–2426
Kennedy MI, Claes S, Fuso FA, Williams BT, Goldsmith MT, Turnbull TL et al (2015) The anterolateral ligament: an anatomic, radiographic, and biomechanical analysis. Am J Sports Med 43(7):1606–1615
Kim DK, Park WH (2015) Sex differences in knee strength deficit 1 year after anterior cruciate ligament reconstruction. J Phys Ther Sci 27(12):3847–3849
Kim SJ, Kumar P, Oh KS (2009) Anterior cruciate ligament reconstruction: autogenous quadriceps tendon-bone compared with bone-patellar tendon-bone grafts at 2-year follow-up. Arthroscopy 25(2):137–144
Kraeutler MJ, Bravman JT, McCarty EC (2013) Bone-patellar tendon-bone autograft versus allograft in outcomes of anterior cruciate ligament reconstruction: a meta-analysis of 5182 patients. Am J Sports Med 41(10):2439–2448
Krosshaug T, Steffen K, Kristianslund E, Nilstad A, Mok KM, Myklebust G et al (2016) The vertical drop jump is a poor screening test for ACL injuries in female elite soccer and handball players: a prospective cohort study of 710 athletes. Am J Sports Med 44(4):874–883
Krych AJ, Jackson JD, Hoskin TL, Dahm DL (2008) A meta-analysis of patellar tendon autograft versus patellar tendon allograft in anterior cruciate ligament reconstruction. Arthroscopy 24(3):292–298
Kuechle DK, Pearson SE, Beach WR, Freeman EL, Pawlowski DF, Whipple TL et al (2002) Allograft anterior cruciate ligament reconstruction in patients over 40 years of age. Arthroscopy 18(8):845–853
Lansdown DA, Riff AJ, Meadows M, Yanke AB, Bach BR Jr (2017) What factors influence the biomechanical properties of allograft tissue for ACL reconstruction? A systematic review. Clin Orthop Relat Res 475(10):2412–2426
Lautamies R, Harilainen A, Kettunen J, Sandelin J, Kujala UM (2008) Isokinetic quadriceps and hamstring muscle strength and knee function 5 years after anterior cruciate ligament reconstruction: comparison between bone-patellar tendon-bone and hamstring tendon autografts. Knee Surg Sports Traumatol Arthrosc 16(11):1009–1016
Lawrence JT, Argawal N, Ganley TJ (2011) Degeneration of the knee joint in skeletally immature patients with a diagnosis of an anterior cruciate ligament tear: is there harm in delay of treatment? Am J Sports Med 39(12):2582–2587
Lemaire M (1980) Technique actuelle de plastie ligamentaire pour rupture ancienne du ligament croisé antérieur. Rev Chir Orthop Repar Appar 66(8):523–525
Lepley AS, Ericksen HM, Sohn DH, Pietrosimone BG (2014) Contributions of neural excitability and voluntary activation to quadriceps muscle strength following anterior cruciate ligament reconstruction. Knee 21(3):736–742
Leppänen M, Pasanen K, Kujala UM, Vasankari T, Kannus P, Äyrämö S et al (2017) Stiff landings are associated with increased ACL injury risk in young female basketball and floorball players. Am J Sports Med 45(2):386–393
Leys T, Salmon L, Waller A, Linklater J, Pinczewski L (2012) Clinical results and risk factors for reinjury 15 years after anterior cruciate ligament reconstruction: a prospective study of hamstring and patellar tendon grafts. Am J Sports Med 40(3):595–605
Logerstedt DS, Scalzitti D, Risberg MA, Engebretsen L, Webster KE, Feller J et al (2017) Knee stability and movement coordination impairments: knee ligament sprain revision 2017. J Orthop Sports Phys Ther 47(11):A1-a47
Lucarno S, Zago M, Buckthorpe M, Grassi A, Tosarelli F, Smith R et al (2021) Systematic video analysis of anterior cruciate ligament injuries in professional female soccer players. Am J Sports Med 49(7):1794–1802
Magnussen RA, Carey JL, Spindler KP (2011) Does autograft choice determine intermediate-term outcome of ACL reconstruction? Knee Surg Sports Traumatol Arthrosc 19(3):462–472
Maletis GB, Inacio MC, Desmond JL, Funahashi TT (2013) Reconstruction of the anterior cruciate ligament: association of graft choice with increased risk of early revision. Bone Joint J 95-B(5):623–628
Mayr HO, Hochrein A, Hein W, Hube R, Bernstein A (2010) Rehabilitation results following anterior cruciate ligament reconstruction using a hard brace compared to a fluid-filled soft brace. Knee 17(2):119–126
Mehran N, Moutzouros VB, Bedi A (2015) A review of current graft options for anterior cruciate ligament reconstruction. JBJS Rev 3(11):e2
Meuffels DE, Favejee MM, Vissers MM, Heijboer MP, Reijman M, Verhaar JA (2009) Ten year follow-up study comparing conservative versus operative treatment of anterior cruciate ligament ruptures. A matched-pair analysis of high level athletes. Br J Sports Med 43(5):347–351
Mohtadi N, Chan D, Barber R, Oddone Paolucci E (2015) A randomized clinical trial comparing patellar tendon, hamstring tendon, and double-bundle ACL reconstructions: patient-reported and clinical outcomes at a minimal 2-year follow-up. Clin J Sport Med 25(4):321–331
Mohtadi NG, Chan DS, Dainty KN, Whelan DB (2011) Patellar tendon versus hamstring tendon autograft for anterior cruciate ligament rupture in adults. Cochrane Database Syst Rev 2011(9)
Murray MM, Flutie BM, Kalish LA, Ecklund K, Fleming BC, Proffen BL et al (2016) The bridge-enhanced anterior cruciate ligament repair (BEAR) procedure: an early feasibility cohort study. Orthop J Sports Med 4(11):2325967116672176
Murray MM, Kalish LA, Fleming BC, Flutie B, Freiberger C, Henderson RN et al (2019) Bridge-enhanced anterior cruciate ligament repair: two-year results of a first-in-human study. Orthop J Sports Med 7(3):2325967118824356
Musahl V, Diermeier T, de Sa D, Karlsson J (2020) ACL surgery: when to do it? Knee Surg Sports Traumatol Arthrosc 28(7):2023–2026
Musahl V, Herbst E, Burnham JM, Fu FH (2018) The anterolateral complex and anterolateral ligament of the knee. J Am Acad Orthop Surg 26(8):261–267
Musahl V, Hoshino Y, Ahlden M, Araujo P, Irrgang JJ, Zaffagnini S et al (2012) The pivot shift: a global user guide. Knee Surg Sports Traumatol Arthrosc 20(4):724–731
Musahl V, Karlsson J (2019) Anterior cruciate ligament tear. N Engl J Med 380(24):2341–2348
Nwachukwu BU, Patel BH, Lu Y, Allen AA, Williams RJ 3rd (2019) Anterior cruciate ligament repair outcomes: an updated systematic review of recent literature. Arthroscopy 35(7):2233–2247
Padua DA, DiStefano LJ, Beutler AI, de la Motte SJ, DiStefano MJ, Marshall SW (2015) The landing error scoring system as a screening tool for an anterior cruciate ligament injury-prevention program in elite-youth soccer athletes. J Athl Train 50(6):589–595
Pallis M, Svoboda SJ, Cameron KL, Owens BD (2012) Survival comparison of allograft and autograft anterior cruciate ligament reconstruction at the United States military academy. Am J Sports Med 40(6):1242–1246
Palmieri-Smith RM, Villwock M, Downie B, Hecht G, Zernicke R (2013) Pain and effusion and quadriceps activation and strength. J Athl Train 48(2):186–191
Papageorgiou CD, Kostopoulos VK, Moebius UG, Petropoulou KA, Georgoulis AD, Soucacos PN (2001) Patellar fractures associated with medial-third bone-patellar tendon-bone autograft ACL reconstruction. Knee Surg Sports Traumatol Arthrosc 9(3):151–154
Park HB, Koh M, Cho SH, Hutchinson B, Lee B (2005) Mapping the rat somatosensory pathway from the anterior cruciate ligament nerve endings to the cerebrum. J Orthop Res 23(6):1419–1424
Park MJ, Lee MC, Seong SC (2001) A comparative study of the healing of tendon autograft and tendon-bone autograft using patellar tendon in rabbits. Int Orthop 25(1):35–39
Park SS, Dwyer T, Congiusta F, Whelan DB, Theodoropoulos J (2015) Analysis of irradiation on the clinical effectiveness of allogenic tissue when used for primary anterior cruciate ligament reconstruction. Am J Sports Med 43(1):226–235
Paterno MV (2017) Non-operative care of the patient with an ACL-deficient knee. Curr Rev Musculoskelet Med 10(3):322–327
Paterno MV, Rauh MJ, Schmitt LC, Ford KR, Hewett TE (2012) Incidence of contralateral and ipsilateral anterior cruciate ligament (ACL) injury after primary ACL reconstruction and return to sport. Clin J Sport Med 22(2):116–121
Paterno MV, Schmitt LC, Ford KR, Rauh MJ, Myer GD, Huang B et al (2010) Biomechanical measures during landing and postural stability predict second anterior cruciate ligament injury after anterior cruciate ligament reconstruction and return to sport. Am J Sports Med 38(10):1968–1978
Persson A, Fjeldsgaard K, Gjertsen JE, Kjellsen AB, Engebretsen L, Hole RM et al (2014) Increased risk of revision with hamstring tendon grafts compared with patellar tendon grafts after anterior cruciate ligament reconstruction: a study of 12,643 patients from the Norwegian Cruciate Ligament Registry, 2004–2012. Am J Sports Med 42(2):285–291
Peterson DC, Ayeni OR (2016) Pediatric anterior cruciate ligament reconstruction outcomes. Curr Rev Musculoskelet Med 9(4):339–347
Pietrosimone BG, Lepley AS, Ericksen HM, Clements A, Sohn DH, Gribble PA (2015) Neural excitability alterations after anterior cruciate ligament reconstruction. J Athl Train 50(6):665–674
Pietrosimone BG, Lepley AS, Ericksen HM, Gribble PA, Levine J (2013) Quadriceps strength and corticospinal excitability as predictors of disability after anterior cruciate ligament reconstruction. J Sport Rehabil 22(1):1–6
Prodromos CC, Han Y, Rogowski J, Joyce B, Shi K (2007) A meta-analysis of the incidence of anterior cruciate ligament tears as a function of gender, sport, and a knee injury-reduction regimen. Arthroscopy 23(12):1320-1325.e1326
Rahr-Wagner L, Thillemann TM, Pedersen AB, Lind M (2014) Comparison of hamstring tendon and patellar tendon grafts in anterior cruciate ligament reconstruction in a nationwide population-based cohort study: results from the danish registry of knee ligament reconstruction. Am J Sports Med 42(2):278–284
Ramski DE, Kanj WW, Franklin CC, Baldwin KD, Ganley TJ (2014) Anterior cruciate ligament tears in children and adolescents: a meta-analysis of nonoperative versus operative treatment. Am J Sports Med 42(11):2769–2776
Reid JS, Hanks GA, Kalenak A, Kottmeier S, Aronoff V (1992) The Ellison iliotibial-band transfer for a torn anterior cruciate ligament of the knee. Long-term follow-up. J Bone Joint Surg Am 74(9):1392–1402
Robertson GA, Coleman SG, Keating JF (2009) Knee stiffness following anterior cruciate ligament reconstruction: the incidence and associated factors of knee stiffness following anterior cruciate ligament reconstruction. Knee 16(4):245–247
Robson AW (1903) VI. Ruptured crucial ligaments and their repair by operation. Ann Surg 37(5):716–718
Salem HS, Varzhapetyan V, Patel N, Dodson CC, Tjoumakaris FP, Freedman KB (2019) Anterior cruciate ligament reconstruction in young female athletes: patellar versus hamstring tendon autografts. Am J Sports Med 47(9):2086–2092
Scholten RJ, Opstelten W, van der Plas CG, Bijl D, Deville WL, Bouter LM (2003) Accuracy of physical diagnostic tests for assessing ruptures of the anterior cruciate ligament: a meta-analysis. J Fam Pract 52(9):689–694
Schreiber VM, van Eck CF, Fu FH (2010) Anatomic double-bundle ACL reconstruction. Sports Med Arthrosc Rev 18(1):27–32
Schutte MJ, Dabezies EJ, Zimny ML, Happel LT (1987) Neural anatomy of the human anterior cruciate ligament. J Bone Joint Surg Am 69(2):243–247
Schwartz HE, Matava MJ, Proch FS, Butler CA, Ratcliffe A, Levy M et al (2006) The effect of gamma irradiation on anterior cruciate ligament allograft biomechanical and biochemical properties in the caprine model at time zero and at 6 months after surgery. Am J Sports Med 34(11):1747–1755
Sheean AJ, Lian J, Tisherman R, Meredith SJ, de Sa D, Lynch A et al (2020) Augmentation of anatomic anterior cruciate ligament reconstruction with lateral extra-articular tenodesis does not significantly affect rotatory knee laxity: a time zero, in vivo kinematic analysis. Am J Sports Med 48(14):3495–3502
Shelbourne KD, Gray T (2009) Minimum 10-year results after anterior cruciate ligament reconstruction: how the loss of normal knee motion compounds other factors related to the development of osteoarthritis after surgery. Am J Sports Med 37(3):471–480
Shelbourne KD, Urch SE, Gray T, Freeman H (2012) Loss of normal knee motion after anterior cruciate ligament reconstruction is associated with radiographic arthritic changes after surgery. Am J Sports Med 40(1):108–113
Slone HS, Romine SE, Premkumar A, Xerogeanes JW (2015) Quadriceps tendon autograft for anterior cruciate ligament reconstruction: a comprehensive review of current literature and systematic review of clinical results. Arthroscopy 31(3):541–554
Smith TO, Postle K, Penny F, McNamara I, Mann CJ (2014) Is reconstruction the best management strategy for anterior cruciate ligament rupture? A systematic review and meta-analysis comparing anterior cruciate ligament reconstruction versus non-operative treatment. Knee 21(2):462–470
Snaebjörnsson T, Hamrin-Senorski E, Svantesson E, Karlsson L, Engebretsen L, Karlsson J et al (2019) Graft diameter and graft type as predictors of anterior cruciate ligament revision: a cohort study including 18,425 patients from the Swedish and Norwegian National Knee Ligament Registries. J Bone Joint Surg Am 101(20):1812–1820
Snaebjörnsson T, Hamrin Senorski E, Ayeni OR, Alentorn-Geli E, Krupic F, Norberg F et al (2017) Graft diameter as a predictor for revision anterior cruciate ligament reconstruction and KOOS and EQ-5D values: a cohort study from the Swedish National Knee Ligament Register based on 2240 patients. Am J Sports Med 45(9):2092–2097
Solomon DH, Simel DL, Bates DW, Katz JN, Schaffer JL (2001) The rational clinical examination. Does this patient have a torn meniscus or ligament of the knee? Value of the physical examination. JAMA 286(13):1610–1620
Song GY, Ni QK, Zheng T, Zhang ZJ, Feng H, Zhang H (2020) Slope-reducing tibial osteotomy combined with primary anterior cruciate ligament reconstruction produces improved knee stability in patients with steep posterior tibial slope, excessive anterior tibial subluxation in extension, and chronic meniscal posterior horn tears. Am J Sports Med 48(14):3486–3494
Sonnery-Cottet B, Haidar I, Rayes J, Fradin T, Ngbilo C, Vieira TD et al (2021) Long-term graft rupture rates after combined ACL and anterolateral ligament reconstruction versus isolated ACL reconstruction: a matched-pair analysis from the SANTI study group. Am J Sports Med 49(11):2889–2897
Sonnery-Cottet B, Thaunat M, Freychet B, Pupim BH, Murphy CG, Claes S (2015) Outcome of a combined anterior cruciate ligament and anterolateral ligament reconstruction technique with a minimum 2-year follow-up. Am J Sports Med 43(7):1598–1605
Spencer L, Burkhart TA, Tran MN, Rezansoff AJ, Deo S, Caterine S et al (2015) Biomechanical analysis of simulated clinical testing and reconstruction of the anterolateral ligament of the knee. Am J Sports Med 43(9):2189–2197
Stein DA, Hunt SA, Rosen JE, Sherman OH (2002) The incidence and outcome of patella fractures after anterior cruciate ligament reconstruction. Arthroscopy 18(6):578–583
Takeuchi S, Rothrauff BB, Taguchi M, Kanto R, Onishi K, Fu FH (2021) In situ cross-sectional area of the quadriceps tendon using preoperative magnetic resonance imaging significantly correlates with the intraoperative diameter of the quadriceps tendon autograft. Knee Surg Sports Traumatol Arthrosc 29(3):742–749
Tan SH, Lau BP, Khin LW, Lingaraj K (2016) The importance of patient sex in the outcomes of anterior cruciate ligament reconstructions: a systematic review and meta-analysis. Am J Sports Med 44(1):242–254
Tejwani SG, Chen J, Funahashi TT, Love R, Maletis GB (2015) Revision risk after allograft anterior cruciate ligament reconstruction: association with graft processing techniques, patient characteristics, and graft type. Am J Sports Med 43(11):2696–2705
Thein R, Boorman-Padgett J, Stone K, Wickiewicz TL, Imhauser CW, Pearle AD (2016) Biomechanical assessment of the anterolateral ligament of the knee: a secondary restraint in simulated tests of the pivot shift and of anterior stability. J Bone Joint Surg Am 98(11):937–943
Theologis AA, Kuo D, Cheng J, Bolbos RI, Carballido-Gamio J, Ma CB et al (2011) Evaluation of bone bruises and associated cartilage in anterior cruciate ligament-injured and -reconstructed knees using quantitative t(1ρ) magnetic resonance imaging: 1-year cohort study. Arthroscopy 27(1):65–76
Thomas AC, Wojtys EM, Brandon C, Palmieri-Smith RM (2016) Muscle atrophy contributes to quadriceps weakness after anterior cruciate ligament reconstruction. J Sci Med Sport 19(1):7–11
Thompson SM, Salmon LJ, Waller A, Linklater J, Roe JP, Pinczewski LA (2016) Twenty-year outcome of a longitudinal prospective evaluation of isolated endoscopic anterior cruciate ligament reconstruction with patellar tendon or hamstring autograft. Am J Sports Med 44(12):3083–3094
Tian S, Wang B, Liu L, Wang Y, Ha C, Li Q et al (2016) Irradiated hamstring tendon allograft versus autograft for anatomic double-bundle anterior cruciate ligament reconstruction: midterm clinical outcomes. Am J Sports Med 44(10):2579–2588
Tiderius CJ, Olsson LE, Nyquist F, Dahlberg L (2005) Cartilage glycosaminoglycan loss in the acute phase after an anterior cruciate ligament injury: delayed gadolinium-enhanced magnetic resonance imaging of cartilage and synovial fluid analysis. Arthritis Rheum 52(1):120–127
Tomita F, Yasuda K, Mikami S, Sakai T, Yamazaki S, Tohyama H (2001) Comparisons of intraosseous graft healing between the doubled flexor tendon graft and the bone-patellar tendon-bone graft in anterior cruciate ligament reconstruction. Arthroscopy 17(5):461–476
Tsoukas D, Fotopoulos V, Basdekis G, Makridis KG (2016) No difference in osteoarthritis after surgical and non-surgical treatment of ACL-injured knees after 10 years. Knee Surg Sports Traumatol Arthrosc 24(9):2953–2959
van Melick N, van Cingel RE, Brooijmans F, Neeter C, van Tienen T, Hullegie W et al (2016) Evidence-based clinical practice update: practice guidelines for anterior cruciate ligament rehabilitation based on a systematic review and multidisciplinary consensus. Br J Sports Med 50(24):1506–1515
Vaswani R, Meredith SJ, Lian J, Li R, Nickoli M, Fu FH et al (2020) Intercondylar notch size can be predicted on preoperative magnetic resonance imaging. Arthrosc Sports Med Rehabil 2(1):e17–e22
Ventura A, Legnani C, Terzaghi C, Borgo E (2012) Single- and double-bundle anterior cruciate ligament reconstruction in patients aged over 50 years. Arthroscopy 28(11):1702–1709
Viskontas DG, Giuffre BM, Duggal N, Graham D, Parker D, Coolican M (2008) Bone bruises associated with ACL rupture: correlation with injury mechanism. Am J Sports Med 36(5):927–933
Webster KE, Feller JA (2016) Exploring the high reinjury rate in younger patients undergoing anterior cruciate ligament reconstruction. Am J Sports Med 44(11):2827–2832
Webster KE, Hewett TE (2018) Meta-analysis of meta-analyses of anterior cruciate ligament injury reduction training programs. J Orthop Res 36(10):2696–2708
Webster KE, Nagelli CV, Hewett TE, Feller JA (2018) Factors associated with psychological readiness to return to sport after anterior cruciate ligament reconstruction surgery. Am J Sports Med 46(7):1545–1550
Wellsandt E, Failla MJ, Axe MJ, Snyder-Mackler L (2018) Does anterior cruciate ligament reconstruction improve functional and radiographic outcomes over nonoperative management 5 years after injury? Am J Sports Med 46(9):2103–2112
Wellsandt E, Khandha A, Capin J, Buchanan TS, Snyder-Mackler L (2020) Operative and nonoperative management of anterior cruciate ligament injury: differences in gait biomechanics at 5 years. J Orthop Res 38(12):2675–2684
West RV, Harner CD (2005) Graft selection in anterior cruciate ligament reconstruction. J Am Acad Orthop Surg 13(3):197–207
Wiggins AJ, Grandhi RK, Schneider DK, Stanfield D, Webster KE, Myer GD (2016) Risk of secondary injury in younger athletes after anterior cruciate ligament reconstruction: a systematic review and meta-analysis. Am J Sports Med 44(7):1861–1876
Williams GN, Buchanan TS, Barrance PJ, Axe MJ, Snyder-Mackler L (2005) Quadriceps weakness, atrophy, and activation failure in predicted noncopers after anterior cruciate ligament injury. Am J Sports Med 33(3):402–407
Wilson WT, Hopper GP, Byrne PA, MacKay GM (2016) Anterior cruciate ligament repair with internal brace ligament augmentation. Surg Technol Int 29:273–278
Wright RW, Haas AK, Anderson J, Calabrese G, Cavanaugh J, Hewett TE et al (2015) Anterior cruciate ligament reconstruction rehabilitation: MOON guidelines. Sports Health 7(3):239–243
Wylie JD, Marchand LS, Burks RT (2017) Etiologic factors that lead to failure after primary anterior cruciate ligament surgery. Clin Sports Med 36(1):155–172
Xie X, Liu X, Chen Z, Yu Y, Peng S, Li Q (2015) A meta-analysis of bone-patellar tendon-bone autograft versus four-strand hamstring tendon autograft for anterior cruciate ligament reconstruction. Knee 22(2):100–110
Yamaguchi KT, Cheung EC, Markolf KL, Boguszewski DV, Mathew J, Lama CJ et al (2018) Effects of anterior closing wedge tibial osteotomy on anterior cruciate ligament force and knee kinematics. Am J Sports Med 46(2):370–377
Yoon KH, Yoo JH, Kim KI (2011) Bone contusion and associated meniscal and medial collateral ligament injury in patients with anterior cruciate ligament rupture. J Bone Joint Surg Am 93(16):1510–1518
Zaffagnini S, Marcheggiani Muccioli GM, Grassi A, Roberti di Sarsina T, Raggi F, Signorelli C et al (2017) Over-the-top ACL reconstruction plus extra-articular lateral tenodesis with hamstring tendon grafts: prospective evaluation with 20-year minimum follow-up. Am J Sports Med 45(14):3233–3242
Zakko P, van Eck CF, Guenther D, Irrgang JJ, Fu FH (2017) Can we predict the size of frequently used autografts in ACL reconstruction? Knee Surg Sports Traumatol Arthrosc 25(12):3704–3710
Zebis MK, Aagaard P, Andersen LL, Hölmich P, Clausen MB, Brandt M et al (2021) First-time anterior cruciate ligament injury in adolescent female elite athletes: a prospective cohort study to identify modifiable risk factors. Knee Surg Sports Traumatol Arthrosc. https://doi.org/10.1007/s00167-021-06595-8
Zeng C, Gao SG, Li H, Yang T, Luo W, Li YS et al (2016) Autograft versus allograft in anterior cruciate ligament reconstruction: a meta-analysis of randomized controlled trials and systematic review of overlapping systematic reviews. Arthroscopy 32(1):153-163.e118
Zink EJ, Trumper RV, Smidt CR, Rice EL, Reiser RF 2nd (2005) Gender comparison of knee strength recovery following ACL reconstruction with contralateral patellar tendon graft. Biomed Sci Instrum 41:323–328
Acknowledgements
The authors dedicate this work to the life and legacy of Dr. Freddie Fu. Through his charisma, love of orthopaedics, and passion for teaching, Dr. Fu directly touched the lives of tens of thousands of orthopaedic surgeons, young and old, domestic and from afar. His lessons of respecting the past, doing the right thing for patients, and working hard with a smile on your face are qualities that each of us strive to achieve in both our careers as well as our personal lives. Mentors like Dr. Fu come once in a lifetime. Although he is irreplaceable, he has forged a path for many of us to continue his legacy and carry forth the lessons he so graciously taught us during his lifetime.
Funding
There was no funding for this manuscript.
Author information
Authors and Affiliations
Contributions
VM, IDE, EMN, JFD, GAL, JDH, and FDV contributed to manuscript writing and editing. SZ, JJI, and JK contributed to editing. FHF contributed to the groundwork for this manuscript and inspiration for its production. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Musahl, V., Engler, I.D., Nazzal, E.M. et al. Current trends in the anterior cruciate ligament part II: evaluation, surgical technique, prevention, and rehabilitation. Knee Surg Sports Traumatol Arthrosc 30, 34–51 (2022). https://doi.org/10.1007/s00167-021-06825-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-021-06825-z